Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Non-thermal plasma restructuring of electrocatalyst surfaces for efficient hydrogen and oxygen reactions
Chu
Qin
 a,
Zhenglong
Tao
a,
Zhenglong
Tao
 a,
Xuanhao
Wu
a,
Xuanhao
Wu
 *b and
Zhongqing
Jiang
*b and
Zhongqing
Jiang
 *a
*a
aZhejiang Key Laboratory of Quantum State Control and Optical Field Manipulation, Department of Physics, Zhejiang Sci-Tech University, Hangzhou 310018, China. E-mail: zhongqingjiang@zstu.edu.cn
bState Key Laboratory of Soil Pollution Control and Safety, Zhejiang University, Hangzhou 310058, China. E-mail: xuanhao.wu@zju.edu.cn
First published on 10th March 2026
Abstract
Electrocatalysts for water electrolysis, fuel cells, and metal–air batteries are largely governed by processes occurring within the outermost few nanometres of their surface. Low-temperature plasmas provide an effective means to tailor this region, using energetic electrons and reactive species to enable rapid, surface-confined modification while preserving the underlying scaffold. In this Feature Article, we review plasma engineering strategies for HER, OER and ORR electrocatalysts, including examples from zinc–air batteries and direct methanol fuel cells. We classify plasma effects into five generic design modes: phase transformation and plasma-assisted derivation; defect and vacancy engineering; plasma-induced doping; plasma-enabled deposition and interface construction; and single-atom anchoring with strong metal–support interactions. We also propose simple design rules that connect controllable plasma parameters to targeted active sites and device-level performance.
1. Introduction
Electrochemical energy technologies, including water electrolysis, hydrogen production, fuel cells, and metal–air batteries, critically depend on catalysts that can drive the hydrogen evolution (HER), oxygen evolution (OER), and oxygen reduction (ORR) reactions with high activity, durability, and efficient use of precious metals. Despite rapid progress, sluggish multi-electron kinetics and instability under industrially relevant conditions still constrain device performance and cost. In this context, plasma-assisted nanocatalyst engineering has emerged as a practical interface-engineering tool to tailor active phases, interfaces, and defect chemistries under comparatively mild conditions.9,10Low-temperature plasmas provide a non-equilibrium environment, in which energetic electrons generate a dense flux of radicals, ions and excited species while the heavy species remain comparatively cool. Here, “low-temperature” is used in the plasma-physics sense to denote non-thermal, non-equilibrium discharges, where electron-driven chemistry proceeds while the heavy species remain far cooler than in thermal plasmas.11,12 Under these conditions, conversion is confined to a shallow “skin” (few-tens of nanometers), so a parent scaffold can be chemically recast at the surface without losing its architecture.20 For electrocatalysts, this “skin-depth” conversion is advantageous: the outermost atomic layers control adsorption and reaction barriers, while the unconverted bulk maintains mechanical integrity, electrical percolation, and open porosity. With short treatments often in the range of seconds to minutes, the plasma exposure can simultaneously induce reduction/oxidation, etching, doping, and thin-film growth, offering a versatile and tunable approach for catalyst construction.25–28
The practical value of this approach shows up across hydrogen and oxygen electrodes. For HER, converting oxide or hydroxide precursors to thin metallic-like phosphide skins (or to nitride skins with high carrier density) lowers the energy cost of water dissociation and tunes ΔGH* toward the optimal range.30,31 For OER and ORR, vacancy-rich chalcogenide or sub-oxide layers adjust the binding energy of OOH*/OH*/O* intermediates, while adjacent nitride or metallic regions provide fast electron delivery and help stabilize the working oxidation states during potential swings.35 In overall water splitting, pairing these two functions on a common 3D current collector (carbon cloth, foams, MOF-derived carbons) reduces ohmic losses and improves durability at high current densities.36 In Zn–air batteries and direct methanol fuel cells, conformal plasma-grown skins help maintain activity under cycling, fuel crossover, and carbonation.37–39
Several characteristics of low-temperature plasmas make these outcomes reproducible and tunable. First, the time-temperature leverage: non-equilibrium chemistry opens reaction paths, so phase conversion, anion incorporation, and gentle reduction finish quickly and at low gas temperature. Second, depth control: because the reaction front is self-limited by short radical lifetimes and slow solid-state diffusion at low temperature, conversions stop naturally after a few nanometers, yielding thin and uniform shells on complex 3D supports. Third, fast anion exchange and near-surface redox produce non-stoichiometry (oxygen/chalcogen vacancies) and mixed valence, and the shallow conversion maximizes the density of phase boundaries, which often raise intrinsic activity and improve charge transfer when kept within a narrow thickness window.
Plasma processing also offers a set of controllable parameters that map directly onto catalytic requirements. The feed gas governs the dominant surface chemistry. For example, O2 for oxidation and oxygen-vacancy tuning,40 H2/NH3 for reduction and nitridation,41 PH3 or organophosphorus vapors for phosphidation,42 and S/Se-containing feeds for chalcogenization.43 Applied power, duty cycle and treatment time determine the radical flux and thus the depth of conversion, while pressure and flow rate control mean free paths and residence times, which in turn dictate how conformally porous scaffolds are modified.44 In capacitively coupled plasmas (e.g. atmospheric-pressure plasma jets, dielectric-barrier discharges, or low-pressure RF/ICP reactors), applying a substrate bias allows the ion bombardment energy to be tuned, providing an additional handle over surface activation and defect generation. In practice, a sequence of short, mild plasma exposures often outperforms a single aggressive treatment, producing thinner shells with higher interfacial area and fewer blocked pores, while minimizing stress and particle growth.
A large body of literature documents the promise of plasmas across energy chemistries (e.g., hydrogen production, ammonia synthesis, and broader plasma catalysis), but most surveys are organized by application or by reactor chemistry. This can obscure the fact that the same plasma effect (e.g., phosphidation or N-doping) is portable across materials families and reactions, and that performance gains often trace back to a small number of recurring structural motifs, like phase conversion, vacancies, heterointerfaces, and metal–support interaction (MSI). What is needed, therefore, is a clear link between tunable plasma parameters, the structural changes they produce, and the resulting catalytic behavior (as shown in Table 1), so that insights can be transferred across different systems.
| Reference | Plasma parameters | Structural descriptor | Catalytic metric |
|---|---|---|---|
| Li et al.45 | RF glow discharge N2/H2, N2/H2 = 40![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1; 24 Pa; RF power 120 W; 1 h; 250 °C 1; 24 Pa; RF power 120 W; 1 h; 250 °C |
Ni(OH)2 → porous Ni3N while keeping nanosheet macrostructure; Ni–N hybridized states | HER: η = 44 mV, Tafel = 46 mV dec−1 |
| Xu et al.1 | Ar/NH3 RF plasma; 20 Pa; 100 W; 500 °C; 1 h | Se-vacancy EPR g ≈ 2.003; Raman ID/IG 0.87 (plasma) vs. 0.79; BET 46.1 vs 18.4 m2 g−1 | OERR E1/2 = 0.80 V; OER η = 311 mV@30 mA cm−2 |
| Wei et al.48 | H2/N2 RF plasma; H2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) N2 = 1 N2 = 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1; 20 sccm; 30 Pa; 200 W; 1 h 1; 20 sccm; 30 Pa; 200 W; 1 h |
Surface Ni3N/Mo2N heterostructure on nanorods; crystalline shell (∼12 nm) over amorphous core; EPR g = 2.31 | HER: η100 = 66 mV; η500 = 134 mV; Tafel = 38 mV dec−1 |
| Ouyang et al.14 | RF N2 plasma; 13.56 MHz; 500 W; N2 40 sccm; ∼30 Pa; 300 s | Ni plate → Ni3N nano-framework; facet control: dominant (2–10) (cooling plasma) vs. (2–11) (normal plasma) | HER: η10 = 58 mV; η100 = 183 mV; Tafel = 64 mV dec−1; stable 72 h@−0.4 V vs. RHE |
| Zha et al.3 | RF N2 plasma; 100 W; 20 Pa; N2 5 sccm; phosphorization at 300 °C, 90 min | Ni-Doped CoP3 NPs on N-doped carbon nanofibers; rich P vacancies (g = 2.003); BET 44.4 m2 g−1 (vs. 20.6 m2 g−1 thermal) | HER: η100 = 107 mV, Tafel 68 mV dec−1; OER: η100 = 306 mV, Tafel 97 mV dec−1 |
| Salem et al.15 | RF-PECVD PH3/Ar plasma; PH3/Ar = 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 99; 10 sccm; 980 mTorr; 100 W; 250 °C; 3 h 99; 10 sccm; 980 mTorr; 100 W; 250 °C; 3 h |
MnNiCoP nanoflowers on NF; Ni–P/Co–P formation | HER: η10 = 14 mV, Tafel = 58 mV dec−1; OER: η10 = 289 mV; overall: 1.48 V@10 mA cm−2 |
| Xu et al.2 | Ar RF plasma (commercial 13.56 MHz); 100 W; 40 Pa; 120 s (0–240 s tested) | BET area 160.26 vs. 95.27 m2 g−1 (engraved vs. pristine); surface Co2+/Co3+ ratio 1.2 vs. 1.0 | OER: η10 = 0.30 V; Tafel 68 mV dec−1; TOF 0.21 s−1@η = 0.3 V |
| Han et al.17 | Ar or O2 plasma; 100 W; 130 Pa; 240 s | O vacancy ratio (A/B): 0.48 (L), 0.70 (P), 1.60 (H) | CO2RR: FE(CO) 94.8% at −0.62 V vs RHE; Tafel 77 mV dec−1 |
| Ye et al.21 | O2 RF plasma; 100 W; 20 Pa; 20 min | Co/CoO heterojunction with abundant oxygen vacancies; EPR signal present; ECSA 351.75 cm2 (vs. 247.25 cm2 without plasma) | ORR E1/2 = 0.855 V; OER 1.607 V@10 mA cm−2; ΔE 0.752 V; liquid ZAB 133.5 mW cm−2, 1000 cycles |
| Wang et al.6 | Ar plasma, 18 W, 0.6 Torr | Exposure of PtSe2 (101) facet (60 s); Se loss; formation of Pt-rich amorphous layer | 17.6 mA cm−2 (60 s, 50 mV s−1); Tafel slope: 177 mV dec−1 |
| Zhang et al.13 | Ar microwave plasma, 100 W, 0.5–1.0 Pa, 15–45 min | Defect-engineered CoP3/Ni2P heterostructure (vacancy-type/O-refilling defects at interface) | HER: η10 = 21 mV (acidic), 37 mV (alkaline); OER: η10 = 300 mV |
| Li et al.4 | NH4F RF plasma, 500 W and 13.56 MHz for 1 min | Dual N, F incorporation in Co3O4, increased M–O covalency; O 2p upshift; lattice-oxygen activation | OER: η10 = 254 mV |
| Zheng et al.58 | DBD cold plasma; 100 V, 1.5 A; 30 min; gases: Ar, O2, air; T < 140 °C | Oxygen vacancies; Co3+ → Co2+ partial reduction; surface CoO formation; N doping up to 10.1 at% | ORR onset shift +0.03 V; j = 4.9 mA cm−2 at 0.2 V (vs. RHE); e− transfer number ≈ 3.99 |
| Chen et al.19 | Focused plasma jet system with AC power: 0–30 kV, 8.00–30.0 kHz | Electrode-emerged NiCo(OH)x/NiCo2O4 nanointerfaces grown directly on Ni foam | At j = 100 mA cm−2: HER η = 306 mV, OER η = 484 mV |
| Hoek et al.68 | O2 plasma steps; RF power 200 W; 0.1 Torr | Ultrathin NiOx@Co3O4 (amorphous/defect-rich NiOx shell) | OER: η10 = 360 mV (vs. 420 mV for Co3O4/CC) |
| Zhu et al.22 | H2/N2 plasma; 200 W; 80 mL min−1; 20 min | Defective TiO2 nanowires (oxygen vacancies); ultrafine Ir nanoparticles; Sr single atoms stabilized on defective TiO2 | OER: η10 = 250 mV; HER: η10 = 32 mV |
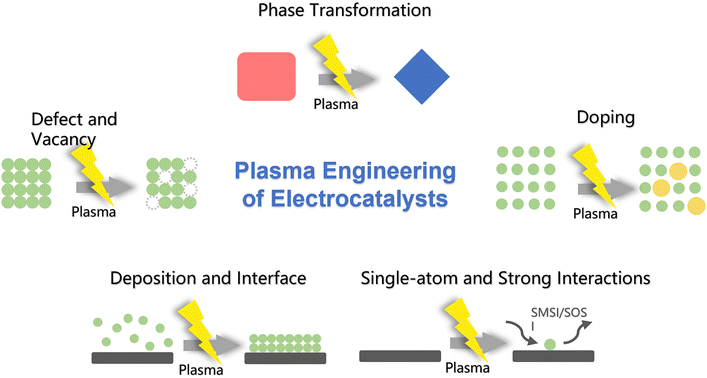
Building on our group's sustained work with plasma-engineered electrocatalysts spanning HER, OER, ORR, zinc–air batteries (ZABs), and direct methanol fuel cells (DMFC) cathodes, we frame plasma's role through five fundamental effect categories (Fig. 1): (i) phase transformation and plasma-assisted derivation; (ii) defect and vacancy engineering; (iii) plasma-induced doping; (iv) plasma-enabled deposition and interface construction; (v) single-atom anchoring and strong metal–support interactions. Across HER, OER, ORR, overall water splitting, ZABs, and DMFCs, the recurring theme is the same: write the chemistry where the reaction happens and keep the physics that carries current. Low-temperature plasmas make that balance practical, scalable, and compatible with binder-free electrodes and device-level current densities.
For each effect, we describe how plasma parameters drive structural evolution, and how these structures in turn modulate in situ binding and transport to deliver device-level advantages. By emphasizing a structure-first causality that links controllable plasma conditions to characteristic structural signatures and then to catalytic performance, we aim to provide portable design rules that can be applied across HER, OER and ORR systems rather than treating each application in isolation. This mechanism-oriented view is intended to complement application-focused reviews and to guide predictive, scalable catalyst design that leverages the distinctive capabilities of non-thermal plasmas.
2. Plasma effects in catalyst engineering
2.1. Phase transformation and derivation
Phase transformation is among the most fundamental yet effective plasma-induced effects for tailoring catalytic materials. This section covers cases where plasma produces a discernible near surface phase or conversion layer, evidenced by new phase signatures such as XRD or SAED, or a clearly resolved shell. Compared with conventional thermal conversion routes, non-thermal plasma enables phase transformation to be confined to the near-surface region. Non-thermal electrons, radicals and ions drive rapid, near-surface anion exchange and lattice rearrangement, which enables chemically intensive transformations of the active phase, including conversion into nitrides, phosphides, sulfides, selenides or defect-rich oxides. Two features are particularly important for electrocatalysis: the emergence of a new surface phase and the formation of closely coupled phase boundaries with distinct electronic structures. Together, these reshape adsorption energetics at the active surface while ensuring efficient mass and electron transport through the electrode.Plasma nitridation provides one of the clearest demonstrations of this decoupling between surface chemical change and retaining architectures. As reported by Li et al., Ni(OH)2 nanosheets were converted into porous Ni3N under an N2/H2 radio-frequency (RF) glow-discharge plasma, which was chosen for its non-thermal, radical-rich environment that enables rapid nitridation while preserving the nanosheet framework.45 In this discharge, adding H2 promotes the formation of NH* species and simultaneously assists the reduction of Ni(OH)2 to reactive Ni, together accelerating Ni–N bond formation and the Ni(OH)2 → Ni3N transformation. Replacing OH− with N3− strengthens metal–ligand covalency and introduces Ni–N hybridized states near the Fermi level, which manifests as lower charge-transfer resistance and enhanced HER kinetics. Liu et al. showed that NiCoO nanostructures exposed to N2 plasma develop NiCoN-rich shells surrounding NiCoO cores.46 The resulting oxide–nitride junctions create internal electric fields arising from the electronegativity contrast between N and O and the coexistence of multiple Ni/Co valence states, effectively lowering the barriers for OH− adsorption and deprotonation in zinc–air ORR/OER cycles.
Building on this observation, Xu et al. extended the plasma-induced phase-transformation concept to chalcogenides.1 They first prepare Ni/NCNT@CC by electrodeposition, then obtain p-Ni@Ni3N/NCNT@CC by Ar/NH3 RF plasma nitridation, a configuration typically chosen to provide a high flux of reactive nitrogen/hydrogen species for rapid, near-surface nitride formation without collapsing the porous carbon scaffold. Subsequent high-temperature selenization produces a ternary p-NiSe2/Ni@Ni3N/NCNT@CC catalyst in which NiSe2/Ni-Ni3N heterojunctions are uniformly anchored on N-doped CNTs (Fig. 2a–c). In the RF Ar/NH3 discharge, the interfacial nitridation is governed by plasma-generated NH* (and related NHx*) species together with Ar* activation/etching, which promotes Ni–N bond formation and stabilizes a thin Ni3N skin and nitrogen-enriched interface that survives the subsequent high-temperature selenization. These multiple heterointerfaces generate band offsets that redistribute charge across the junctions, thereby optimizing both H2O activation and H* binding relative to any single phase. Liu et al. further showed that nitridation of NiCo2S4 generates a NiCo2S4@Co2N heterostructure, producing sulfide–nitride boundaries populated with coordinatively unsaturated metal sites that facilitate water dissociation.47
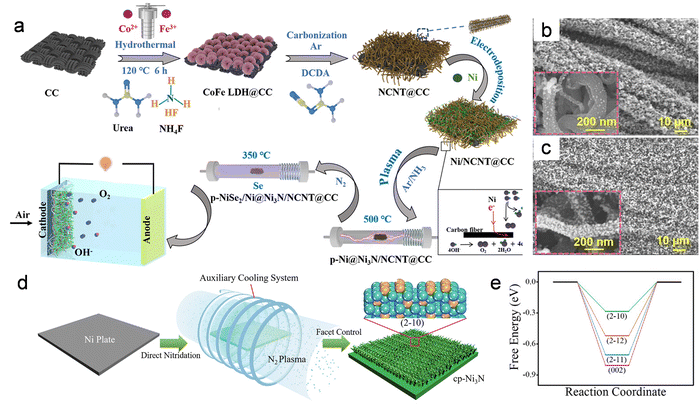 | ||
| Fig. 2 (a) Schematic illustration of the synthetic process of the p-NiSe2/Ni@Ni3N/NCNT@CC. SEM images of (b) p-Ni@Ni3N/NCNT@CC and (c) p-NiSe2/Ni@Ni3N/NCNT@CC. Reproduced with permission.1 Copyright 2025, Wiley-VCH. (d) Schematic Illustration of the as-prepared Ni3N nano-frameworks through cooling-assisted plasma process. (e) Free energy for HER on typical facets of hexagonal Ni3N model. Reproduced with permission.14 Copyright 2022, Wiley-VCH. | ||
Likewise, Wei et al. showed that near-room-temperature H2/N2 plasma converts Ni–Mo oxide hydrate nanorods into a conformal Ni3N/Mo2N shell, where reactive H species first promote reduction and Mo–O/Ni–O bond breaking, followed by activated N species permeation to form M–N bonds.48 The resulting nitride–oxide heterostructure lowers the barrier for nitride formation and couples Ni3N sites for H adsorption with Mo2N sites for water activation, thus enhancing alkaline HER. At higher plasma reactivity, non-thermal nitridation can even steer crystallographic outcomes: Ouyang et al. reports that a cooling-assisted plasma process suppresses surface heating and thereby enables preferential exposure of the metastable Ni3N(2–10) facet rather than the thermodynamically favored (2–11) facet.14 Notably, this was achieved in a RF nitridation plasma, where auxiliary cooling tunes the plasma-surface energy coupling instead of changing the precursor or reaction chemistry. Operando plasma diagnostics further show that cooling reduces surface temperature and moderates key plasma parameters, shifting the nitridation regime away from thermally stabilized growth and toward metastable facet exposure. DFT further identifies Ni3N(2–10) as having a nearly optimal ΔGH* for alkaline HER, explaining the superior kinetics observed experimentally (Fig. 2d and e). Collectively, these studies show that plasma nitridation rewires the surface electronic structure by introducing more metallic d–p hybrid bands, internal fields and fast charge-transfer channels, while leaving the micron-scale geometry effectively untouched.
On the same principle of surface-confined anion exchange, plasma phosphidation converts hydroxide, oxide and chalcogenide precursors into interfacially rich phosphides whose electronic properties differ markedly from their parent phases. In our recent work, Zha et al. showed that with NaH2PO2 and RF plasma, the sheet-like NiCo LDH collapses into intertwined nanowires composed of Ni-doped CoP3 nanoparticles uniformly anchored on N-doped carbon nanotube frameworks3 (Fig. 3a and b). This low-pressure N2 RF discharge was selected to supply a high flux of reactive P-containing species from NaH2PO2 decomposition while limiting bulk heating, enabling a rapid, surface-confined phosphidation that preserves the conductive carbon backbone. Mechanistically, the dominant plasma effect is the generation of energetic radicals/ions that accelerate M–O(H) bond rupture and M–P bond formation, while concurrent plasma etching promotes P-defect formation and surface roughening, which collectively enhance charge transfer and accelerating H*/OH* adsorption at the phosphide surface.
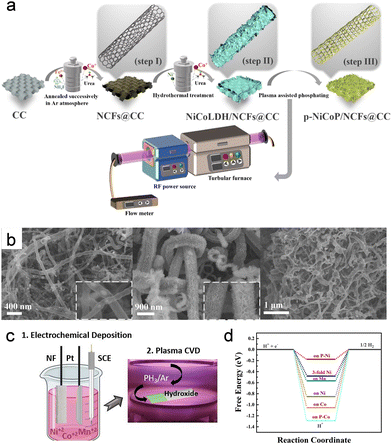 | ||
| Fig. 3 (a) Schematic of the synthesis process of the p-NiCoP/NCFs@CC. (b) SEM images of NCFs@CC, NiCoLDH/NCFs@CC, and p-NiCoP/NCFs@CC. Reproduced under the terms of CC-BY 4.0 license.3 (c) Schematic of the design of the MnNiCoP alloy on nickel foam as electrolytic water splitting bifunctional catalyst. (d) Free-energy diagram of intermediate state for H* adsorption on the MnNiCoP (0001) surface. Reproduced with permission.15 Copyright 2023, Wiley-VCH. | ||
Plasma phosphidation can push MoS2-based hybrids into more strongly coupled metal–phosphide junctions, rather than simply completing an anion-exchange step. Fu et al. used RF plasma-assisted phosphorization to synthesize MoS2@Ni12P5/ZnP2, where finer Ni12P5 and ZnP2 nanocrystallites uniformly decorate MoS2 and create dense nanoscale heterojunctions with strengthened electronic coupling.49 Zhang et al. reported that plasma-assisted phosphidation transforms NiMoO4 nanorods into a tri-phase MoP2/Ni3P/MoO2 composite composed of small sub-nanometer crystallites embedded within the preserved rod architecture.50 In this discharge, energetic N/N2+ species promote the generation of reactive PH· and H· from NaH2PO2-derived PH3, accelerating phosphidation and simultaneously enriching oxygen vacancies in MoO2.
As reported by Salem et al., ternary Mn–Ni–Co hydroxides phosphidized using a PH3 plasmas (plasma-enhanced chemical vapor deposition, PECVD) process, yielding porous flower-like MnNiCoP that retains the hierarchical framework while shifting from a hydroxide-like, weakly conductive state to a more metallic phosphide surface15 (Fig. 3c and d) The key plasma contribution is the high flux of reactive species that accelerates M–O(H) bond removal and M–P bond formation with uniform phosphidation across the 3D architecture, thereby improving conductivity and exposing dense catalytic sites for fast alkaline HER/OER kinetics.
Plasma phosphidation can be paired with prior plasma oxidation to stack phases with complementary roles. Zhang et al. created VNiCoPy/NiFeOx heterostructures by using an O2 atmospheric-pressure plasma jet (APPJ) to form vacancy-rich NiFeOx, followed by plasma-assisted phosphating of the overlying VNiCo layer.51 The resulting phosphide/oxide junction shows interfacial charge redistribution, where the phosphide supplies conductive pathways and the oxide stabilizes high-valent states; importantly, the plasma steps preserve the 3D architecture while tightening interfacial coupling.
Plasma-assisted oxidation and oxide deposition offer a third pathway to surface phase engineering, particularly for ultrathin catalytic layers and dynamic reconstruction. As reported by Mallmann et al., atmospheric-pressure plasma-enhanced CVD is used to produce Co3O4 films at reduced substrate temperatures, where the plasma provides reactive oxygen-containing species and simultaneously promotes rapid nucleation and controlled crystallization.52 In this AP-PECVD setup, the blown-arc plasma torch produces a reactive afterglow in which long-lived oxygen-containing radicals (notably NO and OH) dominate precursor activation and oxide growth. Yang et al. showed that O2 plasma pretreatment combined with plasma-enhanced ALD yields NiOx@Co3O4 electrodes with an ultrathin amorphous NiOx shell, where an RF O2 plasma step supplies O species for surface activation and the PE-ALD plasma pulse enables low-temperature, conformal oxidation during growth.53 XPS reveals abundant Ni3+ and non-lattice oxygen, indicating a defect-rich sub-oxide that can dynamically reconstruct into Ni(oxy)hydroxide during OER operation. In all of these cases, the oxide phase formed by plasma is electronically and structurally primed for further transformation under operation, lowering the barrier to reconstruct into catalytically competent (oxy)hydroxide states.
Taken together, these nitridation, phosphidation and oxidation studies point to a unified mechanism. Non-thermal plasmas selectively rewrite the chemistry and electronic structure of the outer reaction layer, generating new phases, heterointerfaces and defect configurations that govern intermediate binding and reaction kinetics, while the mesoscopic scaffold remains largely unchanged and continues to support efficient electron and mass transport. Across systems, SEM and low-magnification TEM consistently reveal inherited nanosheet, nanorod, nanoarray and foam morphologies, even as HRTEM and XPS uncover thin transformed shells enriched in nitride, phosphide or sub-oxide species, modified metal valence states and redistributed charge densities. A conceptual workflow that begins with LDH, oxide or chalcogenide precursors, proceeds through a radical- and electron-rich plasma environment, and emerges as multi-phase, interface-dense electrocatalysts captures the essence of these transformations. The recurrence of these plasma-driven motifs across alkaline water splitting, overall electrolysis, zinc–air batteries and fuel-related reactions underscores the versatility of non-thermal plasmas in re-engineering surface phases and electronic landscapes without compromising the advantages inherent to rational nanoarchitectural design.
2.2. Defect and vacancy engineering
Plasma-induced defect engineering represents a second, complementary axis to the phase transformations discussed above. This section focuses on situations where the parent phase is largely retained while defect populations or coordination disorder are tuned, as indicated by EPR signals, defect components in XPS, or local structure changes in EXAFS. Instead of converting the parent phase into nitrides or phosphides, non-thermal plasmas can leave the crystallographic identity and nanoarchitecture essentially unchanged while densely decorating the near-surface region with vacancies, distorted coordination environments and disordered shells. These local imperfections are highly effective in reshaping the electronic structure, tuning adsorption energetics and opening new reaction channels, yet they are confined to only a few nanometres so that the underlying scaffold continues to provide mechanical integrity and efficient electron and mass transport. In contrast to ion implantation or high-temperature reduction, which can introduce deep lattice damage, non-thermal plasma generates defects and vacancies through surface-localized chemical reactions, making it particularly suitable for engineering electrocatalytically active interfaces with minimal bulk disruption.The clearest picture emerges from oxides in which plasma engraving generates oxygen vacancies. In a widely cited study on Co3O4 nanosheets by Xu et al., low-power plasma bombardment roughens the sheet surfaces, removes lattice oxygen and partially reduces Co3+ to Co2+ while leaving the spinel framework intact.2 (Fig. 4a and b) Specifically, an RF Ar plasma was used as an ion-assisted engraving/etching treatment, where energetic Ar species primarily drive surface sputtering and oxygen removal, so the resulting oxygen vacancies are largely confined to the near-surface region. XPS, EPR and EXAFS collectively show the appearance of vacancy-related oxygen components, changes in Co coordination and a more disordered Co–O environment, which together increase electronic conductivity and introduce defect states that promote lattice-oxygen mediated OER pathways.
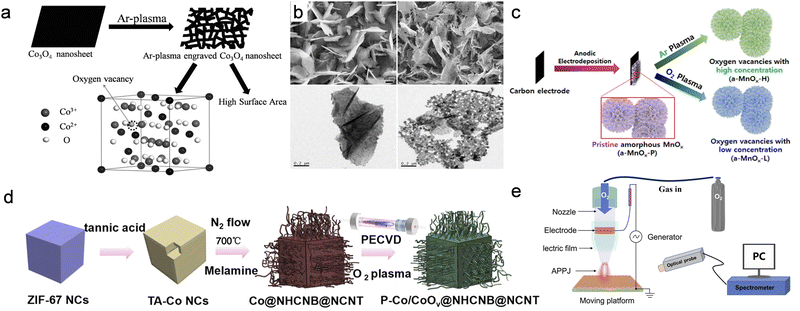 | ||
| Fig. 4 (a) Fabrication of oxygen-vacancy-rich Co3O4 with high surface area via Ar plasma engraving. (b) SEM and TEM images of pristine (left) and Ar-plasma engraved (right) Co3O4, respectively. Reproduced with permission.2 Copyright 2016, Wiley-VCH. (c) Schematic of the plasma treatment for the a-MnOx catalysts with different concentrations of oxygen vacancies. Reproduced with permission.17 Copyright 2020, Elsevier. (d) The synthesis process of the P-Co/CoOV@NHCNB@NCNT. Reproduced under the terms of CC-BY 4.0 license.21 (e) Schematic illustration showing the APPJ setup and the treatment of the MnOx film. Reproduced with permission.32 Copyright 2024, Wiley-VCH. | ||
A related strategy was adopted in amorphous MnOx by Han et al.17 (Fig. 4c). Here, controlled Ar and O2 plasma treatments were used to tune vacancy density: Ar plasma favors oxygen removal via energetic-species-driven surface activation/etching, whereas O2 plasma provides oxygen-containing reactive species that suppress or passivate VO sites. Electrochemical CO2 reduction on these vacancy-rich MnOx films shows markedly higher CO faradaic efficiency and lower overpotentials, consistent with the idea that VO sites stabilize CO2− and facilitate subsequent proton–electron transfer. Liang et al. extended this concept to crystalline WO3 nanowires for acidic OER: a brief Ar plasma sputtering step creates a high density of surface oxygen vacancies, lowers the average W valence and distorts surface WO6 octahedra, as seen by atomic-resolution microscopy and XAFS.54
Similar oxygen-defect behavior also emerges in more compositionally complex systems, including those developed in our Zn–air and OER studies. Ye et al. used plasma activation on Co-based air electrodes, showing that O2-containing plasmas generate oxygen non-stoichiometry and high-valent Co species at the surface without collapsing the porous framework21 (Fig. 4d). In their work, optical emission spectroscopy (OES) confirms the presence of energetic O species and O2+ in the O2 RF plasma discharge. Mechanistically, these reactive O species partially oxidize the Co surface to form a thin CoO skin, while concurrent energetic bombardment/etching helps create oxygen-vacancy-rich Co/CoO heterojunctions. The resulting electrodes exhibit enhanced bifunctional ORR/OER activity and improved full-cell performance, with XPS and EPR fingerprints that closely mirror the VO signatures in Co3O4 and MnOx.
Zhang et al. pursued a related strategy in Ni-based OER catalysts, where atmospheric-pressure plasma treatments increase the fraction of high-valent metal sites and introduce disordered oxygen environments that favour rapid redox cycling under OER conditions.55 Qin et al. further demonstrated that plasma-generated defective oxide skins exhibit exceptional structural and compositional stability. Post-operation analyses revealed that the catalyst, with its inherently high concentration of stabilized Mn3+ and oxygen vacancies, maintains its active state during the oxygen evolution reaction, directly contributing to the high activity and long-term durability32 (Fig. 4e).
Beyond oxygen, non-thermal plasmas can also sculpt chalcogen and pnictogen vacancy landscapes. In the case of layered PtSe2 crystals by Wang et al., RF plasma exposure preferentially removes Se from the outermost layers, creating Se vacancies and driving partial reduction or segregation of Pt at the surface6 (Fig. 5a). Because the feed is inert Ar, the dominant role of the plasma is ion/electron-assisted physical etching, which selectively depletes the more weakly bound chalcogen from the topmost layers and is naturally self-limited in depth. The resulting Pt-enriched, vacancy-populated skin therefore arises from a near-surface compositional “reset” rather than bulk phase conversion. Structural and spectroscopic analysis reveal a Pt-rich, defect-populated skin coherently coupled to the underlying PtSe2 lattice.
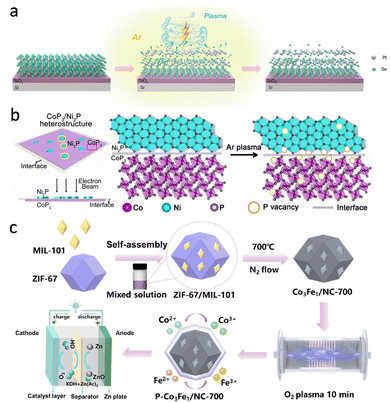 | ||
| Fig. 5 (a) Schematic illustration of PtSe2 nanosheet under Ar plasma treatment. Reproduced with permission.6 Copyright 2025, Wiley-VCH. (b) Schematic illustration of the synthesis process of CoP3/Ni2P. Reproduced with permission.13 Copyright 2021, Wiley-VCH. (c) Schematic illustration of the synthesis approach of P-Co3Fe1/NC-700. Reproduced with permission.23 Copyright 2023, Elsevier. | ||
In our Zn–air systems, Hu et al. and Feng et al. demonstrated that dielectric-barrier discharge (DBD) and RF plasma treatment of NiFe/NiSe2 and CoNiSe-based air cathodes induces the formation of non-stoichiometric surface layers rich in oxygen vacancies or selenium vacancies, along with modulated metal valence states (e.g., Ni2+/Ni3+, Co2+/Co3+) and enhanced electronic conductivity.56,57 These structural and electronic modifications lead to improved ORR/OER activities, higher limiting current densities, lower overpotentials, and superior cycling stability in both liquid and flexible all-solid-state Zn–air batteries. These works underscore that plasma-induced vacancy engineering is a universal strategy for enhancing the performance of chalcogenide-based electrocatalysts, analogous to its role in oxides.
In many cases, defects and interfaces are generated together, and their interplay becomes the dominant control knob for activity. Zhang et al. provide a representative CoP3/Ni2P example, where an Ar microwave plasma treatment targets the ultrathin 2D heterostructure and drives interfacial P escape, generating P-vacancy defects preferentially at the CoP3/Ni2P junction13 (Fig. 5b). Meanwhile, a fraction of these vacancies is occupied by external oxygen to form O-refilling defects, producing a mixed defective interface rather than a single clean junction. Such vacancy-decorated heterointerfaces reconstruct the local electronic structure, intensify charge redistribution across Co- and Ni-centred sites, and lower kinetic barriers – thereby enhancing HER via improved H* binding and promoting alkaline steps by facilitating interfacial charge transfer and water activation.
Similar defect-rich heterojunctions arise in our Co-based Zn–air catalysts after plasma activation, where Co/CoO, Co–N–C and carbon nanotube frameworks are intimately coupled, as reported by Xiong et al.23 (Fig. 5c). Here, an oxygen RF plasma was used to activate the air electrode, because O-containing radicals/ions can introduce oxygen non-stoichiometry and interfacial disorder without the thermal budget that would sinter Co-based sites or collapse the CNT network. In these systems, plasmas introduce vacancies and disordered coordination at the metal/oxide or metal/carbon interfaces, leading to enhanced electronic coupling and more favorable ORR/OER kinetics at the composite level rather than at any single phase in isolation.
Taken together, these studies show that plasma-induced defects are not random damage but structured design elements. Oxygen, chalcogen and pnictogen vacancies, coordination distortions, defect-rich interfaces and ultrathin disordered shells all arise from the same basic features of non-thermal plasmas: a flux of energetic electrons and reactive radicals that act within only a few surface layers, far from thermodynamic equilibrium. The resulting defect ensembles tune metal valence, introduce new electronic states, increase covalency in metal–ligand bonds and redistribute charge across junctions, thereby modulating the adsorption and transformation of key intermediates in HER, OER, ORR and CO2 reduction. In combination with the phase transformation pathways discussed earlier, plasma defect engineering therefore provides an orthogonal yet synergistic handle for reprogramming surface chemistry and electronic landscapes.
2.3. Plasma-induced doping and surface functionalization
Low-temperature plasmas provide a third mode of control over electrocatalysts beyond phase transformation and vacancy creation: they can selectively introduce heteroatoms into the near-surface region and thereby rewrite the local electronic structure without sacrificing the underlying phase or nanoarchitecture. Unlike bulk doping achieved by high-temperature diffusion or solution-based routes, plasma-induced doping is inherently shallow and interface-focused, enabling controlled modification of surface electronic structure without altering the bulk composition. Energetic electrons and reactive radicals act within only a few nanometers, breaking specific metal–ligand or C–C bonds and creating transient open sites that can be occupied by N, F, P, B or oxygenated fragments. This section discusses plasma enabled incorporation of heteroatoms or functional groups, identified by composition changes and bonding motifs related signatures.A first, and perhaps clearest, manifestation of this behavior is seen in N/F doping of transition-metal oxides and chalcogenides, where the primary role of plasma is to tune the covalency and energy alignment of metal–ligand bonds. Li et al. used an NH4F-containing RF plasma to co-introduce N and F into Co3O4 nanowire arrays while preserving the spinel phase and 1D architecture4 (Fig. 6a). The key outcome is electronic: dual anion incorporation strengthens Co–O covalency and shifts the O 2p band closer to the Fermi level, lowering the barrier for lattice-oxygen participation and pushing OER from a purely adsorbate-evolution route toward a more lattice-oxygen-involved pathway, without changing the nanowire backbone. Density functional calculations corroborate that dual N, F incorporation pulls the O 2p band closer to the Fermi level and increases Co–O covalency, which lowers the free-energy barrier for lattice-oxygen participation.
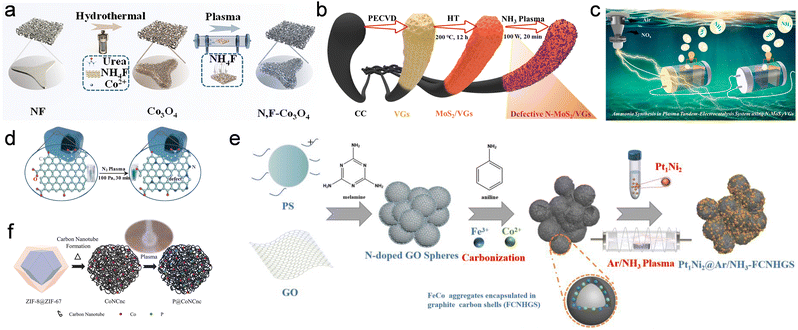 | ||
| Fig. 6 (a) Schematic illustration of the synthesis process of the N,F-Co3O4. Reproduced with permission.4 Copyright 2025, Wiley-VCH. (b) Schematic illustration for N-MoS2/VGs engraved by RF NH3 plasma. (c) The plasma tandem-electrolysis system. Reproduced under the terms of CC-BY 4.0 license.8,9 (d) Schematic illustration showing the synthesis of N-doped quasi-graphene by N2 Plasma. Reproduced with permission.16 Copyright 2020, American Chemical Society. (e) Illustration of the fabrication of Pt1Ni2@Ar/NH3-FCNHGS. Reproduced with permission.29 Copyright 2023, Elsevier. (f) Schematic showing the P soft doping of CoNCnc by microwave plasma. Reproduced with permission.34 Copyright 2025, Elsevier. | ||
Plasma-enabled N doping can also be used to construct multi-parameter surface motifs, in which heteroatoms, vacancies and even metastable phases cooperate. Zheng et al. treated MoS2 with RF NH3 plasma to produce N-doped, vacancy-rich and partially 1T-phase MoS2 for tandem plasma–electrocatalytic nitrogen chemistry8 (Fig. 6b and c) The plasma step introduces N into the S sublattice, creates S vacancies and induces a local 2H to 1T transition, again confined to the near-surface region. Calculations show that these features collectively enhance charge transfer to nitrogenous intermediates and suppress hydrogen evolution by shifting the Mo d states and creating electron-rich traps at N/S-defective sites. In this case, the dopant is not a passive spectator but an integral part of a complex defect-phase ensemble, yet the macroscopic morphology of the MoS2 support is preserved.
Analogous principles apply in carbon-based systems. Li et al. exposed quasi-graphene to N2 plasma, introducing pyridinic, pyrrolic and graphitic nitrogen into the topmost carbon layers while retaining the overall sheet-like morphology16 (Fig. 6d). The nitrogen functionalities increase the density of edge-like sites, improve electronic conductivity and adjust the redox potential of surface carbon atoms, which together translate into markedly enhanced peroxidase-mimicking activity towards H2O2. Here, the active phase is the doped carbon itself rather than a transition metal, but the underlying logic is the same: plasma-installed N dopants and associated defects reshape the local density of states at the Fermi level and create chemically distinct adsorption sites, without the need for high-temperature bulk treatments.
Within our own work, Gong et al. used atmosphere-programmed plasma to tune N doping and defect density in Fe/Co/N-decorated hollow graphene spheres as supports for PtNi2 nanoparticles29 (Fig. 6e). The source is an RF plasma operated at low pressure, allowing the reactive environment to be switched simply by changing the feed gas. Among the atmospheres, Ar/NH3 is the most effective because it couples Ar-driven etching with NHx chemistry: OES shows NH- and H-related species during discharge, consistent with simultaneous surface roughening/defect creation and the formation of pyridinic/graphitic N and Fe(Co)–Nx motifs. These N-rich defects act as anchoring sites that suppress PtNi2 agglomeration and strengthen metal–support electronic coupling, as reflected by the binding-energy shifts of Pt and Ni. The resulting catalysts deliver improved ORR/HER activity and fuel-cell durability compared with undoped or thermally doped counterparts, highlighting plasma N-doping as a practical route to engineer carbon supports for higher noble-metal utilization.
In many plasma-treated systems, doping is intimately coupled to defect formation so that vacancies act as entry points for heteroatoms. This vacancy-mediated route provides a natural bridge between the defect engineering discussed earlier and the doping strategies considered here. In the NiCo2O4 study by Zheng et al.58 DBD plasma first generates abundant oxygen vacancies in the spinel lattice, partially reducing Co3+ to Co2+ and disordering the local Co–O environment. Subsequent nitrogen incorporation occurs preferentially at these defective sites, forming Ni–Nx and Co–Nx coordinations that stabilize the vacancies and further modify the electronic structure. A similar sequence arises in the NiCoP system of Zhang et al., where vacuum annealing and plasma treatment create oxygen-deficient NiCoOx precursors, and N doping during phosphidation yields an N-doped, vacancy-rich NiCoP surface.59 In both cases, dopants and vacancies form coupled ensembles: vacancies lower the barrier for substitution, and the heteroatoms in turn stabilize and electronically activate the defective sites.
Huang et al. reached a related conclusion in N, Fe co-incorporated CoO nanoarrays: the N, Fe co-bonded CoO was formed via a nitrogen-plasma step carried out in a N2 PECVD reactor, and the reactive nitrogen species (e.g., N/excited N2/N2+) generated in the discharge both etch the surface and enable near-surface N incorporation. As a result, N acts as a chemical bridge to bind Fe and Co centers while simultaneously promoting oxygen non-stoichiometry and higher-valent Co at the surface, pointing to a cooperative adjustment of vacancies, dopants and metal valence that optimizes the energetics of lattice-oxygen-involved OER.60
Beyond directly modifying active phases, plasma doping can be applied in a “soft” fashion to carbon and interfacial layers, where the principal outcome is to re-engineer metal–support interactions, wettability and charge-transfer pathways rather than bulk band structure. Ai et al. used a microwave plasma to gently dope phosphorus into Co nanoparticle-embedded, nest-like N-doped carbon frameworks34 (Fig. 6f). Mechanistically, energetic electrons in the microwave discharge volatilize the phosphate salt into short-lived P-containing radicals/ions (P*/POx/PHx*), which graft onto defected carbon and Co–N sites to form covalent P–C and P–O–C motifs. The P functionalities introduce additional defects, increase hydrophilicity and adjust the local electron density around Co–N centers, leading to improved oxygen reduction activity and stability compared with undoped samples. Lin et al. employed oxygen plasma to activate carbon nanotube-interconnected Prussian blue analogue nanoarrays.61 The treatment decorates CNTs with oxygenated groups and partially oxidizes the PBA surface, thereby lowering interfacial resistance and introducing more redox-active metal–oxygen moieties for the OER.
Zhao et al. show that plasma is useful not just as a “doping step”, but as a sequence controller for building multi-heteroatom interfaces in porous carbon/NiFe hybrids for zinc–air batteries.62 An initial Ar plasma etch activates the carbon scaffold – creating defects and opening access to embedded NiFe domains – so that subsequent B doping and NH3 treatment can be directed to the most reactive carbon sites and, critically, to the metal–support boundary. The outcome is a coupled B/N environment (B–C, B–O and interfacial M–Nx motifs) in which plasma-created defects act as nucleation/anchoring points, stabilizing high-valent Ni/Fe centers while improving charge transport through the porous carbon network. As a result, the co-doped architecture delivers stronger ORR/OER bifunctionality and improved zinc–air cell performance than singly doped or undoped counterparts, underscoring plasma's value as a practical platform for constructing electronically synergistic interfaces rather than isolated dopant sites.
Taken together, these studies demonstrate that plasma-induced dopants are not random impurities but precisely placed design elements that operate alongside phases and vacancies. In the N/F-doped oxides and chalcogenides, heteroatoms directly tune metal–ligand covalency and shift band positions, thereby optimizing the adsorption energies of key intermediates in OER, ORR and nitrogen reduction. In vacancy-assisted systems, dopants and defects are co-localized, with vacancies enabling heteroatom incorporation and the heteroatoms stabilizing and electronically activating the defective sites. In soft-doped carbons and interfacial layers, heteroatoms such as N, P and B reshape metal–support interactions, wettability and local charge distributions, improving utilization and stability of the catalytically active phase. Across all cases, the distinctive advantage of non-thermal plasmas lies in their ability to sculpt these dopant landscapes within only a few surface layers, far from thermodynamic equilibrium and without sacrificing the integrity of carefully designed nanoarchitectures. In combination with the phase-transformation and defect-engineering pathways discussed earlier, plasma-enabled doping thus provides a complementary and highly versatile lever for reprogramming surface chemistry and electronic structure in electrocatalysts.
2.4. Deposition and interface engineering
Plasma processing does more than transform bulk phases or create vacancies; it actively constructs the interfacial landscape through which electrons, ions and molecules exchange. This section highlights cases where plasma is used to deposit, reduce, or assemble a secondary component and the main outcome is a designed junction or conformal coating, tracked by dispersion, thickness, and interfacial electronic fingerprints. Compared with wet-chemical deposition or thermal growth, plasma-enabled deposition allows reactive species to be delivered directly to the surface under non-equilibrium conditions, facilitating the construction of ultrathin interfaces and heterostructures without prolonged thermal exposure. In many representative systems, plasma acts simultaneously as a surface activator and a non-thermal driver for metal nucleation, enabling catalytic metals or secondary phases to be anchored onto predesigned architectures with interfacial chemistries that are purposefully engineered rather than incidentally formed.Li et al. illustrated this using Pt-decorated NiCo2O4 nanowire arrays for bifunctional oxygen electrocatalysis and Zn–air batteries.63 Specifically, a RF Ar plasma is used to activate the oxide mainly through energetic Ar+ bombardment/electron-stimulated oxygen removal, followed by a brief H2/Ar plasma that supplies H˙/e− for rapid in situ reduction of Pt precursors and immediate interfacial anchoring. Compared with thermal treatment, the plasma route produces smaller and more uniformly dispersed Pt domains and strengthens the Pt–NiCo2O4 interfacial interaction. These features lower the charge-transfer resistance and enhance ORR/OER kinetics, consistent with stronger electronic coupling at the Pt–oxide interface that optimizes oxygen-intermediate binding rather than simply increasing Pt loading.
A similar interplay between deposition and interface activation appears across a family of plasma-engineered fuel-cell electrodes. In the work by Zha et al., PtNi nanoparticles were assembled on nitrogen-doped carbon nanotubes (NCFs) grown from CoFe-layered double hydroxide precursors, using Ar/NH3 RF plasma treatment.64 The plasma served to create defects and oxygen vacancies in the carbon support, enhance PtNi loading, and reduce the oxidation state of Pt and Ni, leading to improved catalytic activity for both ORR and MOR in alkaline direct methanol fuel cells. Hu et al. combined RF Ar/H2 plasma pretreatment of N-doped carbon nanotubes with plasma reduction of Pt precursors, showing that defects introduced into the carbon framework provide high-affinity anchoring sites and that plasma reduction yields Pt nanoparticles with an optimized fraction of oxidized surface atoms.65 Wang et al. used direct-current plasma magnetron sputtering to deposit an ultralow loading of Pt onto self-standing CoFe@NCNT electrodes, achieving uniform coverage along the nanotube array and excellent trifunctional activity7 (Fig. 7a). Across these examples, the role of the plasma is not limited to “putting metal onto a support”; it prepares specific chemical and topological anchoring sites, suppresses agglomeration and constructs extended metal–support contact networks, so that noble metals are wired into conductive oxide or carbon scaffolds with minimal dead surface.
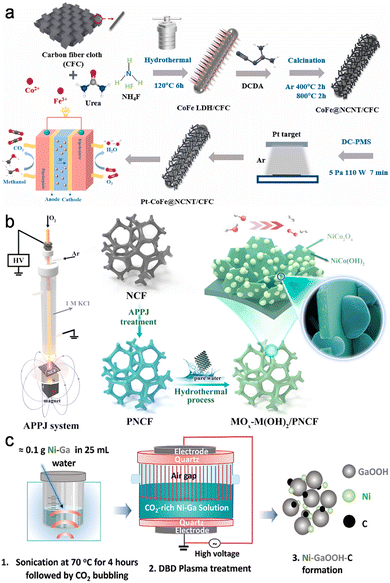 | ||
| Fig. 7 (a) Schematic illustration of the synthesis process of Pt-CoFe@NCNT/CFC electrode. Reproduced with permission.7 Copyright 2022, Elsevier. (b) The schematic illustration for the fabrication of the electrode-emerged nanointerfaced bimetallic NiCo hydroxide–oxide active system. Reproduced with permission.19 Copyright 2021, American Chemical Society. (c) Schematic showing the synthesis of Ni-GaOOH-C catalyst. Reproduced under the terms of CC-BY 4.0 license.24 | ||
Plasmas can also deposit or trigger the growth of oxide shells that are electronically coupled to their underlying metallic or mixed-metal cores. In our group's DMFC work and in related Zn–air systems, short plasma exposures convert the outer layers of transition-metal sulfides, nitrides or carbons into thin, defective oxides while preserving the inner phase. When noble metals or additional functional phases are subsequently deposited, they sit on top of an electronically active oxide interlayer rather than directly on the parent phase. In this stacked structure, the deposited metal, the plasma-formed oxide layer and the conductive core work together to generate internal potential gradients and redox-buffer regions that enhance charge separation and stabilize high-valent states under operating conditions. The interfacial chemistry is again set by the plasma: its radicals and ions determine how deeply oxidation proceeds, which cations are oxidized and how many vacancies or hydroxyl groups remain to bind the next layer.
Another group of studies uses plasmas to fabricate or modify non-noble catalytic films and their junctions with light absorbers or conductive backbones. Benedet et al. employed PECVD to grow three-dimensional, flower-like MnO2 nanoarchitectures on Ni foam and then coupled them with g-C3N4via electrophoretic deposition (EPD), forming an extended oxide/semiconductor junction for photo-assisted OER. The plasma controls both the Mn oxidation state and the film porosity, ensuring that the g-C3N4 flakes contact a defect-rich MnO2 surface rather than a compact, poorly hydrated oxide.66 Chen et al. developed a “plasma-triggered growth” route to fabricate NiCo hydroxide–oxide nanointerfaces directly on nickel foam19 (Fig. 7b). In this case, the plasma first oxidizes and roughens the metal surface; exposure to water then drives spontaneous growth of intertwined NiCo(OH)x/NiCo2O4 domains. The resulting interpenetrating hydroxide/oxide network exhibits fast redox kinetics and enhanced charge storage, highlighting how plasma-triggered surface chemistry can be harnessed to grow nanointerfaces directly from a current collector without separate precursors or binders. They first applied a magnetically focused atmospheric-pressure plasma jet in an Ar/O2 mixture to locally activate the NiCo foam surface. Subsequent immersion/hydrothermal treatment in pure water without added reagents then drives the electrode to “sprout” intertwined NiCo(OH)x nanosheets that are densely decorated with strongly bonded NiCo oxide nanoparticles, yielding an interpenetrating hydroxide/oxide network with fast redox kinetics and robust high-current overall water-splitting performance.
Babikir et al. extended this interfacial thinking to liquid-metal-derived Ni–Ga system, where a dielectric barrier discharge plasma reduces dissolved CO2 into highly dispersed activated carbon that deposits directly onto in situ-formed Ni–GaOOH particles24 (Fig. 7c). The resulting material is not merely “Ni–GaOOH plus carbon” but a tightly intergrown Ni–O–C region containing metallic Ni, NiOx and graphitic carbon domains. DFT shows that the Ni–C and NiOx–C interfaces significantly lower the free-energy barriers for nitrate reduction to ammonia relative to either Ni or NiO alone, indicating that the plasma-generated carbon phase constructs the true ensemble of active interfacial sites. Deas et al. demonstrated a related concept for WO3−x films produced by aerosol-assisted atmospheric-pressure plasma jets: pre-formed oxide colloids are assembled into hierarchical, porous layers whose internal grain boundaries, necks and pore walls are fused by the plasma.67 Even without additional metals, the resulting network has a high density of electrically connected internal interfaces that facilitate charge percolation and ion access, underscoring that “deposition” in a plasma context often means welding together a three-dimensional interfacial scaffold rather than laying down a flat film.
At still thinner length scales, PEALD offers atomic-level control over interfacial composition and wetting. Hoek et al. compared plasma-enhanced and purely thermal ALD of In2S3 thin films on gas-diffusion electrodes for CO2 electroreduction.68 The plasma process produces films with different surface terminations, defect densities and wettability, which in turn tune the gas–liquid–solid triple-phase boundary and stabilize high formate partial current densities. In a similar research, Li et al. used plasma to engineer Pt–O–W interfaces on tungsten-based supports at extremely low Pt loadings.69 The plasma not only reduces Pt precursors but also creates oxygen-bridged Pt–O–W linkages at the contact region, modifying the local electronic structure so that hydrogen adsorption and desorption kinetics are optimized for HER. In both cases, the key advantage of the plasma is its ability to set the chemical identity and polarity of just a few atomic layers at the interface – whether by controlling ligand terminations in ALD cycles or by forming specific metal–oxygen–metal bridges – while leaving the bulk of the support untouched.
Taken together, these deposition and interfacial studies show that non-thermal plasmas are effective tools for producing catalytic junctions directly onto three-dimensional scaffolds. Noble metals can be anchored as ultrasmall particles at plasma-generated defect sites on oxides or carbons; oxide and oxyhydroxide shells can be grown from plasma-activated surfaces; carbon or chalcogenide domains can be nucleated in situ on oxide backbones; and atomic-scale junctions can be defined through PEALD or plasma-driven ligand chemistry. In all of these cases, the active region is not a single phase, but a thin, chemically complex interfacial zone where electronic coupling, local fields and wetting properties are all set by the preceding plasma exposure. Because these modifications are confined to the outer few nanometers, the underlying nanoarchitecture continues to provide mechanical support and long-range charge and mass transport. When viewed alongside the phase and defect engineering routes discussed earlier, plasma-assisted deposition and interfacial design complete a coherent picture in which low-temperature plasmas are used to sculpt not only what phases are present, but also how they touch and communicate, thereby controlling where the true catalytic work is done.
2.5. Single atom anchoring and strong interactions
In the previous sections we have seen how plasmas can reshape phases, inject defects, introduce heteroatoms and sculpt interfaces. A natural end point of this evolution is the regime where these structural motifs no longer act independently but instead lock into one another through strong electronic interactions and atomically precise coordination. In this regime, the catalytically relevant unit is no longer a “particle on a support” but a coupled ensemble of vacancies, single atoms, clusters and oxide frameworks whose behavior is defined by interfacial charge redistribution. This section addresses plasma strategies that stabilize atomically dispersed metals or strongly coupled metal support contacts, supported by single atom evidence from HAADF STEM or EXAFS and by clear interfacial charge transfer or enhanced stability under operation. Relative to conventional impregnation or high-temperature anchoring strategies, non-thermal plasma is particularly effective at creating surface-confined anchoring sites and strong metal–support interactions, stabilizing single atoms and interfacial charge transfer motifs under electrochemical operating conditions.A recurring theme across several systems is that plasma first creates or reorganizes anion vacancies, which then serve as chemically selective anchoring sites for isolated metals. In the Pt/TiO2−x single-atom catalyst reported by Tian et al., alternate plasma sputtering was used to deposit Pt atoms on a pre-deposited TiO2−x support5 (Fig. 8a). The TiO2−x support contained a low concentration of Ti3+ and oxygen vacancies, which, together with the Pt–O coordination, helped stabilize Pt in a linear O–Pt–O configuration, prevent aggregation, and enhance electron transfer. This resulted in a catalyst with ultra-low Pt loading (1.69 wt%) that outperformed commercial Pt/C in mass activity and TOF for the HER. A similar vacancy-templated strategy underlies the Pt–Co–Se coordinated sites in CoSe2 described by Zhuang et al.: Ar plasma generates Se vacancies that, during subsequent photochemical reduction, guide Pt into well-defined Pt–Co–Se configurations with highly asymmetric charge distribution and lowered barriers for key OER intermediates18 (Fig. 8b). In a more complex perovskite host, Han et al. showed that plasma-enhanced CVD simultaneously induces A-site deficiency in LaFeO3 and drives Ru incorporation, yielding a mixed population of Ru single atoms and Ru clusters embedded in the lattice.70 Operando analysis indicates that these two Ru species communicate through the perovskite oxygen network, jointly sustaining high N2 reduction rates. In all three cases, the plasma step does more than “activate” a surface: it writes a vacancy landscape that dictates where and how single atoms and clusters are stabilized, and how they subsequently talk to the support.
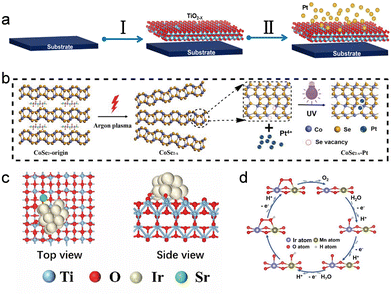 | ||
| Fig. 8 (a) Schematic illustration of the synthesis process of the Pt/TiO2−x catalyst. Reproduced with permission.5 Copyright 2021, Elsevier. (b) Schematic of the Ar-plasma-induced exfoliation of CoSe2-origin. Reproduced with permission.18 Copyright 2018, Wiley-VCH. (c) Top and side views of Ir@Sr-d-TiO2(004). Reproduced with permission.22 Copyright 2024, Wiley-VCH. (d) OER process by the IMO@p-ATO through OPM. Reproduced with permission.33 Copyright 2025, Wiley-VCH. | ||
Once these anchoring environments are in place, the interaction between metal and host often enters the strong metal–support interaction (SMSI) regime, in which charge transfer and orbital hybridization become as important as geometric confinement. A mechanistic template for such behavior is provided by Pt/NbOx in the work of Jia et al., where the Pt–NbOx interface switches between Pt–O and Pt–Nb coordination depending on oxygen coverage, yet in all cases electrons flow from Pt into the oxide.71 This electron depletion stabilizes low-coordinated Pt sites, contracts Pt–Pt bonds and suppresses dissolution under ORR conditions. Plasma-engineered systems map naturally onto this picture. In the Ir/TiO2 nanowire catalyst of Zhu et al., H2/N2 plasma first generates oxygen-vacancy-rich TiO2, which tightly anchors Ir nanoparticles; the vacancy-rich skin also enforces strong interfacial electronic coupling, so that Ir is stabilized by a more polar/ionic metal–oxide contact rather than weak physisorption22 (Fig. 8c). These VO sites further act as preferred adsorption/coordination traps for the Sr promoter, making the subsequent Sr single-atom introduction spatially correlated with the Ir–oxide contact region. Sr single atoms are then introduced at or near these vacancies. The resulting Ir–Sr–TiO2 triple interface shows pronounced charge redistribution in theory, stabilizes high-valent Ir–O intermediates in experiment, and dramatically reduces Ir loss during acidic OER/HER and PEM electrolysis.
In a carbon-based analogue, Yi et al. used N2 plasma to introduce pyridinic and graphitic N into graphene, which then form Ir–N coordination shells around ultra-small Ir particles.72 The associated negative shift in Ir binding energy and enhanced durability point again to a SMSI-like electronic coupling: the plasma-modified support donates and redistributes charge in a way that optimizes adsorbate binding and protects the noble metal.
These material-specific examples can be viewed through a broader conceptual lens. Zhou et al. has argued that in oxide electrocatalysts, strong oxide–support interactions (SOSI) and defect–interface coupling drive interfacial electronic reconstruction: shifting the O 2p band center, modulating M–O covalency and steering the balance between adsorbate evolution mechanisms and lattice oxygen pathways33 (Fig. 8d). Vacancy-rich sub-oxide skins, heteroatom-decorated carbon shells and perovskite lattices populated by embedded clusters and single atoms all fit naturally into this SOSI/SMSI framework. What plasma processing adds is a kinetic shortcut to such states: it can simultaneously create vacancies, reduce or partially oxidize metals, and install single-atom promoters in the same near-surface region, thereby collapsing multi-step wet-chemical protocols into a single, controllable interface-writing operation.
Taken together, the systems illustrate how non-thermal plasmas can be used not only to disperse metals, but to sculpt the interaction landscape around them. By designing vacancy types and densities, choosing supports capable of SOSI/SMSI behavior, and judiciously adding single-atom promoters, it becomes possible to assemble interfacial motifs in which geometry, valence and reaction pathway are co-determined. This perspective suggests that future progress in plasma-assisted electrocatalysis will depend less on pushing any single lever – phase, defect, doping or interface – in isolation, and more on deliberately integrating the strong, atom-scale interactions that emerge when all of these levers are pulled together.
3. Conclusions
The design logic of electrocatalysts with non-thermal plasma is straightforward. One first selects a scaffold that is worth preserving – oxide nanowires, MOF-derived carbons, carbon textiles, or pre-grown chalcogenide sheets – so that the bulk architecture continues to provide electronic and mass-transport pathways. The plasma discharge is then used to place an appropriate phase at the surface: phosphides to combine metallic conduction with optimized H* binding, nitrides to provide conductivity and charge-transfer support, and vacancy-rich chalcogenides or sub-oxides to tune the chemistry of oxygen intermediates. The conversion layer is kept thin so that internal interfaces are dense while transport through the backbone remains fast. When precious metals are required, plasma activation can be exploited to generate anchoring defects, followed by a brief plasma reduction or sputtering step to control dispersion and oxidation state, thereby improving utilization and stability. Where strong interactions are desired, defect-rich skins are combined with subsequent metal loading or single-atom decoration so that charge transfer and coordination environments are established from the outset and preserved under electrochemical load.Equally important are the limits and common pitfalls. Not every system benefit from deeper penetration; beyond the first few nanometers, gains in active area are often offset by increased resistance, poorer wetting, or accumulated mechanical stress. Morphology collapse can occur if gas heating or exposure times are excessive, and is best avoided by modest power, short treatment steps and, when necessary, cooling flows. Non-thermal plasma processing is inherently surface-confined and therefore not suited for bulk modification, and its effectiveness depends on well-defined process windows. As such, plasma should be regarded as a complementary interface-engineering tool rather than a universal substitute for conventional materials processing methods. Robust recipes arise when process variables are explicitly correlated with surface composition and structure, and these in turn are linked to kinetic response, allowing those relationships to be condensed into simple and reproducible operating windows. To support this point, Table 1 was provided that collates representative plasma parameters, structural fingerprints and catalytic metrics, helping readers identify reproducible processing windows across systems.
Looking ahead, the most important opportunities lie in tightening the loop between plasma physics, surface structure and electrochemical function. Operando spectroscopy and microscopy, coupled with realistic modelling of non-thermal discharges at catalyst interfaces, will be essential to follow how plasma-written phases, defects and strong interactions evolve under current and potential. At the same time, translating these ideas beyond model electrodes to porous gas-diffusion layers, membrane–electrode assemblies and large-area electrodes will require closer integration of reactor design, diagnostics and device testing. If these threads can be woven together, non-thermal plasmas should move from being occasional activation steps to becoming genuinely predictive tools for constructing robust, low-PGM electrodes, with the active chemistry written exactly where the electrochemical work is done.
Author contributions
Chu Qin: conceptualization; investigation; visualization; writing – original draft; writing – review and editing. Zhenglong Tao: investigation; data curation; resources; writing – review and editing. Xuanhao Wu: supervision; writing – review and editing. Zhongqing Jiang: supervision; project administration; writing – review and editing.Conflicts of interest
There are no conflicts to declare.Data availability
No primary research results, software or code have been included, and no new data were generated or analyzed as part of this review.Acknowledgements
Authors acknowledge the supports from the National Natural Science Foundation of China (No. 12505288, 12275239, and 52400098), the Zhejiang Provincial Natural Science Foundation (No. QN26A050005, LR22E070001, and LZ25B070003).Notes and references
- Z. Xu, J. Wu, W. Chen, Z. Jiang, J. Cao, G. Chen and Z.-J. Jiang, Adv. Funct. Mater., 2025, e11117 CrossRef.
- L. Xu, Q. Jiang, Z. Xiao, X. Li, J. Huo, S. Wang and L. Dai, Angew. Chem., Int. Ed., 2016, 55, 5277–5281 CrossRef PubMed.
- D. Zha, R. Wang, S. Tian, Z.-J. Jiang, Z. Xu, C. Qin, X. Tian and Z. Jiang, Nano-Micro Lett., 2024, 16, 250 CrossRef PubMed.
- C. Li, B. Ye, B. Ouyang, T. Zhang, T. Tang, Z. Qiu, S. Li, Y. Li, R. Chen, W. Wen, M. Song, B. Mei, X. Xia and Y. Zhang, Adv. Mater., 2025, 37, 2501381 CrossRef PubMed.
- Y. Tian, L. Yu, C. Zhuang, G. Zhang and S. Sun, Mater. Today Energy, 2021, 22, 100877 CrossRef.
- L. Wang, J. Qi, Y. Zhang, Y. Dai, K. Bao, W. Wang, J. Wu, C. Ma, Z. Yin, C. Ma, Y. Chen, J. Bao, R. Ye, Y. Liu, Z. Lin, Z. Wang and Q. He, Adv. Mater., 2025, 37, 2502047 CrossRef PubMed.
- W. Wang, Z. Jiang, X. Tian, T. Maiyalagan and Z.-J. Jiang, Carbon, 2023, 201, 1068–1080 CrossRef.
- J. Zheng, H. Zhang, J. Lv, M. Zhang, J. Wan, N. Gerrits, A. Wu, B. Lan, W. Wang, S. Wang, X. Tu, A. Bogaerts and X. Li, J. Am. Chem. Soc. Au, 2023, 3, 1328–1336 Search PubMed.
- J. Huang, H. Sheng, R. D. Ross, J. Han, X. Wang, B. Song and S. Jin, Nat. Commun., 2021, 12, 3036 CrossRef PubMed.
- H. Liang, A. N. Gandi, C. Xia, M. N. Hedhili, D. H. Anjum, U. Schwingenschlögl and H. N. Alshareef, ACS Energy Lett., 2017, 2, 1035–1042 CrossRef.
- Z. Shao, Q. Zhu, Y. Sun, Y. Zhang, Y. Jiang, S. Deng, W. Zhang, K. Huang and S. Feng, Adv. Mater., 2022, 34, 2110172 CrossRef PubMed.
- X. Wu, X. Wang, Y. Wu, H. Xu, Z. Li, R. Hong, K. Rigby, Z. Wu and J.-H. Kim, Nat. Commun., 2025, 16, 1122 CrossRef PubMed.
- L. Zhang, L. Zhuang, H. Liu, L. Zhang, R. Cai, N. Chen, X. Yang, Z. Zhu, D. Yang and X. Yao, Small Science, 2021, 1, 2000027 CrossRef PubMed.
- B. Ouyang, Y. Zhang, X. Wang, Y. Deng, F. Liu, Z. Fang, R. S. Rawat and E. Kan, Small, 2022, 18, 2204634 CrossRef PubMed.
- K. E. Salem, A. A. Saleh, G. E. Khedr, B. S. Shaheen and N. K. Allam, Energy Environ. Mater., 2023, 6, e12324 CrossRef.
- S. Li, X. Zhao, R. Gang, B. Cao and H. Wang, Anal. Chem., 2020, 92, 5152–5157 CrossRef PubMed.
- H. Han, S. Jin, S. Park, Y. Kim, D. Jang, M. H. Seo and W. B. Kim, Nano Energy, 2021, 79, 105492 CrossRef.
- L. Zhuang, Y. Jia, H. Liu, X. Wang, R. K. Hocking, H. Liu, J. Chen, L. Ge, L. Zhang, M. Li, C.-L. Dong, Y.-C. Huang, S. Shen, D. Yang, Z. Zhu and X. Yao, Adv. Mater., 2019, 31, 1805581 CrossRef PubMed.
- G. Chen, D. Chen, J. Huang, C. Zhang, W. Chen, T. Li, B. Huang, T. Shao, J. Li and K. K. Ostrikov, ACS Appl. Mater. Interfaces, 2021, 13, 45566–45577 CrossRef PubMed.
- X. Li, H. Jiang, C. Ma, Z. Zhu, X. Song, H. Wang, P. Huo and X. Li, Appl. Catal., B, 2021, 283, 119638 CrossRef.
- L. Ye, W. Chen, Z. J. Jiang and Z. Jiang, Carbon Energy, 2024, 6, e457 CrossRef.
- H. Zhu, Y. Wang, Z. Jiang, B. Deng, Y. Xin and Z.-J. Jiang, Adv. Energy Mater., 2024, 14, 2303987 CrossRef.
- Y. Xiong, Z. Jiang, L. Gong, X. Tian, C. Song, T. Maiyalagan and Z.-J. Jiang, J. Colloid Interface Sci., 2023, 649, 36–48 CrossRef CAS PubMed.
- A. H. Babikir, X. Mao, A. Du, J. D. Riches, K. Ostrikov and A. P. O'Mullane, Small, 2024, 20, 2311302 CrossRef PubMed.
- B. Ouyang, S. Peng, H. Qin, Y. Du, J. Xu, Y. Cao, E. Kan and R. S. Rawat, Appl. Catal., B, 2025, 366, 125061 CrossRef.
- K. Huang, D. Peng, Z. Yao, J. Xia, B. Zhang, H. Liu, Z. Chen, F. Wu, J. Wu and Y. Huang, Chem. Eng. J., 2021, 425, 131533 CrossRef.
- X. Yan, L. Zhuang, Z. Zhu and X. Yao, Nanoscale, 2021, 13, 3327–3345 RSC.
- M. Li, X. Wang, K. Liu, H. Sun, D. Sun, K. Huang, Y. Tang, W. Xing, H. Li and G. Fu, Adv. Mater., 2023, 35, 2302462 CrossRef PubMed.
- L. Gong, Z. Jiang, Y. Xiong, X. Tian and Z.-J. Jiang, Int. J. Hydrogen Energy, 2023, 48, 32860–32874 CrossRef.
- H. Song, J. Yu, Z. Tang, B. Yang and S. Lu, Adv. Energy Mater., 2022, 12, 2102573 CrossRef.
- J. Fan, L. K. Arrazolo, J. Du, H. Xu, S. Fang, Y. Liu, Z. Wu, J.-H. Kim and X. Wu, Environ. Sci. Technol., 2024, 58, 12823–12845 CrossRef PubMed.
- C. Qin, S. Tian, J. Wu, J. Mou, L. Feng and Z. Jiang, ChemCatChem, 2024, 16, e202401033 CrossRef.
- K. Zhou, Y. Wang, Z. Jiang, B. Dai and Z.-J. Jiang, Adv. Mater., 2025, 37, 2420159 CrossRef PubMed.
- J. Ai, N. Wang, F. Li, W. He, Z. Qu, X. Ma, L. Huang and Q. Pu, Appl. Surf. Sci., 2025, 688, 162448 CrossRef.
- K. Fan, H. Zou, Y. Lu, H. Chen, F. Li, J. Liu, L. Sun, L. Tong, M. F. Toney, M. Sui and J. Yu, ACS Nano, 2018, 12, 12369–12379 Search PubMed.
- M. Zhang, Q. Dai, H. Zheng, M. Chen and L. Dai, Adv. Mater., 2018, 30, 1705431 CrossRef PubMed.
- X. Wang, L. R. Winter, X. Wu, Y. Fan, Y. Zhao, J.-H. Kim and M. Elimelech, Sci. Adv., 2025, 11, eads6943 CrossRef PubMed.
- L. Li, X. Tang, B. Wu, B. Huang, K. Yuan and Y. Chen, Adv. Mater., 2024, 36, 2308326 CrossRef PubMed.
- K. Chen, S. Kim, M. Je, H. Choi, Z. Shi, N. Vladimir, K. H. Kim and O. L. Li, Nano-Micro Lett., 2021, 13, 60 CrossRef PubMed.
- J.-Y. Kim, M.-J. Jin, B. Hou, M. P. Kim, D.-S. Um and C.-I. Kim, Appl. Surf. Sci., 2023, 639, 158271 CrossRef.
- T. Zhang, J. Wu, J. Chen, Q. Pan, X. Wang, H. Zhong, R. Tao, J. Yan, Y. Hu, X. Ye, C. Chen and J. Chen, ACS Appl. Mater. Interfaces, 2021, 13, 24682–24691 CrossRef PubMed.
- S. Song, M. Guo, S. Zhang, K. Zhan, Y. Yan, J. Yang, B. Zhao and M. Xu, Electrochim. Acta, 2020, 331, 135431 CrossRef.
- R. Wang, Z. Guo, X. Tan, J. Zhang, L. Yang, W. Wang, L. Cao and B. Dong, Int. J. Hydrogen Energy, 2021, 46, 32425–32434 CrossRef.
- F. Yu, M. Liu, C. Ma, L. Di, B. Dai and L. Zhang, Nanomaterials, 2019, 9, 1436 CrossRef PubMed.
- G. Li, X. Wu, H. Guo, Y. Guo, H. Chen, Y. Wu, J. Zheng and X. Li, ACS Appl. Mater. Interfaces, 2020, 12, 5951–5957 CrossRef PubMed.
- Y. Liu, Z. Jiang and Z.-J. Jiang, Adv. Funct. Mater., 2023, 33, 2302883 CrossRef.
- H. Liu, S. Jiang, J. Li, W. Chen, Z.-J. Jiang, X. Tian and Z. Jiang, Small, 2025, 21, 2504987 CrossRef PubMed.
- Y. Wei, L. Yi, S. Zhang, C. Ni, X. Cai, W. Sun and W. Hu, J. Mater. Chem. A, 2024, 12, 8534–8542 RSC.
- Z. Fu, Z. Jiang, T. Hu and Z.-J. Jiang, Electrochim. Acta, 2022, 419, 140392 CrossRef CAS.
- B. Zhang, Z. Jiang, X. Shang, S. Li and Z.-J. Jiang, J. Mater. Chem. A, 2021, 9, 25934–25943 RSC.
- J. Zhang, W. Nie, R. Wang, C. Qin, Z.-J. Jiang, G.-R. Jin and Z. Jiang, J. Alloys Compd., 2024, 1004, 175900 CrossRef CAS.
- J. Mallmann, J.-B. Chemin, D. Cardenas Morcoso, A.-M. Philippe, S. Bulou, N. Chaabane, F. Rouillard, P. Choquet and N. D. Boscher, ACS Appl. Energy Mater., 2025, 8, 7038–7051 Search PubMed.
- G. Yang, H. Xiang, M. Rauf, H. Mi, X. Ren, P. Zhang and Y. Li, J. Power Sources, 2021, 481, 228925 CrossRef CAS.
- H. Liang, Z. Cao, C. Xia, F. Ming, W. Zhang, A.-H. Emwas, L. Cavallo and H. N. Alshareef, CCS Chem., 2021, 3, 1553–1561 CrossRef CAS.
- B. Zhang, X. Shang, Z. Jiang, C. Song, T. Maiyalagan and Z.-J. Jiang, ACS Appl. Energy Mater., 2021, 4, 5059–5069 CrossRef CAS.
- T. Hu, Z. Jiang, Z. Fu and Z.-J. Jiang, J. Mater. Chem. A, 2022, 10, 8739–8750 RSC.
- Y. e Feng, W. Chen, L. Zhao, Z. J. Jiang, X. Tian and Z. Jiang, Small Methods, 2024, 8, 2400565 CrossRef CAS PubMed.
- J. Zheng, X. Peng and Z. Wang, Phys. Chem. Chem. Phys., 2021, 23, 6591–6599 RSC.
- Y. Zhang, Z. Zhu, X. Guo, J. Qi and X. Li, Chem. Eng. J., 2023, 452, 139230 Search PubMed.
- K. Huang, Y. Yan, Y. Yu, T. Yang, L. Qiao, J. Tu, J. Sui, W. Cai, S. Liu and X. Zheng, EES Catal., 2025, 3, 1044–1054 RSC.
- Y.-C. Lin, C.-H. Chuang, L.-Y. Hsiao, M.-H. Yeh and K.-C. Ho, ACS Appl. Mater. Interfaces, 2020, 12, 42634–42643 CrossRef CAS PubMed.
- L. Zhao, J. Zhang, G. Jin, Z.-J. Jiang and Z. Jiang, J. Colloid Interface Sci., 2024, 668, 525–539 CrossRef CAS PubMed.
- H. Li, L. Hansen, A. Aliyeva, J. Wang, H. Qiu, M. Müller, S. Chen, C. Aktas, L. Kienle, B. Hartke and J. Benedikt, Appl. Catal., B, 2025, 361, 124607 CrossRef CAS.
- D. Zha, S. Jiang, Q. Zhang, J. Li, Z.-J. Jiang, C. Qin, X. Tian, T. Maiyalagan and Z. Jiang, Chem. Eng. J., 2025, 522, 166892 CrossRef CAS.
- T. Hu, W. Chen, Y. Liu, L. Gong, Z. Jiang, D. Bhalothia, T. Maiyalagan and Z.-J. Jiang, Small, 2023, 19, 2304076 CrossRef CAS PubMed.
- M. Benedet, A. Gallo, C. Maccato, G. A. Rizzi, D. Barreca, O. I. Lebedev, E. Modin, R. McGlynn, D. Mariotti and A. Gasparotto, ACS Appl. Mater. Interfaces, 2023, 15, 47368–47380 CrossRef CAS PubMed.
- R. Deas, T. Lolohea, A. Chan, G. I. N. Waterhouse, G. Henshaw, D. J. McGillivray and D. E. Williams, ACS Appl. Nano Mater., 2023, 6, 5613–5622 CrossRef CAS.
- J. Van den Hoek, F. Mathew, L. Hintjens, B. De Coen, E. Solano, M. M. Minjauw, R. Blomme, N. Daems, D. Choukroun, C. Detavernier, J. Dendooven and T. Breugelmans, Adv. Energy Mater., 2025, 15, 2404178 CrossRef.
- J. Li, Q. Zhang, H. Liu, Z.-J. Jiang, X. Tian and Z. Jiang, Appl. Phys. Lett., 2025, 127, 203903 CrossRef.
- Z. Han, D. Tranca, F. Rodríguez-Hernández, K. Jiang, J. Zhang, M. He, F. Wang, S. Han, P. Wu and X. Zhuang, Small, 2023, 19, 2208102 CrossRef CAS PubMed.
- Q. Jia, S. Ghoshal, J. Li, W. Liang, G. Meng, H. Che, S. Zhang, Z.-F. Ma and S. Mukerjee, J. Am. Chem. Soc., 2017, 139, 7893–7903 CrossRef PubMed.
- L. Yi, B. Feng, N. Chen, W. Li, J. Li, C. Fang, Y. Yao and W. Hu, Chem. Eng. J., 2021, 415, 129034 CrossRef.
| This journal is © The Royal Society of Chemistry 2026 |