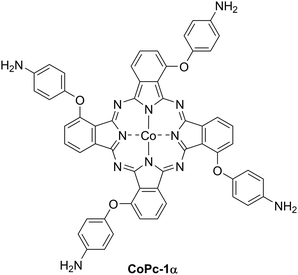
A polymeric α-tetrasubstituted CoII-phthalocyanine catalyst for stable and selective electrochemical carbon dioxide reduction
Kornkamol
Mangmoon
a,
Permsak
Chairat
a,
Yijiao
Jiang
 b,
Poobodin
Mano
c,
Supawadee
Namuangruk
b,
Poobodin
Mano
c,
Supawadee
Namuangruk
 c and
Patchanita
Thamyongkit
c and
Patchanita
Thamyongkit
 *a
*a
aDepartment of Chemistry, Faculty of Science, Chulalongkorn University, Bangkok, 10330, Thailand. E-mail: patchanita.v@chula.ac.th
bSchool of Engineering, Macquarie University, NSW 2109, Sydney, Australia
cNational Nanotechnology Center (NANOTEC), National Science and Technology Development Agency (NSTDA), Pathum Thani 12120, Thailand
First published on 6th January 2026
Abstract
The development of efficient molecular catalysts for electrochemical CO2 reduction (ECO2R) remains a key challenge for scalable carbon utilization. Herein, we report the synthesis and electropolymerization of an α-tetraamino-substituted CoII-phthalocyanine monomer (CoPc-1α) to yield robust polymer films (p(CoPc-1α)) on various conductive substrates. The resulting films were characterized by UV-visible spectrophotometry, Raman and attenuated total reflection-Fourier transform infrared (ATR-FTIR) spectroscopy, scanning electron microscopy (SEM) equipped with energy dispersive X-ray spectroscopy (EDX), variable frequency square-wave voltammetry (VF-SWV), and density functional theory (DFT), confirming successful polymer network formation via phenazine linkages and efficient electron transfer across macrocyclic units. In H-cell electrolysis at −1.15 V vs. NHE, a p(CoPc-1α)-modified carbon paper electrode exhibited high selectivity for CO generation with a faradaic efficiency (FECO) of 97%, along with a current density of 3.8 mA cm−2 and a turnover number (TONCO) and a turnover frequency (TOFCO) of 6.0 × 104 and 0.37 s−1, respectively, over 45 h. At an applied potential of −1.54 vs. NHE in a flow-cell system, the p(CoPc-1α) film on a microporous layer of a carbon fiber paper exhibited remarkable catalytic performance, achieving an average current density of 151 mA cm−2 with 98% FECO for 42 h, corresponding to a TONCO of 1.9 × 106 and a TOFCO of 12.6 s−1. This study demonstrates that p(CoPc-1α) offered a balanced profile of high selectivity, long-term stability and practical current output, establishing it as a promising material for scalable CO2-to-CO conversion.
1. Introduction
Achieving the net-zero greenhouse gas emission goal has become a global agenda driving the development of low-carbon technologies. Among these, carbon dioxide (CO2) conversion offers a sustainable approach to mitigate climate change while producing value-added chemicals and fuels. Electrochemical CO2 reduction (ECO2R) enables conversion of CO2 by using electricity from renewable energy sources into various chemicals, such as carbon monoxide (CO), methane (CH4), and formic acid (HCOOH), depending on types of electrocatalysts employed.1,2 ECO2R offers several advantages, including mild operating conditions, tunable reaction rates and product selectivity via applied potential, and broad scalability.2–4 To overcome kinetic and thermodynamic barriers associated with the reduction of the inert CO2 molecule, the use of an efficient catalyst is very necessary.5 Metallophthalocyanines (MPcs) have demonstrated high efficiency as electrocatalysts for ECO2R.6 Among them, CoII-phthalocyanine (CoPc) is widely recognized as one of the most active MPc-based catalysts for ECO2R.7–10 Several studies reported the use of monomeric CoPc-modified electrodes, but they suffered from limited stability under operating conditions, often leading to catalyst degradation.11 To address this, binders, such as polystyrene12 and Nafion,13 have been commonly used to immobilize the catalyst onto the electrode substrate and improve film stability. Nonetheless, these binders have been reported to adversely impact catalytic performance by blocking active sites, limiting electron conductivity, causing microstructural destruction and impeding mass transport. Moreover, binder degradation under working conditions could deteriorate film stability.14–16 To address these limitations, the direct polymerization of CoPc monomers on the electrode surfaces has become a promising option. Among various techniques, electropolymerization stands out as a highly effective method for producing reproducible, uniform thin films with tunable thickness, morphology and orientation in a single step.17–19 By this process, polymeric CoPc films can be prepared from suitably functionalized monomers containing electropolymerizable groups, such as pyrrole,20 thiophene21 or aniline.22 The substitution patterns on MPc molecules are known to have a significant influence on their electronic and physical properties. Recently, our group reported polymeric ECO2R catalysts based on β-tetrasubstituted CoPc bearing bithiophenyl,23 amino24 and 4-aminophenoxy25 groups that exhibited outstanding catalytic activity, selectivity and stability. According to previous studies, α-substitution on Pc macrocycles substantially suppressed cofacial π to π association relative to β-substitution, and induced out of plane distortion, thereby reducing aggregation tendency and preserving the exposure of the catalytic sites during the electrolysis.26 Moreover, α-functionalization could induce greater changes in the physical, optical and electronic properties than β-substitution.27–30 Building upon these findings, this work aims to synthesize an α-aminophenoxy-tetrasubstituted CoPc monomer (CoPc-1α, Fig. 1) and prepare its corresponding electropolymer (p(CoPc-1)). Electrochemical properties, charge transfer behavior and electrocatalytic performance of p(CoPc-1α) toward heterogeneous ECO2R in both H-type and flow electrochemical cells were investigated, with a focus on improving catalytic selectivity, productivity and stability.2. Results and discussion
2.1 Monomer synthesis and polymer film preparation
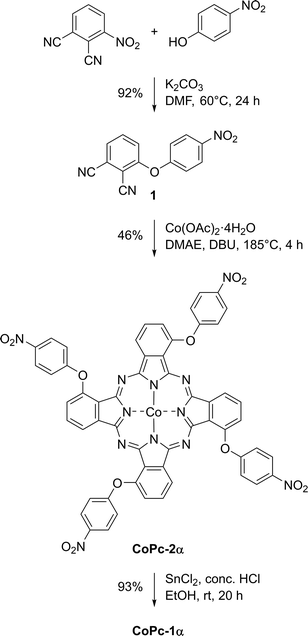
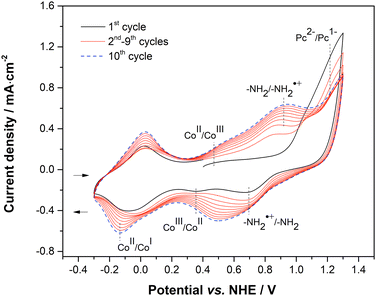
Synthesis of CoPc-1α was carried out via a three-step process following previously published procedures,31–33 starting from a nucleophilic aromatic nitro displacement reaction of 3-nitrophthalonitrile with 4-nitrophenol in dimethylformamide (DMF) at 60 °C for 24 h, leading to 3-(4-nitrophenooxy)phthalonitrile (1) containing a newly formed ether linkage in 92% yield (Scheme 1). Cyclotetramerization of 1 in the presence of Co(OAc)2·4H2O and 1,8-diazabicyclo[5.4.0]undec-7-ene (DBU) in dimethylethanolamine (DMAE) under reflux for 4 h resulted in 1,8,15,22-tetra(4-nitrophenoxy)phthalocyaninatocobalt(II) (CoPc-2α) in 46%. Then, a nitro-reduction of CoPc-2α with a mixture of SnCl2 in concentrated HCl and ethanol at room temperature for 20 h gave CoPc-1α in 93% yield. Formation of CoPc-2α and CoPc-1α was confirmed by their molecular ion peaks at m/z 1119.1291 and 999.2314 in high-resolution matrix-assisted laser desorption/ionization time-of-flight (HR-MALDI-TOF) mass spectra.CoPc-1α was coated on various types of substrates, namely indium tin oxide-coated glass (ITO-glass), gold-coated glass (Au-glass), carbon paper (CPs) and a microporous layer of the commercial carbon fiber paper (MPL-CFP), by means of cyclic voltammetry (CV). Electropolymerization was performed in a 0.1 M tetrabutylammonium hexafluorophosphate (TBAPF6) solution in DMF containing 0.1 mM CoPc-1α in a three electrode one-compartment cell consisting of the ITO-glass, Au-glass, CP or MPL-CFP as a working electrode (WE), an AgCl-coated Ag wire (Ag/AgCl) as a quasi-reference electrode (QRE), and a Pt wire as a counter electrode (CE). A potential range of −0.33 V to 1.27 vs. normal hydrogen electrode (NHE) was applied at a scan rate of 50 mV s−1. Fig. 2 presents the cyclic voltammograms collected during the electropolymerization of CoPc-1α on the CP substrate. In the anodic scans, the oxidation of the primary amine group of CoPc-1α was observed at 0.89 V vs. NHE and gave the corresponding radical cation, initiating the electropolymerization.34,35 The characteristic anodic signals of CoII/CoIII, –NH2/–NH2˙+ and Pc2−/Pc− oxidation were detected at 0.44 V, 0.89 V and 1.19 V vs. NHE, respectively.3 On the reverse scan, the CoII/CoI reduction peak was observed at −0.16 V vs. NHE.36,37 Upon increasing the scanning cycles, the anodic peaks shifted toward more positive potentials and the cathodic ones toward more negative potentials, consistent with the growth of the polymer film on the electrode, which increased the ohmic (iR) drop and amplified the potential shifts.34,38 In addition, the progressive increase in the anodic peak current of the –NH2/–NH2˙+ couple corroborated the continuous polymer deposition on the electrode.36
2.2 Polymer film characterization and electrochemical behavior study
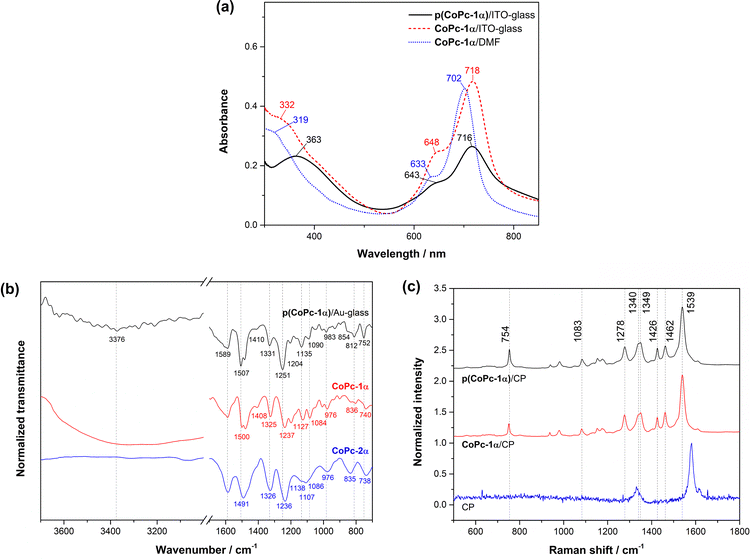
Formation of the resulting polymer films of CoPc-1α (p(CoPc-1α)) was confirmed by UV-visible (UV-vis) spectrophotometry, attenuated total reflection-Fourier transform infrared (ATR-FTIR) spectroscopy, Raman spectroscopy and scanning electron microscopy (SEM) equipped with energy dispersive X-ray spectroscopy (EDX). The UV-vis spectrum of the p(CoPc-1α) film on the ITO-glass (p(CoPc-1α)/ITO-glass) showed a broad B-band at 363 nm, and Q-bands at 643 and 716 nm, corresponding to those observed from a CoPc-1α solution in DMF (CoPc-1α/DMF) and a dropcast CoPc-1α film on the ITO-glass (CoPc-1α/ITO-glass) as shown in Fig. 3a. Peak broadening and slightly red-shifted absorption observed in p(CoPc-1α)/ITO-glass and CoPc-1α/ITO-glass, compared to the absorption of CoPc-1α/DMF, was attributed to H-aggregation among the Pc macrocycles in the polymer film.39–42 As shown in Fig. 3b and Table S1 (SI), the ATR FT-IR spectrum of the p(CoPc-1α) film on the Au-glass (p(CoPc-1α)/Au-glass) was collected and compared with those of CoPc-1α and CoPc-2α. The p(CoPc-1α) film exhibited C–O–C stretching peaks at 854, 1090 and 1251 cm−1,43–47 C–C stretching vibration peaks of pyrrole and isoindole units at 1331 and 1410 cm−1,43,48 and macrocyclic C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C stretching peaks at 1507 and 1589 cm−1.43 These signals were consistent with those observed in CoPc-1α and CoPc-2α, and with those of their β-substituted derivatives (CoPc-1β), described in our previous study.25 Attenuation of the N–H stretching signal in the range of 3000–3600 cm−1,49,50 together with appearance of a peak of phenazine (PNZ) ring vibration with the C–H out-of-plane at 752 cm−1 and a skeletal deformation peak at 812 cm−1 indicate the possible formation of PNZ bridges within the p(CoPc-1α) network.51 The Raman spectrum of p(CoPc-1α)/CP exhibits a similar peak pattern to that of CoPc-1α/CP with slightly broader features (Fig. 3c and Table S2, SI). The signals at 754 and 1340 cm−1 were assigned to Co–N stretching coupled with pyrrole ring expanding,52 and C–H in-plane bending and isoindole vibration,52 respectively. The peaks at 1083 and 1278 cm−1 were also attributed to C–H in-plane bending.53 The vibration peak at 1349 cm−1 was a characteristic feature of the Pc ring and corresponded to the Cβ–Cβ, Cα–Cβ–Cβ, Cγ–Cδ and Cβ–Cγ stretching of the pyrrole group,54 with an additional Cβ–Cβ stretching signal found at 1462 cm−1.55 A peak at 1426 cm−1 was assigned to C–Nm–C stretching (where Nm is an N atom coordinated with a Co center) accompanied by pyrrole expanding,55 while the one at 1539 cm−1 corresponded to C–Nm–C stretching together with pyrrole expanding coupled with C–H in-plane bending.52 Unlike the Raman profiles of the electropolymer of CoPc-1β (p(CoPc-1β)) reported in our previous work,25 a distinct feature of the PNZ linkage could not be clearly observed by this technique. SEM-EDX showed uniform Co distribution across the substrate surface with minor local agglomerates, suggesting that p(CoPc-1α) was deposited over the entire CP substrate (Fig. S8, SI).
C stretching peaks at 1507 and 1589 cm−1.43 These signals were consistent with those observed in CoPc-1α and CoPc-2α, and with those of their β-substituted derivatives (CoPc-1β), described in our previous study.25 Attenuation of the N–H stretching signal in the range of 3000–3600 cm−1,49,50 together with appearance of a peak of phenazine (PNZ) ring vibration with the C–H out-of-plane at 752 cm−1 and a skeletal deformation peak at 812 cm−1 indicate the possible formation of PNZ bridges within the p(CoPc-1α) network.51 The Raman spectrum of p(CoPc-1α)/CP exhibits a similar peak pattern to that of CoPc-1α/CP with slightly broader features (Fig. 3c and Table S2, SI). The signals at 754 and 1340 cm−1 were assigned to Co–N stretching coupled with pyrrole ring expanding,52 and C–H in-plane bending and isoindole vibration,52 respectively. The peaks at 1083 and 1278 cm−1 were also attributed to C–H in-plane bending.53 The vibration peak at 1349 cm−1 was a characteristic feature of the Pc ring and corresponded to the Cβ–Cβ, Cα–Cβ–Cβ, Cγ–Cδ and Cβ–Cγ stretching of the pyrrole group,54 with an additional Cβ–Cβ stretching signal found at 1462 cm−1.55 A peak at 1426 cm−1 was assigned to C–Nm–C stretching (where Nm is an N atom coordinated with a Co center) accompanied by pyrrole expanding,55 while the one at 1539 cm−1 corresponded to C–Nm–C stretching together with pyrrole expanding coupled with C–H in-plane bending.52 Unlike the Raman profiles of the electropolymer of CoPc-1β (p(CoPc-1β)) reported in our previous work,25 a distinct feature of the PNZ linkage could not be clearly observed by this technique. SEM-EDX showed uniform Co distribution across the substrate surface with minor local agglomerates, suggesting that p(CoPc-1α) was deposited over the entire CP substrate (Fig. S8, SI).
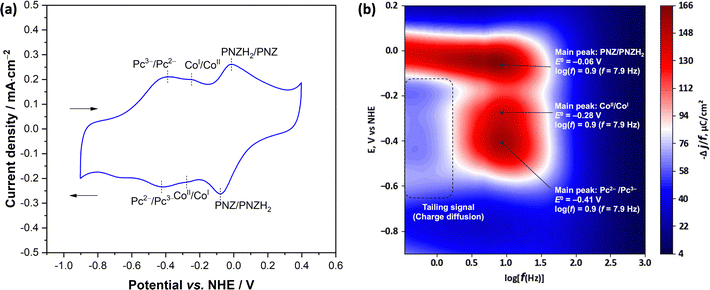
Furthermore, the electrochemical behavior of the p(CoPc-1α) film was characterized by CV in a one-compartment three-electrode electrochemical cell consisting of p(CoPc-1α)/CP as the WE, Ag/AgCl (saturated KCl) as the reference electrode (RE) and a Pt wire as the CE. 0.1 M KOH solution in deionized water was used as the electrolyte to clearly observe the characteristic redox responses of electrochemically active components in the polymer film, while suppressing interference from oxygen reduction and water splitting reactions.56,57 The potential was scanned between −0.90 V and 0.40 V vs. NHE at a scan rate of 100 mV s−1 as shown in Fig. 4a. During the cathodic sweep, the resulting cyclic voltammogram of the p(CoPc-1α)/CP exhibited reduction peaks corresponding to the CoII/CoI and Pc2−/Pc3− couples at −0.27 V and −0.44 V vs. NHE, respectively. These peaks were correlated with those observed in its monomer solution (Fig. S9, SI), thereby indicating the presence of electrochemically active centers for the ECO2R in the p(CoPc-1α) film. Furthermore, a prominent reduction peak assigned to the two-electron reduction of the PNZ linkage was observed at −0.08 V vs. NHE. These three reduction peaks were consistent with those described for the β-substituted analog in our previous report.25 To investigate electron transfer behavior of the above-mentioned electrochemically active components in the p(CoPc-1α) film, variable frequency square-wave voltammetry (VF-SWV) was subsequently conducted in the same electrochemical setup, following the previous works.25,56,57 The charge transfer kinetics of the p(CoPc-1α)/CP was investigated by recording 76 SWV curves within a potential window between −1.00 V and 0.20 V vs. NHE across frequencies (f) from 0.33 Hz to 1250 Hz, corresponding to log(f) values between −0.5 and 3.0. As shown in Fig. 4b, the p(CoPc-1α) film exhibited the main responses corresponding to the CoII/CoI and Pc2−/Pc3− reductions at the potentials of −0.28, and −0.41 V vs. NHE, respectively. In addition, the highest intensity response of the PNZ linkage appeared at a potential of −0.06 V vs. NHE, consistent with the PNZ/PNZH2 reduction peak observed in Fig. 4a. In terms of the frequency response that directly depends on the electron transfer rate in the polymer film, p(CoPc-1α)/CP displayed a wide range of frequency responses between the log(f) of 2.2 and −0.40 by exhibiting the major responses of the Co metal center, Pc ring and PNZ linkage at the log(f) of 0.90 (f = 7.9 Hz) with the tail-like response diffused into the lower log(f) of −0.4 (f = 0.4 Hz). These features were originated from charge penetration from the first layer to the bulk catalyst layers as proposed for the β-substituted analog in our previous works. These observations confirmed the formation of a PNZ-conjugated CoPc network from the oxidative electropolymerization of CoPc-1α and indicated that the intermacrocyclic electron transfer was facilitated by the newly formed PNZ bridge in the p(CoPc-1α) film.
2.3 Electrocatalytic performance
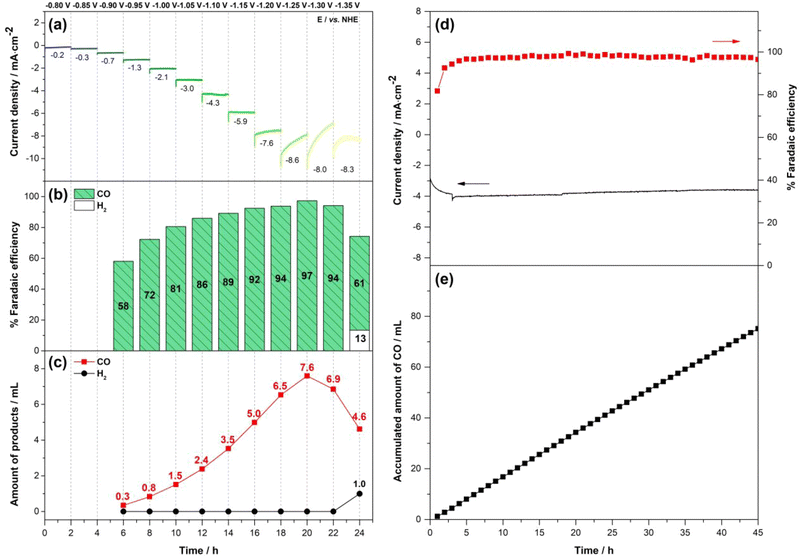
After that, controlled potential electrolysis (CPE) was performed at various potentials, starting from −0.80 V to −1.35 V vs. NHE with 0.05 V increments, for 2 h at each potential. The formation of headspace gas products was analyzed via gas chromatography (GC), while the dissolved species were investigated via nuclear magnetic resonance (NMR). Fig. 6a–c show that no significant product formation was detected until the potential of −0.90 V vs. NHE, consistent with Ered,onset observed in the above-mentioned CV experiment. Upon increasing the applied potential from −0.90 V to −1.20 V vs. NHE, CO was detected as a sole product with Faradaic efficiency of the CO formation (FECO) increasing from 58% to 94% and the average current density increasing from 1.3 mA cm−2 to 7.6 mA cm−2. At −1.25 V to −1.30 V vs. NHE, CO remained the only detectable product, but the current density was unstable despite comparable FECO, likely due to the partial detachment of the p(CoPc-1α) film from the substrate. This effect was more pronounced at −1.35 V vs. NHE, where the FECO dropped to 61% and hydrogen (H2) was generated at 13% FE. According to GC analysis, the CO production reached up to 7.6 mL in the 2-h interval at an applied potential of −1.25 V vs. NHE and then drastically inclined when the reduction potential was increased further. Based on these results, the reduction potentials of −1.15 V and −1.20 vs. NHE were selected for further stability studies, which will be described later.
The stability of p(CoPc-1α) was evaluated at the selected potentials of −1.15 V and −1.20 V vs. NHE under the ECO2R conditions used for the CPE. The results showed that at −1.20 V vs. NHE, p(CoPc-1α)/CP gave CO as the sole product at the FECO above 90% in the first 20 h with the gradually decreasing current density (Fig. S11, SI). After that, the FECO dropped to 63% after the 40th hour and then to 56 at the 45th hour, when the experiment was terminated. The average current density and the accumulated CO amount were 4.4 mA cm−2 and 79.5 mL, respectively. At −1.15 V vs. NHE, the current density and the FECO were quite stable at approximately 3.8 mA cm−2 and 97%, respectively, for at least 45 h with a comparable accumulated CO amount, i.e. 75.1 mL, to that obtained from the ECO2R at −1.20 V vs. NHE (Fig. 6d–e). According to inductively coupled plasma-optical emission spectroscopy (ICP-OES), total catalyst loading of the p(CoPc-1α) film on the CP was determined to be 52 nmol cm−2 (Table S3, SI). Therefore, for the 45-h ECO2R at −1.15 V vs. NHE, the turnover number (TONCO) and a turnover frequency of the CO formation (TOFCO) were calculated to be 6.0 × 104 and 0.37 s−1, respectively
Interestingly, although CoPc-1α and CoPc-1β shared structural similarities, p(CoPc-1α) exhibited a higher FECO (97% vs. 94%) at slightly less negative potential than p(CoPc-1β) (−1.15 V vs. −1.19 V, vs. NHE). However, p(CoPc-1β) clearly outperformed p(CoPc-1α) in terms of the current density (5.9 vs. 3.8 mA cm−2) and long-term operational stability (120 h vs. 45 h) (Table S4, SI). The lower TOFCO observed for p(CoPc-1α) (0.37 vs. 1.1 s−1) was likely attributed to higher catalyst loading on the CP (52 vs. 25 nmol cm−2), as a result of greater solubility of CoPc-1α, compared to that of CoPc-1β. This higher loading led to a thicker catalyst film, which might hinder CO2 diffusion to the active sites close to the electrode surface where electron transfer was most efficient. The weaker frequency-dependent CoII/CoI feature in the above-mentioned VF-SWV response of the p(CoPc-1α)/CP, compared with that of the previously studied p(CoPc-1β)/CP,25 suggested the slower charge transfer kinetics at the Co center in p(CoPc-1α). This observation further supported that the effective turnover was limited by interfacial and interlayer charge transport, and mass transport within the thicker p(CoPc-1α) film. Such charge transfer kinetics could also be governed by the local interfacial ion environment and electric double layer structure, which modulated interfacial charge transfer and consequently electrocatalytic rates.58 Furthermore, electrochemical impedance spectroscopy (EIS) of the p(CoPc-1α) films prepared by CV with cycle numbers of 10, 20, and 30 showed a clear increase in the Nyquist plot semicircle size with increasing cycle number (Fig. S12, SI). This trend indicated higher charge transfer resistance and greater transport limitation in the thicker films from higher deposition cycles, thereby reducing the ECO2R performance. While mechanistic insight was addressed in a later section, these EIS and VF SWV results suggested that the charge transport across the interface and the mass transport within the catalyst film were key contributors to the lower TOF in the thicker films.59 Moreover, when compared to other catalyst architectures, such as the previously reported bithiophenyl-substituted CoPc polymer23 and CoPc-based covalent organic frameworks (COFs),60,61p(CoPc-1α) exhibited superior product selectivity and operational durability, despite showing lower intrinsic activity, i.e. TOFCO, than the COF-based materials. This study supported that p(CoPc-1α)/CP was the stable and effective WE for the batch ECO2R process. However, due to low solubility of CO2 in aqueous solution and diffusion-limited current in the H-cell, we extended our study to a continuous ECO2R using a gas diffusion electrode (GDE), allowing direct feed of gaseous CO2 to the catalyst surface and enabling rigorous evaluation of activity, selectivity, and durability under practical conditions, as described in the next section.
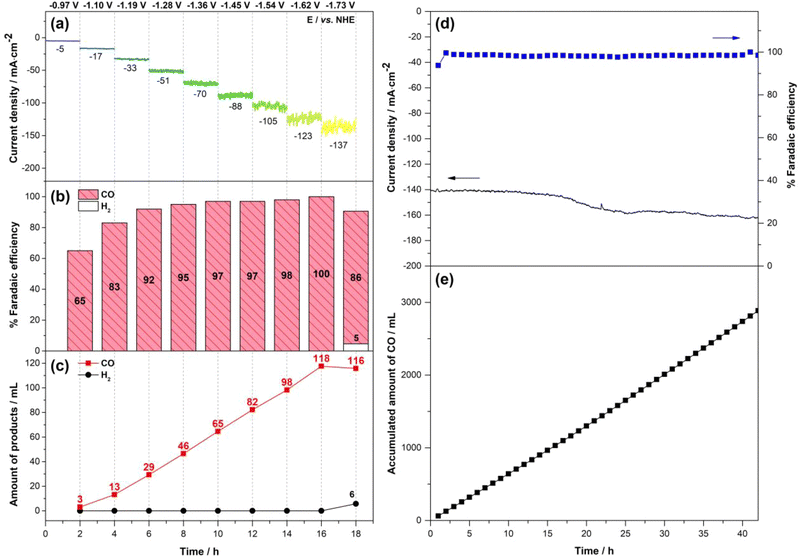
Fig. 7d and e show that by using the p(CoPc-1α)/MPL-CFP as the GDE in the flow cell, the ECO2R at −1.54 V vs. NHE exhibited stable current density approximately 140 mA cm−2 in the first 20 h. After that, the current density slightly increased and became stable again at approximately 160 mA cm−2 by the 25th hour before drastically dropping after the 42th hour due to electrolyte leakage from the cell. Over 42 h, the ECO2R in this flow cell was highly productive with average FECO of 98% and the accumulated CO amount of 2.84 L with the average current density of 151 mA cm−2. Accordingly, TONCO and TOFCO were enhanced to 1.9 × 106 and 13 s−1, respectively, compared with the ECO2R in the H-cell. According to the SEM-EDX analyses, the p(CoPc-1α)/MPL-CFP before and after the electrolysis appeared similar, showing uniform catalyst distribution and the expected existence of carbon, oxygen and cobalt atoms (Fig. S14, SI). The increase in the current density and the subsequent electrolyte leakage arose from hydroxide (OH−) generation during the ECO2R at the cathode, which migrated through the AEM to recombine with protons (H+) produced from water oxidation at the anode consistent with this ion flux, and we observed a decrease in the catholyte volume, while the anolyte volume increased during extended operation. At a high CO production rate, OH− transport became rate-limited, leading to OH− accumulation and subsequent reaction with CO2 at the GDE to form HCO3−/CO32−.62–66 This resulted in an ionic strength increase and thus lowered electrolyte resistance, as observed during the CPE, causing the current density to rise over time. Simultaneously, KHCO3/K2CO3 precipitated within the porous GDE, promoting the leakage and impeding CO2 transport to the catalytic sites.67–72 Consequently, mass-transport limitations became intensified and the FeCO declined.
When compared to the published ECO2R flow system catalyzed by a tetraamino-substituted CoPc electropolymer,24p(CoPc-1α) demonstrated higher FECO (98% vs. 93%) and comparable stability, although with a notably lower TOF (13 vs. 140 s−1). Moreover, p(CoPc-1α) offered significantly higher current density than the CoPc-based COF reported by Han et al.,60 with similar CO selectivity at slightly more negative potential. Overall, p(CoPc-1α) thus presented a well-balanced profile, combining high selectivity, practical current output and decent stability, making it a strong candidate for the scalable electrochemical CO2-to-CO conversion.
3. Theoretical study on charge redistribution and catalytic behavior
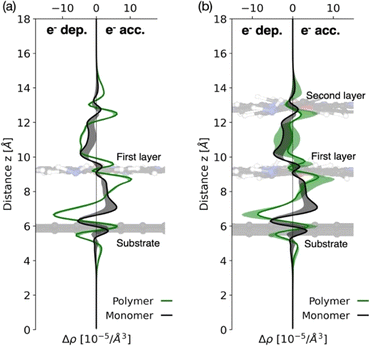
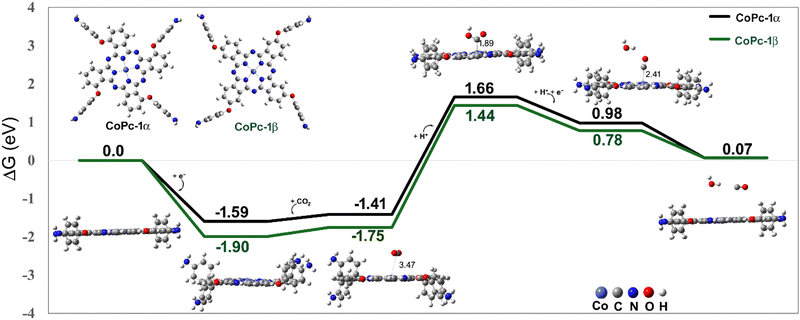
In this section, density functional theory (DFT) calculations were performed to investigate the interfacial and interlayer charge redistribution of CoPc-1α and p(CoPc-1α) on the CFP substrate (Fig. 8). The optimized structures exhibited good lattice matching between the polymer network (22.3 Å × 22.3 Å) and the graphene surface with the most stable configuration found at the hollow site with a 75° orientation, consistent with favorable π–π stacking interaction (Fig. S15–S19, SI). The planar-averaged charge density difference (Δρxy) profiles revealed distinct electronic characteristics between the two systems. In the monomeric one, electron accumulation occurred mainly at the interface between the CoPc macrocycle and graphene, indicating charge transfer from the substrate to the molecules. In contrast, the polymeric network showed pronounced charge accumulation near the two CoPc layers and diminished substrate interaction, suggesting that the PNZ linkages promoted electron delocalization in the polymer layers and enhanced through-plane electronic coupling. This theoretical observation agreed well with the VF-SWV results, demonstrating efficient charge transport through the Co, Pc and PNZ redox centers. Redistribution of the electron density around the Co and PNZ sites accounted for the experimentally observed CoII/CoI and PNZ/PNZH2 reduction peaks at −0.28 V and −0.06 V vs. NHE, respectively, as well as enhanced conductivity of the p(CoPc-1α) film. Furthermore, the results were consistent with those of our previous mechanistic studies presenting the crucial role of the CoI redox center in a key [CoI–Pc2−]− intermediate during the ECO2R.23 Overall, electropolymerization converted the localized interfacial charge of CoPc-1α into a vertically delocalized PNZ-conjugated CoPc network in p(CoPc-1α), thereby facilitating electron transfer while preserving weak substrate binding. This aligned with the high film stability and sustained catalytic current observed in both H-cell and flow-cell experiments.To gain mechanistic insights into the catalytic pathway of the p(CoPc-1α)-catalyzed ECO2R and the effect of the α-functionalization on the Pc macrocycle, computed free-energy (ΔG) profiles for CO2-to-CO conversion catalyzed by CoPc-1α in comparison with our previously reported CoPc-1β were examined as shown in Fig. 9. The reaction started with the CoII/CoI reduction, followed by CO2 adsorption on the CoI center with a small adsorption energy of 0.18 eV, forming a bent *CO2 intermediate stabilized by π-back-donation from metal 3d orbitals. During the CO2 adsorption, the electron transfer upon CO2 activation was not localized only on the Co center. Instead, a substantial fraction of the transferred charge was delocalized over the conjugated π-system of the phthalocyanine macrocycle, indicating that the phthalocyanine macrocyclic ligand functioned as an electronic reservoir that buffers charge during the catalysis (Fig. S20, SI). Subsequent protonation to yield a *COOH intermediate was a rate-determining step, with a slightly lower energy barrier for CoPc-1α (3.07 eV) than for CoPc-1β (3.19 eV), indicating higher catalytic competence of CoPc-1α. Formation of *CO and the following desorption were both downhill in energy, indicating thermodynamically favorable CO release. These theoretical results were consistent with experimental observations showing that p(CoPc-1α) exhibited higher CO selectivity and slightly lower overpotential compared to p(CoPc-1β). The α-functionalization on the Pc macrocycle therefore played a crucial role in strengthening metal–ligand electronic communication, promoting charge transfer and lowering the activation energy for the electrochemical CO2-to-CO conversion. The calculated stacking configuration (Fig. S15–S19, SI) indicated that the distance between the CoPc macrocycles in the first p(CoPc-1α) film layer and the carbon-based substrate was 3.30 Å, while the interplanar distance between adjacent CoPc units within the polymer bulk was 3.77 Å. Both values were slightly larger than those obtained for the previously reported p(CoPc-1β) analogue, i.e. 3.27 and 3.59 Å, respectively.25 These data agreed with the previously reported data showing that α-alkoxy substitution on the Pc macrocycles effectively suppressed the aggregation,73 thereby promoting the accessibility to the catalytically active sites during the ECO2R. The same study further reported that the α-alkoxy groups could induce a Q-band redshift by narrowing the highest occupied molecular orbital (HOMO)–lowest unoccupied molecular orbital (LUMO) gap. In line with this study, the Q-bands of the p(CoPc-1α) film were observed at 643 and 715 nm, redshifted relative to those of the p(CoPc-1β) one (631 and 700 nm).22
4. Conclusions
The desirable α-tetrasubstituted monomer CoPc-1α was successfully synthesized and electropolymerized to form the polymer film of p(CoPc-1α) on the ITO-glass, Au-glass, CP and MPL-CFP substrates. The formation of the p(CoPc-1α) films was confirmed by UV-vis spectrophotometry, Raman and ATR-FTIR spectroscopy, and SEM-EDX analysis. ATR-FTIR spectroscopy showed the characteristic signals corresponding to the newly formed PNZ linkage in the polymer network, while VF-SWV indicated the efficient intermacrocyclic electron transfer facilitated by the PNZ bridge. At a potential of −1.15 V vs. NHE in the H-cell, p(CoPc-1α) demonstrated the optimal catalytic performance by giving the current density, FECO, TONCO and TOFCO of 38 mA cm−2, 97%, 6.0 × 104 and 0.37 s−1, respectively, for at least 45 h. Compared with its β-tetrasubstituted analog CoPc-1β, CoPc-1α exhibited higher solubility in DMF and, according to the ICP-OES, the catalyst loading of p(CoPc-1α) was approximately twice that of p(CoPc-1β) on the CP. As a result, the more hindered CO2 diffusion possibly occurred in p(CoPc-1α) led to lower current density and TOFCO observed in the H-cell, despite achieving higher FECO at the lower reduction potential, compared to the case of p(CoPc-1β). When used in the flow cell, p(CoPc-1α) exhibited high catalytic activity for the CO2-to-CO conversion by giving a current density of 151 mA cm−2 with 97% FECO for 42 h, corresponding to TONCO and TOFCO of 1.9 × 106 and 13 s−1, respectively. The DFT calculations confirmed efficient charge transport through the Co, Pc and PNZ redox centers and revealed that the ECO2R under catalysis of CoPc-1α had a lower energy barrier for the rate determining step than that catalyzed by CoPc-1β, reflecting the beneficial effect of the α-substitution of the Pc macrocycles.5. Experimental
5.1 Materials and methods
All chemicals were purchased from commercial sources and used without further purification, unless noted otherwise. The CPs (AvCarb MGL190), CFPs (avcarb-gds3250) and Nafion membranes (117) were supplied by Fuel Cell Store (USA). The ITO-glass was purchased from Semiconductor wafer, Inc. The Sustainion® X37-50 Grade RT AEM was supplied by dioxide materials, and polytetrafluoroethylene (PTFE) 60 wt% dispersion in water and the Pt wire with a purity of 99.99% were obtained from Sigma Aldrich. 1H- and 13C-NMR spectra were recorded in CDCl3 using a Bruker Avance 400 operated at 400 MHz and 100 MHz, respectively. Chemical shifts (δ) are reported in parts per million (ppm) relative to residual CHCl3 peaks (δ = 7.26 ppm and 77.16 ppm for 1H- and 13C-NMR spectroscopy, respectively). Mass spectra were obtained by HR-MALDI-TOF mass spectrometry (HR-MALDI-TOF-MS). The absorption spectra were measured in DMF at room temperature using an Agilent Cary 60 UV-vis spectrophotometer, and molar extinction coefficients (ε) were reported in L mol−1 cm−1. The ATR-FTIR spectra were recorded using s a Nicolet iS50-ATR spectrometer (Thermo Fisher Scientific, U. S.). Raman measurements were conducted at room temperature using a Horiba LabRAM HR800 micro-Raman spectrometer with a 633 nm line of a He–Ne laser. All Raman spectra were calibrated with a signal of a silicon wafer in the Raman spectrum, which typically shows a peak at 520.7 nm. The SEM-EDX images were obtained using a JEOL JSM-IT100. The amount of cobalt on polymeric catalysts was determined through the ICP-OES analysis using a Thermo iCAP 6500 Duo (Thermospectrometer Scientific). The electrochemical experiments were performed using a MetrohmAutolab/PGSTAT101 potentiostat with NOVA software and an AMETEK, Inc, VersaSTAT3 potentiostat with VersaStudio software. The flow rate of 99.995% pure CO2 gas was controlled using a BRONKHORST select mass flow controller (EL-FLOW Select series). The reduction products in the gas phase were determined using an Agilent 8890 GC system equipped with a thermal conductivity detector (TCD) and two parallel columns (CP-Molsieve 5 Å and CP-PoraBOND Q). These columns separated and quantified the gas mixture, including H2, O2, N2, CO, and CO2, in a single run. Mixtures of each standard gas with N2 were used for calibration. Ultra-high purity helium (99.999%) was used as a carrier gas for CO and H2 analysis, respectively.5.2 Synthesis of 3-(4-nitrophenooxy)phthalonitrile (1)
Following a reported procedure31 with modification of the types of staring materials and purification conditions, a mixture of 3-nitrophthalonitrile (514 mg, 2.97 mmol), 4-nitrophenol (426 mg, 3.06 mmol), potassium carbonate (399 mg, 2.89 mmol) and dimethylformamide (DMF) (7 mL) was heated at 60 °C for 24 h under a N2 atmosphere. After cooling to room temperature, the reaction mixture was poured into cold water (50 mL). The resulting precipitate was collected by suction filtration, then washed with water and dried in a vacuum oven at 60 °C for 24 h, affording 3-(4-nitrophenoxy)phthalonitrile (1) as a white solid (723 mg, 92%). m.p. 177–178 °C, 1H NMR (CDCl3) (δ): 8.36–8.31 (m, 2H), 7.77–7.69 (m, 1H), 7.64 (dd, J = 7.7, 1.0 Hz, 1H), 7.29 (dd, J = 8.7, 1.1 Hz, 1H), 7.23–7.17 (m, 2H); 13C NMR (CDCl3) (δ): 159.6, 158.5, 145.0, 135.1, 129.2, 126.6, 123.0, 119.6, 118.0, 112.1, 108.3. Anal. calcd for C14H7N3O3: C, 63.40; H, 2.66; N, 15.84. Found: C, 62.32; H, 2.44; N, 15.08.5.3 Synthesis of 1,8,15,22-tetra(4-nitrophenoxy)phthalocyaninatocobalt(II) (CoPc-2α)
Following a reported procedure32 with modification of types of staring materials and the purification process, a solution of 3-(4-nitrophenoxy)phthalonitrile (1, 239 mg, 0.225 mmol) and Co(OAc)2·4H2O (131 mg, 0.53 mmol) in DMAE (5.0 mL) in the presence of DBU (0.5 mL) was refluxed under a N2 atmosphere for 4 h. After cooling to room temperature, the precipitate was collected by centrifugation at 4500 rmp for 5 min and washed by sonication–centrifugation in water (40 mL) and ethanol (40 mL) twice. The resulting precipitate was dried in a vacuum oven at 60 °C for 24 h to afford CoPc-2α as a dark green solid (115 mg, 46%). UV-vis: λabs (ε) 610 (9.3 × 104), 672 (8.8 × 104) nm; HR-MALDI-TOF m/z (%): found, 1119.1291 [M+]; calcd, 1119.1284 (M+, M = C56H28CoN12O12).5.4 Synthesis of 1,8,15,22-tetra(4-aminophenoxy)phthalocyaninatocobalt(II) (CoPc-1α)
Following a reported procedure33 with modification of types of staring materials and purification conditions, a suspension of CoPc-2α (101 mg, 0.091 mmol) in ethanol (16.0 mL) was slowly treated with a mixture of SnCl2 (5.0 g, 0.03 mmol) in concentrated HCl (7.0 mL) at room temperature under a N2 atmosphere. After 20 h, the reaction mixture was filtered by a filter paper and the resulting dark blue crude was dissolved in a 1.75 M acetic acid aqueous solution (150 mL). Then, a 0.008 M ammonium hydroxide aqueous solution (40 mL) was added. The resulting dark green precipitate was collected by centrifugation at 4500 rmp for 5 min and washed by sonication–centrifugation in water (40 mL) and ethanol (40 mL) twice. After drying in a vacuum oven at 60 °C for 24 h, CoPc-1 was obtained as a dark green solid (84 mg, 93%). UV-vis: λabs (ε) 633 (2.2 × 104), 702 (6.4 × 104) nm; HR-MALDI-TOF m/z (%): found, 999.2314 [M+]; calcd, 999.2314 (M+, M = C56H36CoN12O4).5.5 Electropolymerization of CoPc-1α
The ITO-glass substrate was cut into 0.8 cm × 4.0 cm pieces and cleaned by consecutive sonication in toluene, acetone, isopropanol and deionized water. The Au-glass substrate was cut into 1.0 cm × 1.0 cm pieces and cleaned by sonication in acetone. The CP was cut into 1.0 cm × 2.0 cm pieces and cleaned by sonication in acetone. The MPL-CFP substrate was prepared according to a previously reported procedure.24 After the cleaning process, all substrates were dried in an oven at 100 °C for 15 min. The electrochemical polymerization of CoPc-1α was performed in a one-compartment three-electrode system by means of CV. The electrochemical cell consisted of the ITO-glass, Au-glass, CP or MPL-CFP substrates as the WE, the Pt wire as the CE and the Ag/AgCl as QRE. The Ag/AgCl QRE was calibrated using a ferrocene/ferrocenium (Fc/Fc+) redox couple in a 0.1 M TBAPF6 solution in DMF as an external standard. A standard potential of 0.72 V vs. NHE in DMF for the Fc/Fc+ couple was used to convert the potentials recorded against the Ag/AgCl QRE to the NHE scale.74 The electropolymerization of CoPc-1α was conducted in a 1.0 mM CoPc-1α solution in DMF containing 0.1 M TBAPF6 under a N2 atmosphere. The potential was swept in a range from −0.33 V to 1.27 V vs. NHE at a scan rate of 50 mV s−1 for 80, 50, 10 and 40 cycles on the ITO-glass, the Au-glass, the CP and the MPL-CFP substrates, respectively. For the catalytic performance studies, the deposited coverage area on the CP and the MPL-CFP substrates was controlled at 1.0 cm × 1.0 cm. After the electropolymerization of CoPc-1α, the resulting polymer films were washed with DMF to remove the excess monomer and electrolyte salt, and then dried in an oven at 60 °C for 24 h.5.6 Determination of the Co content in p(CoPc-1α) on substrates
The Co content in the polymer films was determined by ICP-OES following a previously reported procedure.75 The polymer samples were digested using a 9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 volume ratio of 65% aqueous HNO3
1 volume ratio of 65% aqueous HNO3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 30% v/v H2O2 in a 45-mL Teflon-lined autoclave and heated at 185 °C for 5 h. Co standard solutions for the ICP were prepared at suitable concentrations using Milli-Q water and filtered through 0.22 µm nylon membrane filters before measurement.
30% v/v H2O2 in a 45-mL Teflon-lined autoclave and heated at 185 °C for 5 h. Co standard solutions for the ICP were prepared at suitable concentrations using Milli-Q water and filtered through 0.22 µm nylon membrane filters before measurement.
5.7 Study on electrocatalytic performance of CoPc-1α for the ECO2R
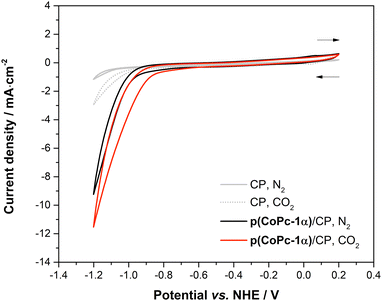
The electrocatalytic activity of CoPc-1α was studied using the CV and CPE in both H-cell and flow cell electrochemical setups. The H-cell consisted of a 50-mL two-compartment three-electrode system with the cathodic and anodic chambers separated from each other by an activated proton exchange membrane (Nafion 117).23 The cathodic chamber contained p(CoPc-1α)/CP as the WE and commercial Ag/AgCl (saturated KCl) as the RE, while the anodic chamber contained a Pt wire as the CE. The CV experiments were conducted in N2- and CO2-saturated 0.5 M KHCO3 electrolyte solution in Milli-Q water (45 mL). For the potential optimization experiment, the CPE was performed at the potential ranging between −0.80 V and −1.35 V vs. NHE with 0.05 V increments for 2 h at each potential. CO2 was continuously purged through a catholyte solution at a flow rate of 5 mL min−1. For a long-term stability study, a constant potential of −1.15 V or −1.20 V vs. NHE was applied, and the solutions in both chambers were stirred at 250 rpm during the entire experiment. In the H-cell experiments, the potentials were converted to the NHE scale using the following equation.74ENHE = EAg/AgCl![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) (saturated (saturated![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) KCl) + 0.197 KCl) + 0.197 |
The flow cell consisted of three customized PTFE plates stacked and tightened together with the silicone rubber sheets, stainless steel bolts and nuts, followed by configuration as described in the previous work.24 In the cathodic chamber, the p(CoPc-1α)/MPL-CFP connected to a titanium (Ti) plate current collector and the commercial Ag/AgCl (saturated KCl) were used as the WE and RE, respectively. The Pt wire connected to another Ti plate current collector served as the CE and put in the anodic chamber. Both chambers were separated by the Sustainion® X37-50 Grade RT membrane. The 1.0 M KHCO3 electrolyte solution in Milli-Q water was circulated between each chamber and its external 250-mL electrolyte reservoir at a flow rate of 15 mL min−1 using a peristaltic pump. CO2 was continuously supplied from a backside of the GDE through a serpentine flow field with a flow rate of 15 mL min−1. For the potential optimization experiment, the potentials ranging from −0.97 V to −1.73 V vs. NHE were applied with 0.05 V increments for 2 h at each potential. For the stability test, the constant potential of −1.54 V vs. NHE was applied with 80% iR compensation and converted to the NHE scale using the following equation.74
ENHE = EAg/AgCl![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) (saturated (saturated![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) KCl) + 0.197 − (0.8) × iRu KCl) + 0.197 − (0.8) × iRu |
To conduct online gas product analysis, headspace gas samples (200 μL) were collected from the cathodic chamber of the H-cell or from the cathodic reservoir of the flow cell, and real-time analyzed by the GC every 20 min throughout the experiment.
5.8 VF-SWV study
The electrochemical behavior of p(CoPc-1α) was first examined in the one-compartment three-electrode electrochemical setup consisting of p(CoPc-1α)/CP, Ag/AgCl (saturated KCl) and a Pt wire as the WE, RE and CE, respectively. The Ar-saturated 0.1 M KOH solution in Milli-Q water (20 mL) was used as the electrolyte. The cyclic voltammogram of the p(CoPc-1α) film was recorded by sweeping the potential between −0.90 V and 0.40 V vs. NHE at a scan rate of 100 mV s−1. The VF-SWV experiment of p(CoPc-1α)/CP was carried out using a previously reported procedure56,57,76 in the same one-compartment three-electrode electrochemical setup. The potential between −0.90 V and 0.40 V vs. NHE was applied using a perturbation amplitude and staircase steps of 25 mV and 10 mV. The perturbation frequency (f) was varied from 1250 Hz to 0.33 Hz, corresponding to pulse times from 0.4 µs to 1.5 s. A second-order Savitzky−Golay digital filter of seven points was applied to smoothen the signal in the low-frequency region.5.9 Electrochemical impedance spectroscopy (EIS)
Following the previously reported procedure.77 electrochemical impedance spectroscopy (EIS) was performed in the 0.5 M KHCO3 aqueous electrolyte solution in the one-compartment electrochemical cell containing p(CoPc-1α)/CP, Ag/AgCl (sat. KCl), and a Pt wire as the WE, RE and CE, respectively. After purging of CO2 into the electrolyte solution for 15 min, the EIS spectra were recorded at −0.95 V vs. NHE with the frequency ranging from 0.1 Hz to 100 000 Hz and an AC voltage of 5 mV on Versastat3.5.10 Computational details
The theoretical investigations were conducted within the DFT using GPAW software.78 The projector augmented wave (PAW) method79 was used to represent core electrons, while the Perdew–Burke–Ernzerhof (PBE) functional80 was selected for the exchange–correlation functional. To consider the dispersion correction, the Grimme's DFT-D381 method was applied. The real space grid was set to (192, 192, 96) throughout the calculations, while energy cutoff for wavefunctions was set to 450 eV. Periodic boundary condition was imposed to reproduce the 2D-graphene substrate where the z axis of the simulation box was set to 18 Å throughout the calculations. K-point sampling was done only at the Γ point. Dipole correction was considered in the z-direction to treat asymmetric stacking along the z-direction. To ensure the localization of the 3d orbital as well as the magnetic moment of the Co 5atom, the Hubbard U correction was imposed with U = 6.0 eV to the 3d orbital, following the previous study.82,83In this study, the adsorption energy Eads was described by the equation.
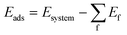 | (1) |
 | (2) |
To construct the ΔG profiles of the CO2-to-CO conversion on CoPc-1α, its molecular model was employed. All quantum chemical calculations were carried out using the Gaussian 16 software package. Geometry optimizations were performed with the B3LYP exchange–correlation functional and the 6-31G(d,p) basis set. No symmetry constraints were imposed on the structures. Subsequent frequency analyses were carried out at the 6-311++G(d,p) level to obtain thermochemical corrections and ΔG. The ΔG of the proton–electron pair (H+ + e−) was referenced to one-half of the total energy of an isolated H2 molecule following the computational hydrogen electrode (CHE) approach proposed by Noskov.84
Author contributions
Kornkamol Mangmoon: writing – original draft, data curation, investigation, and visualization. Permsak Chairat: writing – original draft, data curation, investigation, and methodology. Yijiao Jiangb: validation. Poobodin Mano and Supawadee Namuangruk: writing – data curation and investigation. Patchanita Thamyongkit: writing – review and editing, resources, supervision, validation, and project administration.Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article have been included as part of the supplementary information (SI). Supplementary information: the characterization data of the target monomers and polymer films, an H-cell configuration, the electrochemical and catalytic performance data of the H and the flow cells, the theoretical calculation data, the ICP-OES data, and the comparison table of the catalytic performance. See DOI: https://doi.org/10.1039/d5cp04205d.Acknowledgements
This research has received funding support from the NSRF via the Program Management Unit for Human Resources & Institutional Development, Research and Innovation [grant number B13F680070], the 90th Anniversary of Chulalongkorn University Fund, and Overseas Research Experience Scholarship for Graduate Student from Graduate School of Chulalongkorn University, and Thailand Science Research and Innovation Fund Chulalongkorn University (IND_FF_68_341_6200_012).References
- L. J. R. Nunes, Environments, 2023, 10, 66 Search PubMed.
- K. Wiranarongkorn, K. Eamsiri, Y.-S. Chen and A. Arpornwichanop, J. CO2 Util., 2023, 71, 102477 CrossRef CAS.
- A. Kumar, L. M. Aeshala and T. Palai, J. Appl. Electrochem., 2023, 53, 1295–1319 CrossRef CAS.
- Q. Chen, P. Tsiakaras and P. Shen, Catalysts, 2022, 12, 1348 Search PubMed.
- J. Shen, M. J. Kolb, A. J. Göttle and M. T. M. Koper, J. Phys. Chem. C, 2016, 120, 15714–15721 Search PubMed.
- A. Kumar, G. Zhang, W. Liu and X. Sun, J. Electroanal. Chem., 2022, 922, 116799 CrossRef CAS.
- Z. Zhang, J. Xiao, X.-J. Chen, S. Yu, L. Yu, R. Si, Y. Wang, S. Wang, X. Meng, Y. Wang, Z.-Q. Tian and D. Deng, Angew. Chem., Int. Ed., 2018, 57, 16339–16342 CrossRef CAS PubMed.
- S. Yu, D. Menga, A. Moranka, S. Muy, M. Busch, V. Tileli and Y. Shao-Horn, J. Am. Chem. Soc., 2025, 147, 34183–34198 Search PubMed.
- L. Yao, J. Ding, X. Cai, L. Liu, N. Singh, C. C. L. McCrory and B. Liu, ACS Nano, 2024, 18, 21623–21632 CrossRef CAS PubMed.
- Q. Feng, Y. Sun, X. Gu and Z. Dong, Electrolysis, 2022, 13, 675–690 CAS.
- Y.-H. Tse, P. Janda, H. Lam, J. Zhang, W. J. Pietro and A. B. P. Lever, J. Porphyrins phthalocyanines, 1997, 01, 3–16 CrossRef CAS.
- S. Meshitsuka, M. Ichikawa and K. Tamaru, J. Chem. Soc., Chem. Commun., 1974, 158–159, 10.1039/C39740000158.
- X. Zhang, Z. Wu, X. Zhang, L. Li, Y. Li, H. Xu, X. Li, X. Yu, Z. Zhang, Y. Liang and H. Wang, Nat. Commun., 2017, 8, 14675 CrossRef PubMed.
- H. Yang, M. Driess and P. W. Menezes, Adv. Energy Mater., 2021, 11, 2102074 CrossRef CAS.
- M. Asad, A. Shah, F. J. Iftikhar, R. Nimal, J. Nisar and M. A. Zia, Sustainable Chem., 2022, 3, 286–299 CrossRef CAS.
- G. Barati Darband, M. Aliofkhazraei and A. S. Rouhaghdam, J. Colloid Interface Sci., 2019, 547, 407–420 Search PubMed.
- M. Josowicz and J. Janata, in Applications of Electroactive Polymers, ed. B. Scrosati, Springer Netherlands, Dordrecht, 1993, pp. 310–343 DOI:10.1007/978-94-011-1568-1_10.
- A. Pailleret and F. Bedioui, in N4-Macrocyclic Metal Complexes, ed. J. H. Zagal, F. Bedioui and J.-P. Dodelet, Springer New York, New York, NY, 2006, pp. 363–438 DOI:10.1007/978-0-387-28430-9_8.
- L. Liu, in Solution-Processed Organic Light-emitting Devices, ed. G. Xie, Woodhead Publishing, 2024, pp. 477–494 DOI:10.1016/B978-0-323-95146-3.00004-5.
- H. A. Abd El-Rahman and H. H. Rehan, J. Appl. Electrochem., 1993, 23, 827–834 Search PubMed.
- T. Shimidzu, React. Polym., Ion Exch., Sorbents, 1987, 6, 221–227 CrossRef CAS.
- M. Ando, Y. Watanabe, T. Iyoda, K. Honda and T. Shimidzu, Thin Solid Films, 1989, 179, 225–231 Search PubMed.
- J. Luangchaiyaporn, D. Wielend, D. Solonenko, H. Seelajaroen, J. Gasiorowski, M. Monecke, G. Salvan, D. R. T. Zahn, N. S. Sariciftci and P. Thamyongkit, Electrochim. Acta, 2021, 367, 137506 CrossRef CAS.
- J. Luangchaiyaporn, P. Chairat, R. Sodkhomkhum, N. S. Sariciftci and P. Thamyongkit, ChemCatChem, 2024, 16, e202400281 CrossRef CAS.
- P. Chairat, J. Luangchaiyaporn, A. S. Kochubei, A. N. Marianov, P. Mano, S. Namuangruk, Y. Jiang and P. Thamyongkit, ChemSusChem, 2025, 18, e202501266 CrossRef CAS PubMed.
- A. W. Snow, in The Porphyrin Handbook, ed. K. M. Kadish, K. M. Smith and R. Guilard, Academic Press, Amsterdam, 2003, pp. 129–176 DOI:10.1016/B978-0-08-092391-8.50009-1.
- R. R. Cranston and B. H. Lessard, RSC Adv., 2021, 11, 21716–21737 Search PubMed.
- C. G. Claessens, U. Hahn and T. Torres, Chem. Rec., 2008, 8, 75–97 CrossRef CAS PubMed.
- N. Kobayashi, H. Ogata, N. Nonaka and E. A. Luk’yanets, Chem. – Eur. J., 2003, 9, 5123–5134 Search PubMed.
- T. Ikeuchi, J. Mack, T. Nyokong, N. Kobayashi and M. Kimura, Langmuir, 2016, 32, 11980–11985 Search PubMed.
- B. Amir, H. Zhou, F. Liu and H. Aurangzeb, J. Polym. Sci., Part A: Polym. Chem., 2010, 48, 5916–5920 Search PubMed.
- N. Hamdi, R. Medyouni, H. Bilel, L. Mansour and A. Romerosa, Molecules, 2017, 22, 605 Search PubMed.
- D. Wöhrle and V. I. Bregadze, J. Porphyrins phthalocyanines, 2005, 9, 268–274 Search PubMed.
- Y. Wei, G. W. Jang, C. C. Chan, K. F. Hsueh, R. Hariharan, S. A. Patel and C. K. Whitecar, J. Phys. Chem., 1990, 94, 7716–7721 Search PubMed.
- K. S. Lokesh and A. Adriaens, Dyes Pigm., 2015, 112, 192–200 Search PubMed.
- H. Li and T. F. Guarr, J. Chem. Soc., Chem. Commun., 1989, 832–834, 10.1039/C39890000832.
- N. Manjunatha, M. Imadadulla, K. S. Lokesh and K. R. Venugopala Reddy, Dyes Pigm., 2018, 153, 213–224 Search PubMed.
- J. Roncali, A. Yassar and F. Garnier, J. Chem. Soc., Chem. Commun., 1988, 581–582, 10.1039/C39880000581.
- R. Georgescu, C. Boscornea, I. Calinescu and R. State, Rev. Chim., 2015, 66, 1554–1557 Search PubMed.
- K. Serbest, İ. Değirmencioğlu, Y. Ünver, M. Er, C. Kantar and K. Sancak, J. Organomet. Chem., 2007, 692, 5646–5654 Search PubMed.
- M. Tian, T. Wada and H. Sasabe, J. Heterocycl. Chem., 1997, 34, 171–176 CrossRef CAS.
- M. Tian, T. Wada and H. Sasabe, Dyes Pigm., 2002, 52, 1–8 Search PubMed.
- A. V. Ziminov, S. M. Ramsh, E. I. Terukov, I. N. Trapeznikova, V. V. Shamanin and T. A. Yurre, Semiconductors, 2006, 40, 1131–1136 Search PubMed.
- O. Tsaryova, A. Semioshkin, D. Wöhrle and V. I. Bregadze, J. Porphyrins Phthalocyanines, 2005, 09, 268–274 CrossRef CAS.
- V. Ahsen, E. Yilmazer, M. Ertas and Ö. Bekâroğlu, J. Chem. Soc., Dalton Trans., 1988, 401–406, 10.1039/DT9880000401.
- O. E. Sielcken, M. M. Van Tilborg, M. F. M. Roks, R. Hendriks, W. Drenth and R. J. M. Nolte, J. Am. Chem. Soc., 1987, 109, 4261–4265 CrossRef CAS.
- M. Koçak, A. Cihan, A. I. Okur, A. Gül and Ö. Bekaroğlu, Dyes Pigm., 2000, 45, 9–14 Search PubMed.
- A. Kumar, S. Samanta, S. Latha, A. K. Debnath, A. Singh, K. P. Muthe and H. C. Barshilia, RSC Adv., 2017, 7, 4135–4143 RSC.
- K. S. Lokesh and A. Adriaens, Dyes Pigm., 2013, 96, 269–277 CrossRef CAS.
- M. Tammer, Colloid Polym. Sci., 2004, 283, 235 CrossRef CAS.
- C. Stammer and A. Taurins, Spectrochim. Acta, 1963, 19, 1625–1654 CrossRef CAS.
- R. D. Rodriguez, C. J. Villagómez, A. Khodadadi, S. Kupfer, A. Averkiev, L. Dedelaite, F. Tang, M. Y. Khaywah, V. Kolchuzhin, A. Ramanavicius, P.-M. Adam, S. Gräfe and E. Sheremet, ACS Photonics, 2021, 8, 2243–2255 CrossRef CAS.
- B. J. Palys, D. M. W. van den Ham, W. Briels and D. Feil, J. Raman Spectrosc., 1995, 26, 63–76 CrossRef CAS.
- C. A. Melendres and V. A. Maroni, J. Raman Spectrosc., 1984, 15, 319–326 CrossRef CAS.
- Z. Liu, X. Zhang, Y. Zhang and J. Jiang, Spectrochim. Acta, Part A, 2007, 67, 1232–1246 CrossRef PubMed.
- A. N. Marianov, A. S. Kochubei, T. Roman, O. J. Conquest, C. Stampfl and Y. Jiang, Anal. Chem., 2021, 93, 10175–10186 CrossRef CAS PubMed.
- A. S. Kochubei, A. N. Marianov, O. J. Conquest, T. Lu, Y. Liu, C. Stampfl and Y. Jiang, J. Mater. Chem. A, 2025, 13, 1874–1886 RSC.
- H. Tao, S. Wang, H. Liu and C. Lian, Angew. Chem., Int. Ed., 2025, 64, e202418447 CrossRef CAS PubMed.
- Y. Niu, H. Tao, J. Li, C. Lian and H. Liu, J. Phys. Chem. C, 2025, 129, 19371–19379 CrossRef CAS.
- B. Han, X. Ding, B. Yu, H. Wu, W. Zhou, W. Liu, C. Wei, B. Chen, D. Qi, H. Wang, K. Wang, Y. Chen, B. Chen and J. Jiang, J. Am. Chem. Soc., 2021, 143, 7104–7113 Search PubMed.
- B. Han, Y. Jin, B. Chen, W. Zhou, B. Yu, C. Wei, H. Wang, K. Wang, Y. Chen, B. Chen and J. Jiang, Angew. Chem., Int. Ed., 2022, 61, e202114244 CrossRef CAS PubMed.
- F. Habibzadeh, P. Mardle, N. Zhao, H. D. Riley, D. A. Salvatore, C. P. Berlinguette, S. Holdcroft and Z. Shi, Electrochem. Energy Rev., 2023, 6, 26 Search PubMed.
- P. Zhu and H. Wang, Nat. Catal., 2021, 4, 943–951 CrossRef CAS.
- S. M. Hosseini, S. S. Madaeni and A. R. Khodabakhshi, Sep. Sci. Technol., 2012, 47, 455–462 Search PubMed.
- K. Solonchenko, A. Kirichenko and K. Kirichenko, Membranes, 2023, 13, 52 CrossRef CAS PubMed.
- H. J. Kwon, B. Kim, G. Lim and J. Han, J. Mater. Chem. A, 2018, 6, 7714–7723 Search PubMed.
- M. E. Leonard, L. E. Clarke, A. Forner-Cuenca, S. M. Brown and F. R. Brushett, ChemSusChem, 2020, 13, 400–411 Search PubMed.
- D. G. Wheeler, B. A. W. Mowbray, A. Reyes, F. Habibzadeh, J. He and C. P. Berlinguette, Energy Environ. Sci., 2020, 13, 5126–5134 Search PubMed.
- S. Haj-Bsoul, J. R. Varcoe and D. R. Dekel, J. Electroanal. Chem., 2022, 908, 116112 Search PubMed.
- S. Willdorf-Cohen, A. Zhegur-Khais, J. Ponce-González, S. Bsoul-Haj, J. R. Varcoe, C. E. Diesendruck and D. R. Dekel, ACS Appl. Energy Mater., 2023, 6, 1085–1092 CrossRef CAS PubMed.
- Y. Wang, J. Qiu, J. Peng, L. Xu, J. Li and M. Zhai, J. Membr. Sci., 2011, 376, 70–77 CrossRef CAS.
- E. Fontananova, D. Messana, R. A. Tufa, I. Nicotera, V. Kosma, E. Curcio, W. van Baak, E. Drioli and G. Di Profio, J. Power Sources, 2017, 340, 282–293 Search PubMed.
- T. Furuyama, K. Satoh, T. Kushiya and N. Kobayashi, J. Am. Chem. Soc., 2014, 136, 765–776 Search PubMed.
- T. J. Smith and K. J. Stevenson, in Handbook of Electrochemistry, ed. C. G. Zoski, Elsevier, Amsterdam, 2007, pp. 73–110 DOI:10.1016/B978-044451958-0.50005-7.
- W. Boonta, W. Sangkhun, C. Suppaso, N. Chantanop, W. Panchan, K. Chainok, P. Thamyongkit, T. Sudyoadsuk, K. Maeda, T. Butburee and J. Unruangsri, ACS Catal., 2023, 13, 12391–12402 CrossRef CAS.
- A. N. Marianov and Y. Jiang, Acc. Mater. Res., 2022, 3, 620–633 Search PubMed.
- S. Zhou, L.-J. Zhang, L. Zhu, C.-H. Tung and L.-Z. Wu, Adv. Mater., 2023, 35, 2300923 CrossRef CAS PubMed.
- J. J. Mortensen, A. H. Larsen, M. Kuisma, A. V. Ivanov, A. Taghizadeh, A. Peterson, A. Haldar, A. O. Dohn, C. Schäfer, E. Ö. Jónsson, E. D. Hermes, F. A. Nilsson, G. Kastlunger, G. Levi, H. Jónsson, H. Häkkinen, J. Fojt, J. Kangsabanik, J. Sødequist, J. Lehtomäki, J. Heske, J. Enkovaara, K. T. Winther, M. Dulak, M. M. Melander, M. Ovesen, M. Louhivuori, M. Walter, M. Gjerding, O. Lopez-Acevedo, P. Erhart, R. Warmbier, R. Würdemann, S. Kaappa, S. Latini, T. M. Boland, T. Bligaard, T. Skovhus, T. Susi, T. Maxson, T. Rossi, X. Chen, Y. L. A. Schmerwitz, J. Schiøtz, T. Olsen, K. W. Jacobsen and K. S. Thygesen, J. Chem. Phys., 2024, 160, 092503 CrossRef CAS PubMed.
- G. Kresse and D. Joubert, Phys. Rev. B: Condens. Matter Mater. Phys., 1999, 59, 1758–1775 Search PubMed.
- J. P. Perdew, K. Burke and M. Ernzerhof, Phys. Rev. Lett., 1996, 77, 3865–3868 Search PubMed.
- S. Grimme, J. Antony, S. Ehrlich and H. Krieg, J. Chem. Phys., 2010, 132, 154104 Search PubMed.
- S. Bhattacharjee, B. Brena, R. Banerjee, H. Wende, O. Eriksson and B. Sanyal, Chem. Phys., 2010, 377, 96–99 CrossRef CAS.
- S. L. Dudarev, G. A. Botton, S. Y. Savrasov, C. Humphreys and A. P. Sutton, Phys. Rev. B: Condens. Matter Mater. Phys., 1998, 57, 1505–1509 CrossRef CAS.
- J. K. Nørskov, J. Rossmeisl, A. Logadottir, L. Lindqvist, J. R. Kitchin, T. Bligaard and H. Jónsson, J. Phys. Chem. B, 2004, 108, 17886–17892 CrossRef PubMed.
| This journal is © the Owner Societies 2026 |