Photo-induced intramolecular alkyl/aryl group transfer and SO2 insertion: a new strategy for the synthesis of 3-(alkyl/arylsulfonyl)benzothiophenes†
Tiantian
Xu
a,
Fen-Dou
Wang
a,
Wen-Chao
Yang
 *b,
Tong
Lu
a,
Min
Wang
*b,
Tong
Lu
a,
Min
Wang
 *a and
Pinhua
Li
*a and
Pinhua
Li
 *a
*a
aCollege of Chemical and Environmental Engineering, Anhui Polytechnic University, Wuhu, Anhui 241000, P. R. China. E-mail: wangmin204@chnu.edu.cn; pphuali@126.com
bSchool of Plant Protection, Yangzhou University, Yangzhou, 225009, P. R. China. E-mail: wccyang@126.com
First published on 4th February 2025
Abstract
A new and efficient strategy for the synthesis of 3-(alkyl/arylsulfonyl)benzothiophenes and benzoselenophenes via a photo-induced tandem cyclization of 2-alkynylthioanisoles or -selenoanisoles with DABSO through intramolecular alkyl/aryl group transfer and SO2 insertion was developed. The reaction proceeded smoothly under visible-light irradiation without any external photocatalyst, and generated the desired products in excellent yields with good functional-group tolerance.
Green foundation1. We present an efficient and eco-friendly methodology for the preparation of 3-(alkyl/arylsulfonyl)benzothiophenes and benzoselenophenes via a photoinduced tandem cyclization of 2-alkynylthioanisoles or -selenoanisoles with DABSO through intramolecular alkyl/aryl group transfer and SO2 insertion. This approach can offer the desired products in excellent yields with the leaving groups (sulphur atom attached groups) reutilized.2. Our work exhibits various advantages, including photocatalyst-free, metal-free, external addition free, high atom-economy and mild reaction conditions. 3. For greener future research, it is necessary to enhance the rates and efficiency of this photochemical process and expand the scope of 2-alkynylthioanisoles or -selenoanisoles. |
Introduction
Thioheterocycles, as important scaffolds, are present in many bioactive molecules, such as thiophene compounds, which exhibit significant activity in pharmaceuticals and agrochemicals (Scheme 1a).1 As a result, the construction of thioheterocycles has attracted considerable attention, and chemists have also witnessed significant advancements in this field over the past few decades.2 Among them, the cascade cyclization of 2-alkynylthioanisoles was recognised as one of the most efficient and convenient approach for the preparation of functionalized benzothiophenes.3 For example, Yu and co-workers reported a visible-light-induced metal-catalyst-free cascade reaction of alkynes for the construction of phosphorylated benzothiophenes.4 In 2018, an AgNO3-catalyzed radical cascade cyclization of 2-alkynylthioanisoles with α-oxocarboxylic acids has been described by Xia, Zhou and Wu's group.5 Meanwhile, other radical-triggered 1,2-difunctionalization of 2-alkynylthioanisoles with various radical precursors has also been utilized for the synthesis of functionalized benzothiophenes.6 However, all of these reactions have at least one drawback, such as the use of metal catalysts, oxidants, and harsh reaction conditions, low atom-economy (waste of methyl groups, Scheme 1b), etc. Green chemistry has emerged as a compelling research field, focusing on 12 principles, including atom economy, the use of environmentally friendly solvents, step economy, waste prevention, low-hazard synthesis and so on.7 Therefore, developing a new strategy to address the aforementioned issues would be highly meaningful. | ||
| Scheme 1 Bioactive sulfur-containing heterocycles and the developed synthetic strategies for the C3 substituted benzothiophenes. | ||
Synthetic chemists have utilized visible light to activate organic compounds, achieving various transformations that have altered traditional reaction pathways and promoted the development of new catalytic models.8 Since most organic molecules can only absorb visible light radiation in the high-energy ultraviolet region, it is often necessary to use costly dyes or transition metal-based photocatalysts to absorb low-energy visible light radiation and trigger the desired radical reactivity.9 This does not perfectly meet the demands of green chemistry development. Even though most of the above transformations have abandoned traditional metal catalysts, the reliance on photocatalysis still exhibits limitations in certain specific reactions. Recently, a photochemical strategy has been proposed that leverages charge transfer interactions between two colourless organic molecules, an electron-rich donor and an electron-deficient acceptor, to form EDA-complexes (electron donor–acceptor).10 This method bypasses the use of noble metal catalysts or dye-based photocatalysts. In addition to these reactions, achieving homolysis of specific covalent bonds through substrate interactions represents another promising strategy for green synthesis.11 Recently, DABSO (1,4-diazoniabicyclo[2.2.2]octane-1,4-disulfinate) has been considered as an indispensable sulfur dioxide surrogate, widely applied in organosulfonyl syntheses.12 In the past years, a variety of concise and robust synthetic protocols have been developed to access 3-(arylsulfonyl)benzothiophenes via a radical process, including the multi-component cascade reaction of 2-alkynylthioanisoles using DABSO, Na2S2O5, and K2S2O5 as sulfur dioxide surrogates.13 As part of our ongoing interests in the synthesis of heterocycles via radical cascade reactions,14 herein, we described an unprecedented radical cascade cyclization of 2-alkynylthioanisoles with DABSO through photo-induced intramolecular alkyl/aryl group transfer and SO2 insertion processes (Scheme 1c). We propose that the interactions of DABSO with 2-alkynylthioanisoles maybe formed the EDA complex, and they could weaken the C–S bond and result in the cleavage of the C–S bond more easily, facilitating the tandem transformation to access 3-organosulfonyl benzothiophenes with the sulphur atom protecting groups reutilized.
Results and discussion
For reaction optimization, methyl(2-(phenylethynyl)phenyl)-thioalkane (1a) and DABSO (1,4-diazoniabicyclo[2.2.2]octane-1,4-disulfinate, 2a) were selected as model substrates. Gratifyingly, the desired reaction occurred in acetonitrile with a 40 W blue light-emitting diode (LED) (427 nm) irradiation under an argon atmosphere (the reaction temperature is about 40–45 °C), affording the target product 3a in 80% yield. However, when a fan was used to reduce the reaction temperature, the yield of 3a dramatically decreased (Table 1, entry 1). In the absence of visible-light irradiation, no 3a was detected (entry 2), and 3a was generated in a lower yield when the reaction was performed under an air atmosphere (entry 3). To improve the synthetic efficiency, a series of solvents were evaluated, and CH3COOH was the best medium for the reaction, and the examined solvents such as chloroform, dichloroethane (DCE), chlorobenzene and methanol were all feasible for this reaction, affording 3a in 46–83% yields (entries 4–8). Unfortunately, no corresponding product was formed when acetone, DMF, THF and toluene were used as solvents (entries 9–12). The wavelength and power of the light source were also investigated, and it was found that a 427 nm LED (40 W) was the best choice (entries 13–15). The ratio of 1a to 2a as well as the reaction time were optimized, which are outlined in Table 1 (entries 16 and 17).| Entry | LED | Solvent | Yieldb (%) |
|---|---|---|---|
| a Reaction conditions: 1a (0.20 mmol) and DABSO (0.24 mmol, 1.2 equiv.) in solvent (2.0 mL) were irradiated with a 40 W blue LED in a reaction tube under a N2 atmosphere at 40–45 °C for 24 h unless otherwise stated. b Isolated yield. c Room temperature (a fan was used to reduce the reaction temperature). d In the dark at 50 °C (oil bath). e Under air atmosphere. f DABSO (0.20 mmol, 1.0 equiv.). g DABSO (0.30 mmol, 1.5 equiv.). h 18 h. i 30 h; n.d. = not detected. | |||
| 1 | 427 nm (40 W) | MeCN | 80, 12c |
| 2 | 427 nm (40 W) | MeCN | n.d.d |
| 3 | 427 nm (40 W) | MeCN | 33e |
| 4 | 427 nm (40 W) | CHCl3 | 83 |
| 5 | 427 nm (40 W) | DCE | 67 |
| 6 | 427 nm (40 W) | PhCl | 53 |
| 7 | 427 nm (40 W) | MeOH | 46 |
| 8 | 427 nm (40 W) | CH3COOH | 91 |
| 9 | 427 nm (40 W) | CH3COCH3 | Trace |
| 10 | 427 nm (40 W) | DMF | Trace |
| 11 | 427 nm (40 W) | THF | n.d. |
| 12 | 427 nm (40 W) | PhCH3 | n.d. |
| 13 | 427 nm (20 W) | CH3COOH | 51 |
| 14 | 440 nm (40 W) | CH3COOH | 85 |
| 15 | 390 nm (40 W) | CH3COOH | n.d. |
| 16 | 427 nm (40 W) | CH3COOH | 81f, 90g |
| 17 | 427 nm (40 W) | CH3COOH | 70h, 87i |
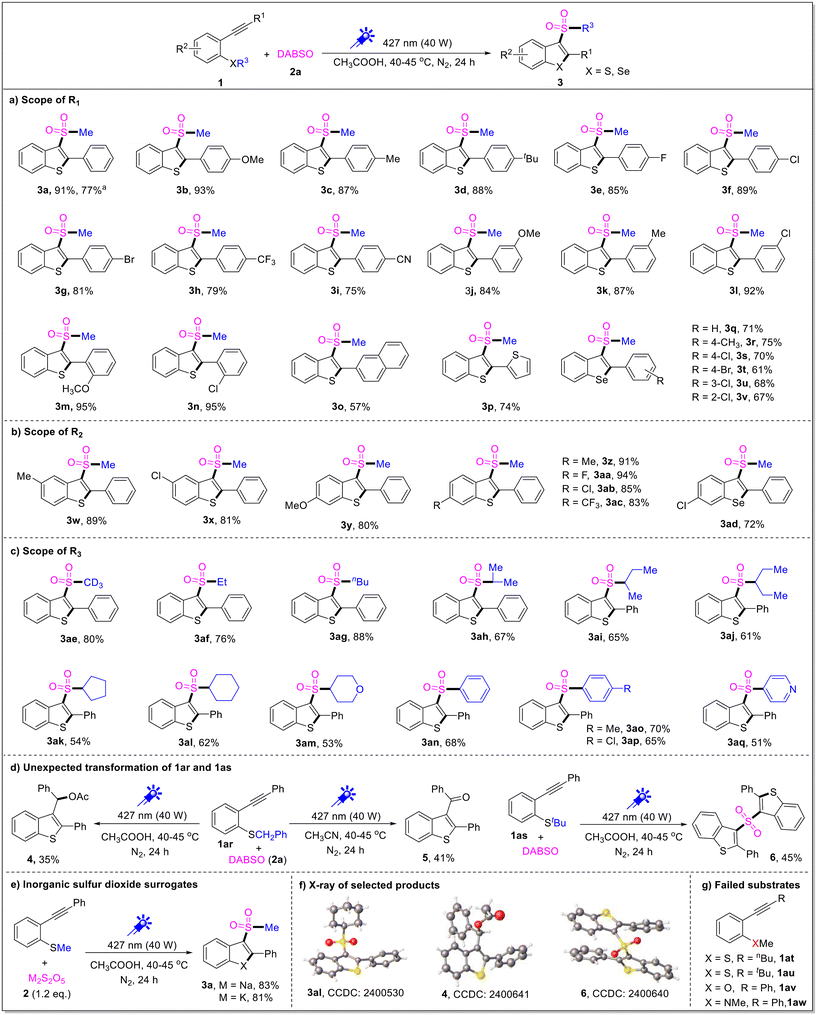
Next, the generality of this tandem cyclization reaction was investigated under the optimal reaction conditions. As shown in Scheme 2a, a variety of 2-alkynylthioanisoles with different groups on the aryl rings (R1) were examined, and both electron donating groups and electron-withdrawing groups on the phenyl ring (R1) showed great compatibility to give the corresponding products (3b–3n) in 75–91% yields. An electron-donating substituent (methoxy, tert-butyl and methyl etc.) favored the product formation, whereas a strongly electron-withdrawing group (cyano and trifluoromethyl) slightly hindered the reaction. Moreover, when R1 was replaced with naphthyl and thiophenyl groups, the two substrates were also amenable for the tandem reaction and the desired products 3o and 3p were obtained in 57% and 74% yields, respectively. However, when R1 was replaced with n-butyl (1at) or tert-butyl (1au) in the substrates, no desired products were detected and the starting materials were recovered (Scheme 2g). This result indicates that the R1 group must be an aromatic group in this transformation under current conditions. It is gratifying to note that the substituted 2-alkynylselenoanisoles were also suitable substrates to deliver the desired products (3q–3v) in 61–75% yields. Moreover, different substituents (R2) on the aromatic ring of thioanisole were also investigated, which is outlined in Scheme 2b; it seems that the electronic and steric effects could be ignored, and the targeted products (3w–3ac) were generated in 81–94% yields. Furthermore, (5-chloro-2-(phenylethynyl)phenyl)(methyl)-selane was also a good candidate for this reaction, giving the desired product 3ad in 72% yield.
To further extend the scope of this transformation, the R3 groups connected to the sulfur atom were also examined, as shown in Scheme 2c. Generally, the reaction proceeded smoothly under the standard conditions, both alkyl groups (methyl-d3, ethyl, isopropyl, cyclohexyl etc.) and aryl groups (phenyl, p-methylphenyl, p-chlorophenyl and 4-pyridyl) attached to the sulfur atom could be well-tolerated, and afforded the desired products (3ae–3aq) in 51–88% yields.
To our surprise, when benzyl(2-(phenylethynyl)phenyl)sulfane (1ar) was employed in this transformation under standard conditions, no desired product was obtained, but a tandem reaction product 4 was isolated in 35% yield. It is interesting that when acetonitrile was used as solvent, an oxidative cyclization product 5 became the main product (41% yield). It is also strange that tert-butyl(2-(phenylethynyl)phenyl)sulfane (1as) conducted unexpected transformation under standard conditions, and diaryl sulfone (6) was isolated in 45% yield (Scheme 2d). In addition, the structures of 3al, 4, and 6 were further confirmed by X-ray single crystal analysis (Scheme 2f). Unfortunately, 1-methoxy-2-(phenylethynyl)benzene (1av) and 1-(2-(phenylethynyl)phenyl)ethan-1-one (1aw) were no longer the effective substrates for this reaction; they failed in this transformation and the starting materials were recovered.
Notably, sodium pyrosulfate and potassium pyrosulfate could also be used as the sulfur dioxide surrogate instead of DABSO to deliver the desired product 3a in 83% and 81% yields, respectively (Scheme 2e). Additionally, the tandem reaction could be easily scalable, and when the reaction was carried out on a 5.0 mmol scale, 3a was isolated in 77% yield.
To understand the pathway of this intramolecular alkyl/aryl transfer and SO2 insertion tandem cyclization, mechanistic investigations and control experiments were performed, and the results are listed in Scheme 3 and the ESI.† At first, ultraviolet-visible spectroscopic measurements were performed on the different reaction components, and evidence for the interaction (maybe formation of an EDA complex) between 1an and DABSO was obtained, where a bathochromic shift was observed for a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
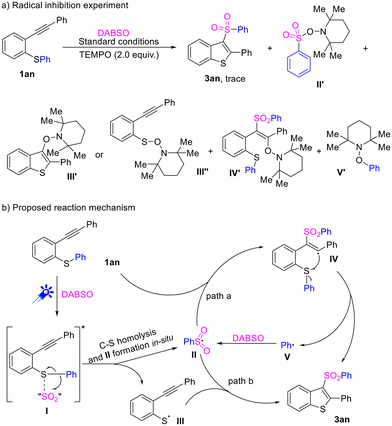
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 mixture of 1an and DABSO (Fig. S2†). The interaction of 1an and DABSO was also apparent from the obvious change in colour observed upon mixing their solutions. The fluorescence quenching experiments of 1a in the presence of DABSO were then performed (Fig. S3†), and the results revealed the expected decrease of emission after the addition of increasing amounts of DABSO. The Stern–Volmer plot of 1a fluorescence produces a linear fit with a quenching constant of Ksv = 46.67 mM−1, and this also confirms the formation of an EDA complex. Moreover, a light on/off experiment demonstrated that the transformation required continuous irradiation of visible light (Fig. S4†). Moreover, when the model reaction was conducted with the addition of 2.0 equiv. TEMPO (2,2,6,6-tetramethyl-1-piperidinyloxy) as a radical scavenger under the standard conditions, only a trace amount of product 3an was detected, and the adducts of radical intermediates (II′, III′, IV′, and V′) were detected by HRMS analysis of the reaction mixtures (see the ESI† for details), implying that a free-radical process might be involved in the reaction.
1 mixture of 1an and DABSO (Fig. S2†). The interaction of 1an and DABSO was also apparent from the obvious change in colour observed upon mixing their solutions. The fluorescence quenching experiments of 1a in the presence of DABSO were then performed (Fig. S3†), and the results revealed the expected decrease of emission after the addition of increasing amounts of DABSO. The Stern–Volmer plot of 1a fluorescence produces a linear fit with a quenching constant of Ksv = 46.67 mM−1, and this also confirms the formation of an EDA complex. Moreover, a light on/off experiment demonstrated that the transformation required continuous irradiation of visible light (Fig. S4†). Moreover, when the model reaction was conducted with the addition of 2.0 equiv. TEMPO (2,2,6,6-tetramethyl-1-piperidinyloxy) as a radical scavenger under the standard conditions, only a trace amount of product 3an was detected, and the adducts of radical intermediates (II′, III′, IV′, and V′) were detected by HRMS analysis of the reaction mixtures (see the ESI† for details), implying that a free-radical process might be involved in the reaction.
According to the above mechanistic studies, a plausible mechanism for the photo-induced intramolecular alkyl/aryl transfer and SO2 insertion tandem cyclization was depicted with 1an and DABSO, as shown in Scheme 3. Initially, 1an interacts with DABSO, maybe to generate an EDA complex I. At the same time, tertiary amine cations in DABSO interact with the sulfur atom in 1an and thus make the cleavage of the C–S bond easier. Subsequently, the intermediate I was excited under the irradiation of visible light, and it underwent a direct homolytic cleavage to produce free radicals III and V. The released phenyl radical (V) captures sulfur dioxide and generates methanesulfonyl radical II, then II attacks the triple bond of 1an and produces a free radical intermediate IV, which forms a C–S bond to deliver the desired product 3an releasing phenyl radicals (V), which continue to participate in the further free radical transfer process (path a). On the other hand, the formed radical intermediates II and III could undergo a radical addition and coupling sequence to afford the desired product (path b).
Conclusions
In summary, an efficient photo-induced intramolecular alkyl/aryl transfer and SO2 insertion tandem cyclization of 2-alkynylthioanisoles or -selenoanisoles with DABSO was developed. Following this protocol, a variety of 3-(alkyl/arylsulfonyl)benzothiophenes and benzoselenophenes were synthesized in moderate to excellent yields. This straightforward method exhibits several advantages, such as the use of photocatalyst-free, metal-free, and mild reaction conditions, high atom economy, and broad substrate scope. The preliminary mechanism study shows that the reaction is carried out through a free-radical process. Further studies to gain an in-depth understanding of the reaction mechanism and the new synthetic applications of this strategy are ongoing in our laboratory.Data availability
The data supporting this article have been included as part of the ESI.†Conflicts of interest
There are no conflicts to declare.Acknowledgements
We gratefully acknowledge the Research Fund of Anhui Polytechnic University (2023YQQ0001) and the University Synergy Innovation Program of Anhui Province (No. GXXT-2020-078) for financial support of this work.References
- (a) I. P. Beletskaya and V. P. Ananikov, Chem. Rev., 2022, 122, 16110–16293 Search PubMed; (b) W. Yang, M. Zhang, W. Chen, X. Yang and J. Feng, Chin. J. Org. Chem., 2020, 40, 4060–4070 Search PubMed; (c) K. Abe, T. Myoda and S. Nojima, J. Agric. Food Chem., 2021, 69, 1020–1026 Search PubMed; (d) W. Zhao, S. Zheng, J. Zou, Y. Liang, C. Zhao and H. Xu, J. Agric. Food Chem., 2021, 69, 5798–5803 Search PubMed; (e) P. Devendar and G.-F. Yang, Top. Curr. Chem., 2017, 375, 82–126 Search PubMed; (f) M. Wang and X. Jiang, ACS Sustainable Chem. Eng., 2022, 10, 671–677 Search PubMed.
- (a) S. Huang, M. Wang and X. Jiang, Chem. Soc. Rev., 2022, 51, 8351–8377 Search PubMed; (b) N. N. Makhova, L. I. Belen'kii, G. A. Gazieva, I. L. Dalinger, L. S. Konstantinova, V. V. Kuznetsov, A. N. Kravchenko, M. M. Krayushkin, O. A. Rakitin and A. M. Starosotnikov, Russ. Chem. Rev., 2020, 89, 55–124 Search PubMed; (c) S. Gharge and S. G. Alegaon, Chem. Biodiversity, 2024, 21, 202301738 Search PubMed; (d) M. Wu, C. Yan, D. Zhuang and R. Yan, Org. Lett., 2022, 24, 5309–5313 Search PubMed; (e) Y.-G. Ji, Z.-H. Li, Y.-Q. Yang, X. Yang and W.-C. Yang, Chem. – Eur. J., 2024, 30, e202402891 Search PubMed; (f) S.-P. Zhang, D.-W. Guo, M.-L. Yang, Y.-T. Xia and W.-C. Yang, J. Org. Chem., 2024, 89, 10614–10623 Search PubMed; (g) W.-C. Yang, K. Wei, X. Sun, J. Zhu and L. Wu, Org. Lett., 2018, 20, 3144–3147 Search PubMed; (h) Y. Lv, J. Meng, C. Li, X. Wang, Y. Ye and K. Sun, Adv. Synth. Catal., 2021, 363, 5235–5265 Search PubMed; (i) K. Sun, X. Wang, C. Li, H. Wang and L. Li, Org. Chem. Front., 2020, 7, 3100–3119 Search PubMed; (j) W.-C. Yang, Y. Sun, L.-Y. Shen, X. Xie and B. Yu, Mol. Catal., 2023, 535, 112819 Search PubMed; (k) F.-L. Zeng, K.-C. Xie, Y.-T. Liu, H. Wang, P.-C. Yin, L.-B. Qu, X.-L. Chen and B. Yu, Green Chem., 2022, 24, 1732–1737 Search PubMed.
- for reviews see: (a) J. Huang, Z. Chen and J. Wu, ACS Catal., 2021, 11, 10713–10732 Search PubMed; (b) T. Shang, C. Ma, M. Xie, Y. Gao and T. Cai, Eur. J. Org. Chem., 2024, e202401059 Search PubMed.
- X.-Y. Yuan, F.-L. Zeng, H.-L. Zhu, Y. Liu, Q.-Y. Lv, X.-L. Chen, L. Peng and B. Yu, Org. Chem. Front., 2020, 7, 1884–1889 Search PubMed.
- W. Liu, Y.-Q. Hu, X.-Y. Hong, G.-X. Li, X.-B. Huang, W.-X. Gao, M.-C. Liu, Y. Xia, Y.-B. Zhou and H.-Y. Wu, Chem. Commun., 2018, 54, 14148–14151 Search PubMed.
- (a) Y. Ren, Z. An, P. Zhao, M. Li and R. Yan, Org. Chem. Front., 2022, 9, 4085–4090 Search PubMed; (b) M. Li, T. Wang, Z. An and R. Yan, Chem. Commun., 2020, 56, 11953–11956 Search PubMed; (c) T. Cai, J. Liu, H. Zhang, X. Wang, J. Feng, R. Shen and Y. Gao, Org. Lett., 2019, 21, 4605–4608 Search PubMed; (d) W. Liu, Y.-Q. Hu, X.-Y. Hong, G.-X. Li, X.-B. Huang, W.-X. Gao, M.-C. Liu, Y. Xia, Y.-B. Zhou and H.-Y. Wu, Chem. Commun., 2018, 54, 14148–14151 Search PubMed; (e) D. J. Faizi, A. J. Davis, F. B. Meany and S. A. Blum, Angew. Chem., Int. Ed., 2016, 55, 14286–14290 Search PubMed; (f) D. Yue and R. C. Larock, J. Org. Chem., 2002, 67, 1905–1909 Search PubMed.
- J. B. Zimmerman, P. T. Anastas, H. C. Erythropel and W. Leitner, Science, 2020, 367, 397–400 Search PubMed.
- For selected reviews and examples: (a) G. Yue and B. Liu, Tetrahedron Chem, 2024, 9, 100067 Search PubMed; (b) F. Medici, V. Chiroli, L. Raimondi and M. Benaglia, Tetrahedron Chem, 2024, 9, 100061 Search PubMed; (c) X.-Y. Yu, Q.-Q. Zhao, J. Chen, W.-J. Xiao and J.-R. Chen, Acc. Chem. Res., 2020, 53, 1066–1083 Search PubMed; (d) T.-Y. Shang, L.-H. Lu, Z. Cao, Y. Liu, W.-M. He and B. Yu, Chem. Commun., 2019, 55, 5408–5419 Search PubMed; (e) W.-Q. Liu, T. Lei, S. Zhou, X.-L. Yang, J. Li, B. Chen, J. Sivaguru, C.-H. Tung and L.-Z. Wu, J. Am. Chem. Soc., 2019, 141, 13941–13947 Search PubMed; (f) Y. Liu, K. Zhu, X. Li, X. Wu, J. Feng, A. Roosta, Q. Wu and D. Zhu, Tetrahedron Chem, 2024, 11, 100083 Search PubMed; (g) H. Song, J. Jiang, C. Wu, J. Hou, Y. Lu, K. Wang, T. Yang and W.-M. He, Green Chem., 2023, 25, 3292–3296 Search PubMed; (h) M. Chiminelli, G. Scarica, D. Balestri, L. Marchiò, N. Della Ca’ and G. Maestri, Tetrahedron Chem, 2023, 8, 100053 Search PubMed.
- M. Huang, G. Wang, H. Li, Z. Zou, X. Jia, G. Karotsis, Y. Pan, W. Zhang, J. Ma and Y. Wang, Green Chem., 2025, 27, 413–419 Search PubMed.
- (a) E. Arceo, I. D. Jurberg, A. álvarez-Fernández and P. Melchiorre, Nat. Chem., 2013, 5, 750–756 Search PubMed; (b) K. Sun, A. Shi, Y. Liu, X. Chen, P. Xiang, X. Wang, L. Qu and B. Yu, Chem. Sci., 2022, 13, 5659–5666 Search PubMed; (c) G. E. M. Crisenza, D. Mazzarella and P. Melchiorre, J. Am. Chem. Soc., 2020, 142, 5461–5476 Search PubMed; (d) S. V. Rosokha and J. K. Kochi, Acc. Chem. Res., 2008, 41, 641–653 Search PubMed.
- (a) S. H. Druker and M. D. Curtis, J. Am. Chem. Soc., 1995, 117, 6366–6367 Search PubMed; (b) K. Luo, W. Yang, K. Wei, Y. Liu, J. Wang and L. Wu, Org. Lett., 2019, 21, 7851–7856 Search PubMed; (c) X. Gong, M. Wang, S. Ye and J. Wu, Org. Lett., 2019, 21, 1156–1160 Search PubMed.
- For selected reviews: (a) Z. Wu and D. A. Pratt, Nat. Rev. Chem., 2023, 7, 573–589 Search PubMed; (b) D. Joseph, M. AliyuIdris, J. Chen and S. Lee, ACS Catal., 2021, 11, 4169–4204 Search PubMed; (c) Q. Tang, X. Yin, R. R. Kuchukulla and Q. Zeng, Chem. Rec., 2021, 21, 893–905 Search PubMed; (d) S. Liang, K. Hofman, M. Friedrich and G. Manolikakes, Eur. J. Org. Chem., 2020, 4664–4676 Search PubMed; (e) Y.-J. Zhang, M.-L. Li, H.-X. Hu and F. Teng, Org. Biomol. Chem., 2024, 22, 5868–5885 Search PubMed; (f) M. S. Hashtroudi, V. F. Vavsari and S. Balalaie, Org. Biomol. Chem., 2022, 20, 2149–2163 Search PubMed; (g) G. Qiu, K. Zhou, L. Gao and J. Wu, Org. Chem. Front., 2018, 5, 691–705 Search PubMed.
- (a) J. Xu, X. Yu, J. Yan and Q. Song, Org. Lett., 2017, 19, 6292–6295 Search PubMed; (b) J. Yan, J. Xu, Y. Zhou, J. Chen and Q. Song, Org. Chem. Front., 2018, 5, 1483–1487 Search PubMed; (c) D. Zhang, J. Cai, J. Du, X. Wang, W. He, Z. Yang, C. Liu, Z. Fang and K. Guo, J. Org. Chem., 2021, 86, 2593–2601 Search PubMed; (d) V. S. Bhat and A. Lee, Adv. Synth. Catal., 2023, 365, 1514–1520 Search PubMed.
- (a) F.-D. Wang, J. Jiang, T. Xu, W.-C. Yang, S.-P. Zhang, M. Wang and P. Li, Tetrahedron Chem, 2025, 13, 100119 Search PubMed; (b) W.-C. Yang, Y. Sun, X. Bao, S.-P. Zhang and L.-Y. Shen, Green Chem., 2023, 25, 3111–3116 Search PubMed; (c) S.-P. Zhang, J.-X. Lan, M.-L. Yang, J.-Y. Cao and W.-C. Yang, Org. Lett., 2024, 26, 9990–9995 Search PubMed; (d) F. Meng, Y. Cui, W. Xu and W.-C. Yang, Org. Lett., 2024, 26, 6884–6888 Search PubMed; (e) Z. Wang, J.-L. Li, S.-P. Zhang and W.-C. Yang, Mol. Catal., 2023, 549, 113469 Search PubMed; (f) F.-D. Wang, C. Wang, M. Wang, H. Yan, J. Jiang and P. Li, Org. Biomol. Chem., 2023, 21, 8170–8175 Search PubMed; (g) H. Yan, F.-D. Wang, M. Wang, L. Ye and P. Li, J. Org. Chem., 2023, 88, 15288–15297 Search PubMed.
Footnote |
| † Electronic supplementary information (ESI) available. CCDC 2400530, 2400641 and 2400640. For ESI and crystallographic data in CIF or other electronic format see DOI: https://doi.org/10.1039/d4gc06127f |
| This journal is © The Royal Society of Chemistry 2025 |