Photoelectrochemical nickel-catalyzed carboacylation/silanoylation of alkenes with unactivated C/Si–H bonds†
Lanfen
Wang‡
a,
Xiangyu
Huo‡
c,
Xiaozhi
He
a,
Lutz
Ackermann
 *b and
Dingyi
Wang
*b and
Dingyi
Wang
 *a
*a
aDepartment of Chemistry, College of Sciences, Northeastern University, Shenyang 110819, China. E-mail: wangdingyi@mail.neu.edu.cn
bInstitut für Organische und Biomolekulare Chemie, Georg-August-Universität Göttingen, Tammannstraße 2, Göttingen 37077, Germany. E-mail: Lutz.Ackermann@chemie.uni-goettingen.de
cState Key Laboratory and Institute of Elemento-Organic Chemistry, College of Chemistry, Nankai University, Tianjin 300071, China
First published on 13th February 2024
Abstract
Direct and selective installation of two different functional groups into the unsaturated bonds of alkenes constitutes a long-standing goal in organic synthesis. Herein, we first report a photoelectrochemical approach for the carboacylation/silanoylation of alkenes with unactivated alkanes/hydrosilanes via the LMCT process. A number of ketones with high complexity and diversity were rapidly obtained from commercially available materials in an economical and sustainable manner. Furthermore, this protocol can be scaled up to the gram-level. This paired electrocatalysis strategy features excellent regioselectivity, and ample substrate scope, is external-oxidant-free, and features a high resource economy. A series of mechanistic experiments were performed to determine the preferred pathway for the photoelectrochemical Ni(I)/Ni(II)/Ni(III) cycles in these transformations.
Introduction
Carbon–carbon bonds form the fundamental backbones of organic compounds, so constructing them efficiently is one of the central goals of synthetic chemistry.1 Carbofunctionalization of alkenes can effectively construct carbon–carbon bonds by directly and selectively introducing two different carbon fragments into the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C double bond.2 Recently, impressive progress has been made in the radical-based carbofunctionalization of alkenes by cooperative nickelaphotoredox catalysis. Many chemists have performed representative work in this field.3,4 Despite the success of the direct alkene 1,2-difunctionalization process, these strategies often involve the utilization of prefunctionalized alkyl substrates, such as alkyl halides, alkyl trifluoro borates, alkyl silicates, α-silyl amines, or oxalate esters, which translate into the low efficiency and low atomic economy (Scheme 1a). To overcome these major drawbacks, photochemical alkene difunctionalization by employing unactivated aliphatic C/Si–H bonds as radical precursors has been reported by Kong,5 Wu,6 Li,7 and our group.8 Nevertheless, these methods rely heavily on the use of expensive photocatalysts, such as tetrabutylammonium decatungstates, 2,4,5,6-tetrakis(carbazol-9-yl)-1,3-dicyano benzene, and [Ir(dtbbpy)[dF(CF3)ppy]2]PF6, which lead to uncontrollable economic costs (Scheme 1a). Therefore, developing general and efficient synthetic strategies to achieve direct alkene 1,2-difunctionalization in an economical manner is attractive and urgently needed.
C double bond.2 Recently, impressive progress has been made in the radical-based carbofunctionalization of alkenes by cooperative nickelaphotoredox catalysis. Many chemists have performed representative work in this field.3,4 Despite the success of the direct alkene 1,2-difunctionalization process, these strategies often involve the utilization of prefunctionalized alkyl substrates, such as alkyl halides, alkyl trifluoro borates, alkyl silicates, α-silyl amines, or oxalate esters, which translate into the low efficiency and low atomic economy (Scheme 1a). To overcome these major drawbacks, photochemical alkene difunctionalization by employing unactivated aliphatic C/Si–H bonds as radical precursors has been reported by Kong,5 Wu,6 Li,7 and our group.8 Nevertheless, these methods rely heavily on the use of expensive photocatalysts, such as tetrabutylammonium decatungstates, 2,4,5,6-tetrakis(carbazol-9-yl)-1,3-dicyano benzene, and [Ir(dtbbpy)[dF(CF3)ppy]2]PF6, which lead to uncontrollable economic costs (Scheme 1a). Therefore, developing general and efficient synthetic strategies to achieve direct alkene 1,2-difunctionalization in an economical manner is attractive and urgently needed.
 | ||
| Scheme 1 Alkene functionalization via the LMCT process and the development of (photo)electrochemistry. | ||
Recently, several groups have demonstrated that earth-abundant and inexpensive metal-chloride-based photocatalysts are powerful tools for versatile aliphatic C–H functionalization through the intermolecular hydrogen atom transfer (HAT) (Scheme 1b). Recent progress in this field from the groups of Rovis,9 Walsh and Schelter,10 Zuo,11 Gong,12 Zeng,13 Xia,14 and others15 has involved the use of these metal chlorides to generate Cl radicals via ligand-to-metal charge transfer (LMCT). For instance, Rovis and Gong disclosed two elegant and practical CuCl2/FeCl3-catalyzed approaches for C(sp3)–H bond alkylation by using activated alkenes as alkylation reagents. However, studies on the use of these metal chlorides to difunctionalize alkenes with aliphatic C–H bonds via the photoinduced LMCT process has never been reported. With the rapid development of electrochemistry and photochemistry,16 photoelectrocatalysis has received considerable attention from scientists. The main feature of this approach is that it combines the advantages of electrochemistry and photochemistry in the same reaction. Several groups have systematically studied photoelectrocatalysis, and provided novel synthetic strategies for organic synthetic chemistry. Representative work from the groups of Lambert,17 Lin,18 Lei,19 Xu,20etc.21 has proven the synthetic potential of these strategies. Although significant progress has been made, most methods have focused on two-component nonmetallic radical-mediated cross-coupling reactions or intramolecular cyclization reactions (Scheme 1b). This greatly limits the application value of these methods. In addition, in (photo)electrochemical alkene difunctionalization reactions, the anode supports the catalyst in the catalytic cycle, while the cathode merely generates hydrogen. This results in additional stages and waste creation, as well as decreased efficiency and atom economy (Scheme 1c).
In 2023, Xu and co-workers reported the photoelectro-chemical asymmetric copper-catalyzed heteroarylcyanation of alkenes.22 Very recently, the Lu group disclosed an elegant approach for the selective C(sp3)–H arylation/alkylation of alkanes by photoelectrocatalysis.23 In this context, direct functionalization of C–H bonds, which are the most abundant chemical bonds in organic molecules, by photoelectrocatalytic strategies using inexpensive and readily available catalysts and reagents for building C–H/X bonds would be more attractive. Herein, we report a photoelectrochemical approach for the difunctionalization of alkenes with unactivated alkanes, hydrosilanes, and a range of aryl chlorides via the LMCT process (Scheme 1d). In contrast to previous photochemical nickel-catalyzed alkene difunctionalizations, which require the use of noble photocatalysts, we used the cheap and readily available FeCl3 as a photocatalyst. Furthermore, by combining the advantages of electrochemistry, the catalytic cycling of iron and nickel catalysts can be achieved using green electrons as oxidants. This strategy is more practical and far less economically costly than previous methods. This paired electrocatalysis strategy features excellent regioselectivity, ample substrate scope, and high resource economy.
Results and discussion
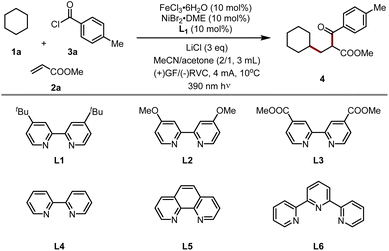
To explore the optimal photoelectrochemical conditions for three-component alkene 1,2-difunctionalizations, we used commercially available cyclohexane (1a), methyl acrylate (2a), and p-toluoyl chloride (3a) as model substrates in the presence of dual catalysts (FeCl3·6H2O and NiBr2·DME) (Table 1). After several experiments, the electrolysis proceeded smoothly in the presence of FeCl3·6H2O as the photocatalyst, NiBr2·DME as the nickel catalyst, and 4,4-di-tert-butyl bipyridine (dtbbpy) as the ligand, a mixture solvent of MeCN/acetone (2/1, 3 mL) at 10 °C in an undivided cell with a constant current (4 mA) exposed to 390 nm LEDs. Under the above conditions, the carbonylation product 4 was isolated in 71% yield with excellent regioselectivity (entry 1). When FeCl3·6H2O was replaced with other photocatalysts, such as CuCl2 and CeCl3, the target product was detected only in trace amounts (entries 2 and 3). This is because these metal salts readily precipitate at the cathode. Switching to other nickel salts as catalysts, such as NiCl2·DME or NiBr2, leads to shutting down the reactivity (entries 4 and 5). Additionally, 4,4′-di-tert-butyl-2-2′-bipyridine (dtbbpy) shows the best reactivity compared to the other pyridine-type ligands bearing different electronic effects, such as L2, L3, L4, L5, and L6 (entry 6, please see the ESI†). Further studies revealed that the addition of chlorine salts increased the concentration of the anion [FeCl4]− and thereby improved the reaction efficiency, and LiCl shows the best outcome (entry 7). The use of other commonly used solvents, such as DMA, DMF, TFE, and HFIP, leads to completely shutting down the transformation (entry 8). It should be noted that the highest yields were obtained when the volume ratio of MeCN/acetone was set to 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, while deviations from this ratio resulted in lower reactivity (entries 9 and 10). Altering the anode or cathode exhibited lower reactivity (entries 11–13). The cascade reaction became inefficient when the current was increased from 5 to 6 mA (entry 14). Finally, control experiments indicated that this electrolysis did not occur without light irradiation, electricity, paired catalysts, or ligands (entries 15 and 16).
1, while deviations from this ratio resulted in lower reactivity (entries 9 and 10). Altering the anode or cathode exhibited lower reactivity (entries 11–13). The cascade reaction became inefficient when the current was increased from 5 to 6 mA (entry 14). Finally, control experiments indicated that this electrolysis did not occur without light irradiation, electricity, paired catalysts, or ligands (entries 15 and 16).
| Entry | Variation from “standard conditions” | Yieldb (%) |
|---|---|---|
| a Reaction conditions: 1a (3.0 mmol), 2a (0.9 mmol), 3a (0.3 mmol), NiBr2·DME (10 mol%), FeCl3·6H2O (10 mol%), dtbbpy (10 mol%), LiCl (3.0 equiv.), MeCN/acetone (3.0 mL, 2/1), 4 mA, 24 h, 390 nm, 10 °C, nitrogen, graphite felt (GF) as an anode, reticulated vitreous carbon (RVC) as a cathode, undivided cell. b GC yields using dodecane as an internal standard. c Isolated yields. | ||
| 1 | None | 71c |
| 2 | CuCl2 instead of FeCl3·6H2O | Trace |
| 3 | CeCl3 instead of FeCl3·6H2O | Trace |
| 4 | NiCl2·DME instead of NiBr2·DME | 62 |
| 5 | NiBr2·DME instead of NiBr2 | 52 |
| 6 | L2–L6 as ligand | 0–55 |
| 7 | TBACl instead of LiCl | 17 |
| 8 | DMF/DMA/DMSO/HFIP as solvent | 0 |
| 9 | MeCN/acetone (1/1) | 64 |
| 10 | MeCN/acetone (1/2) | 55 |
| 11 | (+)GF/(−)GF | 60 |
| 12 | (+)GF/(−)Ni foam | 15 |
| 13 | (+)Fe/(−)RVC | <5 |
| 14 | I = 6 mA | 57 |
| 15 | w/o NiBr2·DME or FeCl3·6H2O or L1 | 0 |
| 16 | w/o light or current | 0 |
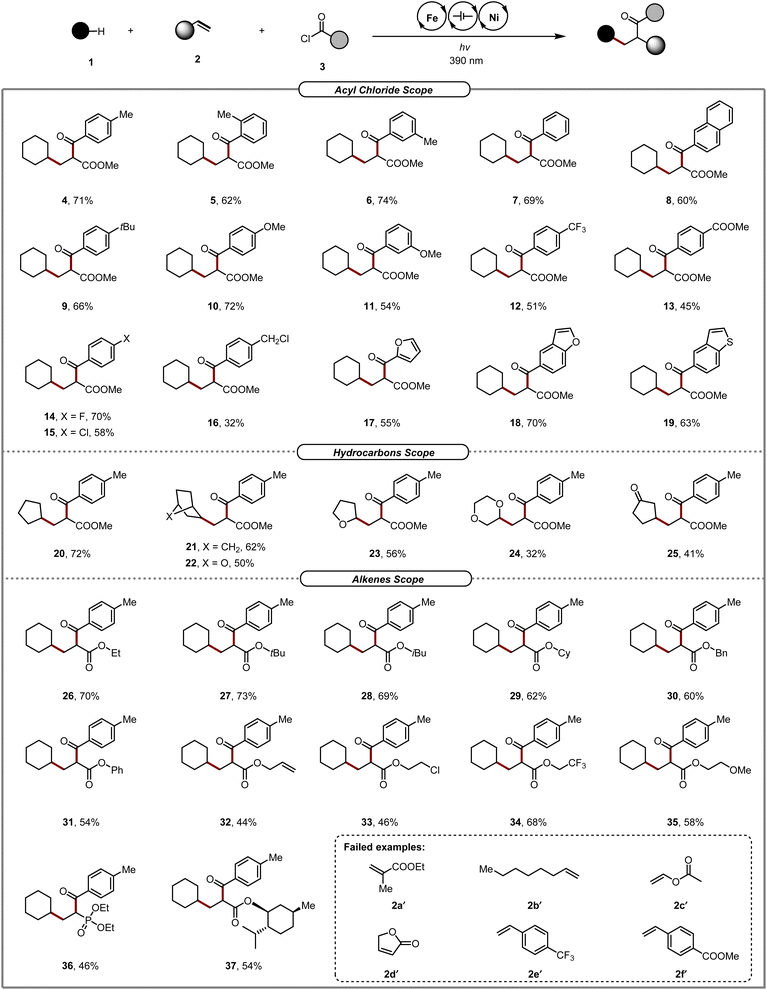
With the optimal reaction conditions in hand, we investigated the range of acyl chloride 3 compatible with the three-component electrophotocatalytic strategy. As shown in Scheme 2, a series of acyl chlorides bearing different groups and a range of electronic properties at the meta- and para-positions of the aromatic ring were compatible with this transformation. Electron-donating groups (Me, tBu, MeO), neutral groups (H, Naphth), electron-withdrawing groups (COOMe, CF3), and halogens (F, Cl) all exhibited modest to good yields and high enantioselectivities (4–15). The sterically hindered o-toluoyl chlorides were also tolerated well, providing the corresponding product (5) in 62%. Notably, the method was also useful in transformations for the more challenging methyl chloride substituents of acyl chlorides (16). In addition to aryl chlorides, various heteroaromatic acyl chlorides, such as furans, benzofurans, and benzothiophenes, were coupled smoothly with cyclohexane 1a and acrylic ester 2a, furnishing the targeted dicarbofunctionalization products (17–19) in good yields. In general, electron-rich acyl chlorides exhibited higher yields than electron-deficient substrates.
Next, the reactivities of hydrocarbons in this electrophotocatalytic system were examined. The electrophotochemical alkene difunctionalization reaction tolerated various unactivated C–H precursors with different skeletons such as cyclopentane and bicyclo[2.2.1]heptane. In addition, a number of oxygen-containing substrates, such as (1s,4s)-7-oxabicyclo[2.2.1]heptane, tetrahydrofuran, and 1,4-dioxane, all participated smoothly in the electrophotocatalytic process, affording the target carbonyl compounds (22–24) with good yields and excellent regioselectivities. Additionally, cyclopentanone was smoothly transformed into the corresponding product 25.
Various alkenes were studied next to further evaluate the substrate scope of this electrophotocatalytic protocol. A broad range of acrylates containing ethyl, tert-butyl, cyclohexyl, phenyl, benzyl, allyl, chloroalkyl, trifluoroalkyl, and alkoxy groups can be employed as effective coupling partners for these transformations. Our method was suitable not only for acrylic esters but also for other alkene acceptors. For instance, diethyl vinyl phosphonate was also well tolerated and delivered the corresponding product (36) in moderate yields. To our delight, this synergistic protocol was successfully applied to complex acrylate derived from natural products such as D-menthol (37). However, sterically hindered acrylates (2a′ and 2d′), unactivated alkenes (2b′ and 2c′), and styrene derivatives (2e′ and 2f′), were not tolerated in this transformation.
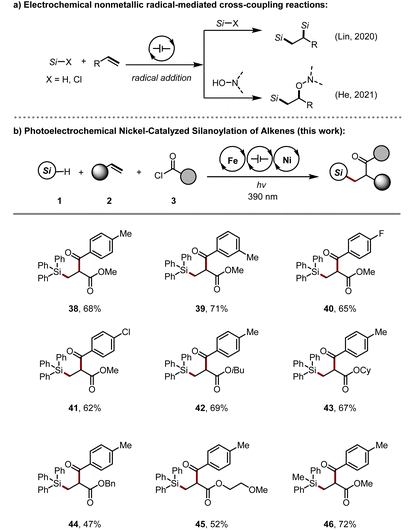
Organosilicon compounds are one of the most attractive structural motifs frequently found in the fields of agrochemistry, materials science, and medicinal chemistry, as well as serve as versatile synthetic intermediates widely used for diverse transformations in synthetic chemistry. Among the many approaches for synthesizing organosilicon compounds,24 electrochemical 1,2-silylfunctionalization of alkenes by using hydrosilanes as substrates are rare. The group of Lin and He disclosed electrochemical alkene silylation reactions under metal-free conditions (Table 2a).25 Since then, there have been no further reports on the silylation of alkenes utilizing hydrosilanes via electrochemical strategies. After the successful carboacylation of alkenes via a radical process, we envisioned that hydrosilanes could also be derived under standard conditions (Table 2b). To our delight, silanoylation of alkenes could be achieved under slightly modified conditions. Subsequently, various silanes and acyl chlorides with different groups were examined, and the results showed good reactivity, affording the corresponding products (38–41, 46) without any problems. Besides, various acrylates with diverse ester O-substituents, including Bn, and alkoxy groups (42–45) are perfectly compatible with this methodology, affording the desired ketones in 47%–72% yields.
| a Reaction conditions: 1 (1.5 mmol), 2 (0.9 mmol), 3 (0.3 mmol), NiBr2·DME (10 mol%), FeCl3·6H2O (10 mol%), dtbbpy (10 mol%), LiCl (3.0 equiv.), MeCN/acetone (3.0 mL, 2/1), 4 mA, 24 h, 390 nm, 25 °C, nitrogen, graphite felt (GF) as an anode, reticulated vitreous carbon (RVC) as a cathode, undivided cell. Isolated yields. |
|---|
|
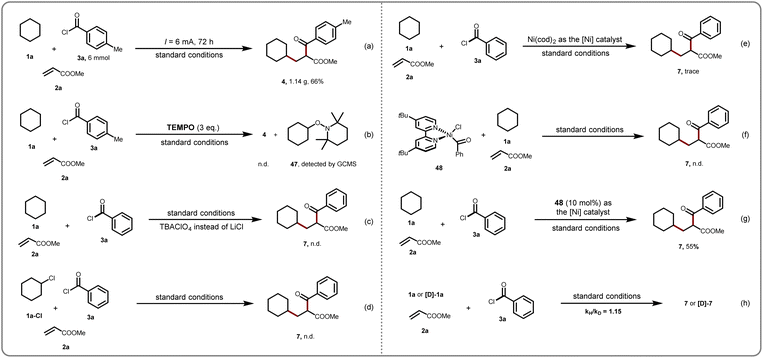
To further illustrate the outstanding potential of this approach, we conducted a gram-scale reaction under slightly modified conditions to synthesize the cascade product in good yield (Scheme 3a). A series of mechanistic experiments probing the mechanism of action have been performed. First, the addition of TEMPO (3.0 equiv.) strongly inhibited these transformations, and product 4 could not be observed. Instead, a cyclohexylated TEMPO 47 was observed by GCMS (Scheme 3b). These findings support the suggestion that this cascade reaction involved a radical mechanism. When the transformation was completed with TBABClO4 to replace LiCl as the electrolyte under the standard conditions, no expected product was obtained (Scheme 3c). This finding demonstrated that LiCl is essential for the formation of [FeCl4]−in situ. The target product was not detected when the reaction was conducted by using cyclohexyl chlorides instead of cyclohexanes as substrates, indicating that the alkyl chloride is not a key intermediate in this transformation (Scheme 3d). A trace amount of additional product was obtained utilizing Ni(COD)2 as a nickel catalyst, suggesting that Ni(0) species may not be involved in the main catalytic cycle (Scheme 3e). In addition, the nickel(II)-complex 48 was subjected to transformation in the presence of a catalytic photocatalyst, and no target carbonyl compound was detected (Scheme 3f). Moreover, when the reaction was performed with 10 mol% of nickel-complex 48 as the catalyst, the desired product 7 was obtained in 55% yield (Scheme 3g). Furthermore, a kinetic isotope effect (KIE) of 1.15 was observed between cyclohexane 1a and [D]-1a with methyl acrylate 2a, and benzoyl chloride 3b from parallel reactions, suggesting that the C–H bond of the cyclohexanes cleavage step is not the turnover-determining step (Scheme 3e). Finally, cyclic voltammetry (CV) experiments further confirmed that the nickel complex (NiBr2·dtbbpy) underwent cathodic reduction at reduction potentials of Ni(II)/Ni(I) (−1.38 V vs. Fc/Fc+) and Ni(I)/Ni(0) (−1.88 V vs. Fc/Fc+) and the photocatalyst (FeCl3·H2O) underwent anodic oxidation at oxidation potentials of Fe(II)/Fe(III) (−0.09 V vs. Fc/Fc+) (please see the ESI† for details).
According to the above mechanistic studies and previous findings,20,23,26 a plausible mechanism for the electrophoto-catalytic alkenes carboacylation is proposed (Scheme 4). Upon visible light irradiation, [FeIIICl4]− (A) is excited to generate a photoexcited state *[FeIIICl4]− (B), which could release chlorine radicals via the LMCT process. This highly active radical species could abstract a hydrogen atom from the C–H compound 1 to form a carbon-centered radical intermediate D, and the [FeIICl3]− (C) undergo anodic oxidation with chloride ions to regenerate the [FeIIICl4]− (A). Concomitantly, the NiI catalyst concerted oxidative addition with acyl chlorides 3 to afford nickelIII species F, which was reduced on the cathode to generate the NiII species G. Then intermediate D was added to alkenes 2 to give the secondary alkyl radical E, which could be intercepted by NiII species G to produce alkyl–NiIII intermediate H. The intermediate H would undergo reductive elimination to deliver the corresponding carbonyl compounds 4 and regenerate the NiI catalyst I.
Conclusions
In summary, by combining photochemistry and electrochemistry, we have successfully developed a nickel-catalyzed three-component carboacylation/silanoylation of alkenes with acyl chlorides, hydrosilanes, and unactivated C(sp3)–H donors. The electrophotocatalytic platform allowed the facile construction of ketones from inexpensive commercial starting materials and showed excellent enantioselectivity. In addition, a series of experimental studies revealed the key mechanistic pathway for this photoelectrocatalytic process. The use of an electrophotocatalytic strategy for developing more challenging functionalization processes of unsaturated bonds is underway in our group.Author contributions
D. W. and L. A. conceived and designed the study, and wrote the paper. L. W., X. H., and X. H. performed the experiments, mechanism study, and analyzed the data.Conflicts of interest
There are no conflicts to declare.Acknowledgements
Generous support by the National Natural Science Foundation of China (NSFC-22301032), and the DFG (Gottfried-Wilhelm Leibniz Award to L. A.) is gratefully acknowledged.References
- (a) J.-S. Zhang, L. Liu, T. Chen and L.-B. Han, Chem. – Asian J., 2018, 13, 2277–2291 CrossRef CAS PubMed; (b) R. K. Dhungana, S. Kc, P. Basnet and R. Giri, Chem. Rec., 2018, 18, 1314–1340 CrossRef CAS PubMed; (c) J. Derosa, O. Apolinar, T. Kang, V. T. Tran and K. M. Engle, Chem. Sci., 2020, 11, 4287–4296 RSC.
- (a) Y. Ping and W. Kong, Synthesis, 2020, 979–992 Search PubMed; (b) X. Zhao, H.-Y. Tu, L. Guo, S. Zhu, F.-L. Qing and L. Chu, Nat. Commun., 2018, 9, 3488 CrossRef PubMed; (c) A. A. Kadam, T. L. Metz, Y. L. Qian and M. Stanley, ACS Catal., 2019, 9, 5651–5656 CrossRef CAS; (d) L. Wang and C. Wang, Org. Lett., 2020, 22, 8829–8835 CrossRef CAS PubMed; (e) M. Liu, T. Feng, Y. Wang, G. Kou, Q. Wang, Q. Wang and Y. Qiu, Nat. Commun., 2023, 14, 6467 CrossRef CAS PubMed; (f) K. Yang, T. Feng and Y. Qiu, Angew. Chem., Int. Ed., 2023, 62, e202312803 CrossRef CAS PubMed.
- (a) L. Liao, R. Jana, K. B. Urkalan and M. S. Sigman, J. Am. Chem. Soc., 2011, 133, 5784–5787 CrossRef CAS PubMed; (b) J.-W. Gu, Q.-Q. Min, L.-C. Yu and X. Zhang, Angew. Chem., Int. Ed., 2016, 55, 12270–12274 ( Angew. Chem. , 2016 , 128 , 12458–12462 ) CrossRef CAS PubMed; (c) L. Zhang, G. J. Lovinger, E. K. Edelstein, A. A. Szymaniak, M. P. Chierchia and J. P. Morken, Science, 2016, 351, 70–74 CrossRef CAS PubMed; (d) T. Qin, J. Cornella, C. Li, L. R. Malins, J. T. Edwards, S. Kawamura, B. D. Maxwell, M. D. Eastgate and P. S. Baran, Science, 2016, 352, 801–805 CrossRef CAS PubMed; (e) B. Shrestha, P. Basnet, R. K. Dhungana, S. Kc, S. Thapa, J. M. Sears and R. Giri, J. Am. Chem. Soc., 2017, 139, 10653–10656 CrossRef CAS PubMed; (f) P. Gao, L. A. Chen and M. K. Brown, J. Am. Chem. Soc., 2018, 140, 10653–10657 CrossRef CAS PubMed; (g) J. Derosa, R. Kleinmans, V. T. Tran, M. K. Karunananda, S. R. Wisniewski, M. D. Eastgate and K. M. Engle, J. Am. Chem. Soc., 2018, 140, 17878–17883 CrossRef CAS PubMed; (h) P. Basnet, S. Kc, R. K. Dhungana, B. Shrestha, T. J. Boyle and R. Giri, J. Am. Chem. Soc., 2018, 140, 15586–15590 CrossRef CAS PubMed; (i) M. Chierchia, P. Xu, G. J. Lovinger and J. P. Morken, Angew. Chem., Int. Ed., 2019, 58, 14245–14249 ( Angew. Chem. , 2019 , 131 , 14383–14387 ) CrossRef CAS PubMed; (j) J. Derosa, V. T. Tran, M. N. Boulous, J. S. Chen and K. M. Engle, J. Am. Chem. Soc., 2017, 139, 10657–10660 CrossRef CAS PubMed.
- (a) X. Wei, W. Shu, A. García-Domínguez, E. Merino and C. Nevado, J. Am. Chem. Soc., 2020, 142, 13515–13522 CrossRef CAS PubMed; (b) A. García-Domínguez, R. Mondal and C. Nevado, Angew. Chem., Int. Ed., 2019, 58, 12286–12290 CrossRef PubMed; (c) A. Lipp, S. O. Badir and G. A. Molander, Angew. Chem., Int. Ed., 2021, 60, 1714–1726 ( Angew. Chem. , 2021 , 133 , 1738–1750 ) CrossRef CAS PubMed; (d) R. S. Mega, V. K. Duong, A. Noble and V. K. Aggarwal, Angew. Chem., Int. Ed., 2020, 59, 4375–4379 ( Angew. Chem. , 2020 , 132 , 4405–4409 ) CrossRef CAS PubMed; (e) A. Dewanji, P. E. Krach and M. Rueping, Angew. Chem., Int. Ed., 2019, 58, 3566–3570 ( Angew. Chem. , 2019 , 131 , 3604–3608 ) CrossRef CAS PubMed; (f) L. Huang and M. Rueping, Angew. Chem., Int. Ed., 2018, 57, 10333–10337 ( Angew. Chem. , 2018 , 130 , 10490–10494 ) CrossRef CAS PubMed; (g) H. Yue, C. Zhu and M. Rueping, Angew. Chem., Int. Ed., 2017, 57, 1371–1375 ( Angew. Chem. , 2017 , 130 , 1385–1389 ) CrossRef PubMed; (h) S. Sun, Y. Duan, R. S. Mega, R. J. Somerville and R. Martin, Angew. Chem., Int. Ed., 2020, 59, 4370–4374 ( Angew. Chem. , 2020 , 132 , 4400–4404 ) CrossRef CAS PubMed; (i) H. Jiang, X. Yu, C. G. Daniliuc and A. Studer, Angew. Chem., Int. Ed., 2021, 60, 14399–14404 ( Angew. Chem. , 2021 , 133 , 14520–14525 ) CrossRef CAS PubMed; (j) T. Yang, X. Chen, W. Rao and M. J. Koh, Chem, 2020, 6, 738–751 CrossRef CAS; (k) X. Shu, L. Huan, Q. Huang and H. Huo, J. Am. Chem. Soc., 2020, 142, 19058–19064 CrossRef CAS PubMed; (l) B. Maity, C. Zhu, H. Yue, L. Huang, M. Harb, Y. Minenkov, M. Rueping and L. Cavallo, J. Am. Chem. Soc., 2020, 142, 16942–16952 CrossRef CAS PubMed; (m) A. Y. Chan, I. B. Perry, N. B. Bissonnette, B. F. Buksh, G. A. Edwards, L. I. Frye, O. L. Garry, M. N. Lavagnino, B. X. Li, Y. Liang, E. Mao, A. Millet, J. V. Oakley, N. L. Reed, H. A. Sakai, C. P. Seath and D. W. C. MacMillan, Chem. Rev., 2022, 122, 1485–1542 CrossRef CAS PubMed; (n) F.-D. Lu, J. Chen, X. Jiang, J.-R. Chen, L.-Q. Lu and W.-J. Xiao, Chem. Soc. Rev., 2021, 50, 12808–12827 RSC; (o) C. Zhu, H. Yue, L. Chu and M. Rueping, Chem. Sci., 2020, 11, 4051–4064 RSC; (p) Y.-Y. Gui, L. Sun, Z.-P. Lua and D.-G. Yu, Org. Chem. Front., 2016, 3, 522–526 RSC.
- (a) S. Xu, H. Chen, Z. Zhou and W. Kong, Angew. Chem., Int. Ed., 2021, 60, 7405–7411 CrossRef CAS PubMed; (b) W. Liu, C. Liu, M. Wang and W. Kong, ACS Catal., 2022, 12, 10207–10221 CrossRef CAS.
- J. Hou, A. Ee, H. Cao, H.-W. Ong, J.-H. Xu and J. Wu, Angew. Chem., Int. Ed., 2018, 57, 17220–17224 CrossRef CAS PubMed.
- M. Zheng, J. Hou, L.-L. Hua, W.-Y. Tang, L.-W. Zhan and B.-D. Li, Org. Lett., 2021, 23, 5128–5132 CrossRef CAS PubMed.
- D. Wang and L. Ackermann, Chem. Sci., 2022, 13, 7256–7263 RSC.
- (a) S. M. Treacy and T. Rovis, J. Am. Chem. Soc., 2021, 143, 2729–2735 CrossRef CAS PubMed; (b) Y. C. Kang, S. M. Treacy and T. Rovis, ACS Catal., 2021, 11, 7442–7449 CrossRef CAS PubMed.
- Q. Yang, Y. H. Wang, Y. Qiao, M. Gau, P. J. Carroll, P. J. Walsh and E. J. Schelter, Science, 2021, 372, 847–852 CrossRef CAS PubMed.
- A. Hu, J. J. Guo, H. Pan and Z. Zuo, Science, 2018, 361, 668–672 CrossRef CAS PubMed.
- Z.-Y. Dai, S.-Q. Zhang, X. Hong, P.-S. Wang and L.-Z. Gong, Chem. Catal., 2022, 2, 1211–1222 CrossRef CAS.
- (a) Z. Zhang, X. Li, D. Zhou, S. Ding, M. Wang and R. Zeng, J. Am. Chem. Soc., 2023, 145, 7612–7620 CrossRef CAS PubMed; (b) Z. Zhang, G. Zhang, N. Xiong, T. Xue, J. Zhang, L. Bai, Q. Guo and R. Zeng, Org. Lett., 2021, 23, 2915–2920 CrossRef CAS PubMed; (c) T. Xue, Z. Zhang and R. Zeng, Org. Lett., 2022, 24, 977–982 CrossRef CAS PubMed.
- J.-L. Tu, A.-M. Hu, L. Guo and W. Xia, J. Am. Chem. Soc., 2023, 145, 7600–7611 CrossRef CAS PubMed.
- (a) Y. Jin, Q. Zhang, L. Wang, X. Wang, C. Meng and C. Duan, Green Chem., 2021, 23, 6984–6989 RSC; (b) L. Troian-Gautier, M. D. Turlington, S. A. M. Wehlin, A. B. Maurer, M. D. Brady, W. B. Swords and G. J. Meyer, Chem. Rev., 2019, 119, 4628–4683 CrossRef CAS PubMed; (c) L. Ding, Y. Liu, K. Niu and Q. Wang, Chem. Commun., 2022, 58, 10679–10682 RSC.
- G. Yang, Y. Wang and Y. Qiu, Chin. J. Org. Chem., 2021, 41, 3935–3947 CrossRef CAS.
- (a) H. Huang, Z. M. Strater, M. Rauch, J. Shee, T. J. Sisto, C. Nuckolls and T. H. Lambert, Angew. Chem., Int. Ed., 2019, 58, 13318–13322 CrossRef CAS PubMed; (b) H. Huang, Z. M. Strater and T. H. Lambert, J. Am. Chem. Soc., 2020, 142, 1698–1703 CrossRef CAS PubMed; (c) T. Shen and T. H. Lambert, Science, 2021, 371, 620–626 CrossRef CAS PubMed; (d) H. Huang and T. H. Lambert, Angew. Chem., Int. Ed., 2020, 59, 658–662 CrossRef CAS PubMed.
- (a) H. Kim, H. Kim, T. H. Lambert and S. Lin, J. Am. Chem. Soc., 2020, 142, 2087–2092 CrossRef CAS PubMed; (b) W. Zhang, K. L. Carpenter and S. Lin, Angew. Chem., Int. Ed., 2020, 59, 409–417 CrossRef CAS PubMed.
- (a) Z. Yang, D. Yang, J. Zhang, C. Tan, J. Li, S. Wang, H. Zhang, Z. Huang and A. Lei, J. Am. Chem. Soc., 2022, 144, 13895–13902 CrossRef CAS PubMed; (b) L. Niu, C. Jiang, Y. Liang, D. Liu, F. Bu, R. Shi, H. Chen, A. D. Chowdhury and A. Lei, J. Am. Chem. Soc., 2020, 142, 17693–17702 CrossRef CAS PubMed.
- (a) H. Yan, Z.-W. Hou and H.-C. Xu, Angew. Chem., Int. Ed., 2019, 58, 4592–4595 CrossRef CAS PubMed; (b) P. Xu, P.-Y. Chen and H.-C. Xu, Angew. Chem., Int. Ed., 2020, 59, 14275–14280 CrossRef CAS PubMed; (c) Z.-W. Hou and H.-C. Xu, ChemElectroChem, 2021, 8, 1571–1573 CrossRef CAS; (d) H. Yan, J. Song, S. Zhu and H.-C. Xu, CCS Chem., 2021, 3, 317–325 CrossRef CAS; (e) C.-Y. Cai, X.-L. Lai, Y. Wang, H.-H. Hu, J. Song, Y. Yang, C. Wang and H.-C. Xu, Nat. Catal., 2022, 5, 943–951 CrossRef CAS; (f) X.-L. Lai, M. Chen, Y. Wang, J. Song and H.-C. Xu, J. Am. Chem. Soc., 2022, 144, 20201–20206 CrossRef CAS PubMed.
- (a) L. Zhang, L. Liardet, J. Luo, D. Ren, M. Grätzel and X. Hu, Nat. Catal., 2019, 2, 366–373 CrossRef CAS PubMed; (b) H. Tateno, Y. Miseki and K. Sayama, Chem. Commun., 2017, 53, 4378–4381 RSC; (c) L. Zou, X. Wang, S. Xiang, W. Zheng and Q. Lu, Angew. Chem., Int. Ed., 2023, 62, e202301026 CrossRef CAS PubMed; (d) F. Wang and S. S. Stahl, Angew. Chem., Int. Ed., 2019, 58, 6385–6390 CrossRef CAS PubMed; (e) Y. Qiu, A. Scheremetjew, L. H. Finger and L. Ackermann, Chem. – Eur. J., 2020, 26, 3241–3246 CrossRef CAS PubMed.
- X.-L. Lai and H.-C. Xu, J. Am. Chem. Soc., 2023, 145, 18753–18759 CrossRef CAS PubMed.
- L. Zou, S. Xiang, R. Sun and Q. Lu, Nat. Commun., 2023, 14, 7992 CrossRef CAS PubMed.
- J. V. Obligacion and P. J. Chirik, Nat. Rev. Chem., 2018, 2, 15–34 CrossRef CAS PubMed.
- (a) L. Lu, J. C. Siu, Y. Lai and S. Lin, J. Am. Chem. Soc., 2020, 142, 21272–21278 CrossRef CAS PubMed; (b) J. Ke, W. Liu, X. Zhu, X. Tan and C. He, Angew. Chem., Int. Ed., 2021, 60, 8744–8749 CrossRef CAS PubMed.
- (a) G. A. Dawson, E. H. Spielvogel and T. Diao, Acc. Chem. Res., 2023, 56, 3640–3653 CrossRef CAS PubMed; (b) Q. Lin, Y. Fu, P. Liu and T. Diao, J. Am. Chem. Soc., 2021, 143, 14196–14206 CrossRef CAS PubMed; (c) C. L. Wagner, G. Herrera, Q. Lin, C. T. Hu and T. Diao, J. Am. Chem. Soc., 2021, 143, 5295–5300 CrossRef CAS PubMed; (d) G. A. Dawson, Q. Lin, M. C. Neary and T. Diao, J. Am. Chem. Soc., 2023, 145, 20551–20561 CrossRef CAS PubMed; (e) M. Rafiee, D. J. Abrams, L. Cardinale, Z. Goss, A. Romero-Arenas and S. S. Stahl, Chem. Soc. Rev., 2024, 53, 566–585 RSC.
Footnotes |
| † Electronic supplementary information (ESI) available. See DOI: https://doi.org/10.1039/d3gc04109c |
| ‡ These authors have contributed equally to this paper. |
| This journal is © The Royal Society of Chemistry 2024 |