Folate-decorated polymeric Pt(ii) prodrug micelles for targeted intracellular delivery and cytosolic glutathione-triggered release of platinum anticancer drugs†
Abstract
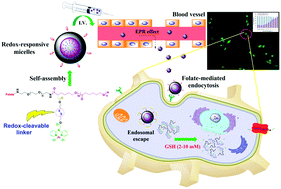
In this work, we have developed redox-responsive platinum(II) prodrug micelles based on folate-poly(ethylene glycol)-b-poly(α-Pt(II)-SS-caprolactone/caprolactone) (FA-PEG-b-P(α-Pt(II)-SS-CL/CL)) copolymers. The FA-PEG-b-P(α-Pt(II)-SS-CL/CL) were conveniently prepared by conjugating the Pt(di(2-pyridyl)amine-CH2CO2H)Cl2 (Pt(dpa-CH2CO2H)Cl2) complex to the folate-poly(ethylene glycol)-b-poly(α-acetic acid-caprolactone/caprolactone) (FA-PEG-b-P(α-AA-CL/CL)) amphiphilic block copolymer with functional side groups on the polyester block through redox-cleavable disulfide linkers. The platinated amphiphilic block copolymer prodrug was self-assembled into nanosized micelles in aqueous solution and the micelles showed an average size of 20 nm. In vitro drug release studies demonstrated that the platinum(II) prodrug micelles are relatively stable under physiological conditions but susceptible to reducing conditions which would trigger the release of conjugated platinum drugs. These results, combined with flow cytometry and fluorescence microscopy images as well as cell viability measurements, suggest the potential of FA-PEG-b-P(α-Pt(II)-SS-CL/CL) prodrug micelles serving as a suitable candidate carrier for targeted intracellular delivery and cytosolic release of potent hydrophobic anticancer drugs.

 Please wait while we load your content...
Please wait while we load your content...