Open Access Article
Open Access ArticleSelective white phosphorus activation and functionalization with inorganic Grignard reagents
Franziska Gilch a,
Jan Brossette
a,
Jan Brossette b,
Franz Westermair
b,
Franz Westermair c,
Wagner Menezes da Silva
c,
Wagner Menezes da Silva c,
Gábor Balázs
c,
Gábor Balázs a,
Ruth M. Gschwind
a,
Ruth M. Gschwind *c,
Hendrik Zipse
*c,
Hendrik Zipse *b and
Robert Wolf
*b and
Robert Wolf *a
*a
aUniversität Regensburg, Institut für Anorganische Chemie, 93040 Regensburg, Germany. E-mail: robert.wolf@ur.de
bLudwig-Maximilians-Universität München, Institut für Chemie, Butenandtstr. 5-13, 81377 München, Germany. E-mail: zipse@cup.uni-muenchen.de
cUniversität Regensburg, Institut für Organische Chemie, 93040 Regensburg, Germany. E-mail: ruth.gschwind@ur.de
First published on 7th April 2026
Abstract
We describe the targeted and selective functionalization of white phosphorus (P4) using ‘inorganic Grignard reagents’. Reactions of the Fe–Mg complexes [(DippBDI)Mg(THF)xFp] (Fp = CpFe(CO)2, 1: x = 0; 1-THF: x = 1, DippBDI = 2,6-diisopropylphenyl-1,3-diketiminate)) and [(DippBDI)MgFp*] (2, Fp* = Cp*Fe(CO)2) afford the compounds [(DippBDI)Mg(THF)xFp(µ-P4)] (3: x = 0; 3-THF: x = 1) and [(DippBDI)MgFp*(µ-P4)] (4) featuring P42− ligands with a ‘butterfly’ structure bridging Fe and Mg. The coordination of the β-diketiminate magnesium cations [(DippBDI)Mg]+ plays a key role in stabilizing these reactive ‘P4-butterfly’ anions through non-covalent Mg–P bonds and dispersion forces. Thermolysis or photolysis of 3-THF and 4 afforded rare tetraphosphacyclopentadienolate complexes 7 and 8. Complex 4 exhibits unmitigated reactivity towards a wide range of main group element electrophiles (iPrNCNiPr, Ph2BCl, Ph3SnCl, (Me3Si)3SiCl and Mes2PCl), furnishing rare examples of stable, mixed-substituted tetraphosphanes 9–13 as well as an octaphosphane 14 featuring a Sn2P8 core.
Introduction
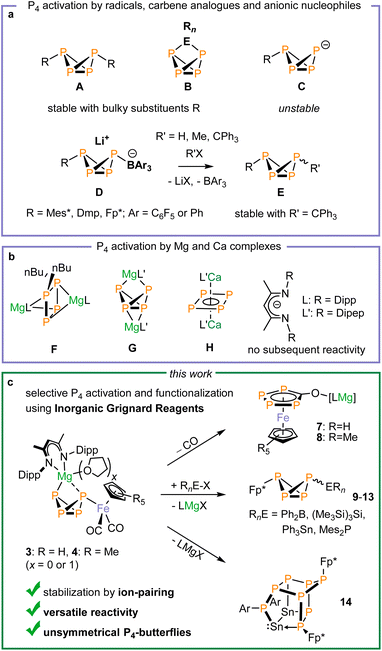
White phosphorus (P4) is the only molecular allotrope of phosphorus stable at room temperature and serves as a crucial feedstock for phosphorus-based chemicals.1 Because of its important role in the phosphorus industry and its multifaceted and often poorly predictable reactivity, the chemistry of P4 is a subject of significant current interest. Although many procedures for P4 activation using main-group elements and transition metals have been reported, the targeted, selective functionalization of the resulting polyphosphorus frameworks lags behind simple P4 activation.2The bicyclo[1.1.0]tetraphosphabutane motive (‘P4-butterfly’) resulting from the cleavage of one P–P bond of the P4 tetrahedron represents the first step in P4 activation.3,4 The synthesis of ‘P4-butterfly’ compounds has been achieved by reaction of P4 with main-group radicals and transition-metal-based radicals,3 as well as using carbenes and heavier carbene analogues.4 Reactions of this type result in the generation of symmetrical P4-butterfly species of types A or B (Fig. 1). Due to the strong, covalent phosphorus-element bonds, these species typically exhibit attenuated reactivity and limited potential for further functionalization. In contrast, the reaction of P4 with charged nucleophiles affords highly reactive anions [RP4]− (C), which tend to decompose quickly.5,6 Nonetheless, alkali metal salts of type M[HP4] have been characterised by 31P NMR spectroscopy at low temperature.5,7 Lammertsma and coworkers have reported that [RP4]− anions (C) containing sterically demanding aryl groups or the Fp*− anion (Fp* = Cp*Fe(CO)2, Cp* = C5Me5) can be stabilized by the coordination of boranes such as BPh3 and B(C6F5)3 to one of the wingtip phosphorus atoms (D, Fig. 1a, R = bulky aryl or Fp*).8–12 However, the reactivity of these species has been limited to protonation and alkylation reactions, a [3 + 1] fragmentation reaction induced by PhNCO and Lewis acid coordination with BH3, BPh3, W(CO)5 and [Au(IPr)]+ [IPr = 1,3-bis(2,6-diisopropylphenyl)imidazolin-2-ylidene]. The P4-butterfly species E resulting from the functionalization of D with R′X decomposes with R′ = H or Me, but can be isolated when sterically demanding alkyl groups such as Ph3C are employed.8,11,12
Recently, we have shown that the combination of a redox-active 3d-metal cation with a main-group element, such as Ga, Si, Sn, or a redox non-innocent d-block element such as Zn, facilitates P4 activation.13 In contrast, the use of transition-metal-magnesium complexes (‘inorganic Grignard reagents’) for P4 activation has hardly been explored,14–16 despite recent studies demonstrating the significant potential of low-valent magnesium and calcium complexes for P4 storage and activation (Fig. 1, complexes F–H, Fig. 1).17,18 Here, we demonstrate that the inorganic Grignard reagents [(DippBDI)Mg(THF)xFp] (1: x = 0; 1-THF: x = 1, Fp = CpFe(CO)2, DippBDI = 2,6-diisopropylphenyl-1,3-diketiminate) and [(DippBDI)MgFp*] (2) selectively afford P4-butterfly complexes [(DippBDI)Mg(THF)xFp(µ-P4)] (3: x = 0; 3-THF: x = 1) and [(DippBDI)MgFp*(µ-P4)] (4). The reversible nature and the mechanism of the P4 activation reaction are investigated using variable-temperature 31P NMR spectroscopy, 1H exchange NMR spectroscopy, and DFT calculations. An analysis of the non-covalent interactions highlights the essential role of the magnesium cation in stabilizing the complexes. Thermolysis or photolysis of 3-THF and 4 generate rare tetraphosphacyclopentadienolate transition metal complexes 7 and 8. Additionally, we show that ‘P4-butterfly’ ligand in 4 is readily functionalized with various electrophiles. These reactions afford unprecedented mixed-substituted bicyclo[1.1.0]tetraphosphabutanes of type Fp*P4R [9–13, R ![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C(NiPr)2 (9), BPh2 (10), SnPh3 (11), Si(SiMe3)3 (12), PMes2 (13)] as well as the octaphosphane [Ar2Sn2P8Fp*2] (14, Ar = 2,6-Dipp2-C6H3). Our results demonstrate that inorganic Grignard reagents act as effective metal-based Lewis pairs, enabling the selective activation of P4 and the targeted functionalization of the resulting P42− ligand.
C(NiPr)2 (9), BPh2 (10), SnPh3 (11), Si(SiMe3)3 (12), PMes2 (13)] as well as the octaphosphane [Ar2Sn2P8Fp*2] (14, Ar = 2,6-Dipp2-C6H3). Our results demonstrate that inorganic Grignard reagents act as effective metal-based Lewis pairs, enabling the selective activation of P4 and the targeted functionalization of the resulting P42− ligand.
Results and discussion
Synthesis and characterization of P4-butterfly complexes
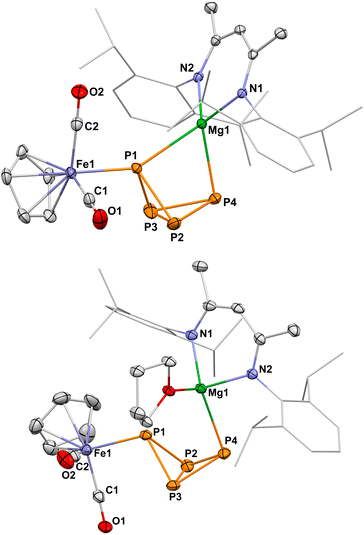
Inspired by the work of Crimmin and Mountford on the use of the inorganic Grignard reagents 1 and 1-THF in C–F activation and heterocumulene insertion,14,15 we investigated their reactivity with P4. Initial 31P NMR spectroscopic studies showed that P4 readily inserts into the Mg–Fe bond of 1-THF in THF-d8 or C6D6 to generate 3-THF, which gives rise to an AMX2 spin system. Likewise, the reaction of 1 with P4 in toluene-d8 afforded the THF-free analogue 3. Complexes 3 and 3-THF can be isolated as air-sensitive yellow-orange crystals in moderate yields (50% and 22%, respectively). The AMX2 spin systems observed in the 31P NMR indicate the formation of a P4-butterfly structure. The 2J31P–31P coupling constants between the wingtip P atoms (47.0 and 36.8 Hz) indicate an endo,exo configuration of the butterfly-P4 ligands,10,12,19 suggesting that there is no through-space interaction between these P atoms.20 The 1H and 31P NMR spectra furthermore indicate that 3 and 3-THF are in an equilibrium with the starting materials 1 and 1-THF and P4 in deuterated benzene and deuterated THF (vide infra).Single-crystal X-ray diffraction (scXRD) studies on 3 and 3-THF confirmed that the P4 molecule has formally inserted into the Mg–Fe bond (Fig. 2). Both complexes crystallize as the endo,exo isomer. One of the wing-tip phosphorus atoms (P1) coordinates to iron. In the structure of 3, the Mg atom bridges the two wing-tip P atoms, P1 and P4, with similar distances (Mg1–P1 2.6529(8) Å, Mg1–P4 2.5623(8) Å). In contrast, a THF molecule is coordinated to the magnesium atom in 3-THF, resulting in a more asymmetric coordination environment (Mg1–P1 2.9427(8) Å, Mg1–P4 2.6320(8) Å). The P–P bond lengths are characteristic of P4-butterfly species, revealing a short P–P bond between the bridgehead P atoms (3: P2–P3 2.163(1) Å; 3-THF: P2–P3 2.1598(8) Å).8,11,19
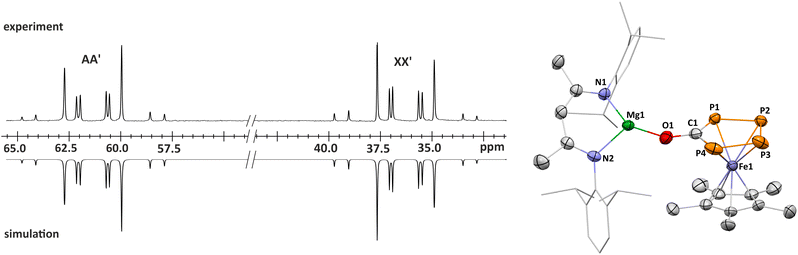
To assess the influence of substituents on the Cp ligand, we synthesized the novel [(DippBDI)MgFp*] complex 2.† Unlike 1, complex 2 forms a dimer in the solid state via the coordination of the CO ligands to magnesium, as previously observed for related complexes9,10.‡ 1H NMR and DOSY NMR studies reveal an equilibrium between monomeric and dimeric species in C6D6 solution (see Fig. S71–S73). The reaction of 2 (0.5 equiv.) with P4 in toluene or benzene cleanly yields 4 as a single endo,exo-configured product, which can be isolated in a high yield (72%) as a yellow-orange crystalline solid (Scheme 1). The molecular structure is very similar to that of 3, as determined by scXRD studies (Fig. 2). In contrast, complex 4 forms a mixture of endo,exo and exo,exo complexes when using THF. Both isomers decompose rapidly in that solvent to the symmetrical [{Fp*}2(µ-P4)] (Fig. S83 and S84, SI), illustrating the stabilizing nature of the ion-pairing between the [(DippBDI)Mg]+ cations and the [Fp*(µ-P4)] anion.
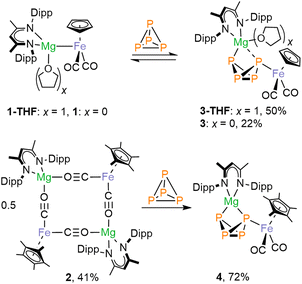 | ||
| Scheme 1 Synthesis of complexes 3-THF, 3 and 4. The reaction is reversible with 1 and 1-THF and irreversible with 2. | ||
Mechanism of the P4 insertion reaction
To gain more insight into the temperature-dependent equilibrium between 1-THF, 3-THF and P4, we performed variable-temperature (VT) 31P{1H} NMR spectroscopic measurements of 3-THF in THF-d8. A van 't Hoff analysis provided the thermodynamic parameters of the P4 activation reaction (ΔG(298 K) = −3.9(±1.7) kJ mol−1, ΔH = −37.8(±0.8) kJ mol−1 and ΔS = −115(±3) J mol−1 K−1; see also Table S8 and Fig. S63). Exchange spectroscopy (EXSY) of the Cp signals in the 1H NMR spectrum revealed a low activation barrier (ΔG‡(298 K) = 73.1(±4.3) kJ mol−1, ΔH‡ = 16.9(±2.9) kJ mol−1, ΔS‡ (298 K) = −188.3(±10.3) J mol−1 K−1; see Table S11 and Fig. S69). 3-THF shows a similar behaviour in toluene-d8 (ΔG(298 K) = −5.0(±6.4) kJ mol−1, ΔH = −43.7(±3.1) kJ mol−1 and ΔS = −129(±11) J mol−1 K−1; see Table S9 and Fig. S63, S64, S76). However, in this case, the van 't Hoff plot shows a nonlinear relationship between 193 K and 233 K, suggesting that the equilibrium was not reached at low temperatures.The donor-free complex [(DippBDI)MgFp] (3) shows a similar temperature-dependent equilibrium in non-coordinating solvents (C6D6 or toluene-d8).15 In toluene-d8, an additional P-containing species is observed at temperatures below 253 K besides P4, which we assign to the exo,exo isomer of 3 (Fig. S79 and S80).
DFT calculations suggest that the mechanism of the P4 activation reaction in THF-d8 likely involves the heterolytic cleavage of the Mg–Fe bond in compound 1-THF (Fig. 3), which appears to be barrierless (see Fig. S128). The coordination of THF generates a solvent-separated ion pair consisting of the Fp− anion and the [(DippBDI)Mg(THF)2]+ cation. Fp− attacks P4 to form the [FpP4]− anion, which is then trapped by [(DippBDI)Mg(THF)2]+. The Gibbs free energy (ΔG0 and ΔG‡) profile calculated by density functional theory (DFT) for this mechanism shown in Fig. 3 qualitatively agrees with the experimental data. However, the calculations slightly overestimate ΔG0 and underestimate ΔG‡. In non-coordinating solvents such as toluene, an alternative mechanism to that depicted in Fig. 3 is likely, in which the CO ligands coordinate to magnesium as previously observed in the molecular structures of magnesium-transition metal carbonylates.9,10 A different reaction pathway, in which P4 coordinates to Mg, but the [(DippBDI)Mg]+ unit does not dissociate from the Fp− anion, is also feasible and features only a slightly higher activation barrier as shown by DFT calculations (see Fig. S129 in the SI). The coordination of P4 to magnesium(II) centers has already been reported.18
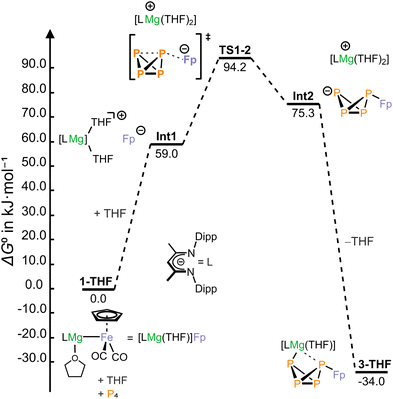 | ||
| Fig. 3 Computed Gibbs free energy profile for P4 activation with 1-THF. Relative Gibbs free energies are presented in kJ mol−1. Level of theory: MN15/def2-TZVP/SMD(THF)//MN15/def2-SVP/SMD(THF). | ||
It is noteworthy that the complexes [(DepBDI)Mg(THF)Fp] (5-THF) and [(MesBDI)Mg(THF)Fp] (6-THF), featuring smaller Mes and Dep substituents on the BDI ligand, gave only traces of the desired P4-butterfly in C6D6 (5% and 3%, respectively), according to qualitative integration of the 31P{1H} NMR spectra (Dep = 2,6-Et2-C6H3, see Table S15 and Fig. S83). This indicates that the strength of the ion-pairing interaction between the Fp− and the magnesium ion significantly influences the equilibrium. The DFT-calculated reaction energies are in full agreement with these findings, showing a greater thermodynamic preference for the formation of the P4-butterfly derivatives with Ar = Dipp by up to −16.2 kJ mol−1, compared to Ar = Dep or Mes (Table S21).
Non-covalent interaction analysis
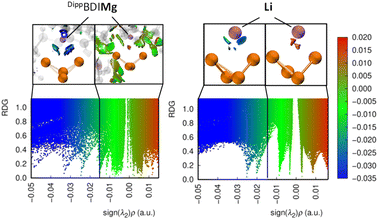
To investigate the influence of the cation on the thermodynamic stability, we performed a non-covalent interaction (NCI) analysis on the complexes 3, 4 and the hypothetical complex [LiFp*(µ-P4)]21.§ The resulting NCI and reduced density gradient (RDG) plots obtained from the wavefunction calculated at the MN15/def2-TZVP/SMD(THF)//MN15/def2-SVP/SMD(THF)22 level of theory are shown in Fig. 4 (see also Fig. S130–S132). Ionic interactions within compound 4 (see Fig. 4 left) and [LiFp*Fe(µ-P4)] (see Fig. 4 right) are visualized in green to blue (sign(λ2)ρ < −0.015 a.u.) in the NCI isosurfaces and RDG scatter plots. The Mg cation in 4 shows a stronger interaction with the wingtip phosphorus atoms compared to the Li cation in [LiFp*(µ-P4)], as indicated by the dark blue color. Dispersion interactions between the [(DippBDI)Mg]+ cation and [Fp*(µ-P4)]− additionally stabilize the complex (Fig. 4).Thermolysis of 3-THF and photolysis of 4: synthesis of 7 and 8
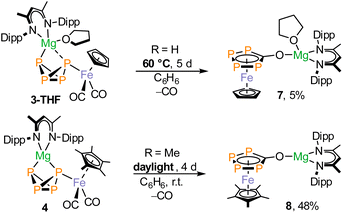
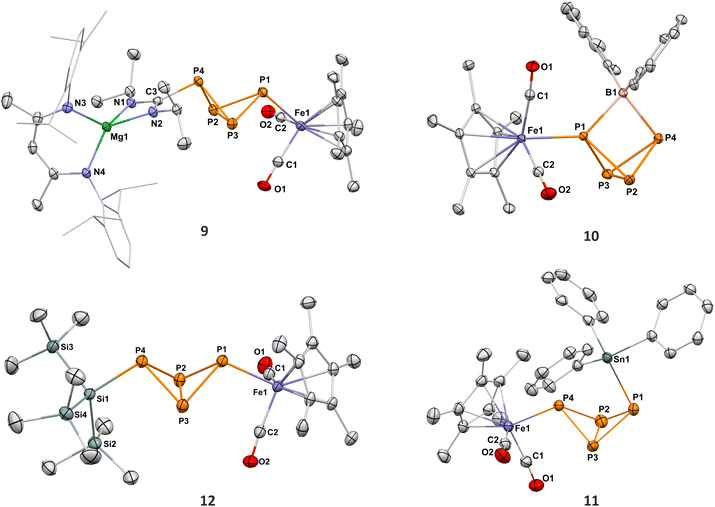
Given the propensity of carbonyl complexes for the dissociation of CO,23 we examined the thermal and photolytic stability of 3-THF. Upon heating of a solution in C6D6 to 60 °C for 5 d, 3-THF was converted to a new species 7 (Scheme 2) characterized by an AA′BB′ spin system in the 31P{1H} NMR spectrum (δ = 44.3 ppm and 30.7 ppm, Fig. S15 and Table S1). It is noteworthy that the presence of the THF ligand in 3-THF is essential for the formation of 7, since the THF-free 3 appears to be completely stable in C6D6 upon heating to +60 °C and under irradiation with a neutral white-light LED (see Fig. S87). In contrast, 4 is also thermally stable but rearranges upon exposure to daylight, forming complex 8 over several days (Scheme 2).The 31P{1H} NMR spectrum of 8 shows an AA′XX′ spin system similar to that of 7 (δ = 61.3 and 36.4 ppm, see Fig. 5). The scXRD analyses of 7 and 8 revealed the formation of ferrocene-like sandwich complexes with η5-coordinated tetraphosphacyclopentadienolate ligands. The planar P4C(O) rings are eclipsed with respect to the co-planar Cp or Cp* moiety. The P–P and P–C bond lengths indicate a delocalized bonding situation (7: P–P2.1013(8) to 2.124(1) Å, P–C1.786(2) and 1.790(2) Å; 8: P–P2.100(2) to 2.135(2) Å, P–C1.781(5) and 1.785(6) Å). Complex 7 crystallizes as a monomer while the THF-free compound 8 forms a polymeric structure via coordination of the P2 atom to a [(DippBDI)Mg]+ cation of an adjacent molecule (Mg1–P2 2.793(2) Å, Fig. S93). To our knowledge, 7 and 8 are the first complexes featuring η5-coordinated tetraphosphacyclopentadienolate P4C(O)− ligands. A similar CO insertion resulting in an η3-P4C(O) ligand has been observed for the reaction of [Fe2(CO)9] with (Cp‴Fe)2(µ-P4) (Cp‴ = C5H2tBu3), which affords the oligonuclear complex [{Cp‴Fe(CO)2}2(µ4,η3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1
1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1
1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1-P4CO){Fe(CO)4}{Fe(CO)3}].24 Additionally, a few related tetraphospholide complexes are known which display similar P–P bond lengths,25–29 e.g. [CpRFe(η5-P4CR′)] (CpR = Cp, R′ = tBu, adamantyl, SiMe3; CpR = Cp*, R′ = Mes, phenyl; CpR = Cp‴, R′ = tBu),25,29 and [Cp*Fe(P4SiL)] (L = PhC{NtBu}2).26
1-P4CO){Fe(CO)4}{Fe(CO)3}].24 Additionally, a few related tetraphospholide complexes are known which display similar P–P bond lengths,25–29 e.g. [CpRFe(η5-P4CR′)] (CpR = Cp, R′ = tBu, adamantyl, SiMe3; CpR = Cp*, R′ = Mes, phenyl; CpR = Cp‴, R′ = tBu),25,29 and [Cp*Fe(P4SiL)] (L = PhC{NtBu}2).26
Reactivity of 4 towards electrophiles
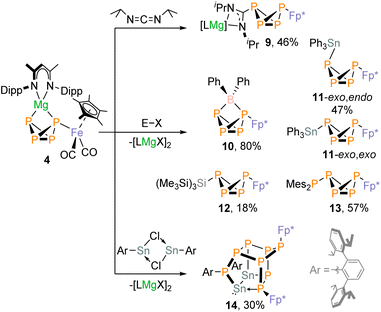
The high-yielding synthesis of complex 4 enabled us to investigate its reactivity towards electrophiles in detail. During these studies, we found that 4 reacts readily and selectively with numerous electrophiles, including main-group element halides and heterocumulenes (Scheme 3). The products exhibit characteristic AMX2 spin systems in the 31P{1H} NMR spectra, which provide evidence for P-functionalized P4-butterfly compounds. Using bulky electrophiles such as iPrNCNiPr, Ph2BCl, Ph3SnCl, (Me3Si)3SiCl, and Mes2PCl we were able to isolate the new P4-butterfly complexes 9–13. Complexes 9, 10 and 12 are stable in solution for several days, while 11 and 13 gradually decompose at ambient temperature (Fig. S54 and S84). It is noteworthy that 11 dimerises slowly to a tricyclic P8 cluster 15, as revealed by 31P NMR data and a preliminary scXRD analysis (Fig. S48). Weigand and co-workers observed a similar dimerization reaction for the pentaphosphane P5(C6F5)2 generated from (DippBDI)GaP4 butterfly with (C6F5)2PBr.30ScXRD studies revealed the molecular structures of 9–13. Complex 9 results from an insertion of the carbodiimide iPrNCNiPr into the Mg–P bond of 4. The two nitrogen atoms coordinate to magnesium, and the carbon atom is bound to phosphorus (Fig. 6). For 9, 12, and 13 (Fig. S1), the butterfly-P4 unit is an exo,exo configuration with the Fp* and carbodiimide, (Me3Si)3Si- and Mes2P-substituents pointing toward the bridgedhead P atoms. In contrast, complex 10 shows a symmetrically bridging Ph2B moiety (P–B 2.016(2) Å and 2.055(2) Å). An endo,exo configuration is likewise observed for the Ph3Sn-substituted complex 11.
The 31P{1H} NMR data are consistent with these crystallographic findings. For complex 9, the 2J31P–31P coupling (258 Hz) is larger than the 1JPP couplings (179 Hz, 185 Hz), indicating an exo,exo butterfly structure in solution. The 13C{1H} NMR spectrum displays a doublet of doublet for the quaternary carbon atom of the carbodiimide unit due to C–P coupling (δ = 175.0 ppm, 1JPC = 103.9 Hz, 3JPC = 23.3 Hz, Fig. S21). The exo,exo isomer is also present for 12 and 13, as evidenced by the high-field shifted PM resonances of the AMX2 spin systems and large 2JPP couplings (2JAM = 235 Hz and 259 Hz, respectively; see Tables S5 and S6), which result from through-space interactions of the lone pairs.10 Additionally, 29Si satellites (1JSiP = 97.5 Hz) were detected for the P atom with the (Me3Si)3Si substituent. In contrast, the small 2JPP coupling constant (2JAM = 33 Hz) indicates that compound 10 retains the endo,exo configuration in solution. The resonances assigned to the wingtip phosphorus atoms P1 and P4 exhibit chemical shifts similar to compound 4, while the signal of the bridgehead phosphorus atoms P2 and P3 is shifted downfield by 50.4 ppm. The 11B{1H} NMR spectrum shows a broad signal (Δν½ = 362 Hz) at 20.5 ppm with unresolved coupling to phosphorus. The 31P{1H} NMR spectra of the Ph3Sn-substituted complex 11 display two distinct AMX2 spin systems in an approximate 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 integral ratio, indicating that the endo,exo and exo,exo isomers coexist in solution (Fig. S30). However, when a sample of isolated 11 was dissolved at −80 °C in toluene-d8 and the 31P{1H} NMR was recorded at −80 °C, only the signals of the endo,exo isomer were observed (2JAM = 16 Hz, Table S3). Variable-temperature 31P{1H} NMR spectroscopy of 11 shows a slow conversion to an equilibrium mixture of the endo,exo and exo,exo isomer at elevated temperature (Fig. S81). The 119Sn NMR spectrum of endo,exo 11 dissolved at −80 °C shows a doublet of doublets with a 1J119Sn–31P coupling of 973 Hz and a smaller coupling of 1J119Sn–31P = 299 Hz in agreement with the 117/119Sn–P coupling constants derived from the 31P{1H} NMR spectrum (see Table S3).
1 integral ratio, indicating that the endo,exo and exo,exo isomers coexist in solution (Fig. S30). However, when a sample of isolated 11 was dissolved at −80 °C in toluene-d8 and the 31P{1H} NMR was recorded at −80 °C, only the signals of the endo,exo isomer were observed (2JAM = 16 Hz, Table S3). Variable-temperature 31P{1H} NMR spectroscopy of 11 shows a slow conversion to an equilibrium mixture of the endo,exo and exo,exo isomer at elevated temperature (Fig. S81). The 119Sn NMR spectrum of endo,exo 11 dissolved at −80 °C shows a doublet of doublets with a 1J119Sn–31P coupling of 973 Hz and a smaller coupling of 1J119Sn–31P = 299 Hz in agreement with the 117/119Sn–P coupling constants derived from the 31P{1H} NMR spectrum (see Table S3).
The reactivity of 4 towards electrophiles strongly depends on the nature and steric bulk of the electrophile. The substituents must provide sufficient steric protection to stabilize the resulting butterfly complex without suppressing reactivity. Thus, 31P{1H} NMR spectroscopic monitoring studies indicate that 4 cleanly reacts with the moderately bulky electrophile Me3SiBr to afford the desired P4-butterfly compound Fp*P4SiMe3. However, this compound is unstable and decomposes overnight at room temperature. In contrast, the reaction of the bulkier tBuMe2SiCl with 4 is slow, resulting in an incomplete substitution reaction. Oxidizing electrophiles such as (2,6-Mes2-C6H3)PCl2 (Mes = C6H2-2,4,6-Me3) produce Fp*2P4 and P4 among other decomposition products.
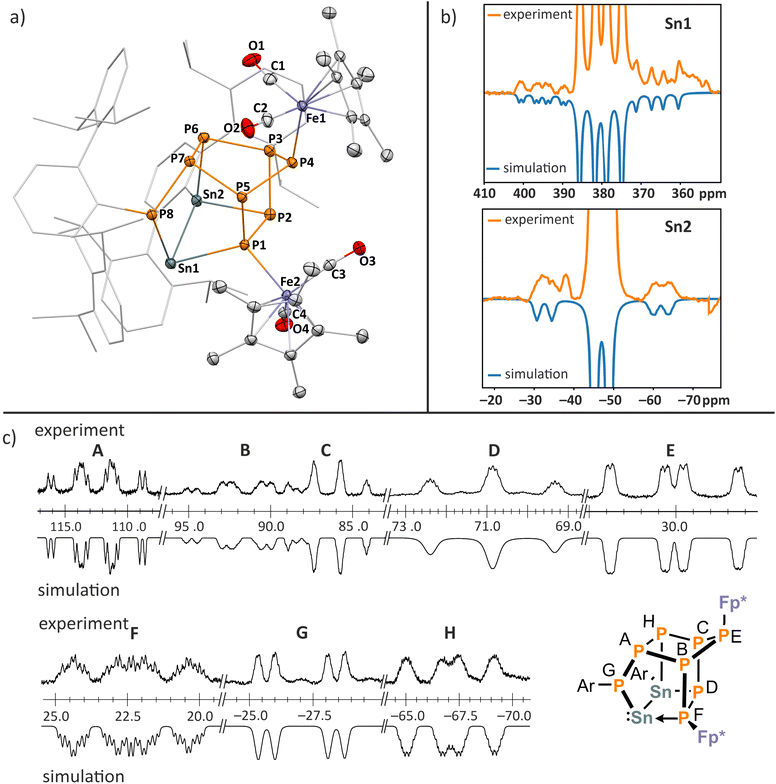
The reaction of 4 with [ArSnCl]2 (0.5 equiv., Ar = 2,6-Dipp2-C6H3, Dipp = 2,6-iPr2-C6H3) affords the polycyclic compound 14 (Fig. 7a), which formally is the dimerization product of the expected P4-butterfly species Fp*P4SnAr. The molecular structure of 14 consists of a pentacyclo[5.2.1.02,6.03,9.04,8]decane-like P8Sn2 core derived from a norbornane-like P7 moiety with an exocyclic ArP unit and a Sn–Sn single bond (2.8591(2) Å).31 The P–P bond lengths (2.1747(6)–2.2483(6) Å) are in the range of P–P single bonds. Notably, the Sn1–P1 bond length 2.6726(4) Å results from a dative interaction involving the lone pair of P1.
The remaining Sn–P distances are in a close range (2.5707(4)–2.5967(4) Å), consistent with Sn–P single bonds. The structural framework of 14 is reminiscent of bishomocubanes derived from dimethyl dicyclopentadienedicarboxylate (Thiele's ester).32 Additionally, some related carbocyclic polyphosphanes have been described, resulting from the dimerization of 1,2,4-triphosphacyclopenta-1,3-dienes or the reaction of R3Sn stabilized 1,2,4-triphosphacyclopenta-1,3-dienes with tert-butylphosphaalkyne.33 Notably, the dimerization of the As4-butterfly complexes [{Cp‴Fe(CO)2}2(µ,η1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1-As4)] or [{Cp*Cr(CO)3}2(µ,η1
1-As4)] or [{Cp*Cr(CO)3}2(µ,η1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1-As4)] afford realgar-like (tricyclo[3.3.0.03,7]octane)-As8 species.34 Similar realgar-like P8 units are also known.35
1-As4)] afford realgar-like (tricyclo[3.3.0.03,7]octane)-As8 species.34 Similar realgar-like P8 units are also known.35
The 119Sn NMR spectrum of 14 shows an apparent doublet of doublets at 380 ppm, and a broad doublet (Δν½ > 700 Hz) with unresolved fine structure at −47 ppm. The assignment of the Sn1 and Sn2 signals is based on the calculated 1J119Sn–31P coupling constants of 1471 Hz for Sn1–P8, 970 Hz for Sn1–P1 and 823 Hz for Sn2–P6 (Fig. 7b; see SI, section 8.3 for details). These data compare well with the experimental values of 1540 Hz, 882 Hz and 830 Hz, respectively. The 117Sn and 119Sn satellites could be detected in the 119Sn NMR spectrum (1J119Sn1–119Sn2 ≈ 6700 Hz; calculated −7663 Hz), confirming that the Sn–Sn bond remains intact in solution. For comparison, the 1J119Sn−119Sn coupling constants in Ar*SnSnPh2Ar* and Ar*SnSn(Me2)Ar* [Ar* = 2,6-(2,4,6-iPr3-C6H2)2-C6H3] are 7237 Hz and 8330 Hz, respectively.36
The 31P{1H} NMR spectrum of 14 displays a set of eight multiplets between 112.4 ppm and −67.1 ppm for the eight distinct P-atoms. The coupling constants were determined by simulating the spectrum using an iterative fitting procedure (Fig. 7c and Table S7, SI). Compound 14 is unstable in solution at ambient temperature. Upon storing a solution at room temperature, the signals of 14 gradually disappear over the course of three weeks, and two new sets of signals appear in the 31P{1H} NMR spectrum in the same chemical shift range as for 14, indicating that it rearranges slowly in solution to form a new complex 16 (see SI, Fig. S60). A preliminary scXRD analysis shows that 14 eliminates one CO ligand. As a result, one of the Fe atoms binds to two adjacent phosphorus atoms (Fig. S49). Based on the very similar chemical shifts and coupling constants in the 119Sn and 31P{1H} NMR spectra (Fig. S57 and S60), we assume that two isomers of 16 coexist in solution.
Conclusions
We have shown that the inorganic Grignard reagents 1 and 2 effectively activate P4 through furnishing P4-butterfly dianions that are coordinated by Fe and the bulky β-diketiminato magnesium cation [(DippBDI)Mg]+. Our theoretical analysis emphasizes the importance of non-covalent and dispersion interactions in stabilizing these complexes, which appear to be stronger for [(DippBDI)Mg]+ than for Li+.§ The reversible dissociation of the Fe–Mg Lewis pairs 1 and 2 into solvent-separated ion pairs appears to be key. According to DFT calculations, the reactions proceed stepwise via nucleophilic attack by the Fp− or Fp*− anion on P4 and subsequent coordination to [DippBDIMg]+. However, unlike previous studies, these reactions do not require an external Lewis acid, simplifying reactivity investigations. Reactions of 4 with electrophiles generate a variety of new mixed substituted P4-butterfly species 9–13, as well as the novel Sn2P8 cluster 14. Additionally, thermally or photochemically induced rearrangements led to rare tetraphosphole complexes 7 and 8.Our results indicate that combining transition-metalate anions with the [DippBDIMg]+ cation is a promising strategy for deliberate and selective P4 activation and functionalization. When sufficient steric bulk is provided, the resulting P4-butterfly complexes can be easily isolated, marking an ideal starting point for future reactivity studies. In other cases, dimerization to P8 frameworks can be observed. Ongoing work in our group focuses on the reaction chemistry of complexes 9–14 and on the broader applicability of our approach using a wider range of metal-based Lewis pairs.
Author contributions
F. G. conceptualisation, investigation – synthesis and characterisation, writing – original draft; J. B. investigation – DFT studies of the mechanism and NCI analysis; W. M. S. investigation – DOSY and EXSY studies; F. W. investigation – Sn NMR studies and DFT calculations; R. W., G. B. conceptualisation, supervision; R.W., R. G., H. Z. supervision and funding acquisition. All authors contributed to manuscript review, editing and discussion.Conflicts of interest
There are no conflicts to declare.Data availability
CCDC 2529860–2529874 and 2541029 contain the supplementary crystallographic data for this paper.38a–pThe data supporting the findings of this work is available in the main text or the electronic supplementary information (SI). Primary research data for this work is openly accessible on Radar4Chem (https://radar4chem.radar-service.eu/radar/de/home) under the DOI: https://doi.org/10.22000/7wa7f1kdq20s260j. Supplementary information: experimental procedures, details of NMR spectroscopic studies, scXRD data, cyclic voltammetry data, IR and UV-vis spectra, and computational data. See DOI: https://doi.org/10.1039/d6sc01229a.
Acknowledgements
We thank the Deutsche Forschungsgemeinschaft for funding (RTG 2620 Ion Pair Effects in Molecular Reactivity, project number 426795949). J. B. and H. Z. acknowledge the computational and data resources provided by the Leibniz Supercomputing Centre (https://www.lrz.de). F. G. thanks the Fonds der Chemischen Industrie for a Kekulé Fellowship.Notes and references
- (a) D. Corbridge, Phosphorus. Chemistry, Biochemistry and Technology, Sixth Edition, CRC Press, 2013 Search PubMed; (b) M. A. de Boer, L. Wolzak and J. C. Slootweg in Phosphorus Recovery and Recycling, ed. H. Ohtake and S. Tsuneda, Springer Singapore, Singapore, 2019, pp. 75–100 Search PubMed; (c) N. Weferling, S. M. Zhang and C. H. Chiang, Procedia Eng., 2016, 138, 291 CrossRef CAS.
- (a) S. Hauer, T. M. Horsley Downie, G. Balázs, K. Schwedtmann, J. J. Weigand and R. Wolf, Angew. Chem., Int. Ed., 2024, 63, e202317170 CrossRef CAS PubMed; (b) F. Dielmann, M. Elsayed Moussa, C. Riesinger and M. Scheer, Z. Anorg. Allg. Chem., 2024, 650 Search PubMed; (c) S. Reichl, E. Mädl, F. Riedlberger, M. Piesch, G. Balázs, M. Seidl and M. Scheer, Nat. Commun., 2021, 12, 5774 CrossRef CAS PubMed; (d) S. Reichl, F. Riedlberger, M. Piesch, G. Balázs, M. Seidl and M. Scheer, Chem. Sci., 2023, 14, 7285 RSC; (e) M. Peruzzini, L. Gonsalvi and A. Romerosa, Chem. Soc. Rev., 2005, 34, 1038 RSC; (f) B. M. Cossairt, N. A. Piro and C. C. Cummins, Chem. Rev., 2010, 110, 4164 CrossRef CAS PubMed; (g) M. Caporali, L. Gonsalvi, A. Rossin and M. Peruzzini, Chem. Rev., 2010, 110, 4178 CrossRef CAS PubMed; (h) C. M. Hoidn, D. J. Scott and R. Wolf, Chem. Eur J., 2021, 27, 1886 CrossRef CAS PubMed; (i) L. Giusti, V. R. Landaeta, M. Vanni, J. A. Kelly, R. Wolf and M. Caporali, Coord. Chem. Rev., 2021, 441, 213927 CrossRef CAS; (j) K. Trabitsch, S. Hauer, K. Schwedtmann, P. Royla, J. J. Weigand and R. Wolf, Inorg. Chem. Front., 2025, 12, 2013 RSC; (k) M. Scheer, G. Balázs and A. Seitz, Chem. Rev., 2010, 110, 4236 CrossRef CAS PubMed.
- (a) O. J. Scherer, G. Schwarz and G. Wolmershäuser, Z. Anorg. Allg. Chem., 1996, 622, 951 CrossRef CAS; (b) S. Pelties, D. Herrmann, B. de Bruin, F. Hartl and R. Wolf, Chem. Commun., 2014, 50, 7014 RSC; (c) D. W. Agnew, C. E. Moore, A. L. Rheingold and J. S. Figueroa, Angew. Chem., Int. Ed., 2015, 54, 12673 CrossRef CAS PubMed; (d) M. J. Drance, S. Wang, M. Gembicky, A. L. Rheingold and J. S. Figueroa, Organometallics, 2020, 39, 3394 CrossRef CAS; (e) M. Haimerl, C. Graßl, M. Seidl, M. Piesch and M. Scheer, Chem. Eur J., 2021, 27, 18129 CrossRef CAS PubMed; (f) S. Heinl, S. Reisinger, C. Schwarzmaier, M. Bodensteiner and M. Scheer, Angew. Chem., Int. Ed., 2014, 53, 7639 CrossRef CAS PubMed; (g) R. J. Schwamm, M. Lein, M. P. Coles and C. M. Fitchett, Angew. Chem., Int. Ed., 2016, 55, 14798 CrossRef CAS PubMed; (h) J. Haberstroh, C. Taube, J. Fidelius, S. Schulz, N. Israel, E. Dmitrieva, R. M. Gomila, A. Frontera, R. Wolf, K. Schwedtmann and J. J. Weigand, Chem. Commun., 2024, 60, 8537 RSC; (i) I. Kovács, G. Baum, G. Fritz, D. Fenske, N. Wiberg, H. Schuster and K. Karaghiosoff, Z. Anorg. Allg. Chem., 1993, 619, 453 CrossRef; (j) S. Khan, R. Michel, J. M. Dieterich, R. A. Mata, H. W. Roesky, J.-P. Demers, A. Lange and D. Stalke, J. Am. Chem. Soc., 2011, 133, 17889 CrossRef CAS PubMed.
- (a) Y. Xiong, S. Yao, M. Brym and M. Driess, Angew. Chem., Int. Ed., 2007, 46, 4511 CrossRef CAS PubMed; (b) J. W. Dube, C. M. E. Graham, C. L. B. Macdonald, Z. D. Brown, P. P. Power and P. J. Ragogna, Chem. Eur J., 2014, 20, 6739 CrossRef CAS PubMed; (c) C. M. E. Graham, C. L. B. Macdonald, P. P. Power, Z. D. Brown and P. J. Ragogna, Inorg. Chem., 2017, 56, 9111 CrossRef CAS PubMed; (d) G. Prabusankar, A. Doddi, C. Gemel, M. Winter and R. A. Fischer, Inorg. Chem., 2010, 49, 7976 CrossRef CAS PubMed; (e) D. Reiter, P. Frisch, D. Wendel, F. M. Hörmann and S. Inoue, Dalton Trans., 2020, 49, 7060 RSC; (f) M. M. D. Roy, A. Heilmann, M. A. Ellwanger and S. Aldridge, Angew. Chem., Int. Ed., 2021, 60, 26550 CrossRef CAS PubMed; (g) D. Sarkar, C. Weetman, D. Munz and S. Inoue, Angew. Chem., Int. Ed., 2021, 60, 3519 CrossRef CAS PubMed; (h) P. Dabringhaus, T. Heizmann and I. Krossing, Chem. Eur J., 2023, 29, e202302212 CrossRef CAS PubMed; (i) D. Holschumacher, T. Bannenberg, K. Ibrom, C. G. Daniliuc, P. G. Jones and M. Tamm, Dalton Trans., 2010, 39, 10590 RSC; (j) J. J. Weigand, M. Holthausen and R. Fröhlich, Angew. Chem., Int. Ed., 2009, 48, 295 CrossRef CAS PubMed.
- M. Baudler, C. Adamek, S. Opiela, H. Budzikiewicz and D. Ouzounis, Angew. Chem., Int. Ed., 1988, 27, 1059 CrossRef.
- (a) M. M. Rauhut and A. M. Semsel, J. Org. Chem., 1963, 28, 471 CrossRef CAS; (b) M. M. Rauhut and A. M. Semsel, J. Org. Chem., 1963, 28, 473 CrossRef CAS; (c) M. M. Rauhut, R. Bernheimer and A. M. Semsel, J. Org. Chem., 1963, 28, 478 CrossRef CAS.
- K. X. Bhattacharyya, S. Dreyfuss, N. Saffon-Merceron and N. Mézailles, Chem. Commun., 2016, 52, 5179 RSC.
- J. E. Borger, A. W. Ehlers, M. Lutz, J. C. Slootweg and K. Lammertsma, Angew. Chem., Int. Ed., 2014, 53, 12836 CrossRef CAS PubMed.
- J. E. Borger, M. S. Bakker, A. W. Ehlers, M. Lutz, J. C. Slootweg and K. Lammertsma, Chem. Commun., 2016, 52, 3284 RSC.
- J. E. Borger, A. W. Ehlers, M. Lutz, J. C. Slootweg and K. Lammertsma, Angew. Chem., Int. Ed., 2017, 56, 285 CrossRef CAS PubMed.
- J. E. Borger, A. W. Ehlers, M. Lutz, J. C. Slootweg and K. Lammertsma, Angew. Chem., Int. Ed., 2016, 55, 613 CrossRef CAS PubMed.
- J. E. Borger, M. K. Jongkind, A. W. Ehlers, M. Lutz, J. C. Slootweg and K. Lammertsma, ChemistryOpen, 2017, 6, 350 CrossRef CAS PubMed.
- (a) C. G. P. Ziegler, T. M. Maier, S. Pelties, C. Taube, F. Hennersdorf, A. W. Ehlers, J. J. Weigand and R. Wolf, Chem. Sci., 2019, 10, 1302 RSC; (b) C. G. P. Ziegler, C. Taube, J. A. Kelly, G. Hierlmeier, M. Uttendorfer, J. J. Weigand and R. Wolf, Chem. Commun., 2020, 56, 14071 RSC; (c) Y. Liu, F. F. Westermair, I. Becker, S. Hauer, M. Bodensteiner, C. Hennig, G. Balázs, F. Meyer, R. M. Gschwind and R. Wolf, J. Am. Chem. Soc., 2025, 147, 7083 CrossRef CAS PubMed; (d) M. D. Fritz, J. A. Kelly, G. Balázs and R. Wolf, ChemistryEurope, 2026, 4, e202500257 CrossRef CAS.
- M. P. Blake, N. Kaltsoyannis and P. Mountford, Chem. Commun., 2013, 49, 3315 RSC.
- M. Garçon, C. Bakewell, A. J. P. White and M. R. Crimmin, Chem. Commun., 2019, 55, 1805 RSC.
- (a) H. Felkin, P. J. Knowles, B. Meunier, A. Mitschler, L. Ricard and R. Weiss, J. Chem. Soc., Chem. Commun., 1974, 44 RSC; (b) J. A. Kelly, J. Gramüller, R. M. Gschwind and R. Wolf, Dalton Trans., 2021, 50, 13985 RSC; (c) M. Gawron, F. Gilch, D. Schmidhuber, J. A. Kelly, T. M. Horsley Downie, A. Jacobi von Wangelin, J. Rehbein and R. Wolf, Angew. Chem., Int. Ed., 2024, 63, e202315381 CrossRef CAS PubMed; (d) V. R. Landaeta, T. M. Horsley Downie and R. Wolf, Chem. Rev., 2024, 124, 1323 CrossRef CAS PubMed.
- (a) M. Arrowsmith, M. S. Hill, A. L. Johnson, G. Kociok-Köhn and M. F. Mahon, Angew. Chem., Int. Ed., 2015, 54, 7882 CrossRef CAS PubMed; (b) S. Thum, O. P. E. Townrow, J. Langer and S. Harder, Chem. Sci., 2025, 16, 4528 RSC.
- M. J. Evans, D. T. Nguyen, J. M. Parr, J. Mullins, R. Mondal, T. Rajeshkumar, L. Maron and C. Jones, Chem, 2025, 102650 CAS.
- J. Bresien, K. Faust, C. Hering-Junghans, J. Rothe, A. Schulz and A. Villinger, Dalton Trans., 2016, 45, 1998 RSC.
- D. C. Meier, Á. García-Romero, D. González-Pinardo, N. H. Rees, A. Lovstedt, I. Fernández and J. M. Goicoechea, Chem. Sci., 2025, 17, 544 RSC.
- J. Contreras-García, E. R. Johnson, S. Keinan, R. Chaudret, J.-P. Piquemal, D. N. Beratan and W. Yang, J. Chem. Theory Comput., 2011, 7, 625 CrossRef PubMed.
- (a) H. S. Yu, X. He, S. L. Li and D. G. Truhlar, Chem. Sci., 2016, 7, 6278 RSC; (b) F. Weigend and R. Ahlrichs, Phys. Chem. Chem. Phys., 2005, 7, 3297 RSC; (c) A. V. Marenich, C. J. Cramer and D. G. Truhlar, J. Phys. Chem. B, 2009, 113, 6378 CrossRef CAS PubMed.
- (a) J. J. Turner, M. W. George, M. Poliakoff and R. N. Perutz, Chem. Soc. Rev., 2022, 51, 5300 RSC; (b) O. J. Scherer, T. Hilt and G. Wolmershäuser, Organometallics, 1998, 17, 4110 CrossRef CAS; (c) O. J. Scherer, T. Brück and G. Wolmershäuser, Chem. Ber., 1988, 121, 935 CrossRef CAS; (d) M. Scheer and U. Becker, J. Organomet. Chem., 1997, 545–546, 451 CrossRef CAS; (e) O. J. Scherer, R. Winter and G. Wolmershäuser, Z. Anorg. Allg. Chem., 1993, 619, 827 CrossRef CAS; (f) C. Eichhorn, O. J. Scherer, T. Sögding and G. Wolmershäuser, Angew. Chem., Int. Ed., 2001, 40, 2859 CrossRef CAS; (g) O. J. Scherer, J. Schwalb, G. Wolmershäuser, W. Kaim and R. Gross, Angew. Chem., Int. Ed., 1986, 25, 363 CrossRef.
- R. Grünbauer, C. Schwarzmaier, M. Eberl, G. Balázs and M. Scheer, Inorg. Chim. Acta, 2021, 518, 120234 CrossRef.
- M. Scheer, S. Deng, O. J. Scherer and M. Sierka, Angew. Chem., Int. Ed., 2005, 44, 3755 CrossRef CAS PubMed.
- R. Yadav, T. Simler, S. Reichl, B. Goswami, C. Schoo, R. Köppe, M. Scheer and P. W. Roesky, J. Am. Chem. Soc., 2020, 142, 1190 CrossRef CAS PubMed.
- J. A. Kelly, V. Streitferdt, M. Dimitrova, F. F. Westermair, R. M. Gschwind, R. J. F. Berger and R. Wolf, J. Am. Chem. Soc., 2022, 144, 20434 CrossRef CAS PubMed.
- A. S. Ionkin, W. J. Marshall, B. M. Fish, A. A. Marchione, L. A. Howe, F. Davidson and C. N. McEwen, Eur. J. Inorg. Chem., 2008, 2386 CrossRef CAS.
- M. J. Ernst, A. Petrov, M. Weber and C. Müller, ChemistryEurope, 2026, 4, e70252 CrossRef CAS.
- F. Hennersdorf and J. J. Weigand, Angew. Chem., Int. Ed., 2017, 56, 7858 CrossRef CAS PubMed.
- (a) H. Lei, J. C. Fettinger and P. P. Power, Organometallics, 2010, 29, 5585 CrossRef CAS; (b) S. Wang, M. L. McCrea-Hendrick, C. M. Weinstein, C. A. Caputo, E. Hoppe, J. C. Fettinger, M. M. Olmstead and P. P. Power, J. Am. Chem. Soc., 2017, 139, 6586 CrossRef CAS PubMed.
- (a) G. L. Dunn and J. K. Donohue, Tetrahedron Lett., 1968, 9, 3485 CrossRef; (b) G. O. Schenck and R. Steinmetz, Chem. Ber., 1963, 96, 520 CrossRef CAS.
- (a) M. Hofmann, C. Höhn, F. W. Heinemann and U. Zenneck, Chem. Eur J., 2009, 15, 5998 CrossRef CAS PubMed; (b) R. Bartsch, P. B. Hitchcock and J. F. Nixon, J. Chem. Soc., Chem. Commun., 1989, 1046 Search PubMed.
- C. Schwarzmaier, A. Y. Timoshkin, G. Balázs and M. Scheer, Angew. Chem., Int. Ed., 2014, 53, 9077 CrossRef CAS PubMed.
- (a) W. Huang and P. L. Diaconescu, Chem. Commun., 2012, 48, 2216 RSC; (b) S. N. Konchenko, N. A. Pushkarevsky, M. T. Gamer, R. Köppe, H. Schnöckel and P. W. Roesky, J. Am. Chem. Soc., 2009, 131, 5740 CrossRef CAS PubMed; (c) M. Scheer, U. Becker and E. Matern, Chem. Ber., 1996, 129, 721 CrossRef CAS; (d) M. E. Barr, B. R. Adams, R. R. Weller and L. F. Dahl, J. Am. Chem. Soc., 1991, 113, 3052 CrossRef CAS.
- A. D. Phillips, S. Hino and P. P. Power, J. Am. Chem. Soc., 2003, 125, 7520 CrossRef CAS PubMed.
- J. R. Pinkes and A. R. Cutler, Inorg. Chem., 1994, 33, 759 CrossRef CAS.
- (a) CCDC 2529860: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2qxjdl; (b) CCDC 2529861: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2qxjfm; (c) CCDC 2529862: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2qxjgn; (d) CCDC 2529863: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2qxjhp; (e) CCDC 2529864: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2qxjjq; (f) CCDC 2529865: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2qxjkr; (g) CCDC 2529866: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2qxjls; (h) CCDC 2529867: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2qxjmt; (i) CCDC 2529868: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2qxjnv; (j) CCDC 2529869: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2qxjpw; (k) CCDC 2529870: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2qxjqx; (l) CCDC 2529871: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2qxjry; (m) CCDC 2529872: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2qxjsz; (n) CCDC 2529873: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2qxjt0; (o) CCDC 2529874: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2qxjv1; (p) CCDC 2541029: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2r94px.
Footnotes |
| † The Fp*− anion in 2 is more electron-rich, which should favour the insertion product as demonstrated for the reaction of KFp* with CO2.37 |
| ‡ Complex 2 forms an Mg–OC–Fe bonded monomer upon crystallization from DME (Fig. S103) and a dimer with similar Mg–OC–Fe linkages upon crystallization from toluene (Fig. S101). Additionally, monomeric Mg–Fe-bonded structures have been obtained for 5-THF and 6-THF by crystallization from THF (Fig. S99 and S100). In line with previous results by Mountford and co-workers, these data indicate that the aggregation is reversible, depending on the solvent and the crystallization conditions.14 |
| § Lammertsma and co-workers have demonstrated that the reaction of LiFp* and P4 in THF affords a mixture of iron polyphosphides.12 |
| This journal is © The Royal Society of Chemistry 2026 |