Seminormal-BrCH2CH2OH-mediated electrochemical epoxidation of unactivated olefins†
Hong
He
ab,
Yanxia
Lv
ab,
Jing
Hu
a,
Zhong-Wei
Hou
*a and
Lei
Wang
 *abcd
*abcd
aAdvanced Research Institute and School of Pharmaceutical Sciences, Taizhou University, Jiaojiang, Zhejiang 318000, P. R. China. E-mail: zhongwei.hou@tzc.edu.cn; leiwang88@hotmail.com
bDepartment of Chemistry, Huaibei Normal University, Huaibei, Anhui 235000, P. R. China
cCollege of Material Chemistry and Chemical Engineering, Key Laboratory of Organosilicon Chemistry and Material Technology, Ministry of Education, Hangzhou Normal University, Hangzhou, 311121, Zhejiang, P. R. China
dState Key Laboratory of Organometallic Chemistry, Shanghai Institute of Organic Chemistry, Chinese Academy of Sciences, Shanghai, 200032, P. R. China
First published on 4th January 2024
Abstract
An electrochemical epoxidation of unactivated olefins using water as the source of oxygen atoms has been developed. The epoxidation reaction employs seminormal-BrCH2CH2OH as the mediator, which shows good functional group compatibility, and can easily be performed on the gram scale. A wide range of unactivated olefins could be tolerated to give the functionalized epoxides with satisfactory results at room temperature.
Introduction
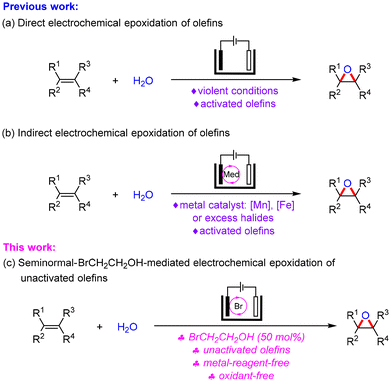
Epoxides fulfill a plethora of important roles in the fields of synthetic chemistry, pharmaceutical science and materials science.1 Olefins are some of the most common organic molecules and are widely used in organic synthesis because of their abundant source, easy availability and diverse reactivity.2 The direct epoxidation of olefins provides an efficient method for rapidly increasing the complexity of molecules to obtain epoxides.3 In this regard, olefin epoxidation has been extensively explored using stoichiometric oxidants4 such as O2, H2O2, m-CPBA, etc. However, these reported methods are frequently limited by their drawbacks such as severe conditions, low efficiency, poor atom economy and huge amounts of waste. Therefore, the development of oxidant-free olefin epoxidation methods is highly desirable.As a green synthetic technology, organic electrochemistry5 uses electrons as reagents and avoids the addition of redox reagents, which has attracted more and more attention.6 The electrochemical epoxidation of olefins has been performed by direct electrolysis or indirect electrolysis. Direct electrochemical epoxidation of olefins could be achieved at high current density (Scheme 1a).7 Metal [e.g. Mn and Fe]-8 and iminium salt-catalyzed9 routes for the epoxidation of styrenes or cyclic alkenes have been reported in recent years. Halides are cheap and readily available activated reagents to achieve electrochemical transformations.10 Halides (e.g. NaCl, KCl, NaBr, n-Bu4NBr and n-Bu4NI) facilitated electrochemical epoxidation of alkenes and demonstrated good efficiency and universality with water as the source of oxygen atoms, but excess halides or activated olefins are often necessary for these transformations (Scheme 1b).11 For example, in 2020, the chloride-mediated electrosynthesis of ethylene and propylene oxides with high selectivity and efficiency at high current density was reported by Sargent.12 In 2022, Zai described a NaBr- and oxygen redox species-mediated selective electrochemical epoxidation of styrenes using a CoS2/CoS modified graphite felt as the anode.13 While we were preparing the article, Qiu and co-workers reported a metal-free electrochemical dihydroxylation and epoxidation of unactivated alkenes using Et4NI (2 equiv.) and NH4I (2 equiv.) at 50 mA and 50 °C for 12 h.14
Although much progress has been made in this field, a mild and efficient approach for the electrochemical epoxidation of unactivated olefins is still worth exploring.15 Recently, our group found that BrCH2CH2OH is an effective brominating reagent to achieve the bromination of arenes and alkenes.16 Inspired by the above progress and our interest in electrochemistry,17 we herein developed a facile and versatile electrochemical approach for the epoxidation of unactivated olefins with seminormal-BrCH2CH2OH as the mediator and water as the source of oxygen atoms (Scheme 1c). This reaction provides a general route for the electrosynthesis of unactivated epoxides with good selectivity and efficiency under metal reagent-free and oxidant-free conditions, and is also highly complementary to previously reported methods.
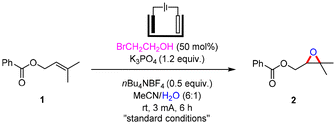
At the beginning, we screened the reaction conditions for the electrochemical epoxidation of alkene 1. The reaction was carried out at 3 mA for 6 h in an undivided cell with reticulated vitreous carbon (RVC) as the anode and a platinum sheet as the cathode (Table 1). (3,3-Dimethyloxiran-2-yl)methyl benzoate (2) was isolated in 83% yield under the standard conditions using BrCH2CH2OH (50 mol%) as the mediator, n-Bu4NBF4 as an electrolyte and K3PO4 as a base in MeCN/H2O (6![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) at room temperature (Table 1, entry 1). Inorganic bromides such as NaBr (Table 1, entry 2) and KBr (Table 1, entry 3) were tested and lower yields were observed. The yield of 2 slightly increased to 85% when employing 1 equivalent of BrCH2CH2OH (Table 1, entry 4). Upon decreasing the equivalent of BrCH2CH2OH to 0.2, the formation of 2 was suppressed (Table 1, entry 5). The product 2 was not obtained in the absence of BrCH2CH2OH (Table 1, entry 6). Bromide-catalyzed epoxidation was still not achieved although great efforts were made. After removing K3PO4 or n-Bu4NBF4, 26% and 36% yields of 2 were generated (Table 1, entries 7 and 8). The reaction solvents were then investigated by adjusting the ratio of MeCN and H2O in mixed solvents. When MeCN/H2O (13
1) at room temperature (Table 1, entry 1). Inorganic bromides such as NaBr (Table 1, entry 2) and KBr (Table 1, entry 3) were tested and lower yields were observed. The yield of 2 slightly increased to 85% when employing 1 equivalent of BrCH2CH2OH (Table 1, entry 4). Upon decreasing the equivalent of BrCH2CH2OH to 0.2, the formation of 2 was suppressed (Table 1, entry 5). The product 2 was not obtained in the absence of BrCH2CH2OH (Table 1, entry 6). Bromide-catalyzed epoxidation was still not achieved although great efforts were made. After removing K3PO4 or n-Bu4NBF4, 26% and 36% yields of 2 were generated (Table 1, entries 7 and 8). The reaction solvents were then investigated by adjusting the ratio of MeCN and H2O in mixed solvents. When MeCN/H2O (13![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) or MeCN/H2O (5
1) or MeCN/H2O (5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2) was used, electrochemical epoxidation occurred with low efficiency (Table 1, entries 9 and 10). Trace amounts of the product 2 were detected when applying MeCN as the reaction solvent (Table 1, entry 11). No product was formed in the absence of electricity (Table 1, entry 12).
2) was used, electrochemical epoxidation occurred with low efficiency (Table 1, entries 9 and 10). Trace amounts of the product 2 were detected when applying MeCN as the reaction solvent (Table 1, entry 11). No product was formed in the absence of electricity (Table 1, entry 12).
| Entry | Variation from the standard conditions | Yieldb (%) |
|---|---|---|
| a Reaction conditions: RVC anode, Pt cathode, undivided cell, 1 (0.2 mmol), BrCH2CH2OH (50 mol%), K3PO4 (0.24 mmol), nBu4NBF4 (0.1 mmol), MeCN (6 mL), H2O (1 mL), rt, 3 mA, 6 h (3.3 F mol−1). b Yield was determined by 1H NMR analysis of the crude reaction mixture using 1,3,5-trimethoxybenzene as the internal standard. c Isolated yield. | ||
| 1 | None | 83c |
| 2 | NaBr (0.5 equiv.) | 61 |
| 3 | KBr (0.5 equiv.) | 60 |
| 4 | BrCH2CH2OH (1 equiv.) | 85 |
| 5 | BrCH2CH2OH (0.2 equiv.) | 60 |
| 6 | No BrCH2CH2OH | 0 |
| 7 | No K3PO4 | 26 |
| 8 | No nBu4NBF4 | 36 |
| 9 | MeCN/H2O (13![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) as solvent 1) as solvent |
57 |
| 10 | MeCN/H2O (5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2) as solvent 2) as solvent |
63 |
| 11 | MeCN as solvent | Trace |
| 12 | No electricity | 0 |
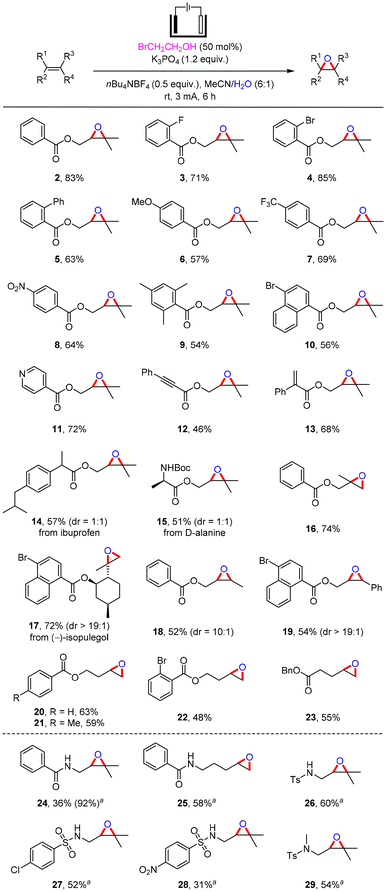
After the reaction conditions were determined, the substrate scope for the electrochemical epoxidation was explored (Scheme 2). We found that this method could be compatible with a variety of ester-linked olefins containing common functional groups such as H (2, 83% yield), F (3, 71% yield), Br (4, 85% yield) and Ph (5, 63% yield) at the ortho-position of the aromatic rings. Substrates with a para-substituent including OMe (6, 57% yield), CF3 (7, 69% yield) and NO2 (8, 64% yield) groups were applicable for the electrochemical epoxidation. (3,3-Dimethyloxiran-2-yl)methyl 2,4,6-trimethylbenzoate (9) was obtained in 54% yield under electrochemical epoxidation conditions. Substrates attached with naphthalene or pyridine were tolerated to give epoxides 10 and 11 in 56% and 72% yields, respectively. The electrochemical epoxidation could selectively occur in an electron-rich alkenyl moiety rather than an alkynyl or alkenyl moiety connected to a carbonyl group, producing 12 and 13 with acceptable efficiency. The ibuprofen derivative 14 containing an epoxy group was formed in 57% yield under the standard conditions. Epoxide 15 containing an amino acid fragment could also be quickly prepared. Furthermore, different types of olefin structures were investigated. The corresponding epoxy products were prepared in satisfactory yields by electrochemical epoxidation of 1,1-disubstituted alkenes (16 and 17 in 74% and 72% yields, respectively), 1,2-disubstituted alkenes (18 and 19, 52 and 54% yields, respectively) and monosubstituted alkenes (20–23, 48–63% yields).
Subsequently, the electrochemical epoxidation of substrates bearing amides and sulfonamides was examined. However, epoxide 24 was only obtained in a yield of 36% under standard conditions. By increasing the amounts of BrCH2CH2OH to 2 equivalents, the yield of 24 was improved to 92%. A series of amide-containing epoxides (24 and 25, 92% and 58% yields, respectively) and sulfonamide-containing epoxides (26–29, 31–60% yields) were synthesized in moderate to good yields, in which the active NH group could be well compatible. Additionally, the electrochemical epoxidation of styrenes, cycloolefins, and α,β-unsaturated ketones was tested, and the production of the corresponding epoxides was either inefficient or entirely unsuccessful. Detailed information on the unsuccessful substrates is provided in the ESI.†
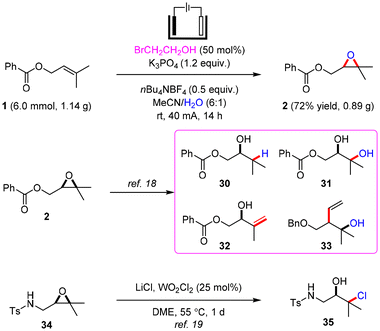
To further highlight the availability of electrochemical epoxidation, the gram-scale electrolysis of 1 was conducted on a 6 mmol scale at a constant current of 40 mA and room temperature for 14 h (Scheme 3). 0.89 g of epoxy product 2 was obtained in 72% yield under simple electrolysis conditions. The epoxy product 2 could undergo diverse transformations into highly functionalized complex structures 30–33via a ring-opening reaction according to the reported methods.18 The regioselective chlorination of 2,3-epoxy sulfonamide 34 was achieved by the Yamamoto group, which then gets converted into chloroaminoalcohol 35.19
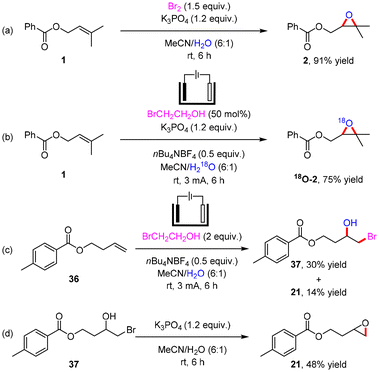
To clarify the reaction process, control experiments were performed (Scheme 5). 91% yield of 2 was formed with Br2 as the activated reagent without electricity (Scheme 4a), showing that the success of the reaction probably depends on the generation of Br2in situ. 18O-2 was primarily obtained in 75% yield when using H218O instead of H2O (Scheme 4b), indicating that the oxygen atom in the epoxy group of the product comes from water. Moreover, the bromohydroxylation product 37 was afforded in 30% yield by electrolyzing 36 without K3PO4 and epoxy product 21 was detected in 14% yield (Scheme 4c), supporting that the epoxidation involves a bromohydroxylation process. In addition, the epoxidation product 21 could be obtained from 37 in 48% yield with K3PO4 in a solution of MeCN/H2O. The addition of K3PO4 could facilitate the conversion of the bromohydroxylation intermediate to the epoxidation product.
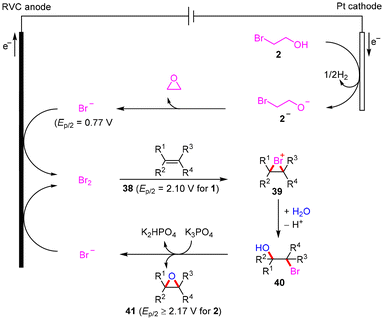
Based on the above-mentioned findings, cyclic voltammograms (Fig. S3–S5, in the ESI†) and previous research studies,11,14,16 a possible mechanism for the electrochemical epoxidation is proposed (Scheme 5). At first, Br− is produced by the cathodic reduction and nucleophilic cyclization of BrCH2CH2OH through ethylene oxide and H2 evolution, and then is oxidized at the anode to form Br2in situ. Subsequently, alkenes 38 react with Br2via electrophilic addition to generate bromonium ions 39, which are trapped by water to give bromohydroxylation intermediates 40.20 Finally, the K3PO4-facilitated intramolecular epoxidation of 40 yields the target products 41 with the release of Br− and K2HPO4. The anodic oxidation of Br− preferentially occurred due to the oxidation potential of Br− (Ep/2 = 0.77 V vs. Ag/AgCl) being obviously lower than that of substrate 1 (Ep/2 = 2.10 V vs. Ag/AgCl) and substrate 2 (Ep/2 ≥ 2.17 V vs. Ag/AgCl), supporting our proposed mechanism shown in Scheme 5. The low concentration of Br− is anodized to regenerate Br2 to allow the reaction to proceed smoothly under seminormal bromine source conditions and effectively avoid the dibromination of olefins.21
Conclusions
In conclusion, we have developed an electrochemical epoxidation of unactivated olefins with seminormal-BrCH2CH2OH as the mediator and water as the oxygen atom source. The method is performed at room temperature in a simple undivided cell and shows good functional group tolerance. A series of functionalized epoxides are obtained in moderate to good yields without the need for metal reagents and oxidants. Further efforts on halogen-catalyzed electrochemical epoxidation of unactivated olefins are ongoing in our laboratory.Conflicts of interest
There are no conflicts to declare.Acknowledgements
We gratefully acknowledge the Natural Science Foundation of Zhejiang Province (LQ22B020005 and LZ22B020003) and the National Natural Science Foundation of China (22101201 and 22071171) for the financial support of this work.References
- (a) J. Herzberger, K. Niederer, H. Pohlit, J. Seiwert, M. Worm, F. R. Wurm and H. Frey, Chem. Rev., 2016, 116, 2170–2243 CrossRef CAS PubMed; (b) J. M. Longo, M. J. Sanford and G. W. Coates, Chem. Rev., 2016, 116, 15167–15197 CrossRef CAS.
- (a) M.-Y. Cao, X. Ren and Z. Lu, Tetrahedron Lett., 2015, 56, 3732–3742 CrossRef CAS; (b) Z.-L. Li, G.-C. Fang, Q.-S. Gu and X.-Y. Liu, Chem. Soc. Rev., 2020, 49, 32–48 RSC; (c) M.-J. Luo, Q. Xiao and J.-H. Li, Chem. Soc. Rev., 2022, 51, 7206–7237 RSC; (d) H. Mei, Z. Yin, J. Liu, H. Sun and J. Han, Chin. J. Chem., 2019, 37, 292–301 CrossRef CAS.
- (a) D. E. De Vos, B. F. Sels and P. A. Jacobs, Adv. Synth. Catal., 2003, 345, 457–473 CrossRef CAS; (b) A. S. Sharma, V. S. Sharma, H. Kaur and R. S. Varma, Green Chem., 2020, 22, 5902–5936 RSC.
- (a) T. Katsuki and K. B. Sharpless, J. Am. Chem. Soc., 1980, 102, 5974–5976 CrossRef CAS; (b) C. Kim, T. G. Traylor and C. L. Perrin, J. Am. Chem. Soc., 1998, 120, 9513–9516 CrossRef CAS; (c) G. Anilkumar, B. Bitterlich, F. G. Gelalcha, M. K. Tse and M. Beller, Chem. Commun., 2007, 289–291 RSC; (d) D. T. Bregante, J. Z. Tan, R. L. Schultz, E. Z. Ayla, D. S. Potts, C. Torres and D. W. Flaherty, ACS Catal., 2020, 10, 10169–10184 CrossRef CAS.
- (a) A. Wiebe, T. Gieshoff, S. Möhle, E. Rodrigo, M. Zirbes and S. R. Waldvogel, Angew. Chem., Int. Ed., 2018, 57, 5594–5619 CrossRef CAS; (b) Y. Jiang, K. Xu and C. Zeng, Chem. Rev., 2018, 118, 4485–4540 CrossRef CAS PubMed; (c) P. Xiong and H.-C. Xu, Acc. Chem. Res., 2019, 52, 3339–3350 CrossRef CAS; (d) Y. Yuan and A. Lei, Acc. Chem. Res., 2019, 52, 3309–3324 CrossRef CAS; (e) K.-J. Jiao, Y.-K. Xing, Q.-L. Yang, H. Qiu and T.-S. Mei, Acc. Chem. Res., 2020, 53, 300–310 CrossRef CAS; (f) S.-H. Shi, Y. Liang and N. Jiao, Chem. Rev., 2021, 121, 485–505 CrossRef CAS; (g) Z.-W. Hou, H.-C. Xu and L. Wang, Curr. Opin. Electrochem., 2022, 34, 100988 CrossRef CAS.
- (a) S. D. Minteer and P. S. Baran, Acc. Chem. Res., 2020, 53, 545–546 CrossRef CAS PubMed; (b) L. Ackermann, Acc. Chem. Res., 2020, 53, 84–104 CrossRef CAS; (c) J.-S. Zhong, Y. Yu, Z. Shi and K.-Y. Ye, Org. Chem. Front., 2021, 8, 1315–1328 RSC; (d) L. F. T. Novaes, J. Liu, Y. Shen, L. Lu, J. M. Meinhardt and S. Lin, Chem. Soc. Rev., 2021, 50, 7941–8002 RSC; (e) Q. Wan, Z. Zhang, Z.-W. Hou and L. Wang, Org. Chem. Front., 2023, 10, 2830–2848 RSC.
- B. R. Buckley, Y. Chan, N. Dreyfus, C. Elliott, F. Markend and P. C. B. Page, Green Chem., 2012, 14, 2221–2225 RSC.
- (a) E. Laura and F. R. James, J. Am. Chem. Soc., 2004, 126, 7676–7682 CrossRef; (b) K. Jin, J. H. Maalouf, N. Lazouski, N. Corbin, D. Yang and K. Manthiram, J. Am. Chem. Soc., 2019, 141, 6413–6418 CrossRef CAS; (c) B. Chandra, K. M. Hellan, S. Pattanayak and S. S. Gupta, Chem. Sci., 2020, 11, 11877–11885 RSC.
- P. C. B. Page, F. Marken, C. Williamson, Y. Chan, B. R. Buckley and D. Bethell, Adv. Synth. Catal., 2008, 350, 1149–1154 CrossRef CAS.
- (a) H.-T. Tang, J.-S. Jia and Y.-M. Pan, Org. Biomol. Chem., 2020, 18, 5315–5333 RSC; (b) F. Lian, K. Xu and C. Zeng, Chem. Rec., 2021, 21, 2290–2305 CrossRef CAS PubMed; (c) L. G. Gombos and S. R. Waldvogel, Sustainable Chem., 2022, 3, 430–454 CrossRef CAS; (d) J. Du, Y.-L. Du and Q.-W. Gui, Synthesis, 2023, 55, 2799–2816 CrossRef CAS.
- (a) S. Torii, K. Uneyama, H. Tanaka, T. Yamanaka, T. Yasuda, M. Ono and Y. Kohmoto, J. Org. Chem., 1981, 46, 3312–3315 CrossRef CAS; (b) A. Shimizu, R. Hayashi, Y. Ashikari, T. Nokami and J.-i. Yoshida, Beilstein J. Org. Chem., 2015, 11, 242–248 CrossRef; (c) Y. Zhang, A. Iqbal, J. Zai, S.-Y. Zhang, H. Guo, X. Liu, I. ul Islam, H. Fazal and X. Qian, Org. Chem. Front., 2022, 9, 436–444 RSC; (d) X. Luo, B. Wu, J. Li, Y. Wang, X. Tang, C. Li, M. Shao and Z. Wei, J. Am. Chem. Soc., 2023, 145, 20665–20671 CrossRef CAS PubMed.
- W. R. Leow, Y. W. Lum and E. H. Sargent, Science, 2020, 368, 1228–1233 CrossRef CAS.
- D. Ma, B. Hu, W. Wu, X. Liu, J. Zai, C. Shu, T. T. Tadesse and T. L. Liu, Nat. Commun., 2019, 10, 3367 CrossRef.
- M. Liu, T. Feng, Y. Wang, G. Kou, Q. Wang, Q. Wang and Y. Qiu, Nat. Commun., 2023, 14, 6467 CrossRef CAS.
- (a) M. Chung, K. Jin, J. S. Zeng and K. Manthiram, ACS Catal., 2020, 10, 14015–14023 CrossRef CAS; (b) H. Tang, J. R. Vanhoof and D. De Vos, Green Chem., 2022, 24, 9565–9569 RSC.
- (a) Y. Lv, Z.-W. Hou, P. Li and L. Wang, Org. Chem. Front., 2023, 10, 990–995 RSC; (b) Z. Zhang, Z.-W. Hou, H. Chen, P. Li and L. Wang, Green Chem., 2023, 25, 3543–3548 RSC.
- (a) Z.-W. Hou, T. Jiang, T.-X. Wu and L. Wang, Org. Lett., 2021, 23, 8585–8589 CrossRef CAS PubMed; (b) Z.-W. Hou, L. Li and L. Wang, Org. Chem. Front., 2022, 9, 2815–2820 RSC; (c) L. Li, Z.-W. Hou, P. Li and L. Wang, J. Org. Chem., 2022, 87, 8697–8708 CrossRef CAS PubMed; (d) Y. Lv, Z.-W. Hou, Y. Wang, P. Li and L. Wang, Org. Biomol. Chem., 2023, 21, 1014–1020 RSC; (e) Q. Wan, Z.-W. Hou, X.-R. Zhao, X. Xie and L. Wang, Org. Lett., 2023, 25, 1008–1013 CrossRef CAS.
- (a) M. Paradas, A. G. Campaña, T. Jiménez, R. Robles, J. E. Oltra, E. Buñuel, J. Justicia, D. J. Cárdenas and J. M. Cuerva, J. Am. Chem. Soc., 2010, 132, 12748–12756 CrossRef CAS; (b) T. Jiménez, A. G. Campaña, B. Bazdi, M. Paradas, D. Arráez-Román, A. Segura-Carretero, A. Fernández-Gutiérrez, J. E. Oltra, R. Robles, J. Justicia and J. M. Cuerva, Eur. J. Org. Chem., 2010, 4288–4295 CrossRef; (c) M. Noji, M. Baba, R. Hirabe, S. Hayashi and T. Takanami, Chem. Commun., 2021, 57, 7104–7107 RSC; (d) J. Justicia, T. Jiménez, S. P. Morcillo, J. M. Cuerva and J. E. Oltra, Tetrahedron, 2009, 65, 10837–10841 CrossRef CAS; (e) S. M. Shafi, J. Chou, K. Kataoka and J. Nokami, Org. Lett., 2005, 7, 2957–2960 CrossRef CAS PubMed; (f) M. Teruaki, I. Kiyomi, Y. Tohru and T. Toshihiro, Chem. Lett., 1992, 21, 231–234 CrossRef.
- C. Wang and H. Yamamoto, Org. Lett., 2014, 16, 5937–5939 CrossRef CAS.
- (a) O. V. Bityukov, V. A. Vil’, G. I. Nikishin and A. O. Terent'ev, Adv. Synth. Catal., 2021, 363, 3070–3078 CrossRef CAS; (b) J. Seitz and T. Wirth, Org. Biomol. Chem., 2021, 19, 6892–6896 RSC.
- (a) Y. Yuan, A. Yao, Y. Zheng, M. Gao, Z. Zhou, J. Qiao, J. Hu, B. Ye, J. Zhao, H. Wen and A. Lei, iScience, 2019, 12, 293–303 CrossRef CAS; (b) L. G. Gombos, L. Werner, D. Schollmeyer, C. A. Martínez-Huitle and S. R. Waldvogel, Eur. J. Org. Chem., 2022, 2022, e202200857 CrossRef CAS.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: https://doi.org/10.1039/d3gc04061e |
| This journal is © The Royal Society of Chemistry 2024 |