Open Access Article
Open Access ArticleNH4+-mediated interfacial chemistry for collaborative dual-pathway high-mass-loading energy storage
Jinxin Wang†
,
Wei Guo†*,
Mingming Sun,
Geng Zhang,
Mengting Cheng,
Wenbin Xie,
Na Hu,
Yuehan Yang and
Qiuyu Zhang *
*
School of Chemistry and Chemical Engineering, Key Laboratory of Special Functional and Smart Polymer Materials of Ministry of Industry and Information Technology, Northwestern Polytechnical University, Xian 710072, China. E-mail: weiguo-nwpu@nwpu.edu.cn; qyzhang@nwpu.edu.cn
First published on 28th April 2026
Abstract
The deposition/dissolution mechanism of MnO2 with two-electron transfer is promising for high-energy aqueous energy storage. However, this reaction is severely limited by the kinetically unfavorable dissolution step, a challenge that is greatly exacerbated as the deposit thickens. Herein, by refining NH4+-mediated interface chemistry, we achieve the precise configuration of MnO2 with controlled hydroxylation, which guides the reversible MnO2/Mn2+ conversion under high-mass-loading conditions. The partially hydroxylated surface further creates a kinetically favorable microenvironment for NH4+ storage, ultimately leading to energetic dual-pathway storage behaviors. This enables a remarkable areal capacitance of 13.8 F cm−2 and sound cycling stability over 6000 cycles under high-mass-loading conditions (27.1 mg cm−2). Theoretical calculations reveal that the controlled partial hydroxylation of MnO2 promotes electronic conduction and lowers the adsorption energy of NH4+, outperforming both highly hydroxylated and pure MnO2. The adsorbed NH4+ delivers intimate interfacial electronic interaction with partially hydroxylated MnO2 to trigger local charge redistribution, substantially lowering the MnO2/Mn2+ conversion energy barrier of the nonspontaneous rate-determining step at the NH4+-proximal site. Our findings highlight the significance of the interfacial microenvironment governing the collaborative dual-pathway storage chemistry, which provides guidance for boosting high-mass-loading energy storage.
Introduction
The escalating global energy crisis and climate change highlight the great demand for developing clean and sustainable energy techniques,1–3 e.g., aqueous electrochemical energy storage with inherent safety, environmental friendliness, and cost-effectiveness.4,5 MnO2 has been widely considered as an economic and highly promising cathode alternative in a near-neutral environment, featuring a wide operating voltage and high theoretical capacitance.6,7 However, the sluggish kinetics and the poor stability occur under commercial-level mass loading (≥10 mg cm−2), posing significant challenges for sustaining efficient practical applications of MnO2 electrodes so far.8,9Tremendous efforts have been made to tailor the reactivity and kinetics behaviors of the electrode materials by controlling the microstructure and local reaction environment toward optimal carrier-intercalation/diffusion and interface behaviors.7,9–13 For instance, it was reported that the structural stability issue can be partially relieved by harnessing the physical confinement effect and abundant interfacial interactions.13,14 In another case, robust electronic coupling between the heteroatom and the Mn atom was developed to suppress the structure deformation and Jahn–Teller effect during cycling.15,16 Despite the progress, under practical scenarios with substantially increased electrode thickness and non-active substances, significant kinetics limitations still exist to compromise the storage capability, stemming from the super-long diffusion distance and complicated interfacial redox process.17–19 Beyond that, the dynamic reconstruction of the surface/interface happens with deactivation and dissolution, creating a varied local reaction microenvironment over time that requires better control.
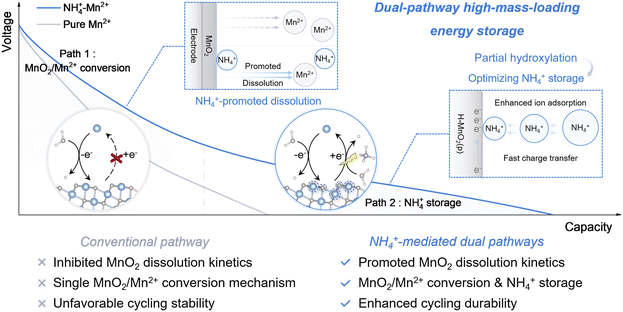
In contrast to intercalation chemistry, the MnO2/Mn2+ deposition/dissolution chemistry provides a new opportunity for highly efficient energy storage. The in situ formation and interface reset during the charge/discharge process can bypass the undesired compositional/structural evolution and considerable local strain accumulation, responsible for breaking the limitations for energetic and sustainable high-mass-loading applications. Moreover, the topological structure and the spatial arrangement of MnO2 can be tailored by refining the cation environment and the reactivity. For better practice, the high concentration of Mn2+ is preferred to boost the deposition behaviors and thereby the charges stored. However, a high-concentration pure Mn2+ electrolyte leads to the suppressed dissolution of MnO2 according to Le Chatelier's principle. At the same time, the deposited thick MnO2 features highly compromised mass/charge-transfer rates with an inferior utilization rate of active species. An open question appears regarding how to enable better control of the deposition/dissolution chemistry with enhanced conversion kinetics under high Mn2+ concentration conditions. Besides, it is predicted that synergistically coupling the cation storage with the MnO2/Mn2+ conversion chemistry may potentially boost high-mass-loading energy storage with ultra-efficient utilization of active species. The mechanistic insights can help the step-forward optimization of high-mass-loading energy storage techniques for better applications.
Herein, by introducing NH4+ into a dynamic environment, we overcome the kinetics limitation of MnO2/Mn2+ conversion and trigger its synergistic coupling with NH4+ (de)intercalation chemistry toward an optimal integrated reaction system. This dual-pathway storage mechanism enables a high areal capacitance of 13.8 F cm−2 for electrodes with a superhigh mass loading of 27.1 mg cm−2, along with a good capacitance retention of 80% after 6000 cycles. The theoretical and experimental results indicate that NH4+ acts as the modulator at the reaction interface to precisely control the hydroxylation degree of MnO2, creating a kinetically favorable reaction microenvironment with fast electronic conduction and enhanced interfacial NH4+ adsorption. Furthermore, the adsorption of NH4+ on the reconstructed interface triggers a local charge redistribution, which substantially reduces the energy barrier of the nonspontaneous rate-determining step during dissolution of MnO2, thereby promoting an energetic dual-pathway storage mechanism. This work offers novel insights into cationic environment engineering for mutual manipulation of deposition/dissolution and intercalation chemistry, and provides a promising avenue for tailoring better high-mass-loading energy storage.
Results
Refining the interfacial microenvironment for favoring MnO2/Mn2+ conversion
Our previous study has demonstrated that dissolved Mnx+ assists in the progressive formation of the electrode–electrolyte interphase, providing the energetic dual-cation storage mechanisms.20 We herein demonstrate the key role of NH4+ in regulating the hydroxylation degree of the derived high-mass-loading MnO2, which is responsible for enhanced intrinsic capability of NH4+ storage (Scheme 1). More importantly, optimized MnO2 dissolution chemistry is revealed with NH4+ in the electrode–electrolyte interface, leading to the collaborative dual-pathway energy storage. The reaction system and the new concept in the collaborative interface chemistry can help break the bottleneck toward energetic high-mass-loading energy storage (Scheme 1).We start with the formation of an active MnO2 substrate through phosphate-assisted in situ reconstruction.20 By studying its charge/discharge properties in 0.5 M Mn2+ electrolyte, we found an increase of the CV curve area and thereby the areal capacitance value for the first 10 cycles (Fig. 1a and b), along with the increased active mass with cycling (SI Fig. 1). However, further cycling leads to a significant performance decline, indicating an unstable storage interface derived from poorly controlled deposition/dissolution chemistry. Increasing the Mn2+ concentration to 1 M leads to an increment of the areal capacitance, but the poor sustainability of the reaction system remains unsolved. In sharp contrast, we discovered that the areal capacitance exhibits a continuous increment during electrochemical cycling in the NH4+–Mn2+ mixed electrolyte, which achieves the highest value after 60 CV cycles (Fig. 1a and b). The derived 60-cycled electrode in NH4+–Mn2+ mixed electrolyte was denoted as Rec-NM-MnO2 for further study.
To further test the superiority of the mixed electrolyte system, we applied the bare carbon substrate (electrochemically exfoliated graphene, EG) to eliminate the influence of active MnO2. A fast increase of areal capacitance with cycling was observed by using the pure EG substrate, ultimately achieving a decent areal capacitance of 9.3 F cm−2 at 2 mA cm−2 (SI Fig. 5). This result reflects the superiority of NH4+–Mn2+ mixed electrolyte to sustain the deposition/dissolution behaviors of MnO2. Of note, a highly improved capacitance was demonstrated by applying the active MnO2 electrode as the substrate (SI Fig. 5), attributed to more abundant nucleation sites and a larger accessible area for better deposition/dissolution chemistry. It delivers an appealing areal capacitance with a high value of 13.8 F cm−2 at 2 mA cm−2 and maintains an areal capacitance of 2.9 F cm−2 at 50 mA cm−2 (Fig. 1c and SI 3d), superior to many state-of-the-art high-mass-loading electrodes (Fig. 1e and SI Table S1).6,21–29 Moreover, it displays good cycling stability with a retention of 80% after 6000 cycles (SI Fig. 3i).
The analysis of capacitive contribution and the b value by Dunn's method indicates a diffusion-controlled behavior during the charge-storage process (Fig. 1f and SI 2b). An asymmetric supercapacitor (Rec-NM-MnO2//active carbon) was configured and investigated in the NH4+–Mn2+ mixed electrolyte. It delivers a high areal capacitance of 3.1 F cm−2 at 4 mA cm−2 (SI Fig. 6d), alongside a maximum energy density of 1070 µWh cm−2, which is superior to many previously reported results (SI Fig. 6e).30–36 Besides, it can operate stably for 500 cycles at a current density of 120 mA cm−2 (SI Fig. 6f). Please refer to SI Note 2 for details.
Revealing the reconstructed high-mass-loading electrode structure
To reveal the reconstructed structure, we first collected scanning electron microscopy (SEM) and high-resolution transmission electron microscopy (HRTEM) images. Before cycling, it demonstrates a core–shell heterostructure with ultra-thin nanosheets uniformly distributed (Fig. 1g and SI 7). It is noted that the unique structure with a large active area creates rich nucleation sites for the energetic MnO2 deposition process, in good agreement with our electrochemical results (SI Fig. 5).20 After 60 cycles in the NH4+–Mn2+ mixed electrolyte, the Rec-NM-MnO2 microstructure features the dense stacking of nanosheets with an enlarged size and thickness compared to before, causing an increase of the overall diameter of the heterostructure from 114 nm to 225 nm and thereby an increment of mass loading from 7.4 mg cm−2 to 27.1 mg cm−2 (Fig. 1g, h, SI 7a and b and 8a–d). The accumulation of mass loading is further indicated by an increase of the charge transfer resistance (Rct) from 1.10 Ω to 6.12 Ω (SI Fig. 9a–c and Table S2). Notably, the system ohmic impedance (Rs) of the Rec-NM-MnO2 electrode is as low as 0.61 Ω (SI Fig. 9a–c and Table S2), compared to the pristine electrode (0.49 Ω). Meanwhile, the Warburg factor (σ) of the samples after different cycles exhibits slight variation in the range of 3.6 to 5.55 (SI Fig. 9d), demonstrating a favorable ion diffusion capability in the thick electrode. The lattice spacing of 0.21 nm in HRTEM images can be indexed to the (101) plane of ε-MnO2 (Fig. 1i and SI 8e).37 One detail is the uniform distribution of N elements on the pristine active substrate, as shown by elemental mapping, which might come from intercalated NH4+ and the dopamine-derived N-doped carbon layer (SI Fig. 8f).Ex situ X-ray diffraction (XRD) measurements were carried out to further reveal the structural evolution with cycling. As shown in Fig. 2a, the pristine active substrate shows diffraction peaks located at 36.2, 43.0, and 65.6°, indexed to the (110), (11-2), and (020) planes of δ-MnO2 (JCPDS card no. 43-1456).20,22,38 However, after cycling, the as-made Rec-NM-MnO2 electrode exhibits diffraction peaks at 37.1, 42.4, and 66.2°, which are matched well with the (100), (101), and (110) planes of ε-MnO2 (JCPDS card no. 30-0820), in good accordance with the HRTEM results.39,40 The transformation of the main structure of the material is further reflected by the gradual shift of diffraction peaks with cycling (SI Fig. 10). Of note, there is an increase in the relative peak intensity of MnO2 compared with that of the EG substrate, attributed to the gradual accumulation of ε-MnO2 upon electrochemical cycling. These results indicate that, despite the enhanced kinetics, the active substrate features a higher deposition rate than that of the dissolution one during the early stage of cycling. The reconstructed reaction interface modulated by the accumulated ε-MnO2 might play a key role in the highly improved electrochemical energy storage behaviors.
Ex situ X-ray photoelectron spectroscopy (XPS) measurement was conducted to study the evolution of the interfacial electronic structure. The peak separation energy (ΔE) of Mn 3 s increases from 4.91 eV for pristine MnO2 to 5.13 eV after the first two cycles (Fig. 2b and SI 11a), suggesting the decrease of the average oxidation state (AOS) of Mn from 3.42 to 3.18 with the accumulation of ε-MnO2. Subsequently, the AOS of Mn increases to 3.49, demonstrating that surface oxidation happens with further cycling. This is also the case in the pure 0.5 M Mn(NO3)2 electrolyte (SI Fig. 12). The O 1s fine spectra can be decoupled and fitted into three components at 530.1, 531.6, and 533.2 eV, assigned to M–O–M, M–OH, and H–O–H, respectively (Fig. 2c and SI 11b).41 The content ratio between M–O–M and M–OH bonds exhibits a reduction after the first two cycles and a gradual increase in the subsequent cycles. This suggests the initial formation of MnO2 with a high hydroxylation degree (H-MnO2(h)), but the hydroxylation degree shows a slight decrease with further cycling, yielding the partially hydroxylated MnO2 (H-MnO2(p)) for the Rec-NM-MnO2 electrode. In the early stage, it is believed that a considerable amount of H+ was produced with the hydrolysis of Mn2+ and the deprotonation effect of NH4+, which promotes the transformation from M–O–M to M–O–H bonds with discharging.42 The similar evolution of M–O–M and M–O–H bonds is found during deposition in the pure 0.5 M Mn2+ electrolyte (SI Fig. 13). However, a relatively low hydroxylation degree was demonstrated, attributed to the inferior H+ supply during hydroxylation.
The N 1s spectra of the pristine active substrate show two main peaks at 400.2 and 407.3 eV, attributed to the –NH– bond derived from dopamine (PDA)-derived carbon and NO3− from the NaNO3 electrolyte, respectively (SI Fig. 11c).20,43,44 Note that a new peak located at 401.4 eV, ascribed to the –N+H– bond, appears at the second cycle, which originates from the NH4+ intercalated in MnO2. Besides, the content ratio between –NH– and –N+H– bonds gradually decreases with cycling (Fig. 2d and SI 11c), indicative of the varied main N source and the gradual accumulation of NH4+-intercalated MnO2, which is also evidenced by XPS full-spectra (SI Fig. 11d and e). Moreover, it is also demonstrated by the ex situ Fourier transform infrared spectra (FTIR), where the characteristic absorptions indexed to the N–H bending vibration (1400 cm−1), N–H⋯O bending vibration (3100–3180 cm−1) and N–H stretching vibration (3180–3300 cm−1) show an increase in intensity, while the peak indexed to the O–H stretching vibration (3300–3600 cm−1) decays with cycling (Fig. 2e and SI 11f), which is consistent with the O 1s results (Fig. 2c).23,45
We focused on the first two cycles to study the structural evolution of the active substrate by ex situ characterization (SI Fig. 14–16). XRD results show periodic changes in the characteristic diffraction peaks (SI Fig. 14). There is a dynamic increase/decrease for the ratio of M–O–M/M–OH with reversible oxidation/reduction (SI Fig. 16), indicating H+ participation in the early stage (please refer to SI Fig. 14–16 for details).
To gain more insights into the microstructure transformation, operando Raman characterization was performed. The pristine active substrate demonstrates three main peaks at 645 cm−1 (ν1), 571 cm−1 (ν2), and 497 cm−1 (ν3), associated with the symmetric stretching vibration of the Mn–O bond along the dz2 orbital within [MnO6] octahedra, the stretching vibration in the basal plane along the dx2–y2 orbital, as well as the weak Mn–O vibration mode, respectively (SI Fig. 17a).46,47 The intensity of these major peaks (ν1, ν2, and ν3) weakened during the first several cycles of deposition (Fig. 2f and SI 17b). This phenomenon might originate from the generation and accumulation of new substances, i.e., hydroxylated MnO2 on the surface of the original MnO2 as discussed (Fig. 2c and SI 11b). As the hydroxylation degree slightly decreases with cycling, the characteristic peaks of MnO2 demonstrate increasing intensity (Fig. 2f and SI 17b). We further focus on the operando Raman spectra during the 10th cycle with the well-defined characteristic peaks. As shown in Fig. 2f and SI 17d, with charging, ν1 and ν2 shift negatively and positively, respectively, and both of them shift back to the original locations upon discharging, indicating a highly reversible lattice deformation with NH4+ insertion/extraction.8,23,47–52 A similar evolution trend was observed during the 1st cycle, except for the spectra in the potential range from 0 to 0.19 V. This unique oscillation phenomenon indicates a significant change in the surface chemical environment with the voltage applied (Fig. 2f and SI 17c).
XPS depth profiles were collected to gain more insights into the surface-accumulated hydroxylated ε-MnO2 of the derived Rec-NM-MnO2 electrode. A high content of Mn–OH (>20%) was found after different etching times, indicative of the considerable hydroxylation degree in the bulk structure upon cycling (Fig. 3f and SI 18b). It is further found that the outermost surface of the electrode demonstrates a higher hydroxylation degree.
The above results have evidenced the interface reconstruction with the accumulation of H-MnO2(p) with high mass loading when cycling in the mixed NH4+–Mn2+ electrolyte (Fig. 2g), responsible for a significant increase in electrochemical performance (Fig. 1e and SI Table S1).
Understanding the role of NH4+ in the reconstructed interface
After understanding the promising nature of H-MnO2(p), we further performed a series of electrochemical analyses and in situ/ex situ characterization experiments to gain insights into two questions: (1) what is the key role of NH4+ in boosting high-mass-loading energy storage? (2) How does NH4+ help overcome the inherent dissolution kinetics limitations?We first studied the electrochemical behaviors of the Rec-NM-MnO2 electrode in the pure 0.5 M (NH4)2SO4 or 0.5 M Mn(NO3)2 electrolyte. The electrode exhibits a higher areal capacitance value in the mixed electrolyte than its counterparts, reinforcing an effective collaborative storage mechanism (Fig. 1c, SI 3a–d and 4). It displays a pronounced oxidation current by using the NH4+–Mn2+ or 0.5 M Mn(NO3)2 electrolyte (Fig. 1d and SI 3e), attributed to Mn2+-to-MnO2 deposition in the Mn2+ rich reaction environment.53,54 In contrast, the reduction current from 1.2 V to 0.9 V increases a lot with NH4+ introduction (Fig. 1d and SI 3e), suggesting a unique role of NH4+ in promoting MnO2 dissolution, which contributes to an increased discharge capacitance.53,54
This is also demonstrated by the charge evolution analysis of the Rec-NM-MnO2 electrode (SI Fig. 3f and g), wherein an obvious charge accumulation was observed in the pure Mn2+ electrolyte, indicative of inadequate dissolution dynamics. In contrast, gradual charge consumption was found when cycling in the pure NH4+ electrolyte, implying promoted MnO2 dissolution with NH4+ introduction. This is further evidenced by significantly weakened charge accumulation in NH4+–Mn2+ mixed electrolyte in comparison to that in the pure Mn2+ electrolyte.55 In addition, with further discharging, an obvious reduction peak was observed in NH4+- Mn2+ or 0.5 M (NH4)2SO4 electrolyte (Fig. 1d and SI 3e), which might originate from the extensive intercalation of NH4+ in the NH4+-rich environments.23,54 We have further studied the electrochemical behaviors in a series of mixed electrolyte environments, evidencing the unique role of NH4+ in boosting charge-storage capability of the reaction system (SI Fig. 3h).
Then, we studied the energy storage mechanism of the Rec-NM-MnO2 electrode in the NH4+–Mn2+ mixed electrolyte. According to ex situ XRD patterns, the diffraction peaks, relative to the (110) and (020) planes of ε-MnO2, demonstrate a positive shift of 0.6° and 0.5° with charging, respectively, and return to the original locations upon discharging, which might originate from the interaction between NH4+ and ε-MnO2 (Fig. 3a).56 This is accompanied by the reversible change of the average oxidation state (AOS) of Mn (Fig. 3b and SI 18a). The characteristic peak indexed to the –NH+- bond displays a negative shift of ∼0.3 eV with charging (Fig. 3c), and shifts back upon discharging, implying the reversible deintercalation/intercalation of NH4+ and its potential interaction with MnO2. Another detail is the negligible change of Mn–O–H content upon discharging, indicative of the negligible contribution of H+ storage after interface reconstruction, which is responsible for the enhanced cycling stability (Fig. 3d).57 This phenomenon is attributed to the accumulation of ε-MnO2 with partial hydroxylation during cycling, which leads to the fast consumption of free H+ in the electrolyte environment. Moreover, the FTIR spectra demonstrate the reversible decrease/increase of peak intensity for the N–H/N–H⋯O bending vibration and N–H stretching vibration during charging/discharging, while the peak indexed to the O–H stretching vibration shows a negligible change, reinforcing that NH4+ serves as the charge carrier during the energy storage process (Fig. 3e and SI 18c).
We further collected the operando Raman spectra of the Rec-NM-MnO2 electrode for tracking the NH4+ storage behaviors. With charging, ν1 shifts positively from 639 cm−1 to 650 cm−1 (Fig. 3g and SI 19), ascribed to the oxidation of the Mn element and enhancement of the Mn–O stretching vibration along the dz2 orbital of [MnO6] octahedra due to the extraction of NH4+.23,47–50 Moreover, the ν2 peak shows an enhanced intensity and a negative shift of 8 cm−1 from 580 cm−1 to 572 cm−1 during charging, originating from the increase of Mn(IV) in the [MnO6] framework and reduced Jahn–Teller disorder, accompanied by NH4+ deintercalation.8,47,52 Both of them shift back to the pristine locations upon discharging, indicative of the reversible structural change with charge carrier deintercalation/intercalation. Operando electrochemical impedance spectra (EIS) of the Rec-NM-MnO2 electrode demonstrate that the semicircle diameter indexed to the charge transfer resistance (Rct) decreases with charging, while the slope in the low-frequency region relative to the ionic diffusion process increases (Fig. 3h and SI 2a). The reversed changes were observed with discharging, further confirming the highly reversible evolution of reaction dynamics.
The above results indicated that NH4+ serves as the charge carrier during the charge/discharge process of the Rec-NM-MnO2 electrode. To further study the effect of NH4+ in the mixed reaction system, the electrochemical behaviors of Rec-NM-MnO2 were further studied in the pure Mn2+ electrolyte. In this case, the capacitance gradually falls behind that in the NH4+–Mn2+ mixed electrolyte with cycling (Fig. 4a). Ex situ XPS results demonstrate the irreversible change of the AOS of Mn after one charge/discharge cycle in the pure Mn2+ electrolyte, resulting in a higher oxidation state of Mn (SI Fig. 20), indicative of an unsatisfactory dissolution dynamics. The fast MnO2 accumulation but unfavorable dissolution kinetics, i.e., an irreversible deposition/dissolution process, serve as the main cause of a fast discharge capacitance decay. The inconspicuous variation in the content of the Mn–O–H bond also manifests the negligible contribution of H+ storage in the pure Mn2+ electrolyte (SI Fig. 21).
Of particular note, the capacitance of Rec-NM-MnO2 in the pure NH4+ electrolyte displays a dramatic decline, with only 52% of the capacitance retained compared with that in the mixed electrolyte after 8 cycles (Fig. 4a). At the same time, we noticed the significantly darkened brown color of the electrolyte (Fig. 4b). This detail indicates the fast dissolution of active Mn species into Mn ions, accompanied by the irreversible disproportionation of Mn3+ in the pure NH4+ electrolyte, which triggers the sharp decrease of capacitance. Operando Raman characterization was employed to further study the microstructure evolution after adopting the pure NH4+ electrolyte. ν1 demonstrates a positive shift of 15 cm−1 during charging (Fig. 4c and SI 22), analogous to the scenario in the mixed electrolyte, suggesting the extraction of NH4+ and the oxidation of Mn centers.23,47–50 Nevertheless, no reverse shift is observed for the ν1 peak in the discharge stage (Fig. 4c and SI 22), indicating the emergence of irreversible oxidation/dissolution processes, in good accordance with our discussion, as shown in Fig. 4a. This is in good agreement with our electrochemical results in the pure NH4+ electrolyte, e.g., the considerable increase of the reduction current (Fig. 1d), the variation of charge accumulation (SI Fig. 3f and g), and the decreased discharge capacitance (Fig. 4a), which concurrently evidence an NH4+-promoted dissolution dynamics.58,59
Through the ex situ XPS measurement, we noticed a considerable change in the content of the Mn–O–H bond during charging/discharging(Fig. 4d and e), indicating a significant proton-storage process along with local structural distortion, which destabilizes the structure and facilitates the dissolution of MnO2 during electrochemical processes. Besides, the most significant variation range of the AOS of Mn (0.47) was found in the pure NH4+ electrolyte (Fig. 4f and SI 23a). The periodic shifts of the –N+H– peak are associated with the intercalation and deintercalation of NH4+ during the cycles (SI Fig. 23b and c). The intercalated NH4+ could also increase local structural disorder, facilitating the interaction of MnO2 with H+ for enhanced dissolution kinetics.47
In addition, in the NH4+–Mn2+ mixed electrolyte system, NH4+ competes with Mn2+ for weakly bound water molecules in the outer solvation sheath through hydrogen bonds and ion–dipole interactions (with solvated H2O), which breaks the compact and ordered solvation configuration of [Mn(H2O)6]2+ in the pure Mn2+ electrolyte.60,61 This reconstruction increases the diversity and weakens the aggregation of solvation structures, thereby elevating the solvation-configurational entropy.62,63 It has been reported that high solvation-configurational entropy can effectively reduce the desolvation energy barrier and accelerate interfacial charge-transfer kinetics.64,65 Meanwhile, the optimized solvation environment is expected to facilitate fast and reversible transport of both NH4+ and Mn2+ by reducing the desolvation energy barrier and weakening the electrostatic aggregation between cationic solvation clusters.61,66 Additionally, the weakly aggregated solvation clusters can also suppress local ion concentration polarization and the subsequent severe interfacial side reactions, thus stabilizing the electrode/electrolyte interface and maintaining the structural integrity of partially hydroxylated MnO2, contributing to good cycling stability.67,68
Therefore, with NH4+ introduced into the reconstructed reaction interface, a synergistic energy storage mechanism integrating the MnO2 deposition/dissolution process coupled with NH4+ storage was enabled (Fig. 4g). This NH4+-assisted cationic environment not only overcomes the dissolution kinetics limitation but also brings additional capacitance contribution through NH4+ storage, leading to sustainable and collaborative dual-pathway high-mass-loading energy storage.
Deciphering the dual-pathway storage mechanism
The X-ray absorption near-edge structure (XANES) characterization was conducted to gain more insights into the evolution of active Mn species. In the Mn K-edge spectra, the absorption edge energy of Rec-NM-MnO2 shows a positive shift to higher energy upon charging (Fig. 5a), indicating the increased oxidation state. Of particular note, the Mn species fabricated by 60 CV cycling in the pure Mn2+ system exhibits a higher valence at 0 V, which is even closer to that in the NH4+–Mn2+ mixed system at 1.2 V (Fig. 5a, SI 24a and b). This is mainly attributed to the sluggish dissolution dynamics for MnO2 in the pure Mn2+ system, resulting in a higher oxidation state at the same discharged state, in good accordance with the ex situ XPS analysis. As shown in the Fourier-transformed (FT) k3-weighted extended XAFS (FT-EXAFS) spectra in Fig. 5b, the peaks located at 1.42 and 2.43 Å indexed to Mn–O and Mn–Mn bonds, respectively, exhibit almost no change in shifts after charging to 1.2 V in NH4+–Mn2+ mixed electrolyte, implying the structural stability of Rec-NM-MnO2 during cycling, in good accordance with the results of the ex situ O 1s spectra (Fig. 3d).69,70 The wavelet transform (WT) analysis further corroborates that the intensity of the Mn–Mn bond experiences a slight increase as the operating voltage ascends (Fig. 5c). In the NH4+–Mn2+ mixed system, the slightly lower intensity of peaks at 0 V than that at 1.2 V is ascribed to insertion of NH4+ and the structural distortion derived from the decline of Mn valence (SI Fig. 24c). Furthermore, the R-space curve in the pure Mn2+ electrolyte shows a high similarity to that in NH4+–Mn2+ mixed electrolyte (SI Fig. 24d), demonstrating the same coordination environment of MnO2 generated, consistent with the XRD analysis (SI Fig. 14c).Our study has demonstrated the formation of partially hydroxylated MnO2 (H-MnO2(p)) as the true active species in the NH4+–Mn2+ mixed system to guide the deposition/dissolution process. For further clarification of the working mechanisms, density functional theory (DFT) simulations were conducted to study the influence of the hydroxylation degree on the interfacial reaction dynamics. As shown in SI Fig. 25, we chose pure ε-MnO2 (named pure MnO2), H-MnO2(p), and H-MnO2(h) (highly hydroxylated MnO2) as models for the calculations. The H-MnO2(p) and H-MnO2(h) structures are modelled as ε-MnO2 with small and large amounts of hydroxyl groups adsorbed on the (101) plane, respectively, according to our aforementioned experimental results. The density of states (DOS) calculation results demonstrate that the band gap decreases in the following sequence: pure MnO2 > H-MnO2(h) > H-MnO2(p) (Fig. 5d), indicating the enhanced electron transfer capability of H-MnO2(p).
To gain more information on the superiority of the storage capacitance of H-MnO2(p) in the NH4+–Mn2+ mixed system, the charge density difference (CDD) analysis and adsorption energy for NH4+ on pure MnO2, H-MnO2(h), and H-MnO2(p) were studied. As depicted in Fig. 5e, at an isosurface level of 0.002 e Å−3, a pronounced charge-transfer behavior appears from the NH4+ side to the MnO2 side, implying an intimate interfacial electronic interaction between NH4+ and H-MnO2(p). In addition, the calculated NH4+ adsorption energy of H-MnO2(p) was considerably lower than that of pure MnO2 and H-MnO2(h) (Fig. 5f), suggesting its kinetically favorable NH4+ adsorption stemming from the tailored interface microenvironment. We further calculated the Gibbs free energy to identify the synergistic modulation mechanism of NH4+ ions during the MnO2 discharge process. As demonstrated in Fig. 5g, this process begins with a proton binding to a lattice oxygen atom on the MnO2 surface, followed by an attack from a second proton. Remarkably, the second proton attack is more favorable at the nearby site, with an energy of −3.03 eV compared to −2.22 eV at the distal site (Fig. 5g). This is followed by the in situ formation of a structural H2O molecule, which is a nonspontaneous process serving as the rate-determining step (RDS). Of note, the energy barrier for this RDS is markedly lower at the NH4+-proximal site (0.51 eV) compared to the distal site (1.13 eV), underscoring the critical role of NH4+ in promoting dissolution kinetics. Also, the proximal site demonstrates a low energy barrier for H2O molecule release from the MnO2 surface, signifying the facilitated dissolution behavior along with the interfacial proton/electron transfer.
Conclusions
In summary, we discovered NH4+-mediated interface chemistry for dynamically reshaping the hydroxylation degree of MnO2 toward an optimal reconstructed interface, which ensures fast Mn2+/MnO2 conversion chemistry, and at the same time, creates a kinetically favorable microenvironment for efficient NH4+ storage. The as-enabled dual-pathway storage mechanism triggers a superior areal capacitance of 13.8 F cm−2 and good cycling stability at a superhigh mass loading of 27.1 mg cm−2. Theoretical calculations indicate that local partial hydroxyl groups help strengthen the electronic conduction and facilitate the interfacial adsorption for NH4+ storage. The adsorbed NH4+ triggers the redistribution of local electrons and thereby results in a considerably reduced uphill energy barrier of the nonspontaneous rate-determining step of the dissolution process, enabling a collaborative dual-pathway storage behavior. This research establishes a study paradigm focusing on the synergistic dual-pathway mechanism by refining the interfacial microenvironment for better high-mass-loading energy storage.Author contributions
W. G. and Q. Z. supervised the research. J. W. and W. G. designed the experimental routes. J. W. carried out the material fabrication and most of the characterization experiments. J. W. and W. G. conducted the main data analysis and the paper writing. J. W., M. S., N. H., and Y. Y. carried out electrochemical measurements. J. W., W. G., G. Z., M. C., and W. X. performed operando Raman characterization. All co-authors contributed to discussions on the relative results and improving the paper.Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article have been included as part of the supplementary information (SI). Supplementary information: the detailed experimental procedures for electrode fabrication, materials characterization, electrochemical measurements, and density functional theory (DFT) computational methods, mass variation data, charge storage dynamics analysis, electrochemical performance comparisons across different electrolytes and substrates, characterization of asymmetric supercapacitors, scanning and transmission electron microscopy images, operando electrochemical impedance spectroscopy, ex situ X-ray diffraction patterns, X-ray photoelectron spectra, Fourier-transform infrared spectra, operando Raman spectra, X-ray absorption near-edge structure data, additional interpretive analysis, performance comparisons with literature reports, and fitted equivalent circuit parameters from impedance measurements. See DOI: https://doi.org/10.1039/d6sc02038k.Acknowledgements
The authors thank the National Natural Science Foundation of China (Grant No. 51673156 for Q. Z., 52202301 for W. G., and 22375161 for W. G.), China Postdoctoral Science Foundation (2022TQ0256 for W. G. and 2022M722587 for W. G.), and Innovation Foundation for Doctor Dissertation of Northwestern Polytechnical University (CX2024093 and CX2025094) for their great support. We also thank eceshi (https://www.eceshi.com) for the theoretical calculations and the Anhui Absorption Spectroscopy Analysis Instrument Co, Ltd for XAFS measurements and analysis.References
- A. Noori, M. F. El-Kady, M. S. Rahmanifar, R. B. Kaner and M. F. Mousavi, Chem. Soc. Rev., 2019, 48, 1272–1341 RSC.
- P. Simon and Y. Gogotsi, Nat. Mater., 2008, 7, 845–854 CrossRef CAS PubMed.
- P. Simon and Y. Gogotsi, Nat. Mater., 2020, 19, 1151–1163 CrossRef CAS PubMed.
- J. Peng, W. Zhang, S. Wang, Y. Huang, J. Z. Wang, H. K. Liu, S. X. Dou and S. L. Chou, Adv. Funct. Mater., 2022, 32, 2111720 CrossRef CAS.
- X. S. Cui, Q. F. Wu, S. Y. Wang, J. Z. Wang, W. L. Qubie, Y. X. Zhang, J. L. Zhang and J. C. Fu, Small, 2025, 21, e05422 CrossRef CAS PubMed.
- Z. Y. Peng, Y. T. Huang, A. G. Bannov, S. L. Li, L. Tang, L. C. Tan and Y. W. Chen, Energy Environ. Sci., 2024, 17, 3384–3395 RSC.
- J. Ma, C. Li, Q. Q. Ji, C. L. Liu, B. Tang, R. Q. Liu, Y. Y. Liu, N. Li, C. Wang, J. R. Zeng, K. Zheng and W. S. Yan, Angew. Chem., Int. Ed., 2025, 64, e202513148 CrossRef CAS PubMed.
- W. Guo, C. Dun, F. P. Yang, C. Zhan, J. J. Urban, J. H. Guo and Q. Y. Zhang, ACS Nano, 2023, 17, 25357–25367 CrossRef CAS PubMed.
- Y. J. Zhao, S. X. Zhang, Y. Y. Zhang, J. R. Liang, L. T. Ren, H. J. Fan, W. Liu and X. M. Sun, Energy Environ. Sci., 2024, 17, 1279–1290 RSC.
- G. J. Lai, X. T. Hu, S. B. Liang, Y. Q. Yang, E. R. Elsharkawy, G. A. M. Mersal, B. A. Lu, Z. M. El-Bahy, Y. Y. Liu and J. Zhou, ACS Energy Lett., 2025, 10, 3437–3444 CrossRef CAS.
- T. Zhang, T. Li, Y. Shen, H. X. Ma, C. Y. Wei, J. H. Cai, Y. Xu, Y. Y. Li, X. J. Dong, S. C. Zhang, F. Q. Huang and T. Q. Lin, Adv. Mater., 2025, 37, 2505082 CrossRef CAS PubMed.
- W. Guo, C. Yu, C. T. Zhao, Z. Wang, S. F. Li, J. H. Yu, X. Y. Tan, Y. Y. Xie, L. Yang, H. L. Huang, R. Fu and J. S. Qiu, Energy Storage Mater., 2020, 31, 172–180 CrossRef.
- X. M. Han, R. Zhao, L. Y. Yu, L. H. Wang, X. Y. Zhang, A. Q. Zhang, J. J. Yang, Z. F. Hu, M. G. Lv, T. X. Miao, F. Wu, Y. Bai and C. Wu, Adv. Mater., 2025, 31, e13548 Search PubMed.
- J. Feng, X. Y. Li, Y. X. Ouyang, H. Y. Zhao, N. Li, K. Xi, J. Y. Liang and S. J. Ding, Angew. Chem., Int. Ed., 2024, 63, e202407194 CrossRef CAS PubMed.
- C. X. Li, W. Guo, J. X. Wang, W. B. Dang and Q. Y. Zhang, Angew. Chem., Int. Ed., 2025, 64, e202422403 CrossRef CAS PubMed.
- W. Guo, F. P. Yang, C. Yu, Y. Y. Xie, J. K. Chen, Y. S. Liu, Y. Zhao, J. Yang, X. F. Feng, S. F. Li, Z. Wang, J. H. Yu, K. L. Liu, K. Qian, M. Tsige, Q. Y. Zhang, J. H. Guo and J. S. Qiu, Matter, 2021, 4, 2902–2918 CrossRef CAS.
- W. Guo, C. Yu, S. F. Li and J. S. Qiu, Energy Environ. Sci., 2021, 14, 576–601 RSC.
- Y. X. Wen, Y. P. Liu, T. Wang, Z. L. Wang, Y. A. Zhang, X. G. Wu, X. T. Chen, S. L. Peng and D. Y. He, ACS Appl. Energy Mater., 2021, 4, 6531–6541 CrossRef CAS.
- D. Geng, S. Zhang, Y. T. Jiang, Z. M. Jiang, M. J. Shi, J. Chang, S. C. Liang, M. Y. Zhang, J. Feng, T. Wei and Z. J. Fan, J. Mater. Chem. A, 2022, 10, 2027–2034 RSC.
- J. X. Wang, W. Guo, M. M. Sun, G. Zhang, Y. Meng and Q. Y. Zhang, Energy Environ. Sci., 2025, 18, 960–971 RSC.
- J. M. Jeong, S. H. Park, H. J. Park, S. B. Jin, S. G. Son, J. M. Moon, H. Suh and B. G. Choi, Adv. Funct. Mater., 2021, 31, 2009632 CrossRef CAS.
- J. X. Wang, W. Guo, Z. X. Liu and Q. Y. Zhang, Adv. Energy Mater., 2023, 13, 2300224 CrossRef CAS.
- Q. Chen, J. L. Jin, M. D. Song, X. Y. Zhang, H. Li, J. L. Zhang, G. Y. Hou, Y. P. Tang, L. Q. Mai and L. Zhou, Adv. Mater., 2022, 34, 2107992 CrossRef CAS PubMed.
- S. F. Zhang, L. Li, Y. L. Liu and Q. L. Li, Carbohydr. Polym., 2024, 326, 121661 CrossRef CAS PubMed.
- Q. H. Cao, J. J. Du, X. W. Tang, X. Xu, L. S. Huang, D. M. Cai, X. Long, X. W. Wang, J. Ding, C. Guan and W. Huang, Research, 2020, 7304767 CAS.
- H. Y. Quan, Z. K. Tu, Z. H. Fang, B. Q. Tan, X. Wang and D. Z. Chen, J. Energy Storage, 2025, 133, 118001 CrossRef CAS.
- L. Chen, F. Wang, Z. W. Tian, H. T. Guo, C. Y. Cai, Q. J. Wu, H. J. Du, K. M. Liu, Z. F. Hao, S. J. He, G. G. Duan and S. H. Jiang, Small, 2022, 18, 2201307 CrossRef CAS PubMed.
- Z. H. Huang, Y. Song, D. Y. Feng, Z. Sun, X. Q. Sun and X. X. Liu, ACS Nano, 2018, 12, 3557–3567 CrossRef CAS PubMed.
- B. R. Li, Y. F. Chu, B. Xie, Y. C. Sun, L. Zhang, H. M. Zhao, L. Zhao, P. F. Liu and J. J. He, J. Mater. Chem. A, 2023, 11, 2133–2144 RSC.
- F. Y. Xie, Y. Du, M. H. Chu, X. Y. Jia, H. Cao, R. Zhang, H. W. Li and M. Zhang, Chem. Eng. J., 2025, 513, 162864 CrossRef CAS.
- Y. J. Lei, W. L. Zhao, Y. P. Zhu, U. Buttner, X. C. Dong and H. N. Alshareef, ACS Nano, 2022, 16, 1974–1985 CrossRef CAS PubMed.
- B. Shi, L. Li, A. B. Chen, T. C. Jen, X. Y. Liu and G. Z. Shen, Nano-Micro Lett., 2022, 14, 34 CrossRef CAS PubMed.
- L. Naderi and S. Shahrokhian, Nanoscale, 2022, 14, 9150–9168 Search PubMed.
- Z. Y. Han, X. Y. Zhang, G. L. Tang, J. Liang and W. Wu, J. Energy Storage, 2024, 91, 112023 CrossRef CAS.
- D. D. Ling, D. H. Zhang and Q. F. Wang, Chem. Eng. J., 2025, 520, 166064 Search PubMed.
- S. J. Xiong, X. X. Li, G. S. Li, Z. L. Wang, Z. J. Wang, M. D. Guo, C. K. Zhang, Y. N. Ma and T. C. Zeng, Adv. Funct. Mater., 2025, 36, e13738 Search PubMed.
- J. Z. Yang, S. S. Bi, H. M. Wang, Y. Y. Zhang, H. X. Yan and Z. Q. Niu, Angew. Chem., Int. Ed., 2024, 63, e202409071 Search PubMed.
- X. H. Zhu, J. Xu, Q. H. Zhang, T. Shen, Y. H. Zhuang, T. T. Chen, S. Li, L. Gu and H. Xia, Nat. Commun., 2025, 16, 3838 CrossRef CAS PubMed.
- H. Y. Chen, P. C. Ruan, H. Zhang, Z. M. El-Bahy, M. M. Ibrahim, B. G. Lu and J. Zhou, Angew. Chem., Int. Ed., 2025, 64, e202423999 CrossRef CAS PubMed.
- W. J. Fan, S. Y. Tian, L. P. Qin, T. S. Alomar, P. C. Ruan, Z. M. El-Bahy, N. AlMasoud, B. G. Lu and J. Zhou, J. Am. Chem. Soc., 2025, 147, 18694–18703 CrossRef CAS PubMed.
- W. L. Zhao, F. H. Xu, L. Q. Liu, M. Liu and B. C. Weng, Adv. Mater., 2023, 35, 2308060 CrossRef CAS PubMed.
- M. X. Li, C. Liu, J. M. Meng, P. Hei, Y. Sai, W. J. Li, J. Wang, W. B. Cui, Y. Song and X. X. Liu, Adv. Funct. Mater., 2024, 34, 2405659 Search PubMed.
- Y. H. Gao, T. Song, X. L. Guo, Y. Zhang and Y. Yang, Green Carbon, 2023, 1, 105–117 Search PubMed.
- W. Q. Wei, Z. Wei, R. Z. Li, H. Yuan, J. S. Chen, J. Lv and S. X. Ouyang, Green Carbon, 2024, 2, 291–299 Search PubMed.
- Y. Song, Q. Pan, H. Z. Lv, D. Yang, Z. M. Qin, M. Y. Zhang, X. Q. Sun and X. X. Liu, Angew. Chem., Int. Ed., 2021, 60, 5718–5722 CrossRef CAS PubMed.
- Q. N. Zhang, M. D. Levi, Q. Y. Dou, Y. L. Lu, Y. G. Chai, S. L. Lei, H. X. Ji, B. Liu, X. D. Bu, P. J. Ma and X. B. Yan, Adv. Energy Mater., 2019, 9, 1802707 CrossRef.
- D. C. Chen, D. Ding, X. X. Li, G. H. Waller, X. H. Xiong, M. A. El-Sayed and M. L. Liu, Chem. Mater., 2015, 27, 6608–6619 CrossRef CAS.
- Y. K. Hsu, Y. C. Chen, Y. G. Lin, L. C. Chen and K. H. Chen, Chem. Commun., 2011, 47, 1252–1254 RSC.
- L. Y. Liu, L. J. Su, Y. L. Lu, Q. N. Zhang, L. Zhang, S. L. Lei, S. Q. Shi, M. D. Levi and X. B. Yan, Adv. Funct. Mater., 2019, 29, 1806778 CrossRef.
- T. Xiao, C. Tang, H. X. Lin, X. R. Li, Y. T. Mei, C. Xu, L. Gao, L. H. Jiang, P. Xiang, S. B. Ni, Y. Q. Xiao and X. Y. Tan, Inorg. Chem., 2024, 63, 17714–17726 CrossRef CAS PubMed.
- L. F. Yang, S. Cheng, X. Ji, Y. Jiang, J. Zhou and M. L. Liu, J. Mater. Chem. A, 2015, 3, 7338–7344 RSC.
- L. F. Yang, S. Cheng, J. H. Wang, X. Ji, Y. Jiang, M. H. Yao, P. Wu, M. K. Wang, J. Zhou and M. L. Liu, Nano Energy, 2016, 30, 293–302 CrossRef CAS.
- X. L. Ye, D. L. Han, G. Y. Jiang, C. J. Cui, Y. Guo, Y. G. Wang, Z. C. Zhang, Z. Weng and Q. H. Yang, Energy Environ. Sci., 2023, 16, 1016–1023 RSC.
- Y. Q. Wang, H. Hong, Z. Q. Wei, D. D. Li, X. R. Yang, J. X. Zhu, P. Li, S. N. Wang and C. Y. Zhi, Energy Environ. Sci., 2025, 18, 1524–1532 RSC.
- J. F. Lei, Y. X. Yao, Z. Y. Wang and Y. C. Lu, Energy Environ. Sci., 2021, 14, 4418–4426 RSC.
- Z. Z. Liu, W. H. Zhou, J. He, H. Chen, R. X. Zhang, Q. Wang, Y. Wang, Y. G. Yan and Y. G. Chen, J. Alloys Compd., 2021, 869, 159279 CrossRef CAS.
- X. L. Li, Z. B. Xu, Y. T. Qian and Z. G. Hou, Energy Storage Mater., 2022, 53, 72–78 CrossRef.
- X. Z. Xue, Z. Liu, S. Chandrasekaran, S. Eisenberg, C. Althaus, M. C. Freyman, A. Pinongcos, Q. Ren, L. Valdovinos, C. T. Hsieh, B. T. Hu, B. Dunn, C. A. Orme, X. Wang, M. A. Worsley and Y. T. Li, Adv. Mater., 2025, 37, 2419505 CrossRef CAS PubMed.
- J. Cui, Z. K. Liu, C. Q. Song, Z. D. Meng and P. Kang, Small, 2025, 21, 2506034 CrossRef CAS PubMed.
- C. Yi, L. Jiao, Y. S. Fu, C. Wang, Z. Q. Ruan, J. J. Liu, X. S. Yang, S. S. Yu, Y. C. Lei, T. Zhang, L. X. Yang, D. K. Shu, S. Yang, C. Y. Li, H. Li, W. J. Zhang and B. W. Cheng, Adv. Energy Mater., 2026, 16, e04881 CrossRef CAS.
- J. T. Yu, X. Y. Ma, X. Y. Zou, Y. Hu, M. C. Yang, Y. L. Cai and F. Yan, Angew. Chem., Int. Ed., 2025, 64, e202416092 CrossRef CAS PubMed.
- B. Liu, G. M. Yao, W. M. Gao, R. X. Wang, J. Xu, C. C. Yuan, B. J. Yang, Q. J. Fu, Y. X. Song, G. M. Zhou and X. J. Wei, J. Am. Chem. Soc., 2026, 148, 7663–7676 CrossRef CAS PubMed.
- F. Ai and Y. C. Lu, Nat. Rev. Mater., 2025, 10, 929–946 CrossRef CAS.
- W. D. Luo, H. W. Fu, P. Peng, C. T. Gao, M. Y. Gu, A. M. Rao, Z. Q. Dai, S. Q. Gu, J. Zhou, F. F. Sun, Y. M. He and B. A. Lu, Joule, 2026, 10, 102271 CrossRef CAS.
- J. W. Huang, X. G. An, Z. L. Cheng, J. L. Jiang, Y. Bai, H. K. Liu, J. Peng, M. H. Wu, S. X. Dou, X. L. Sun and C. Wu, J. Am. Chem. Soc., 2025, 147, 46594–46607 CrossRef CAS PubMed.
- Z. Y. Ou, W. F. Zhang, Y. X. Gang, W. W. Zou, T. M. Ma, Y. F. Su, G. X. Jiang, X. Z. Zhang and L. Du, Adv. Funct. Mater., 2026 DOI:10.1002/adfm.202531692.
- H. Yan, C. G. Hou, Y. M. Wang, Y. C. Guo, Y. M. Bai, H. Fu, X. Q. Li, W. J. Zhang, J. M. Kim and G. C. Liu, Adv. Funct. Mater., 2026 DOI:10.1002/adfm.202530600.
- Y. H. Zhang, J. N. Liu, B. Y. Sun, S. J. Chen, Y. F. Hong, C. T. Lin, J. K. Deng and J. X. Song, Angew. Chem., Int. Ed., 2026, 65, e24476 CrossRef CAS PubMed.
- D. R. Wu, L. M. Housel, S. T. King, Z. R. Mansley, N. Sadique, Y. M. Zhu, L. Ma, S. N. Ehrlich, H. Zhong, E. S. Takeuchi, A. C. Marschilok, D. C. Bock, L. Wang and K. J. Takeuchi, J. Am. Chem. Soc., 2022, 144, 23405–23420 CrossRef CAS PubMed.
- S. Y. Yao, S. Y. Wang, R. C. Liu, X. Liu, Z. Z. Fu, D. W. Wang, H. G. Hao, Z. Y. Yang and Y. M. Yan, Nano Energy, 2022, 99, 107391 CrossRef CAS.
Footnote |
| † These authors contributed equally to this work. |
| This journal is © The Royal Society of Chemistry 2026 |