Greener and scalable MXene fabrication enabled by supercritical CO2: a mini-review
Süleyman Gökhan
Çolak
 *a,
Utku Bulut
Simsek
*a,
Utku Bulut
Simsek
 *b,
Ahmet
Güngör
*b,
Ahmet
Güngör
 cde and
Melis Özge
Alaş Çolak
cde and
Melis Özge
Alaş Çolak
 f
f
aDepartment of Biomedical Engineering, Faculty of Engineering and Natural Sciences, Iskenderun Technical University, Hatay, Turkey. E-mail: sgokhan.colak@iste.edu.tr
bDepartment of Machine Anadolu BIL Vocational School, Istanbul Aydin University, Istanbul, TR-34295, Turkey. E-mail: utkubulutsimsek@aydin.edu.tr
cDepartment of Chemical Engineering, Kocaeli University, Kabaoglu, Izmit, 41001 Kocaeli, Turkey. E-mail: ahmet.gungor@kocaeli.edu.tr
dFaculty of Engineering and Natural Sciences, Sabanci University, Istanbul, TR-34956, Turkey. E-mail: ahmet.gungor@sabanciuniv.edu
eCenter of Excellence for Functional Surfaces and Interfaces for Nano-Diagnostics (EFSUN), Sabancı University, Orhanlı, Tuzla 34956, Istanbul, Turkey
fDepartment of Chemical Engineering, Faculty of Engineering, Mersin University, Mersin, Turkey. E-mail: melis_alas@hotmail.com
First published on 23rd December 2025
Abstract
Supercritical CO2 (scCO2) has a gas-like diffusivity and a liquid-like density, which speeds up transport and allows for tunable termination chemistry during MXene synthesis. This mini review compares scCO2-assisted routes to HF and LiF–HCl systems in terms of time to conversion, monolayer yield, oxidation stability, and readiness for scale-up. Optimized scCO2 protocols show a conversion time of about 0.5 to 3 hours (compared to 12 to 72 hours), a mono/few-layer yield of more than 60%, and better air stability, with kilogram-scale batches presented. We evaluate the effects of pressure, temperature, water activity, and co-solvent polarity, and we demonstrate how RESS blow-down and in situ spectroscopy can speed up the process. Application snapshots (supercapacitors, Li/Na-ion batteries, HER, EMI) demonstrate consistent improvements due to termination control and diminished restacking. There are still route-specific limits, comprising narrow p–T windows, batch-to-batch termination scatter, and compression/heat duties that set techno-economics. We suggest a reporting checklist that includes PMI, E-factor, specific energy, CO2 intensity, and water use. We also describe hybrid scCO2-electrochemical/microwave pathways and closed-loop engineering that are needed to connect the promise of lab-scale research with real-world use. When looked at with clear, metrics-based criteria, scCO2 is a safer, greener, and more scalable way to make MXene.
1. Introduction
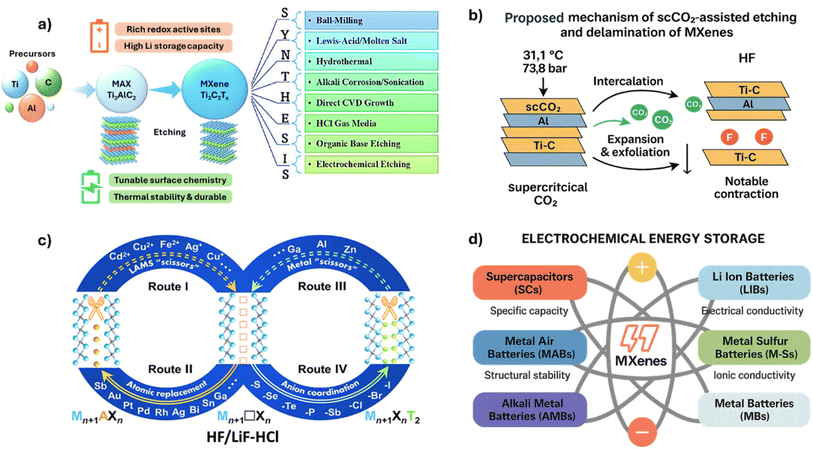
MXenes are two-dimensional (2D) materials, first discovered in 2011 as Ti3C2, and have rapidly emerged as a major research topic in materials science owing to their remarkable electrical, mechanical, and surface-dependent properties.1 These materials follow the general formula Mn+1XnTx, where M represents an early transition metal (e.g., Ti, V, Zr, Hf, Mo, Nb, Cr), X denotes carbon and/or nitrogen, and Tx corresponds to surface terminations such as –O, –OH, –F, or –Cl.2 Here, “n” typically ranges from 1 to 3, defining the number of X atomic layers sandwiched between M layers. Their parent compounds, MAX phases, are described by Mn+1AXn, where A represents elements from groups 13 or 14 (e.g., Al, Ga, or Si). Selective removal of the A element yields MXenes with surface terminations that stabilize the 2D layered structure.3 The process simultaneously introduces surface terminations (–O, –F, –OH) while retaining the layered framework, resulting in materials that combine high electrical conductivity, hydrophilicity, and mechanical robustness key attributes for applications in energy storage, catalysis, and sensing.MXene synthesis is conventionally carried out by removal of the ‘A’ layer (such as Al, Ga, Si) from MAX phases using corrosive chemicals. Hydrofluoric acid (HF) or lithium fluoride (LiF) and hydrochloric acid (HCl) combination (the mild method LiF + HCl) are commonly used for this purpose.4 Fluoride ions (F−) chemically bond with the A element in the MAX phase during HF etching. This breaks M–A bonds (for example, Ti–Al) by moving electrons. These bonds are ruptured via oxidation of Al atoms (Al0 → Al3+) and formation of Al–F bonds with F− ions.
However, these traditional methods suffer from two critical limitations:
• Safety and environmental hazards: utilization of dangerous and corrosive chemicals such as HF poses serious safety risks to scientists and raises concerns regarding environmental sustainability.
• Control challenges: these approaches suppress the accurate control of MXene surface chemistry and interlayer spacing; the corrosive nature of HF may cause random adsorption of functional groups such as F− and OH− ions on the surface along with structural defects.5 Such uncontrolled termination profiles can adversely impact the electrochemical activity and stability of MXenes.
To overcome these limitations, the use of supercritical CO2 (scCO2) offers a green and safer alternative.6 scCO2 enhances penetration of nanomaterials by reducing the liquid–gas surface and enables residue-free processing due to its complete evaporation upon depressurization. This medium also facilitates mechanical exfoliation and delamination of MAX phases under environmentally benign conditions. Therefore, scCO2-assisted synthesis has emerged as a sustainable alternative for both etching and delamination stages, effectively addressing the safety and control challenges of HF-based methods.
scCO2 exists above its critical temperature (31.1 °C) and pressure (7.38 MPa), where it exhibits both gas-like diffusivity (∼10−3 cm2 s−1) and liquid-like density, enabling superior mass transport and solvation behavior.7 These combined properties facilitate enhanced exfoliation, controlled particle size, and high material purity while maintaining a nontoxic and inert reaction medium. In this specific phase, scCO2 has gas-like behavior, with low viscosity and a high diffusion rate of around 10−3 cm2 s−1. It offers better benefits in material synthesis due to liquid-like density and solvent character. scCO2 properties can enhance exfoliation (delamination) of layered compounds, regulate particle size, and enhance material purity. The nontoxicity and inertness of CO2 provide a safe and stable synthesis setting.
The role of scCO2 is particularly important in both etching and delamination stages: (i) etching stage: scCO2 offers improved reaction environment control via the capability to introduce corrosive species (e.g., HF) into the MAX phase in a more uniform and diffusive manner. This leads to more homogeneous etching and uniform surface termination distribution (e.g., –F, –O), reducing defect formation.8 (ii) Delamination stage: scCO2 molecules decrease the van der Waals forces and hydrogen bonds between MXene layers, leading to a gradual delamination of the layers. Rapid depressurization techniques (e.g., RESS) further enhance monolayer yield while preserving structural integrity. Overall, scCO2 routes provide better control of surface chemistry and electrochemical performance compared to conventional HF-based syntheses.
The aim of this mini-review is to provide a critical overview of supercritical CO2-enabled MXene synthesis with an emphasis on how process parameters and reactor design translate into performance and sustainability metrics. Rather than offering a generic historical survey of supercritical CO2 processing, we focus specifically on MXenes and consolidate the scattered reports into a process-knob → metric framework. We compare scCO2-assisted routes with HF-, LiF–HCl-, molten-salt-, and electrochemical etching in terms of etching time, mono-/few-layer yield, oxidation stability, safety and waste generation, and scalability. On this basis, we outline a reporting checklist that includes mass- and energy-based green metrics (PMI, E-factor, electricity and heat demand, CO2 intensity, water use) to support future experimental design and techno–economic analysis. This strategy is consistent with the principles of green chemistry and with the practical needs of industrial scale-up.
Despite notable progress in scCO2-enabled MXene synthesis, key challenges remain unresolved. The effects of process variables such as temperature, pressure, and co-solvent identity are not yet fully understood. Furthermore, scaling beyond laboratory conditions is limited by the lack of reproducibility, throughput optimization, and real-time monitoring. Finally, most reports focus on a narrow subset of MAX chemistries, leaving open questions about universality across different M, A, and X combinations.
Finally, most reported protocols focus on a narrow set of MAX chemistries, leaving it unclear whether a single set of conditions can be reliably applied across different M, A, and X element combinations. Addressing these limitations is essential to transition scCO2-based MXene production from proof-of-concept to robust, industrial-scale synthesis. While scCO2 protocols have been reported in primary papers, the literature lacks a focused review on scCO2 routes. Here we consolidate synthesis mechanisms and process windows while coupling them with a metrics-oriented sustainability discussion. Direct studies on MXene synthesis under supercritical CO2 remain limited. To contextualize and rationalize the trends discussed in this review, we therefore integrate mechanistically relevant evidence from supercritical processing (reduced interfacial tension and enhanced mass transfer), CO2-expanded liquids (polarity and water-activity tuning), rapid expansion of supercritical solutions (RESS), and SCF-assisted exfoliation of layered solids.9–13 This triangulated perspective underpins the process-knob → metric framework formalized in Section 3.4.
2. Traditional methods and limitations in MXene synthesis
2.1. Traditional synthesis methods
MXenes are typically produced through chemical processing of layered ceramic structures known as MAX phases. In this process, specific layers within the MAX phases-particularly the A-layers composed of group IIIA or IVA elements-are selectively removed, yielding two-dimensional MXene materials (Fig. 1(a–d)). Since their discovery, MXenes have primarily been derived through selective etching of A-layers from MAX phases, a process that, while versatile, remains chemically intensive compared to emerging green routes. Among commonly utilized approaches, hydrofluoric acid (HF) etching to remove interlayer elements from MAX phases remains the most frequently employed method.3 For example, when a MAX phase comprising titanium, aluminum, and carbon is treated with HF, the aluminum layer is selectively etched away, resulting in a titanium–carbon-based MXene structure. Although HF is commonly preferred as the etchant, alternative and less aggressive routes using lithium fluoride (LiF) combined with hydrochloric acid (HCl) have also been developed. The etching kinetics and subsequent termination evolution (–F, –OH, –O) in both HF and LiF–HCl systems critically determine the conductivity and interlayer spacing of resultant MXenes. | ||
| Fig. 1 The structural rearrangement protocol of MAX phases and MXenes using, which encompasses four primary chemical intercalation mechanisms. | ||
Additionally, a process involving treatment with molten salts at elevated temperatures, specifically using salts like potassium fluoride (KF), lithium fluoride (LiF), and sodium fluoride (NaF), enables selective removal of elements, facilitating MXene production.4
More recently, electrochemical etching has been introduced as a promising fluoride-free alternative, governed by the following representative reactions.14 This route offers improved termination control and reduced environmental burden; however, it requires precise potential regulation to avoid over-oxidation of Ti sites:
| Ti3AlC2 + 3e− + 3Cl− → Ti3C2 + AlCl3 | (1) |
| Ti3C2 + 2OH− − 2e− → Ti3C2(OH)2 | (2) |
| Ti3C2 + 2H2O → Ti3C2(OH)2 + H2 | (3) |
Alternative mechanisms have been proposed to better understand the thermodynamic and kinetic behavior of A-layer removal and MXene formation.
First, the difference in redox potential between Lewis acidic cations and A-site elements causes the formation of non-van der Waals (non-vdW) gaps within MAX phases by the so-called Lewis Acidic Metal Scissors (LAMS). In this process, cations (such as Cu2+) oxidize the weakly bound interlayer atoms within MAX phases, creating vacancies (e.g., Cu2+ + 2e− → Cu; Mn+1Al Xn → Mn+1Xn + Al3+ + 3e−).15
Second, the spontaneous diffusion of metal atoms into these created vacancies leads to the formation of new MAX phases. This reaction occurs by lowering the system's overall chemical energy. For instance, an isomorphic replacement reaction such as Mn+1Xn + Va + Ga → Mn+1GaXn yields novel MAX phase compositions.
Third, surface terminations of multilayer MXenes are removed, and vdW gaps are opened using metal scissors. This mechanism facilitates the formation of terminated MXenes through coordination of oxidized transition metals with anions in vdW gaps. For example, surface terminations can be removed via reactions like Mn+1Xn → Mn+1Xn2+ + 2e− and Cu2+ + 2e− → Cu.
Finally, oxidized transition metals and anions come together in the opened vdW gaps. This process stabilizes structures through interactions between Lewis's base anions (e.g., Cl−, Br−, I−) and cations. Various MXenes with distinct terminations can be synthesized, as exemplified by the reaction “Mn+1Xn2+ + 2T− → Mn+1XnT2”.16
While these mechanistic frameworks offer details about the underlying chemistry of A-layer removal, the safety concerns associated with reagents used in conventional routes remain a major limitation. The chemicals utilized during MXene synthesis, and the byproducts generated from reactions carry several safety risks. Hydrofluoric acid (HF) is highly toxic and corrosive; skin contact can cause severe burns and potentially life-threatening systemic toxicity. Hydrogen gas (H2) produced during the reaction can pose explosion hazards if accumulated in confined spaces. Moreover, uncontrolled evolution of H2 under closed reaction systems can influence etching kinetics and promote undesired surface oxidation. Fluoride-containing byproducts (such as AlF3 and KAlF4) can lead to serious health issues if inhaled or upon skin contact. Acidic solutions, such as HCl, can damage laboratory equipment and pose long-term health risks if proper precautions are not followed. Therefore, appropriate laboratory safety protocols and ventilation systems are mandatory when performing MXene synthesis through such corrosive routes. Given these challenges, it has become imperative to develop safer, fluoride-free, and environmentally sustainable synthesis routes that retain the electrochemical integrity of the resulting MXenes.
2.2. Limitations of conventional methods
We next delineate route-specific limitations that directly arise from these chemistries and processing steps, reserving quantitative benchmarking for Section 3.5 and Table 3. Conventional MXene syntheses exhibit route-specific constraints that affect safety, reproducibility, and downstream performance:(i) Safety and waste management: HF-based and LiF–HCl routes pose corrosive and toxicity risks, producing fluoride-bearing waste streams that require careful neutralization.
(ii) Termination control: process conditions often bias –F/–OH/–O distributions, leading to defect variability and poor batch-to-batch reproducibility. The predominance of –F terminations in HF-etched MXenes hinders electrochemical accessibility and hampers hydrophilicity tuning, thereby affecting energy storage performance.
(iii) Structural uniformity: liquid-phase transport and post-etch treatments (washing, delamination aids) can cause size variability and restacking, degrading conductivity and oxidation stability. Layer restacking and size variability reduce specific surface area, while uncontrolled oxidation leads to TiO2 formation, compromising conductivity.
(iv) Reporting inconsistency: the absence of standardized mass/energy metrics prevents fair cross-route comparisons.
Hence, the pursuit of green, tunable, and scalable synthesis pathways particularly those leveraging supercritical CO2 systems—has become a focal direction in the evolution of MXene chemistry. These challenges underscore the need for greener and more controllable strategies, setting the foundation for the scCO2-based approaches analyzed in the following sections. These limitations collectively justify the exploration of supercritical CO2 as a controllable, safer, and sustainable reaction medium.
3. Supercritical CO2-assisted MXene synthesis
scCO2 presents a promising green alternative to traditional fluoride-based etching methods for MXene synthesis.1,7 By leveraging the unique physicochemical properties of scCO2, such as gas-like diffusivity and liquid-like solvating power, researchers have demonstrated its efficacy in both the etching and delamination stages of MXene production. In this section, we outline scCO2 fundamentals, mechanistic roles in A-layer removal and delamination, and criteria-based advantages over conventional routes (Table 1).| MAX phase | scCO2 conditions | Etchant | Yield | Ref. |
|---|---|---|---|---|
| Ti3AlC2 | 80 °C, 90 bar | HCl + LiF | ∼82% | 3 |
| Ti3AlC2 | 75 °C, 80 bar | HCl + FeF3 | ∼80% | 6 |
| Ti3AlC2 | 60 °C, 100 bar | HCl + FeCl3 | Not reported | 7 |
| Ti3AlC2 | 50–60 °C, 90 bar | NH4HF2 | ∼78% | 8 |
| Ti3AlC2 | 100 °C, 120 bar | HCl + NaF | ∼76% | 17 |
| Ti3AlC2 | 60 °C, 70 bar | HF | ∼79% | 2 |
| Ti3AlC2 | 85 °C, 85 bar | HCl + LiF | ∼81% | 14 |
| Ti3AlC2 | 70 °C, 100 bar | LiF + HCl | ∼83% | 18 |
3.1. Fundamentals of supercritical CO2
Carbon dioxide reaches its supercritical state at a critical temperature of 31.1 °C and a pressure of 7.38 MPa.1 Above this critical point, CO2 molecules exhibit enhanced solvation entropy and compressibility, which can be exploited to regulate interfacial reactions in confined solids. In this state, scCO2 exhibits a combination of liquid-like density and gas-like viscosity and diffusivity, allowing it to penetrate solid matrices with high efficiency.2 Its zero-surface tension and tunable solvating power make it a unique medium for facilitating chemical reactions and material transformations, especially in layered systems like MAX phases. The low toxicity, non-flammability, and environmental benignity of CO2 further support its use in sustainable materials processing. These attributes also suit scale-up under green-chemistry frameworks when closed-loop CO2 handling and energy integration are implemented. These physicochemical attributes make scCO2 an adaptive medium for both the etching and delamination stages of MXene synthesis, as detailed in the following subsections. These distinctive physicochemical traits also enable precise control over diffusion and reaction equilibria during the conversion of MAX to MXene.3.2. Etching mechanism and reaction control
scCO2 (T ≥ 31.1 °C; p ≥ 7.38 MPa) provides high diffusivity and gas-like viscosity with liquid-like density, which accelerates reagent transport within MAX galleries. In HCl/LiF systems, in situ HF forms and dissolves the A-layer while Li+ intercalation widens the interlayer spacing and promotes delamination; early-stage surface terminations (–F, –O, –OH) and defect formation then govern electronic transport in the resulting MXenes.19,20 In mixed CO2-solvent media, transient carbonic-acid buffering tunes proton activity and etching kinetics, while pressure and temperature co-control solubility and diffusion. Operationally, pressure sets scCO2 density and reagent carry-capacity; temperature co-determines diffusivity and termination kinetics; co-solvent polarity and water activity bias –F/–O/–OH ratios and defect densities.19,20 This tunability allows scCO2 to function simultaneously as a reaction medium and transport enhancer—bridging chemical selectivity with kinetic control. These knobs map to measurable outcomes (etching time, delamination quality, oxidation stability, conductivity) reported comparatively in Tables 2 and 4. A general workflow and route map are summarized in Fig. 2a and b.| Parameter | HF etching | LiF–HCl etching | scCO2-assisted method |
|---|---|---|---|
| a Where HF is present (direct or in situ), standard HF precautions remain mandatory; scCO2 mitigates aqueous acid inventory/exposure rather than eliminating hazards. b “Lower” assumes CO2 recovery/recycle and minimized aqueous acid use; absolute values are process specific. c Demonstrated cases report ∼1 kg batches under supercritical conditions within <5 h. | |||
| HF involved | Yes | Yes | Yes |
| Reaction time | 24–72 h | 12–24 h | 0.5–3 h (often ∼1–3 h) |
| Safety | Highly hazardous | Moderate (still HF in situ) | Improved, route-dependent*a |
| Oxidation stability | Low | Moderate | High |
| Environmental impact | High | Medium | Lower with CO2 recycleb |
| Monolayer yield | ∼30–40% | ∼50% | >60% (reported) |
| Scalability | Limited | Medium | High (1 kg in 5 h)*c |
scCO2-assisted protocols exist in both HF-containing (including in situ HF from LiF + HCl) and HF-free variants. Where HF participates, standard HF precautions remain mandatory; our discussion refers to reduced aqueous acid inventory/exposure, not hazard elimination.
Importantly, we distinguish scCO2-enabled HF-containing protocols from fluoride-free alternatives (Lewis-acid molten salts, electrochemical/hydrothermal routes), emphasizing differences in termination chemistry and safety/waste profiles.8 In scCO2-assisted syntheses, A-site removal in MAX phases (e.g., Al in Ti3AlC2) can be affected by co-delivery of mild etchants (dilute HF, HCl, or FeF3·3H2O) under supercritical conditions, leveraging enhanced diffusivity for more uniform A-layer removal and reduced structural damage.3,21 Under optimized conditions, scCO2-assisted etching has been reported to reach >60% conversion within ∼30 min for selected systems >60% yield within ∼30 min, markedly faster than typical liquid-phase protocols (24–72 h) (often tens of hours).7 Mechanistically, scCO2-driven transport/speciation provides practical “knobs” (pressure, temperature, co-solvent composition, water activity) to steer termination populations and defect densities toward target properties.19,20
To avoid confusion, in this mini-review “LiF–HCl” refers to conventional liquid-phase in situ HF etching at ambient pressure, whereas “scCO2-assisted” denotes protocols in which similar fluoride-containing etchants are combined with supercritical CO2 and rapid depressurization is used to promote delamination and solvent removal. In other words, the comparison in Fig. 3a does not imply a fundamentally different fluoride chemistry, but highlights the additional process knobs introduced by the supercritical medium (enhanced mass transport, controllable water activity, and tunable blow-down rate) and their impact on oxidation stability, solvent removal and scalability.
 | ||
| Fig. 3 (a) Performance radar for MXene preparation routes discussed in Section 3.4, comparing HF etching, LiF–HCl etching, and scCO2-assisted methods across five device-relevant axes: time-to-conversion (fast → high), delamination quality, oxidation stability, safety/environmental (safer → high), and scalability. Scores are normalized and qualitative on a 1–5 scale (higher is better): 1 = weak/slow/risky/poor scalability; 3 = moderate; 5 = strong/fast/safer/highly scalable. Values are indicative and route-dependent; see Table 2 for ranges and notes. Here, “LiF–HCl” denotes conventional liquid-phase in situ HF etching at ambient pressure, whereas “scCO2-assisted” refers to protocols in which similar fluoride-containing etchants are combined with supercritical CO2 and rapid depressurization is used to promote delamination and solvent removal. (b) Overview of MXene synthesis pathways: top–down routes (acid etching—HF or in situ HF generation; alkaline/hydrothermal etching; molten-salt etching; electrochemical etching) and bottom–up routes (CVD, template-assisted, and PLD methods). Analog mechanistic evidence from SCF/CXLs/RESS and layered-solid exfoliation that supports the knob → metric mapping is discussed in Section 3.4.9–13 | ||
Fig. 2 compares scCO2-assisted, HF-based, hydrothermal, molten-salt and ball-milling routes, and maps intercalation/delamination regimes referenced in Sections 3.2 and 3.4.7,8 Where HF is present, the role of scCO2 is to shorten and confine effective etching exposure and to facilitate post-treatment removal; it does not obviate HF hazards. Accordingly, throughout the manuscript we use ‘reduced reliance on corrosive acids rather than categorical ‘HF-free’ claims.
3.3. Layer delamination via scCO2
Following etching, scCO2 also plays a vital role in the delamination of multilayer MXene stacks. Through rapid expansion and depressurization (RESS), scCO2 reduces cohesive forces reducing van der Waals forces between layers and facilitates physical separation, yielding monolayer/few-layer nanosheets with high structural integrity and improved dispersibility.1 Because scCO2 is readily vented/recycled, post-processing residues are minimized relative to liquid-phase routes.6Furthermore, mechanical exfoliation assisted by scCO2 circumvents the need for intercalation with organic molecules or ultrasonication, which are widely used in conventional methods but frequently result in fragmentation, restacking, or surface oxidation. Reports indicate preserved electronic pathways consistent with high conductivity after scCO2 delamination.1,6 We adopt a cradle-to-gate boundary (functional unit: 1 kg MXene) for green-metrics reporting (Table 3). Such preservation of electronic pathways is critical for electrochemical performance in energy storage and catalysis applications. We use a cradle-to-gate boundary (functional unit: 1 kg MXene) and provide green metrics when data is accessible. The reporting fields and metric definitions are aggregated in Table 3. Finally, depressurization rate in RESS governs layer separation and lateral size: faster but controlled blow-down minimizes restacking and preserves conductivity, whereas sluggish release reduces delamination quality. Optimizing this depressurization gradient offers a direct handle to tune MXene lateral dimensions and preserve conductivity.
| Category | What to report (minimum) | HF route | LiF–HCl route | scCO2 route |
|---|---|---|---|---|
| a Definitions follow standard PMI/E-factor usage and energy/CO2 accounting; solvent benchmarking uses CHEM21. Where the cited sources do not provide sufficient mass/energy balances, cells remain blank, and the gap is explicitly noted. See Section 3.5 for boundaries and citation keys.29–32 For clarity, we use PMI = Σm(inputs)/m(product), E-factor = Σm(waste)/m(product), specific energy in kWh kg−1, and carbon intensity in kg CO2e kg−1 under a cradle-to-gate, 1 kg functional unit. | ||||
| Mass balance | Total inputs/outputs; PMI (=Σm(inputs)/m(product)); E-factor (=Σm(waste)/m(product)) | + | + | (Report; show with/without CO2-recycle credit) |
| Energy | Total kWh kg−1 (heat vs. electricity separated); heat-integration/recover y described | + | + | (Report; include compression/recirculation duties) |
| Carbon intensity | kg CO2e kg−1 (grid factor or steam source stated; scope/assumptions explicit) | + | + | (Report; clarify recycling credit and leakage assumptions) |
| Solvents & reagents | Identity, amounts; CHEM21 class; recycle fraction (%) and cycles | + | + | (Report; CO2 loop and any co-solvents) |
| Water use | Process water (L kg−1) and wastewater handling | + | + | (Report; state if negligible) |
| Waste | Hazardous/non-hazardous waste (kg kg−1); neutralization/work-up streams | + | + | (Report) |
| Throughput & mode | Batch size (g–kg), kg h−1, batch vs. (semi)continuous; residence time | + | + | (Report; continuous if applicable) |
| Delamination | Coupled vs. decoupled from etching; delamination yield (%) | + | + | (Report) |
| Reproducibility | Number of runs (n); SD/CI; any scale-up demonstration | + | + | (Report) |
| OHS | Fluoride vs. pressure/mechanical risk archetypes; ISD: closed CO2 loop, interlocks, relief devices, MAWP margins | + | + | (Report) |
3.4. Performance comparisons with conventional methods
The integration of scCO2 in both etching and exfoliation offers substantial advantages over conventional fluoride-based methods. Elevated CO2 pressure and temperature lower interfacial tension and accelerate mass transfer, leading to shorter time-to-conversion.9 CO2-expanded liquids provide a handle to tune solvent polarity and water activity, which can favor monolayer/few-layer delamination and improve termination control.10 In addition, the depressurization rate (dp/dt) in RESS-like steps is known to influence nucleation/growth pathways and thus lateral dimensions and delamination quality.11,22,23 These mechanistic links motivate the qualitative scoring along the axes used here. We use four comparative dimensions: (i) time-to-conversion, (ii) delamination quality/monolayer fraction, (iii) oxidation stability, and (iv) indicative specific energy, under a cradle-to-gate framework (functional unit: 1 kg MXene; CO2-recycle credit stated where used). Where primary papers lack complete balances, entries are marked “not reported (nr)” rather than backcalculated. To strengthen depth and traceability, we group routes into HF/HCl–LiF, molten-salt, alkaline/electrochemical, and scCO2-assisted families and report the four axes where available7,8• Processing time: scCO2-assisted methods can complete conversion in ∼0.5–3 h (system-dependent), whereas liquid-phase HF/LiF–HCl often require tens of hours.
• Safety & environmental impact: on the safety and environmental axes, HF-free or in situ–HF-free routes (alkaline/hydrothermal, microwave-assisted) mitigate hazards while maintaining practical kinetics.24–26 Regarding scalability, circulating or mechanically assisted SCF processing in related layered systems demonstrates kilogram-class feasibility, consistent with the qualitative “high” scores assigned to scCO2-assisted routes.12,13 For fair comparison, we employ indicative energy/solvent considerations and explicitly state route dependence in the figure caption and text. scCO2 is non-toxic/inert; risk shifts from corrosivity to pressure/mechanical integrity and is mitigated by closed-loop handling.
• Oxidation stability: reduced aqueous exposure supports improved air stability for selected terminations. These trends align with the broader MXene literature, where surface terminations and storage conditions dominate degradation pathways and ultimately determine stability windows.27,28
• Delamination quality: > 60% monolayer/few-layer fractions have been reported under optimized scCO2 conditions.
• Scalability: kilogram-scale batches have been reported under supercritical conditions; throughput depends on solids loading, residence time, and recycle/heat-integration assumptions.
Linking knobs to metrics: pressure/temperature → etching time & oxidation stability; co-solvent polarity/water activity → monolayer yield & termination profile; RESS rate → delamination quality & lateral dimensions. We annotate these dependencies per route family in Tables 2 and 6.
Recent implementations report up to ∼1 kg MXene production in < 5 h under supercritical conditions. These attributes collectively position scCO2 as a viable and sustainable method for scalable MXene manufacturing, aligning with industrial green processing frameworks. Such synergy between kinetics and sustainability underscores the potential of scCO2 as a benchmark for next-generation green processing. A compact decision-table aligning these axes with route families is provided in Table 6. Values are indicative; safety and environmental impact are route-dependent and assume CO2 recycle where stated.
Many studies explore a single acid/co-solvent at fixed p–T, limiting generality across MAX chemistries. Limited in situ monitoring further obscures ion-transport, swelling, and termination dynamics under scCO2, constraining mechanism-driven design. We therefore encourage factorial variation of etchant concentration, co-solvent identity, and residence time, coupled with real-time spectroscopy, to elucidate kinetics/thermodynamics guiding delamination and functionalization. Techno–economic metrics (specific energy, recycle fraction, compression duty) should be reported transparently to bridge lab validation and industrial deployment.
3.5. Quantitative green metrics and system boundary
Quantitative definitions and boundaries for PMI, E-factor, specific energy, and CO2 intensity follow the schema in Table 3. When the referenced sources provide sufficient material and energy balances, we qualitatively analyze Process Mass Intensity (PMI) and E-factor.29,30 Specific energy (kWh kg−1) and carbon intensity (kg CO2e kg−1) are estimated only where temperatures, pressures, residence times and utility types are reported; for scCO2 we present values with and without a recycle credit.31 Solvents and etchants are qualitatively benchmarked using the CHEM21 solvent selection guide.323.6. Commercial availability and technology readiness
High-pressure unit operations required for scCO2 processing—pressure-rated reactors, compressors, heat-exchangers, and closed-loop CO2 recycle—are mature and widely available as modular skids. By contrast, turn-key, MXene-specific scCO2 lines remain at an early adoption stage. Literature reports demonstrate kg-scale batches in <5 h under supercritical conditions, yet practical deployment hinges on solids handling, residence-time control, heat integration, and recycle ratios. To avoid over-generalization, we summarize what is demonstrably available:• Core hardware stack & safety envelope: pressure vessels (MAWP-rated), compressor trains, heat-exchange and CO2 recycle loops; relief devices and interlocks define the safety boundary for continuous/semi-continuous operation.
• Demonstrated batch sizes & indicative throughputs: reports of up to ∼1 kg MXene produced within <5 h under supercritical conditions showcase feasibility, albeit with batch-mode constraints.
• Cost/energy levers: compression and heat duty dominate variable costs; without heat integration and high CO2-recycle fractions, OPEX can offset solvent/waste gains. Transparent TEA assumptions are needed for fair comparisons.
4. Applications of scCO2-derived MXenes
The MXenes synthesized via scCO2 assisted processes exhibit enhanced physicochemical and electrochemical properties, making them well-suited for high-performance applications across multiple disciplines.3,7,18 They combine structural integrity, tunable terminations, and favorable processing scalability. This section provides a concise overview of key applications where scCO2-derived MXenes show comparative advantages over conventional routes. Application domains and criteria are summarized in Fig. 2d.4.1. Supercapacitors
Due to their large surface area, controllable surface terminations, and improved oxidation resistance, scCO2-exfoliated MXenes serve as excellent candidates for electrode materials in supercapacitors.3,4Liu et al. have developed a simpler, more environmentally friendly method for the synthesis of Ti3C2Tx MXene using a supercritical CO2-based ternary solution (scCO2/DMSO/HCl) compared to traditional methods.33 It can be applied under mild conditions (32 °C and 20 MPa). During the synthesis process, HCl is trapped within the scCO2 and DMSO phases, penetrating the layers of the material and breaking the Ti–Al bond, thus achieving etching. Increasing the local concentration of CO2 in the supercritical phase significantly increases the etching efficiency of HCl, while DMSO facilitates dispersion by bridging the gap between other components in the system. They reported that the fluorine-free Ti3C2Tx MXenes (S–Ti3C2Tx) synthesized using this method exhibited significantly better electrochemical performance compared to conventional HF-etched Ti3C2Tx structures. In supercapacitor applications, the S–Ti3C2Tx electrodes offer a bulk capacitance of 320 F g−1 at a scan rate of 2 mV s−1, while fluorine-containing Ti3C2Tx samples in the literature generally remain in the range of 50–290 F g−1. They also reported that these new S–Ti3C2Tx electrodes exhibited high cycle stability with a capacitance retention rate of 96.72% and a Coulomb efficiency of over 96% after 10.000 cycles. Roy and co-workers have demonstrated the rapid transformation of Ti3AlC2 MAX phase to Ti3C2Tx MXene nanosheets in a single step within a few minutes using an economical and environmentally friendly supercritical water (SCW) assisted method.3 This method has been shown to successfully remove metallic aluminum interlayers from the MAX phase using the lowest concentration HF up to 4 vol% instead of the conventionally used 40–50 vol% HF etchant. The produced SCW assisted MXene was used in a three-electrode supercapacitor system and the gravimetric specific capacitances at current densities of 0.5, 1.0, 2.0 and 4.0 A g−1 were found to be approximately 380.6, 376.5, 367.5 and 293 F g−1, respectively (Fig. 4a). It was observed that these values were considerably higher than the specific capacitance values obtained at 1.0, 2.0 and 3.0 A g−1 current densities in the supercapacitor systems designed using conventional MXene (Fig. 4b). They also fabricated the SCW-MXene based two-electrode asymmetric supercapacitor (ASC) system based on a three-electrode configuration. With this ASC, they found the highest specific capacitance of 271.3 F g−1 at a current density of 0.75 A g−1. Moreover, when the Ragone plot was examined, they obtained quite high gravimetric energy and power densities of 24.11 Wh kg−1 and 240 W kg−1 (Fig. 4c).
 | ||
| Fig. 4 (a) GCD profiles of three-electrode system based on SCW-MXene, (b) specific capacitances of SCW-MXene and conventional MXene at different current densities, (c) Ragone plot of ASC based on SCW-MXene. Adapted with permission from Roy et al.,3ACS Appl. Nano Mater., 2024, 7, 23, 27628–27639. 2023 ACS, (d) Schematic representation of the semi-solid state supercapacitor device fabricated using PVA/H2SO4 electrolyte and digital image of illuminating an LED light using the fabricated device, and (e) plot of the cycling stability of the NRGM semi-solid-state device in PVA/H2SO4 gel electrolyte up to 100.000 cycles. Adapted with permission from Nasrin et al.,34Chem. Eng. J., 2023, 474, 145505. 2023 Elsevier. | ||
Nasrin et al. have reported a hetero assembly of two-dimensional materials such as reduced graphene oxide (rGO) and Nb2CTx MXene using the supercritical fluid processing (SCF) method for the first time. Also, during the assembly, nitrogen (N) doping was provided to rGO and Nb2CTx simultaneously by using a melamine formaldehyde (MF) source in SCF (N–Nb2CTx/rGO: NRGM). It was aimed at increasing the conductivity of the material by N doping. A high energy and power density of 29 Wh kg−1 and 6000 W kg−1 and a high specific capacitance of 816 F g−1 were achieved with the prepared symmetric full cell in aqueous H2SO4 electrolyte using NRGM, respectively (Fig. 4a). In addition, it was observed that the designed device has an extended cycle life with 100% capacitance retention after 100.000 cycles (Fig. 4e).34 Chetana and co-workers reported that the supercritical fluid (SCF) method was used for the first time in the synthesis of CoS/MXene/PANI and CoS/MXene/PEDOT composites.17 They performed electrochemical analyses of these composites in a triple electrode system. As a result of the measurements, it was observed that CoS/MXene/PANI exhibited a high specific capacitance value of 407 F g−1 and CoS/MXene/PEDOT 630 F g−1 at 2 A g−1. In addition, they produced a symmetric device using the button cell technique with composite materials and observed that the specific capacitance value at 2 A g−1 increased in PEDOT (331.1 F g−1) compared to PANI (246 F g−1). Furthermore, in cyclic stability tests performed for 10.000 cycles, it was observed that CoS/MXene/PANI and CoS/MXene/PEDOT exhibited maximum retention of 96% and 97%, respectively. Here, MXene nanosheets are of vital importance in improving the electrochemical performance of the heterostructure with their high surface area and conductivity. These results highlight the crucial role of MXene nanosheets in enhancing heterostructure performance via high conductivity and surface area.
When the literature studies on MXenes used in supercapacitor applications are examined (Table 4), it clearly reveals that Ti3C2Tx electrodes synthesized in scCO2 environment offer higher electrochemical performance in supercapacitor applications compared to Ti3C2Tx electrodes produced by conventional etching methods (HF or LiF–HCl). For example, S–Ti3C2Tx electrodes prepared using the scCO2 method achieve a capacitance of 320 F g−1 at a scan rate of 2 mV s−1 in 1 M H2SO4 electrolyte and a capacitance retention rate of 96.7% after 10.000 cycles. For Ti3C2Tx electrodes produced using the LiF–HCl method, these values remain at approximately 241–248 F g−1 and 89–97% under the same conditions. In HF-etched electrodes, the capacitance is generally between 52–327 F g−1, and the capacitance retention rate is 90–100%. However, it is known that fluorinated functional groups remaining on the surface of such electrodes can limit electrolyte interaction and ion transport. In contrast, fluorine-free MXene structures obtained with scCO2 exhibit significant superiority in both specific capacitance and cycle life due to their more open layered morphology, faster ion diffusion, and more stable surface chemistry. All these results demonstrate that etching in a scCO2 environment is not only a safer option from an environmental perspective but also a more effective alternative to traditional methods in terms of energy storage performance.
| Materials/Synthesis method | Electrolyte | Voltage window (V) | Capacitance (F g−1) | Capacitance retention | Ref. |
|---|---|---|---|---|---|
| S–Ti3C2Tx/scCO2 | 1 M H2SO4 | −0.9–(0.1) | 320 F g−1 at 2 mV s−1 & 254 F g−1 at 1 A g−1 | 96.7% after 10k cycles | 33 |
| Ti3C2Tx/supercritical water-assisted | 3 M KOH | −0.4–(0.4) | 380.6 F g−1 at 0.5 A g−1 | 91% after 100 cycles | 3 |
| Ti3C2Tx/I2 | 1 M H2SO4 | −0.6–(0.2) | 293 F g−1 at 1 mV s−1 | 95.8% after 10k cycles | 35 |
| Ti3C2Tx/molten-salt-electrochemical etching | 1 M H2SO4 | −0.55–(0.2) | 225 F g−1 at 1 A g−1 | 100% after 10k cycles | 36 |
| Ti3C2Tx/NaOH-etching | 1 M H2SO4 | −0.85–(0.55) | 314 F g−1 at 2 mV s−1 | 89.1% after 10k cycles | 37 |
| Ti3C2Tx/LiF–HCl | 3 M H2SO4 | −1.1–(0.15) | 241 F g−1 at 2 mV s−1 | 97.1% after 10k cycles | 38 |
| Clay-like Ti3C2Tx/HF | 1 M H2SO4 | −0.35–(0.2) | 245 F g−1 at 2 mV s−1 | 100% after 10k cycles | 39 |
| Ti3CTx/HF | 6 M KOH | 0–(0.7) | 327 F g−1 at 1 mV s−1 | 96.2% after 5k cycles | 40 |
| Ti3C2Tx/HF | 1 M TEABF4 | −1.0–(0.4) | 52 F g−1 at 2 mV s−1 | — | 41 |
| Ti3C2Tx/LiF–HCl | 1 M H2SO4 | −0.3–(0.3) | 289 F g−1 at 1 A g−1 | 100% after 20k cycles | 42 |
4.2. Battery electrodes (Li-ion, Na-ion)
The interlayer spacing and improved electronic conductivity of scCO2-derived MXenes enhance ion diffusion and charge storage capabilities, making them attractive as anode materials for lithium-ion and sodium-ion batteries.3,14 Studies have shown that MXenes synthesized using supercritical etching exhibit specific capacities and better rate performance than conventionally etched MXenes, attributed to higher structural integrity and uniform terminations.Tang et al. have presented an environmentally friendly H2O-assisted supercritical etching method to improve the etching efficiency in the fabrication of Nb4C3Tx MXene. In this method, the long etching time (96 h) in the traditional HF etching method was reduced by almost 100 times to 1 h due to the synergistic effect between scCO2 and subcritical H2O.43 The synthesized Nb4C3Tx MXene exhibits an accordion-like morphology and has a large interlayer spacing. When Nb4C3Tx MXene was integrated into Li-ion battery as anode, it showed a high specific capacity of 430 mAh g−1 at 0.1 A g−1, outperforming most of the pure-MXene-based anodes. The electrochemical performance of the Nb4C3Tx electrode was compared with that of Nb4C3Tx MXene prepared via HF-based etching, reported in the literature. They clearly demonstrated that the sample synthesized via H2O-assisted supercritical etching exhibited higher Li storage capacity, with a capacity of 430 mAh g−1, exceeding the 380 mAh g−1 produced via conventional methods. In another study, they reported the scCO2 assisted removal method, which reduced the etching time by 64 times to 1.5 hours by using scCO2 with near-zero surface tension (i.e., effectively zero liquid–gas interfacial tension under supercritical conditions, where distinct phases no longer exist), superior solubility and ultra-fast mass transfer in the synthesis of Mo2Ti2C3Tx MXene containing double transition metals (DTM) (Fig. 5a).44 Morphological examination of Mo2Ti2C3Tx reveals that the material has developed an accordion-like structure and there are large distances between the layers (Fig. 5a inset). Mo2Ti2C3Tx used as an anode in Li-ion battery showed a reversible high specific capacitance value of 380 mA h/g at a current density of 0.1 A g−1 (Fig. 5b).
 | ||
| Fig. 5 (a) Comparison of etching time in Mo2Ti2C3Tx synthesis with previous studies (inset: SEM image of Mo2Ti2C3Tx) (b) cycle performance of Mo2Ti2C3Tx at 0.1 A g−1. Adapted with permission from Yang et al.,44J. Alloys Compd., 2025, 1025, 180087. 2025 Elsevier. (c and d) Cycle performance at 1000 and 100 mA g−1 and (e) cycling performance at 1000 cycles of scCO2 supported Ti3C2Tx based electrodes. Adapted with permission from Chen et al.,8Nano Energy, 2023, 107, 102147. 2023 Elsevier. | ||
Chen et al. demonstrated the reduction of MAX phase etching time and increase of MXene layer spacing by using scCO2.8 They showed that 5 typical MAX materials such as Ti3AlC2, Nb2AlC, Ti2AlC, Mo2Ga2C and Ti3AlCN were converted to MXenes (Ti3C2Tx, Nb2CTx, Ti2CTx, Mo2CTx and Ti3CNTx) within a few hours with high efficiency (∼1 kg). They used the produced Ti3C2Tx MXene as anode in Na-ion batteries to show its electrochemical performance. The Na-ion cell, which was initially designed to be like the capacity of previously reported MXenes at a high current density of 1000 mA g−1, exhibited a capacity of approximately 70 mAh g−1 and excellent coulomb efficiency of 100% (Fig. 5c and d). It was observed that when the current density was reduced to 100 mA g−1, the capacity exceeded 100 mAh g−1. In addition, it was observed that the designed Na-ion cell had excellent cycle stability with almost unchanged capacity even after 1000 cycles (Fig. 5e).
Recently published studies indicated that MXene anodes obtained by conventional etching methods based on HF or HCl/LiF typically offer lithium storage capacities in the range of 150–300 mAh g−1 (Table 5). For example, while this value varies between 137–300 mAh g−1 for Ti3C2Tx and Ti3CNTx structures, the HF-synthesized Nb4C3Tx electrode reaches 380 mAh g−1. In contrast, Nb4C3Tx prepared by a scCO2 and water-assisted etching method offers a significantly higher value of 430 mAh g−1. This difference demonstrates that supercritical etching is not only a safer and more environmentally friendly alternative but also enhances electrochemical performance by accelerating ion transport through more optimal arrangement of surface functionalities and interlayer spacing. Therefore, scCO2-based methods have become an attractive option to produce high-performance MXene for Li-ion batteries.
| Structure | Etching system | Cycle | Rate | Mass loading | Capacity | Ref. |
|---|---|---|---|---|---|---|
| Ti3C2Tx | HCl![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) LiF LiF |
100 | 0.1 A g−1 | ∼2.5 mg cm−2 | 137.9 mAh g−1 | 45 |
| Ti3C2Tx | HF and HCl + LiF | 100 | 0.1 A g−1 | ∼1 mg cm−2 | 255 mAh g−1 | 46 |
| Ti2CTx | HF | 120 | 0.1 A g−1 | Not applicable | 260 mAh g−1 | 47 |
| Ti3CNTx | LiF + HCl | 1000 | 0.5 A g−1 | ∼1 mg cm−2 | 300 mAh g−1 | 48 |
| Ti2NbC2Tx | HF | 400 | 0.1 A g−1 | Not applicable | 196.2 mAh g−1 | 49 |
| TiVCxTx | HCl | 100 | 0.1 A g−1 | Not applicable | 207.3 mAh g−1 | 26 |
| V4C3Tx | HF | 300 | 0.1 A g−1 | ∼1.18 mg cm−2 | 225 mAh g−1 | 50 |
| Mo2TiC2Tx | HF | 100 | 0.1 A g−1 | Not applicable | 52 mAh g−1 | 51 |
| Nb2CTx | HCl + LiF | 100 | 0.1 A g−1 | Not applicable | 205.5 mAh g−1 | 52 |
| Nb4C3Tx | HF | 100 | 0.1 A g−1 | ∼0.875 mg cm−2 | 380 mAh g−1 | 53 |
| Nb4C3Tx | NH4HF2 + SPC-CO2@H2O | 150 | 0.1 A g−1 | ∼0.885 mg cm−2 | 430 mAh g−1 | 43 |
| Mo2Ti2C3Tx | NH4HF2-supercritical-CO2 | 100 | 0.1 A g−1 | Not applicable | 380 mAh g−1 | 44 |
| Category | Advantages | Challenges | Ref. |
|---|---|---|---|
| Etching time and efficiency | The duration can be reduced to 30–60 min, and the conversion rate can reach 99% | — | 3, 43 and 60 |
| Surface termination control | Stability increases with the dominance of –OH/–O groups | Surface group inconsistencies may occur due to batch variation | 7 and 60 |
| Layer delamination | Crystal integrity is preserved through physical exfoliation | — | 6 |
| Environmental safety and green chemistry | Provides a non-toxic, inert and recyclable environment | — | 17, 65 and 66 |
| Composite Configuration and electrode efficacy | Robust dispersion of polymers provides high capacitance (>600 F g−1) | — | 17 and 61 |
| Reactor and equipment specifications | — | Specialized high-pressure systems and safety standards are necessary | 8 |
| Optimization specific to material | — | A specific parameter set may be necessary for each MAX phase | 3 and 43 |
| Industrial scaling | — | The process has limitations for integration into industrial systems | 7 |
4.3. Hydrogen evolution reaction (HER) catalysts
MXenes treated with scCO2, especially those with fluoride and hydroxyl functional groups introduced under controlled conditions, exhibit excellent catalytic activity for HER in alkaline media.7 The increased surface area and high density of active sites improve hydrogen adsorption and desorption kinetics.Feng et al. designed a new and green etching method for the synthesis of two-dimensional Ti3C2Tx MXenes by scCO2 for the first time.7 No liquid was used in the method, FeF3·3H2O was used as an effective etchant and scCO2 as a solvent. It was shown that scCO2-coated FeF3·3H2O could successfully delaminate multilayered MXenes into single-layer or few-layered structures. Furthermore, the obtained MXenes were found to have high surface area, superior crystal quality and fluorine-rich end groups. Thanks to these properties, Ti3C2Tx MXenes produced via scCO2 exhibited enhanced alkali HER catalytic activity with very low overpotential thresholds of 29 and 117 mV at current densities of 10 and 100 mA cm−2, respectively, when loaded with platinum clusters (scCO2-Ti3C2Tx-Pt). The normalized mass activity of scCO2-Ti3C2Tx-Pt (49.1 A mg−1) at an overpotential of 400 mV was found to be 3.3 times superior to that of the commercial Pt/C catalyst (14.8 A mg−1). Furthermore, scCO2-Ti3C2Tx-Pt had a Tafel slope of 67.22 mV/dec, indicating that alkaline HER has a typical Volmer–Tafel mechanism, thus promoting H2 desorption (Tafel step) as a rate-limiting step with respect to H2O dissociation (Volmer step). In 2025, Han and his team reported that Ti3C2Tx-based MXene substrates prepared using scCO2 etching could demonstrate high performance in various catalytic applications.54 The scCO2 Ti3C2Tx structure was intercalated with DMSO, and then RuSe2 nanoparticles were homogeneously loaded onto its surface. Both theoretical calculations and experimental analyses revealed that the fluorine-rich Ti3C2Tx surface exhibits strong binding affinity with RuSe2. The RuSe2@SC-Ti3C2Tx heteroelectrode developed based on this structure exhibited remarkable catalytic activity with an overpotential requirement of only 15 mV and a Tafel slope of 21.84 mV dec−1 at a current density of 10 mA cm−2 in alkaline media. Furthermore, the electrode maintained its performance even at high current densities, maintaining its stability even after 120 hours of continuous operation with 5000 cycles of testing. These findings demonstrate the strong potential of the SC–CO2-based etching method not only in supercapacitors but also in other energy conversion applications such as electrocatalysis.
4.4. Additional emerging applications
MXene-based materials produced using the scCO2 method are not only limited to energy storage and catalytic systems but have also begun to attract attention in diverse areas such as electromagnetic interference (EMI) shielding, biosensor technologies, and water purification.4,16,18,33 One study demonstrated that fluorine-free Ti3C2Tx films produced on scCO2 base exhibited a high electrical conductivity of ≈4920 S cm−1 and an outstanding EMI shielding efficiency of ≈53 dB at a thickness of 2.5 µm.33 While studies are underway for other applications of MXenes produced with conventional HF or LiF–HCl, such as EMI and water purification, scCO2-based MXenes appear to be still under development in these areas. Their tunable surface functionalities, high electrical conductivity, and environmentally friendly production processes that minimize solvent use make these materials suitable for use with both polymer-based composites and biocompatible structures. Therefore, scCO2-derived MXenes are expected to emerge as a strong alternative in rapidly developing technological fields such as flexible electronics, wearable sensor platforms, and biomedical applications.In general, MXenes obtained through the scCO2-assisted etching process significantly overcome the limitations of MXenes prepared using traditional methods, thanks to their controlled morphology, stable surface chemistry, and sustainable production structures, and are increasingly gaining a strategic position in studies focusing on next-generation energy, environmental, and health technologies.
5. Advantages and challenges of using scCO2 in MXene synthesis
Conventional etching (HF or LiF–HCl) suffers from toxicity, uncontrolled termination formation, and long processing.55,56 Thus, green-chemistry alternatives with lower footprints and tighter surface-engineering control are sought. In this context, scCO2-assisted synthesis has emerged as a compelling option,55,57 leveraging near-zero surface tension, low viscosity, and high diffusivity for efficient interlayer penetration.Recent studies show that scCO2-assisted etching offers significant performance and environmental advantages over conventional methods.55–58 These include shorter reaction times, higher conversion rates, and enhanced functionalization control.59 For instance, Roy et al.3 reported that the Ti3AlC2 MAX phase achieved a conversion rate of 61% within just 30 minutes of processing. The transformation of Nb4AlC3 to Nb4C3Tx in scCO2 media in combination with H2O was accomplished in only 60 minutes, reaching a conversion rate of 99.43 This efficiency stems from the quick and efficient transportation of etchant agents to the intermediate layers of the MAX phase, assisted by the higher diffusion coefficient of scCO2. This represents a substantial improvement, particularly in terms of time and energy efficiency.3,43
The additional advantage of scCO2-based systems is their capacity to enable the formation of surface functional groups in a more controlled manner. In traditional HF systems, –F terminations are standard; however, –OH and –O groups play a role in MXenes synthesized within the scCO2–DMSO–HCl ternary system.37,60 This termination profile offers significant benefits for enhanced oxidation resistance and electrostatic stability. The research by Hao et al. observed exceptional capacitance values of 320 F g−1 in Ti3C2Tx samples produced using this method.60 Furthermore, these variations in surface chemistry directly influence the interactions of MXenes inside composite systems and enhance performance.37 scCO2 significantly contributes to the physical separation of layered formations. Kim et al.6 demonstrated that the Ti3AlC2 MAX phase could be mechanically exfoliated using alone scCO2 without a requirement for chemical etching. The materials produced through the RESS technique achieved conductivity values of 457 S cm−1, demonstrating that the crystal structure remained intact without degradation.6
Fig. 6 presents a diagram based on the study of Kim et al. (2024), illustrating the supercritical phase conditions of CO2 (a), the physical exfoliation process (b), and a comparative analysis with conventional chemical methods (c). This structure effectively illustrates the function of scCO2 in achieving high conductivity layered structures while preventing surface degradation.
 | ||
| Fig. 6 (a) CO2 phase diagram highlighting the supercritical region (31.1 °C, 74 bar). (b) Schematic of scCO2-assisted mechanical exfoliation of Ti3AlC2via rapid expansion of supercritical suspension (RESS): CO2 penetrates interlayers and expands upon blow-down, promoting delamination while preserving crystallinity. (c) Side-by-side comparison of scCO2-based physical exfoliation vs. HF-etched chemical exfoliation, emphasizing higher retained conductivity and minimized surface degradation in the scCO2 route. Reading note: panels are independent; conditions and variables are annotated to permit understanding without the main text. Adapted from Kim et al.,6Adv. Mater. Technol., 2024, 9, 2301437. 2024 Wiley-VCH GmbH. | ||
Rapid etching techniques conducted in a scCO2 environment not only diminish reaction time but also enhance the penetration of etchant carriers into the interlayers. Specifically, the research conducted by Chen et al.,8 demonstrated that the treatment of Ti3AlC2 MAX phase with scCO2 in propylene carbonate (PC) combined with NH4HF2 solution boosts molecular-level thermal movements, enabling etchant degradation and making it possible the production of high-purity Ti3C2Tx MXene within a few hours. The study suggests the technique can be scaled to the kg level, yielding chemicals that form stable dispersions across a broad spectrum.8Fig. 7 schematically illustrates the underlying molecular mechanisms and highlights the scale-up potential of this method. The reduction of interlayer tensions by CO2 during pressure release facilitates the simple separation of MXene structures, while the improved dispersion capability of the resulting material enhances the process's application potential.
 | ||
| Fig. 7 (a) Ultrafast etching of Ti3AlC2 using SCF-CO2 + propylene carbonate (PC) with NH4HF2; (b and c) enhanced molecular motion accelerates intercalation/etchant transport; (d) kg-scale Ti3C2Tx powder obtained under supercritical conditions demonstrates scalability; (e and f) interlayer expansion and delamination upon pressure release; (g) stable single-layer dispersions in organic media. Reading note: caption specifies reagents, state points, and the take-home message to enable stand-alone interpretation. Adapted from Chen et al.,8Nano Energy, 2023, 118, 108660. 2023 Elsevier. | ||
Another significant contribution of scCO2 can be observed in composite systems, including MXenes and conductive polymers. Chetana et al.,17 indicated that the specific capacitance values of composites, including MXenes produced in scCO2 medium with PEDOT and PANI, reached 630 F g−1 and 407 F g−1, respectively. The higher performance results from scCO2 enhancing the dispersion interaction with polymers and increasing the surface interaction.17 The review by Jayakumar et al.,61 points out that MXenes produced in scCO2 medium have optimal morphology and surface chemistry for energy storage applications.
Despite these advantages, scCO2-based synthesis methods still have several challenges and constraints. Initially, these approaches require specialized reactor systems that operate under high pressure (70–120 bar), which presents challenges for infrastructure when transferring the process from the laboratory to the industrial scale.8,62–65
Furthermore, the process parameters (temperature, pressure, solvent ratio) need to be carefully optimized for each MAX phase, hence constraining the general applicability of the method.43,62,65 Conversely, the research conducted by Liu et al.,60 revealed substantial discrepancies in the termination groups of MXenes produced in various batches under identical synthesis circumstances, resulting in inconsistencies in electrochemical performance. The incorporation of MXenes produced via the scCO2 technique into continuous manufacturing lines remains constrained. Continuous flow systems must be established to facilitate the transfer of the process from the pilot scale to the industrial production scale.7
In conclusion, scCO2-based synthesis strategies represent a transformative step toward sustainable, efficient, and high-performance MXene production. To facilitate widespread adoption, it is imperative to:
• Standardize protocols across different MAX compositions,
• Achieve precise control over surface termination,
• Develop scalable and cost-effective reactor systems.
Future advancements may arise through hybrid approaches, such as microwave-assisted reactions, plasma activation, or electrochemical co-processing. By combining the intrinsic advantages of scCO2 with complementary technologies, it may be possible to unlock new frontiers in both fundamental science and industrial application of MXenes.
6. Challenges and future outlook
Despite the clear advantages of scCO2-assisted MXene synthesis, several challenges remain that must be addressed to enable widespread adoption and commercialization of this method.Although supercritical CO2-assisted MXene synthesis offers clear advantages in safety, tunability, and environmental compatibility, several key challenges must still be addressed before the method can mature into an industrially viable technology. These challenges span process control, scale-up logistics, data transparency, and the balance between material performance and sustainability.1,2,4,8,16
Process control and delamination dynamics: the kinetics of delamination remain highly sensitive to the depressurization rate (dp/dt) during rapid expansion of supercritical solutions (RESS). Uncontrolled or poorly characterized blow-down profiles can lead to layer restacking or flake fracture, thereby compromising lateral size and conductivity. Future studies should adopt standardized pressure-release protocols, report gas-release hardware specifications, and quantify how dp/dt influences morphology and electrochemical behavior.
Throughput, energy efficiency, and safety: scaling the process from gram to kilogram quantities requires precise management of solids flow, residence time, and compression work. Continuous operation demands efficient heat integration and high CO2-recycle ratios to offset the energetic cost of maintaining supercritical conditions. At the same time, the occupational-health-and-safety (OHS) profile shifts from corrosive-chemical hazards to mechanical-integrity risks associated with high-pressure equipment. Thus, clearly documented maximum allowable working pressures (MAWP), relief-valve sizing, interlocks, and closed-loop CO2-handling protocols should become standard elements of process reporting.
Economic and sustainability metrics: many published studies still lack transparent mass- and energy-balance data. To facilitate cross-route comparison and techno–economic analysis, each work should report, per 1 kg of product, the Process Mass Intensity (PMI), E-factor, specific electricity and heat demand (kWh kg−1), carbon intensity with and without CO2-recycle credit, and total water use. Such disclosure would allow consistent benchmarking of scCO2 routes against HF/LiF–HCl and molten-salt systems. In parallel, capital and operating-cost assumptions including pressure-vessel design, compressor duty, and energy-integration strategy should be explicitly stated to improve reproducibility of techno–economic models.
Structure–property correlations and quality descriptors: tailoring surface terminations remains a central challenge: fluorine-rich surfaces often provide better oxidation resistance but lower conductivity, whereas O/OH-dominated terminations enhance wettability and charge transfer at the expense of air stability. Termination control must therefore be guided by the target application. Beyond the commonly cited “monolayer yield,” researchers should report lateral-size distributions, interlayer spacing, defect density, and dispersion stability to define application-specific acceptance windows for energy-storage or catalytic devices.
Emerging hybrid and integrative routes: coupled processes—such as microwave- or plasma-assisted, and electrochemical–supercritical hybrids—offer pathways toward faster and potentially greener synthesis but introduce new engineering complexities. Pilot-scale validation of such systems should include parallel techno–economic (TEA) and life cycle (LCA) assessments to ensure that performance gains justify the additional energy demand and equipment costs. Integrating experimental data with computational-fluid-dynamics (CFD) reactor modeling could ultimately yield predictive tools for designing continuous-flow supercritical reactors.
6.1. Technical and engineering barriers
The requirement for high-pressure equipment and strict control over temperature and pressure conditions limits the accessibility of scCO2 systems to standard laboratories. Moreover, process optimization for each MAX phase composition is still an ongoing effort. Unlike well-established HF-based protocols, scCO2 routes often need to be customized depending on etching agent, reaction time, and pressure range, making standardization more difficult.6.2. Surface chemistry control
Although scCO2 enables better regulation of termination groups, consistent control across batches remains a challenge. Slight deviations in reaction environment can alter the chemical terminations, affecting electrochemical performance and reproducibility.6.3. Cost and scalability
While some reports demonstrate successful gram-to-kilogram scale synthesis, scaling beyond laboratory environments requires significant investment in supercritical reactors, safety infrastructure, and solvent recovery systems. Cost analysis comparing lifecycle emissions and operating expenses with HF- and molten-salt-based processes is still underdeveloped. While several reports demonstrate successful gram-to-kilogram synthesis under supercritical conditions, translating scCO2 routes beyond laboratory environments requires investments in pressure-rated reactors, safety infrastructure, and solvent-recovery loops. Throughput is governed by solids loading, residence time, and whether delamination is coupled to etching. In practice, batch aqueous etching and recirculating/continuous scCO2 setups occupy different operating windows: continuous or semicontinuous scCO2 configurations can deliver higher mass flow rates when compression and heat integration are properly engineered, whereas batch aqueous routes are typically limited by washing and work-up.7,8 Cost drivers separate naturally into variable and fixed components. Variable costs are dominated by electricity/steam utilities and etchant/solvent make-up (including any co-solvents), while fixed costs reflect pressure-rated vessels, compressors, and heat-exchangers required to maintain supercritical operation. For scCO2 processes in particular, compression work and recirculation duties can offset waste-handling gains unless heat recovery and high recycle fractions are implemented; thus any comparison to HF or LiF–HCl routes should state the energy accounting assumptions explicitly.7,8 From an occupational-health-and-safety perspective, fluoride-based aqueous routes are characterized primarily by corrosive/toxicity hazards, whereas scCO2 routes shift the risk profile toward pressure and mechanical integrity. Inherently safer design elements closed CO2 loop, interlocks, appropriately sized relief devices, and conservative maximum allowable working pressure (MAWP) margins7,8 are therefore central to scale-up in supercritical systems.31,676.4. Integration with other green techniques
Future directions may include combining scCO2 with electrochemical or microwave-assisted synthesis to improve efficiency and selectivity. Additionally, fluorine-free approaches using environmentally benign reagents under supercritical conditions are gaining attention and merit further exploration. Operando XPS/Raman under pressure and neutron methods could resolve termination dynamics in real time under scCO2.6.5. Application-driven MXene design
As application fields diversify from biomedicine to aerospace designing MXenes with application-specific features (e.g., surface charge, biocompatibility, conductivity) using scCO2 synthesis will be a key research frontier. Real-time monitoring of scCO2-assisted reactions using in situ spectroscopic tools could further guide rational synthesis.In conclusion, supercritical CO2-assisted MXene synthesis stands as a transformative method with high promise for safe, scalable, and environmentally responsible material production. With targeted research into overcoming current limitations, this strategy could underpin the next generation of multifunctional 2D materials for diverse technological applications.
6.6. Route-specific challenges and engineering constraints
Narrow operative windows and chemistry specificity: process performance is strongly contingent on pressure–temperature windows and co-solvent polarity, which do not transfer uniformly across MAX chemistries (e.g., Ti3AlC2vs. Nb- and Mo-based systems). Outside the working window, layer integrity and yield degrade rapidly. Consequently, each MAX system requires a structured grid of etchant composition, water activity, and p–T mapping rather than “one-recipe-fits-all” optimization.Surface terminations and reproducibility: batch-to-batch variations in –F/–OH/–O ratios and defect density remain a primary source of data scatter in conductivity, oxidation resistance, and electrochemical response. In situ monitoring (e.g., pressure-cell IR/Raman; on-line pH or conductivity proxies) and feedback control targeting termination ratios can reduce this variability and improve inter-laboratory comparability.
Delamination dynamics and RESS blow-down: the pressure release profile (dp/dt) governs re-stacking and mono/low-layer yield. Faster, yet controlled blow-down improves exfoliation while avoiding structural shock. Standardized blow-down protocols and transparent reporting of gas-release hardware are needed for reproducibility.
Throughput, residence time, and compression work: while kilogram-scale batches are achievable, continuous or semi-continuous integration is limited by solids handling, residence-time control, and the energetic cost of compression/heat duty. Without heat integration and high CO2 recycle ratios, operating expenses can offset solvent and waste advantages.
Shift in OHS risk profile: for HF- or fluoride-assisted variants, corrosive/toxicity hazards persist even when acid inventory is reduced; for high-pressure routes, mechanical integrity and relief systems dominate risk management. Closed-loop CO2 handling, correctly sized relief valves, interlocks, and explicit MAWP margins should be treated as reportable process attributes.
Under-reported green metrics: Many studies omit mass- and energy-based metrics needed for method comparison. We recommend reporting, per 1 kg product: Process Mass Intensity (PMI), E-factor, electricity/thermal energy split (kWh kg−1), carbon intensity with and without CO2 recycle credit, and total water use/effluent management. A brief checklist table can facilitate consistent disclosure.
Reactor architecture and cost levers: Capital intensity is driven by pressure vessels, compressors, and heat exchangers, whereas variable costs hinge on utilities and make-up chemicals. Clear statements of CAPEX/OPEX assumptions, energy integration schemes, and recycle ratios are necessary for techno–economic transparency.
Conductivity–termination trade-offs and oxidation stability: fluorine-rich surfaces may suppress oxidation yet constrain conductivity; OH/O-rich surfaces improve wettability and charge transfer but can exhibit variable air stability. Target termination portfolios should be selected per application and explicitly linked to process parameters.
Application-oriented quality metrics: beyond “monolayer yield”, reporting should include lateral size distributions, defect metrics, interlayer spacing, and dispersion stability. For supercapacitors, batteries, and electrocatalysis, we encourage route-specific acceptance windows rather than a single generic figure of merit.
Standardized terminology and data sharing: we distinguish HF-containing from F-free families and recommend a shared schema for reporting process “knobs” (water activity, co-solvent polarity, pressure profile) along with termination statistics including within-batch and batch-to-batch variability. Such standardization accelerates scale-up and technology transfer.
Hybrid routes and engineering realism: Microwave/plasma-assisted or electrochemical–supercritical hybrids are promising but introduce additional hardware, safety protocols, and energy balances. Pilot-scale validation should couple performance testing with concurrent techno–economic analysis (TEA) and life-cycle assessment (LCA). Linking these insights with CFD-aided reactor design will enable predictive scale-up of continuous scCO2 MXene production.
6.7. Route-specific challenges and engineering constraints
Despite their remarkable potential, supercritical CO2-assisted MXene syntheses still face several process-specific and engineering challenges that currently limit large-scale implementation. These issues can be grouped under four main categories: (i) process parameter sensitivity, (ii) reproducibility and monitoring, (iii) scale-up and safety considerations, and (iv) standardization of data and reporting.Finally, hybrid and coupled routes—such as microwave-, plasma-, or electrochemical–supercritical combinations—represent promising extensions of the scCO2 framework but require realistic engineering assessments. Future pilot-scale studies should integrate performance testing with techno–economic analysis (TEA) and life-cycle assessment (LCA). Linking these data with computational fluid dynamics (CFD)-aided reactor modeling will enable predictive scale-up and facilitate the transition of scCO2-assisted MXene synthesis from laboratory concept to industrial process.
7. Conclusion
The development of scCO2-assisted synthesis marks a paradigm shift in the scalable and environmentally responsible fabrication of two-dimensional MXenes. Leveraging the unique properties of scCO2, such as its gas-like diffusivity, liquid-like solvating power, and near-zero surface tension, researchers have significantly reduced etching times (as low as 30 minutes), reduced reliance on corrosive acids (route-dependent), and achieved precise control over surface terminations.These innovations have directly translated into enhanced oxidation resistance, higher electrical conductivity, and superior performance in supercapacitors, batteries, and electrocatalysis. However, several hurdles—such as protocol standardization across diverse MAX phases, reproducible surface chemistry, and affordable, scalable reactor design must still be addressed for broader industrial integration.
Within this context, the present mini-review consolidates the available scCO2-enabled MXene reports into a process-knob → metric framework and aligns them with explicit green metrics and scalability considerations. By comparing scCO2-assisted protocols with established HF, LiF–HCl, molten-salt, and electrochemical routes, and by proposing a practical reporting checklist (PMI, E-factor, energy use, CO2 intensity, water consumption), it is intended to serve as a decision aid for researchers and engineers designing next-generation MXene processes.
Future directions include the incorporation of in situ monitoring techniques, the use of microwave- or plasma-assisted hybrid systems, and techno–economic assessments that validate the sustainability of scCO2 routes. With continued interdisciplinary research and optimization, scCO2-enabled MXene production holds promise not only for advancing fundamental materials science but also for supporting next-generation energy, environmental, and electronic technologies at scale.
Author contributions
Süleyman Gökhan Çolak developed the overall outline of the mini-review, performed the comprehensive literature search and data curation, drafted the original manuscript in its entirety, and assembled and edited all figures and tables. Utku Bulut Şimşek conceived and supervised the project, provided the core conceptual framework for “Green Pathways to MXenes,” and carried out critical revision and final editing of the text. Ahmet Güngör contributed to the methodological structuring of the review, assisted in the literature search, and participated in writing, reviewing, and polishing the manuscript. Melis Özge Alaş Çolak validated the curated data, prepared and refined the visualizations, and supported the revision and refinement of the manuscript. All authors have read and approved the final version of the manuscript.Conflicts of interest
There are no conflicts of interest to declare.Data availability
Data sharing is not applicable to this article as no new data were created or analyzed in this study.Acknowledgements
This study was partially supported by The Scientific and Technological Research Council of Türkiye (TUBITAK) (grant no. 123M826, grant no. 124M988). Süleyman Gökhan Çolak and Utku Bulut Simsek thanks TUBITAK for financial support. In addition, the authors would like to acknowledge the Istanbul Aydin University Board of Trustees for their financial support.References
-
Y. Gogotsi, MXenes: from Discovery to Applications of Two-Dimensional Metal Carbides and Nitrides, 2023, pp. 1–1069 Search PubMed
.
-
C. Zhang and M. Naguib, in Transition Metal Carbides and Nitrides (MXenes) Handbook: Synthesis, Processing, Properties and Applications, Wiley, 2024, pp. 1–752 Search PubMed
.
- K. Roy, N. Rani M, M. P. Narayanaswamy, T. Sardar, S. Vidyashankar and D. Rangappa, ACS Appl. Nano Mater., 2024, 7(23), 27628–27639 CrossRef CAS
.
- M. Mim, K. Habib, S. N. Farabi, S. A. Ali, M. A. Zaed, M. Younas and S. Rahman, ACS Omega, 2024, 9(30), 32350–32393 CAS
.
- T. N. L. Phan, T. X. Nguyen, T. T. Truong, T. K. N. Duong, V. H. M. Doan, T. T. Vo, M. Misra, J. Choi, U. Pal, J. Oh and S. Mondal, SmartMat, 2025, 6, e70009 CrossRef CAS
.
- H. Kim, D. Gwon, D. Kim, J. Kee and J. Koo, Adv. Mater. Technol., 2024, 9, 2301437 Search PubMed
.
- H. Feng, Q. Tian, J. Huang, X. Cui, J. Jiang, Y. Tian, L. Ye and Q. Xu, Green Chem., 2023, 25, 3966–3973 Search PubMed
.
- N. Chen, Z. Duan, W. Cai, Y. Wang, B. Pu, H. Huang, Y. Xie, Q. Tang, H. Zhang and W. Yang, Nano Energy, 2023, 107, 108147 CrossRef CAS
.
- J. Mouallem, M. Arif, A. Raza, G. Glatz, M. M. Rahman, M. Mahmoud and S. Iglauer, Fuel, 2024, 356, 129575 CrossRef CAS
.
- Z. Sun, Q. Fan, M. Zhang, S. Liu, H. Tao, J. Texter, Z. Sun, Q. Fan, M. Zhang, S. Liu, H. Tao and J. Texter, Adv. Sci., 2019, 6, 1901084 CrossRef CAS PubMed
.
- Y. Wang, Z. Chen, Z. Wu, Y. Li, W. Yang and Y. Li, Langmuir, 2018, 34, 7797–7804 Search PubMed
.
- K. Yu, X. Chen, H. Xiang, B. Lu and J. Yin, J. Supercrit. Fluids, 2022, 186, 105605 CrossRef CAS
.
- H. Zhang, Q. Xiang, Z. Liu, X. Zhang, Y. Zhao and H. Tan, Nat. Commun., 2024, 15, 1–10 Search PubMed
.
- W. Bi, G. Gao, C. Li, G. Wu and G. Cao, Prog. Mater. Sci., 2024, 142, 101227 Search PubMed
.
- H. Ding, Y. Li, M. Li, Z. Chai and Q. Huang, Acc. Mater. Res., 2024, 6, 28–39 CrossRef
.
-
J. Singh, K. R. B. Singh, R. P. Singh and C. O. Adetunji, MXenes: Next-Generation 2D Materials: Fundamentals and Applications, Wiley, 2024, pp. 1–351. Search PubMed
.
- S. Chetana, S. Upadhyay, N. C. Joshi, N. Kumar, P. Choudhary, N. Sharma and V. N. Thakur, FlatChem, 2023, 37, 100456 CrossRef
.
- M. T. Hossaın, M. R. Repon, M. A. Shahid, A. Ali and T. Islam, ChemElectroChem, 2024, 11, e202400008 CrossRef
.
- B. Gurzęda, N. Boulanger, A. Nordenström, C. Dejoie and A. V. Talyzin, Adv. Sci., 2024, 11(48), 2408448 CrossRef
.
- A. Iakunkov, N. Boulanger, B. Gurzeda, G. Li, C. Hennig, V. Svitlyk, M. R. V. Jørgensen, I. Kantor, I. A. Baburin, M. M. Hamedi and A. V. Talyzin, Chem. Mater., 2025, 37, 1132–1142 CrossRef CAS
.
- X. Wang, C. Garnero, G. Rochard, D. Magne, S. Morisset, S. Hurand, P. Chartier, J. Rousseau, T. Cabioc’H, C. Coutanceau, V. Mauchamp and S. Célérier, J. Mater. Chem. A, 2017, 5, 22012–22023 RSC
.
- D. W. Matson, R. C. Petersen and R. D. Smith, J. Mater. Sci., 1987, 22, 1919–1928 CrossRef CAS
.
- P. G. Debenedetti, J. W. Tom, X. Kwauk and S. D. Yeo, Fluid Phase Equilib., 1993, 82, 311–321 CrossRef CAS
.
- K. Mahabari, R. D. Mohili, M. Patel, A. H. Jadhav, K. Lee and N. K. Chaudhari, Nanoscale Adv., 2024, 6, 5388–5397 RSC
.
- U. Khan, B. Gao, L. B. Kong, Z. Chen and W. Que, Electrochim. Acta, 2024, 475, 143651 CrossRef CAS
.
- K. Feng, Y. Li, C. Xu, M. Zhang, X. Yang, Y. Cheng, Y. Wang, L. Yang and S. Yin, Electrochim. Acta, 2023, 444, 142022 CrossRef CAS
.
- A. Iqbal, J. Hong, T. Y. Ko and C. M. Koo, Nano Convergence, 2021, 8, 1–22 CrossRef
.
- F. Cao, Y. Zhang, H. Wang, K. Khan, A. Khan Tareen, W. Qian, H. Zhang, H. Ågren, F. Cao, Y. Zhang, H. Wang, K. Khan, A. K. Tareen, H. Zhang, W. Qian and H. Ågren, Adv. Mater., 2022, 34, 2107554 Search PubMed
.
- R. A. Sheldon, Green Chem., 2007, 9, 1273–1283 RSC
.
- C. Jimenez-Gonzalez, C. S. Ponder, Q. B. Broxterman and J. B. Manley, Org. Process Res. Dev., 2011, 15, 912–917 CrossRef CAS
.
- S. D. Manjare and K. Dhingra, Mater. Sci. Energy Technol., 2019, 2, 463–484 Search PubMed
.
- D. Prat, A. Wells, J. Hayler, H. Sneddon, C. R. McElroy, S. Abou-Shehada and P. J. Dunn, Green Chem., 2015, 18, 288–296 RSC
.
- H. Liu, B. Q. Chen, C. Y. Li, C. J. Fang, R. K. Kankala, S. Bin Wang and A. Z. Chen, Small, 2024, 20, 2305321 Search PubMed
.
- K. Nasrin, M. Arunkumar, N. Koushik Kumar, V. Sudharshan, S. Rajasekar, D. Mukhilan, M. Arshad and M. Sathish, Chem. Eng. J., 2023, 474, 145505 CrossRef CAS
.
- H. Shi, P. Zhang, Z. Liu, S. Park, M. R. Lohe, Y. Wu, A. Shaygan Nia, S. Yang, X. Feng, H. Shi, P. Zhang, S. Park, M. R. Lohe, A. S. Nia, S. Yang, X. Feng, Z. Liu and Y. Wu, Angew. Chem., Int. Ed., 2021, 60, 8689–8693 CrossRef CAS PubMed
.
- M. Shen, W. Jiang, K. Liang, S. Zhao, R. Tang, L. Zhang and J.-Q. Wang, Angew. Chem., 2021, 133, 27219–27224 CrossRef
.
- T. Li, L. Yao, Q. Liu, J. Gu, R. Luo, J. Li, X. Yan, W. Wang, P. Liu, B. Chen, W. Zhang, W. Abbas, R. Naz and D. Zhang, Angew. Chem., Int. Ed., 2018, 57, 6115–6119 CrossRef CAS PubMed
.
- Y. Deng, T. Shang, Z. Wu, Y. Tao, C. Luo, J. Liang, D. Han, R. Lyu, C. Qi, W. Lv, F. Kang, Q.-H. Yang, Y. Deng, C. Luo, R. Lyu, W. Lv, F. Kang, T. Shang, Z. Wu, Y. Tao, J. Liang, D. Han, C. Qi and Q. Yang, Adv. Mater., 2019, 31, 1902432 CrossRef CAS
.
- M. Ghidiu, M. R. Lukatskaya, M. Q. Zhao, Y. Gogotsi and M. W. Barsoum, Nature, 2015, 516, 78–81 CrossRef PubMed
.
- Y. Yoon, M. Lee, S. Ku Kim, G. Bae, W. Song, S. Myung, J. Lim, S. Sook Lee, T. Zyung, K.-S. An, Y. Yoon, M. Lee, S. K. Kim, G. Bae, W. Song, S. Myung, J. Lim, S. S. Lee, T. Zyung and K. S. An, Adv. Energy Mater., 2018, 8, 1703173 CrossRef
.
- J. Wang, J. Tang, B. Ding, V. Malgras, Z. Chang, X. Hao, Y. Wang, H. Dou, X. Zhang and Y. Yamauchi, Nat. Commun., 2017, 8, 1–9 CrossRef
.
- H. Liu, X. Chang, L. Li and M. Zhang, Molecules, 2025, 30, 241 CrossRef CAS PubMed
.
- Q. Tang, Y. Wang, N. Chen, B. Pu, Y. Qing, M. Zhang, J. Bai, Y. Yang, J. Cui, Y. Liu, B. Zhou and W. Yang, Small Methods, 2024, 8, 2300836 CrossRef CAS
.
- Y. Yang, Y. Wang, B. Zhou, Q. Tang, B. Pu, J. Cui, Y. Liu and W. Yang, J. Alloys Compd., 2025, 1025, 180087 CrossRef CAS
.
- T. Zhang, L. Pan, H. Tang, F. Du, Y. Guo, T. Qiu and J. Yang, J. Alloys Compd., 2017, 695, 818–826 CrossRef CAS
.
- R. Zheng, L. Wu, J. Zhao, C. Zhu and H. Gao, Nano, 2020, 15(04), 2050051 CrossRef CAS
.
- L. X. Yang, Y. B. Mu, R. J. Liu, H. J. Liu, L. Zeng, H. Y. Li, G. Q. Lin, C. L. Zeng and C. Fu, Electrochim. Acta, 2022, 432, 141152 CrossRef CAS
.
- F. Du, H. Tang, L. Pan, T. Zhang, H. Lu, J. Xiong, J. Yang and C. John) Zhang, Electrochim. Acta, 2017, 235, 690–699 Search PubMed
.
- W. Liu, J. Cao, F. Song, D. D. Zhang, M. Okhawilai, J. Yi, J. Q. Qin and X. Y. Zhang, Rare Met., 2023, 42, 100–110 CrossRef CAS
.
- J. Bai, B. Zhao, S. Lin, K. Li, J. Zhou, J. Dai, X. Zhu and Y. Sun, Nanoscale, 2020, 12, 1144–1154 RSC
.
- C. Chen, X. Xie, B. Anasori, A. Sarycheva, T. Makaryan, M. Zhao, P. Urbankowski, L. Miao, J. Jiang and Y. Gogotsi, Angew. Chem., Int. Ed., 2018, 57, 1846–1850 CrossRef CAS PubMed
.
- M. P. Li, C. B. Zhang, Y. De Li, D. T. Zhang, C. G. Chang and M. C. Liu, Ionics, 2022, 28, 5099–5108 CrossRef CAS
.
- S. Zhao, X. Meng, K. Zhu, F. Du, G. Chen, Y. Wei, Y. Gogotsi and Y. Gao, Energy Storage Mater., 2017, 8, 42–48 Search PubMed
.
- S. L. Han, Q. Y. Tian, B. Gao, X. Q. Sui, H. P. Liu, W. J. Yao, W. Z. Wu and Q. Xu, Rare Met., 2025, 44, 7373–7384 CrossRef CAS
.
- A. M. Amani, L. Tayebi, E. Vafa, M. Abbasi, A. Vaez, H. Kamyab, S. Chelliapan, M. J. Azizli and R. Bazargan-Lari, J. Clean. Prod., 2024, 436, 140606 CrossRef
.
- S. Kumar, Small, 2024, 20, 2308225 Search PubMed
.
- S. Siddique, A. Waheed, M. Iftikhar, M. T. Mehran, M. Z. Zarif, H. A. Arafat, S. Hussain and F. Shahzad, Prog. Mater. Sci., 2023, 139, 101183 Search PubMed
.
- X. Zhang, S. Heinonen and E. Levänen, RSC Adv., 2014, 4, 61137–61152 RSC
.
- C. A. Jones, D. Yang, E. A. Irene, S. M. Gross, M. Wagner, J. DeYoung and J. M. DeSimone, Chem. Mater., 2003, 15, 2867–2869 CrossRef CAS
.
- H. Liu, B. Q. Chen, C. Y. Li, C. J. Fang, R. K. Kankala, S. Bin Wang and A. Z. Chen, Small, 2024, 20, 2305321 Search PubMed
.
- S. Jayakumar, P. C. Santhosh, S. Ramakrishna and A. V. Radhamani, J. Energy Storage, 2024, 97, 112741 CrossRef
.
- A. L. B. Dias, P. dos Santos and J. Martínez, J. CO2 Util., 2018, 23, 159–178 CrossRef CAS
.
- L. Baldino and E. Reverchon, J. Supercrit. Fluids, 2018, 134, 269–273 CrossRef CAS
.
- Z. Jiang, Y. Zeng, R. Guo, L. Lin, R. Luque and K. Yan, Renew. Sustain. Energy Rev., 2024, 203, 114756 CrossRef CAS
.
-
C. D. Wood, B. Tan, H. Zhang and A. I. Cooper, Thermodynamics, Solubility and Environmental Issues, 2007, pp. 383–396 Search PubMed
.
- X. Li, M. Zhan, Y. Liu, W. Tu and H. Li, Chem.–Eur. J., 2024, 30, e202400874 CrossRef CAS PubMed
.
- G. Horvat, K. Aladić and S. Jokić, Teh. Vjesn., 2017, 24(3), 925–934 Search PubMed
.
| This journal is © The Royal Society of Chemistry 2026 |