Tm3+-mediated energy bridge in lead-free double perovskites: suppressing multiphonon relaxation for multifunctional photonic applications
Jiaqi
Zhao
a,
Zheng
Fu
a,
Kejie
Li
a,
Mengmeng
Dai
a,
Zhiying
Wang
 *b,
Guiying
Liang
c,
Yanling
Wei
*c and
Zuoling
Fu
*b,
Guiying
Liang
c,
Yanling
Wei
*c and
Zuoling
Fu
 *a
*a
aKey Laboratory of Physics and Technology for Advanced Batteries, College of Physics, Jilin University, Changchun 130012, China. E-mail: zlfu@jlu.edu.cn
bMinistry of Education Key Laboratory for Cross-Scale Micro and Nano Manufacturing, Nanophotonics and Biophotonics Key Laboratory of Jilin Province, School of Physics, Changchun University of Science and Technology, Changchun, 130022, China. E-mail: zhiyingw@cust.edu.cn
cSchool of Data Science and Artificial Intelligence, Jilin Engineering Normal University, Changchun 130052, China. E-mail: weiyanling@jlenu.edu.cn
First published on 18th December 2025
Abstract
The growing demand for efficient and sustainable photonic materials has spurred significant interest in eco-friendly lead-free double perovskites, which exhibit exceptional photoluminescence properties for next-generation sensing and display technologies. However, the emission performance of the material is limited by multiphonon relaxation, which leads to low energy efficiency under complex operating conditions. In this study, the 3H5 energy level of Tm3+ is strategically engineered as an energy bridge within lead-free double perovskites, effectively overcoming the constraints imposed by inefficient multiphonon relaxation in Er3+ (4I11/2 → 4I13/2). This targeted modulation of energy transfer pathways results in a significant 50-fold enhancement in the red-to-green (R/G) emission ratio of Er3+, increasing from 0.09 to 4.58, accompanied by a pronounced chromaticity shift from green (0.278, 0.709) to red (0.678, 0.320). This tunable emission behavior is further modulated by laser power density and excitation wavelength (808 or 980 nm). Moreover, Gd2MgTiO6 (GMTO):Er3+,Tm3+ demonstrates excellent temperature-measuring ability based on the luminescence intensity ratio (2H11/2/4F9/2 → 4I15/2), achieving a maximum relative sensitivity of 1.03% K−1 at 313 K. This work not only demonstrates a generalizable strategy to suppress multiphonon relaxation via lanthanide energy bridges but also paves the way for multifunctional photonic devices tailored to emerging applications in smart sensing, secure communication, and next-generation optoelectronics.
1. Introduction
Lanthanide-doped luminescent materials have garnered considerable attention in various fields, including solid-state lighting, bio-imaging, information encryption, and solar energy conversion, due to their unique optical properties, such as narrow emission bands, long lifetimes, and excellent photostability.1–6 The energy transfer (ET) process between lanthanide ions is a key mechanism that regulates the luminescent performance of these materials, as it enables the manipulation of emission intensity, color, and even multi-functional integration to be manipulated.7 However, traditional ET strategies often suffer from limitations such as low efficiency, single functionality and difficulty in achieving dynamic tuning, which restricts the further development of lanthanide-doped materials in advanced applications.8–10 Therefore, exploring novel ET strategies to overcome these bottlenecks has become a pressing research goal. Recently, double perovskites with the general formula A2BB′O6 have emerged as promising host matrices for lanthanide doping, owing to their superior structural stability, flexible chemical composition, and unique crystal field environment.11–13 Compared to conventional single perovskites (ABO3) or other host materials (e.g., oxides, fluorides), double perovskites offer more adjustable cation sites (A, B, and B′) with diverse coordination numbers and ionic radii, providing a versatile platform to design and optimize the spatial distribution and interaction of lanthanide ions.14,15 This structural flexibility not only facilitates the construction of efficient ET pathways but also allows for the integration of multiple functionalities by tailoring the doping configuration and host composition. Thus, double perovskites hold great potential to serve as ideal hosts for realizing innovative ET strategies.16,17 Currently, down-conversion (DC) luminescence in rare-earth-doped double perovskites has been extensively studied,18–20 however, up-conversion (UC) luminescence properties remain underexplored. Among halide perovskites, materials such as Cs2NaBiCl6 and other Cs-based compounds exhibit excellent UC luminescent performance, but their synthesis often requires prohibitively harsh conditions (HCl vapors, T > 150 °C) and degrades under standard environmental stressors (RH > 30%, T > 85 °C).21 These challenges, coupled with potential toxicity concerns associated with certain halide components,22 create a critical gap in the development of robust, scalable UC materials.In this study, the oxide-based and lead-free halide perovskite luminescent material Gd2MgTiO6 (GMTO) was selected as a promising host due to its low toxicity, easy preparation, and exceptional optoelectronic properties. Rare earth ions (Er3+,Tm3+ and Yb3+) were strategically doped into the GMTO, enabling tunable UC luminescence under two near-infrared light sources (980 nm or 808 nm). Notably, the Tm3+ ions act as an ET-bridge, thereby effectively modulating the red-to-green (R/G) emission ratio of the Er3+ ions. Importantly, the UC luminescence can be controlled by adjusting the laser power density, allowing for the innovative design of a series of anti-counterfeiting and information storage patterns. Meanwhile, a series of highly sensitive thermometers based on GMTO:Yb3+,Er3+,Tm3+ have been developed based on the luminescence intensity ratio (LIR) temperature sensing technology, which shows promise for real-time monitoring and temperature measurement in extreme working environments. These rare earth-doped GMTO materials exhibit exceptional optical properties and significant application potential, laying a solid foundation for the advancement of multifunctional luminescent materials.
2. Experimental section
2.1 Material and reagents
Gd2O3 (99.8%, Beijing Chemical Factory), TiO2 (AR, Sino pharm Chemical Reagent Co., Ltd), MgO (99.8%, Beijing Chemical Factory), H3BO3 (99.9%, Produced in Shanghai, China), Yb2O3 (99.99%, Aladdin), Er2O3 (99.99%, Produced in Shanghai, China), Tm2O3 (99.99%, Produced in Shanghai, China), anhydrous ethanol (99.99%, Aladdin). All the chemicals are used without any further purification.2.2 Synthesis of Gd2MgTiO6:2%Er3+,xTm3+(x = 0%, 0.5%, 2%, 3%)
A series of Er3+ and Tm3+ co-doped GMTO compounds were synthesized by a high-temperature solid-state method. During sample preparation, 2% (by mass) H3BO3 was added as a flux, followed by the addition of an appropriate amount of anhydrous ethanol during grinding. Since MgO was easily volatilized at high temperature, its content in the total mixture was set to be 10% in excess. The remaining raw materials were precisely weighed in accordance with the predetermined stoichiometric ratio. Subsequently, the accurately measured samples were transferred to an agate mortar and subjected to thorough grinding for 30 minutes. The heating process was carried out in two steps. Initially, the temperature was raised from ambient to 1000 °C at a controlled rate of 10 °C min−1 in air. Subsequently, it was further increased to 1400 °C at a slower rate of 5 °C min−1 in air, and the samples were maintained at this elevated temperature for 8 h continuously. After completion of the heat treatment, the samples were left to cool naturally to room temperature. They were then retrieved and placed back into the agate mortar for an additional round of intensive grinding to ensure homogeneity. The well-prepared samples were then ready for subsequent testing and analysis.2.3 Synthesis of GMTO:7%Yb3+,2%Er3+,yTm3+(y = 0.5%, 2%, 3%)
The sample preparation conditions and process were the same as those of GMTO:Er3+,Tm3+, with raw materials still weighed according to the stoichiometric ratio.2.4 Characterization
Powder X-ray diffraction (XRD) measurements were performed using an X-ray diffractometer equipped with Cu Kα radiation (λ = 0.15405 nm). To visualize the morphological features and analyze the elemental distribution within the samples, a field-emission scanning electron microscope (FE-SEM, model: Regulus-8100, Hitachi) integrated with an energy-dispersive X-ray spectroscopy (EDS) detector was employed. The Raman spectrum was recorded by the Renishaw in Via Raman spectrometer (VERTEX 80v, λex = 532 nm). The luminescence spectra were recorded by an Andor SR-500i spectrometer (Andor Technology Co, Belfast, U.K.) equipped with a SR830 DSP lock-in amplifier and a DInGaAs1700-TE detector (1100 nm–1700 nm) and a DSi300-detector (380 nm–800 nm). The copper-constantan thermocouple and a temperature control system (TAP-02, orient-KOJI) were used to conduct temperature sensing. The decay lifetime curves of the samples were measured using an Omni-λ500 (Zolix, China) spectrometer with 808 and 980 nm (MDL-III-808/980-2 W, China) laser excitation.3. Results and discussion
3.1 Material characterization
Ultra-bright and tunable double perovskite UC phosphors GMTO:Ln3+ have been synthesized via a high-temperature solid-state method. It can be clearly observed that the reflection peaks of the samples completely match those of ICSD#117584 of GMTO (Fig. 1a and b). Considering that the dopant ions such as Er3+ (0.89 Å), Tm3+ (0.88 Å), and Yb3+ (0.87 Å) have a smaller radius than Gd3+ (0.94 Å), the ionic radius percentage formula eqn (1)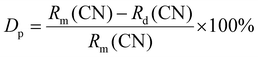 | (1) |
3.2 Suppression of Er3+ cross relaxation by introducing Tm3+–3H5 level under 980 nm excitation
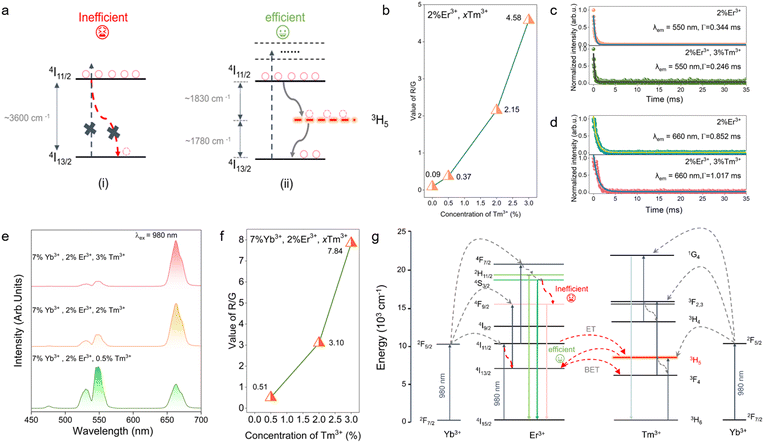
In Er3+-only systems, the large energy gap between the 4I11/2 and 4I13/2 levels is predominantly depopulated via multiphonon relaxation, a nonradiative process that dissipates the excitation energy before efficient radiative emission can occur (Fig. 2a(i)). In contrast, the 3H5 level of Tm3+ is well matched to the Er3+ 4I11/2 → 4I13/2 splitting and can act as an effective energy bridge, providing a competitive energy-transfer pathway that partially suppresses multiphonon relaxation. As a result, population of the Er3+ emitting levels is enhanced and Er3+-related radiative transitions are significantly promoted (Fig. 2a(ii)). As shown in Fig. S3a, the UC emission spectra of GMTO:2%Er3+,xTm3+(x = 0–3%) clearly exhibit the characteristic emission bands of Er3+ without detectable Tm3+ luminescence. The emissions at 530, 550 and 660 nm are attributed to the 2H11/2, 4S3/2 and 4F9/2 to the ground state (4I15/2) transitions of Er3+, respectively.32 Without the Tm3+-doping, the UC emission spectrum exhibits a dominant green emission. Fig. 2b shows that, in 2%Er3+,xTm3+(x = 0–3%) samples under 980 nm excitation, increasing Tm3+ concentration leads to a progressive dominance of red emission accompanied by a pronounced rise in the R/G intensity ratio. To further verify the effective energy transfer introduced by the Tm3+ levels, we measured the UC decay lifetimes of GMTO:2%Er3+ and GMTO:2%Er3+,3%Tm3+. As shown in Fig. 2c and d, co-doping with Tm3+ shortens the Er3+ green (550 nm) lifetime from 0.344 to 0.246 ms, whereas the red (660 nm) lifetime is prolonged from 0.852 to 1.017 ms. These opposite trends provide compelling evidence that Tm3+ serves as an efficient energy bridge that redistributes population from the green-emitting to the red-emitting levels of Er3+. Taken together, these results demonstrate that Tm3+ acts as an effective mediator of Er3+ UC luminescence in the co-doped system, functioning exclusively as a spectral regulator rather than an emissive activator.However, under 980 nm excitation, the emission of Tm3+ is inefficient. To overcome this limitation, Yb3+ ions are introduced as an intermediate energy bridge, which not only effectively enhances the Tm3+ emission but also enables an additional blue emission band of Tm3+ (1G4 → 3H6). This dual-sensitization strategy highlights the synergistic role of Yb3+ in broadening the spectral tunability of lanthanide-doped UC luminescent materials.33,34 Similarly, as illustrated in Fig. 2f and S3c, the R/G intensity ratio of Yb3+, Er3+,Tm3+ tri-doped system also exhibits an increase with the rising Tm3+ concentration. In order to investigate the intrinsic mechanism in GMTO:Er3+,Tm3+ and GMTO:Yb3+,Er3+,Tm3+ system under 980 nm excitation, the correlation between the integrated emission intensity (I) and the laser power (P) is illustrated in Fig. S3b and d. This relationship can be adequately described by the following eqn (2):35
| I∝Pn | (2) |
We therefore propose that introducing Tm3+ creates an intermediate 3H5 level between the Er3+ 4I11/2 and 4I13/2 states, acting as an energy bridge. The original 3600 cm−1 gap is effectively split into two smaller gaps of ∼1830 and ∼1780 cm−1 by the 3H5(Tm3+) level. Subsequent energy transfer (ET) and back energy transfer (BET) between Er3+ 4I11/2 and Tm3+ 3H5 promote population build-up in the Er3+ 4I13/2 level, enhancing the probability of excitation to 4F9/2 (and other higher states) and thereby increasing the population density at 4F9/2. Overall, incorporation of the Tm3+–3H5 level effectively mitigates inefficient multiphonon relaxation between 4I11/2 and 4I13/2, establishes a bidirectional ET channel between Er3+ and Tm3+, and significantly boosts population in the key intermediate 4I13/2 state, leading to more efficient UC and intensified red emission. Furthermore, as shown in Fig. 2d–f, additional sensitization by Yb3+ enables Tm3+ to undergo three-photon UC to yield blue emission (1G4 → 3H6). Taken together, these results demonstrate a rational strategy for tuning UC luminescence by constructing optimized ET channels that exploit the complementary energy-level structures of Er3+,Tm3+, and Yb3+.
3.3 Photoluminescence properties upon 808 nm excitation
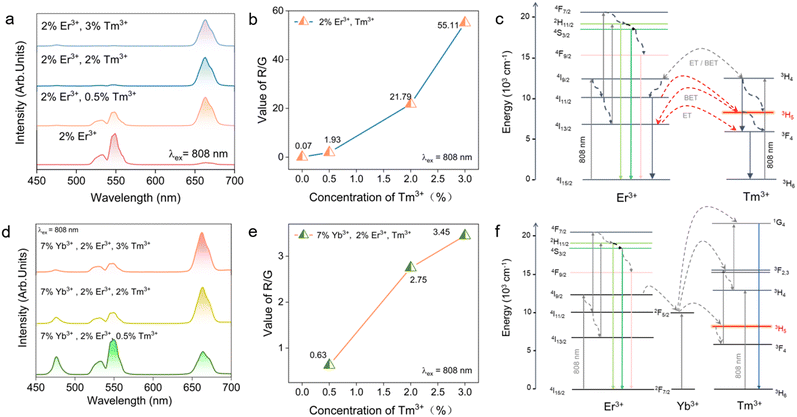
Owing to the rich ladder-like energy-level structure of Er3+, different excitation wavelengths can selectively address distinct intermediate states, offering an additional degree of freedom for tailoring UC pathways. Building on the 980 nm pumping results, we therefore explored how exploiting this multilevel structure under 808 nm excitation could further regulate the UC behavior. Remarkable UC luminescence was observed in GMTO co-doped with Er3+,Tm3+ and Yb3+ under 808 nm excitation. A systematic study of the UC emission spectra and associated ET dynamics revealed significant modulation of the emission color. As shown in Fig. 3a, b and S4a, we observed a systematic increase in the R/G emission ratio. A systematic study of the UC emission spectra and associated ET dynamics revealed significant modulation of the emission. Notably, lifetime analysis (Fig. S5) confirms this finding, under 808 nm excitation, the introduction of Tm3+ ions regulates the energy level populations of Er3+. The lifetime of Er3+ green emission (550 nm, 4S3/2 → 4I15/2) shortens from 0.546 ms (single-doped) to 0.473 ms (co-doped), attributed to efficient energy transfer from Er3+ 4S3/2 to Tm3+–3H5. In contrast, the red emission lifetime (660 nm, 4F9/2 → 4I15/2) extends from 0.937 ms to 1.738 ms, as Tm3+ acts as an “energy bridge” to split the large energy gap between Er3+ 4I11/2 and 4I13/2, suppressing non-radiative multiphonon relaxation and enhancing the population of the 4F9/2 level. The power-dependent studies (Fig. S4b) provide critical insights into the UC mechanisms in GMTO:2%Er3+,3%Tm3+. The n values for Er3+ emissions at 530 nm (n = 2.3), 550 nm (n = 2.01) and 660 nm (n = 1.75) confirm a two-photon excitation process. Under 808 nm irradiation, the higher photon energy enables direct population of the 4I9/2 state in Er3+. Subsequent NR relaxation processes combined with the ET and BET between Er3+ and Tm3+, facilitate efficient energy redistribution. Notably, the Tm3+–3H4 and 3H5 states are found to mediate ET from Er3+–4I9/2 to the 4I13/2 and 4I11/2 states. This is supported by the systematic decrease in Er3+ infrared emission intensity with increasing Tm3+ concentration (Fig. S6), indicating Tm3+-mediated depletion of the 4I11/2 population through BET processes. The observed concentration-dependent quenching behavior confirms the critical role of Tm3+ in modifying the Er3+ excited state population.In contrast to the absorption and pumping dynamics observed under 980 nm excitation, 808 nm photon absorption shows a significantly higher probability of populating the 2H11/2 and 4F7/2 states compared to the 4F9/2 level in Er3+ ions. As shown in Fig. 3c, under 808 nm excitation, red emission predominantly occurs via the relaxation of electrons from the high-energy 4F7/2 and 2H11/2 states rather than through a direct radiative transition from the 4F9/2 state (4F9/2 → 4I15/2). The introduction of Tm3+ ions plays a crucial role in modifying the energy landscape, significantly reducing the energy difference between the 4I11/2 and 4I13/2 states of Er3+. This reduction facilitates enhanced population at the 4I13/2 energy level through BET. In addition, Tm3+ ions efficiently absorb 808 nm photons, promoting transitions from the 3H6 ground state to the 3H4 excited state. Subsequent RET from Tm3+ (3H4) to Er3+ (4I9/2) and NR relaxation processes populate the 4I11/2 state of Er3+, ultimately increasing the population at the 4I11/2 energy level. Thus, under 808 nm excitation, the enhanced red luminescence results from the combined effects of (i) direct Er3+ excitation, (ii) efficient ET from Tm3+ to Er3+ and (iii) multi-pathway energy migration processes. These mechanisms promote the population of high-energy emitting states, thereby optimizing red light generation.
Fig. 3d demonstrates that the incorporation of Yb3+ ions enables blue emission from Tm3+ under 808 nm excitation. The doping concentrations of Yb3+ and Tm3+ were found to effectively modulate Er3+ luminescence, and the variation in the R/G emission ratio (Fig. S4c and Fig. 3e). Notably, the presence of Yb3+ necessitates consideration of ET processes between Yb3+ and Tm3+. The power-dependent studies (Fig. S4d) provide critical insights into the UC mechanisms. The n values for Er3+ emissions at 530 nm (n = 2.44), 550 nm (n = 2.08) and 660 nm (n = 1.87), as well as the n values for Er3+ emissions at 475 nm (n = 2.06), confirm a two-photon excitation process. Therefore, a proposed ET mechanism for the Yb3+, Er3+,Tm3+ system under 808 nm excitation is illustrated in Fig. 3f. Our results suggest that the concentration of Tm3+ plays a dominant role in tuning the color of Er3+ UC emission. Additionally, we postulate that laser power is another critical parameter for luminescence modulation. Systematic investigations reveal that UC emission in both Er3+,Tm3+ and Yb3+,Er3+,Tm3+ systems can be effectively tuned through the selection of excitation wavelength (980 nm or 808 nm) coupled with precise modulation of the laser power (Fig. S7), the R/G emission ratio in the GMTO:2%Er3+,0.5%Tm3+ phosphor displays a distinct decreasing trend as the laser power density increases from 0.5 to 4 W, which differs from the concentration-dependent tuning effects. The multimodal and tunable luminescence characteristics of GMTO:Ln3+ samples provide a robust theoretical and experimental foundation for future developments in information storage and fluorescence-based anti-counterfeiting technologies.36–39 CIE coordinate analysis (Fig. S8) further quantified this wavelength-dependent and power-dependent chromaticity shift, revealing that the magnitude of color change was significantly greater under 808 nm excitation than under 980 nm excitation. These results also demonstrate the existence of an excitation wavelength/power-selective ET mechanism, highlighting the crucial role of pump wavelength optimization in tuning UC luminescence.
3.4 Temperature sensing and optical information storage
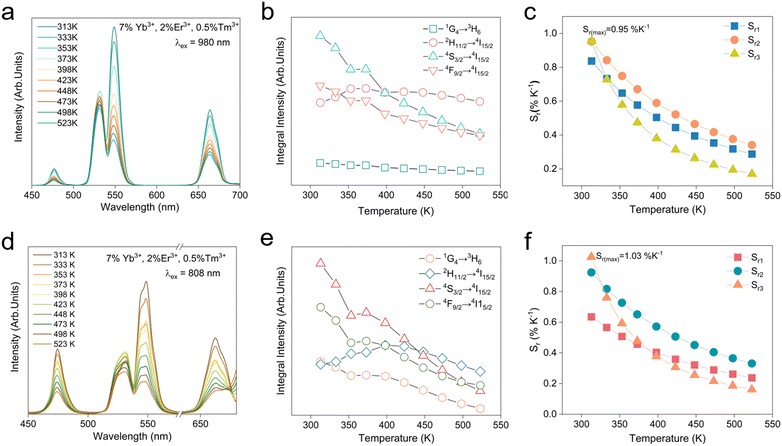
It is well established that lanthanide ions (Ln3+) and transition metal ions exhibit considerable potential for applications in temperature sensing and optical anti-counterfeiting owing to their distinctive luminescence characteristics. The luminescence intensity ratio (LIR) technique relies on the thermally induced redistribution of electrons between thermally coupled levels (TCLs) or non-thermally coupled levels (non-TCLs) of these ions. Nevertheless, such systems are inherently constrained by the relatively narrow energy gaps (typically ΔE = 200–2000 cm−1), which can limit their sensitivity and operational range.40 Although non-TCL-based LIR techniques overcome the limitation of ΔE, they introduce another significant challenge: the absence of a Boltzmann distribution between the two monitored energy levels prevents the establishment of thermal equilibrium. Consequently, the ET efficiency from higher to lower energy levels adversely affects the achievable temperature sensitivity. These fundamental limitations highlight that conventional single-approach LIR thermometry inevitably faces intrinsic drawbacks, underscoring the need for innovative alternative strategies.Building upon these facts, we conducted a comprehensive investigation of LIR thermometry by synergistic integration of both TCLs and non-TCLs, thereby achieving enhanced temperature sensing accuracy. For experimental validation, we selected GMTO:7%Yb3+,2%Er3+,0.5%Tm3+ phosphors, and monitored their luminescence over a broad temperature range (313–523 K) under 980 nm excitation (Fig. S9). As depicted in Fig. 4a, the temperature-dependent UC emission spectra revealed distinct thermal responses for different transitions (Fig. 4b). Specifically, the emissions from the 1G4 → 3H6, 4S3/2 → 4I15/2, and 4F9/2 → 4I15/2 transitions exhibited significant thermal quenching with increasing temperature, while the 2H11/2 → 4I15/2 transition demonstrated remarkable thermal stability. Based on these observations, we developed a triple-mode LIR thermometric model that incorporates both non-TCLs (LIR1 and LIR3 to eqn (3)) and TCLs mechanisms (LIR2 to eqn (4)). In particular, LIR2 utilized the classic Boltzmann-distribution-based TCL approach (2H11/2/4S3/2):32,41
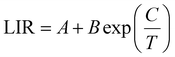 | (3) |
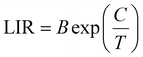 | (4) |
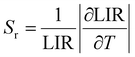 | (5) |
As presented in Fig. 4c, the Sr(max) value of the three modes above reaches up to 0.95% K−1 in LIR2 and LIR3 at 313 K.
The temperature-dependent UC emission spectra of GMTO:7%Yb3+,2%Er3+,0.5%Tm3+ under 808 nm excitation were systematically investigated (Fig. 4d). Notably, the thermal response trends of the emissions closely resembled those observed under 980 nm excitation (Fig. 4e). Distinct thermal behaviors were identified for the transitions originating from 1G4 → 3H6 (Tm3+), 4S3/2 → 4I15/2 (Er3+), and 4F9/2 → 4I15/2 (Er3+), all of which exhibited pronounced thermal quenching. In contrast, the 2H11/2 → 4I15/2 (Er3+) transition demonstrated an initial increase in emission intensity with rising temperature, followed by quenching at elevated temperatures. These characteristics enabled the development of a robust multimodal LIR thermometry system under 808 nm excitation, similar to the three-mode LIR temperature sensing achieved under 980 nm excitation (Fig. S11(a–c)). Notably, as shown in Fig. 4f, the optimized Sr for LIR3 reached 1.03% K−1 at 313 K. A comparison of the Sr obtained in this study with those reported in previously published works was provided in Table S2, further confirming the excellent temperature-sensing performance of the proposed thermometer. In addition, GMTO:Yb3+,Er3+,Tm3+ exhibits outstanding thermal stability, as demonstrated by its performance over heating–cooling cycles between 333 K and 423 K. And the temperature uncertainty (δT) is a critical parameter for optical temperature sensing, the formula is as follows:44
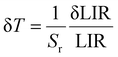 | (6) |
The δT of GMTO:Yb3+,Er3+,Tm3+ reflects the precision of its temperature measurement ability. As displayed in Fig. S12, the LIR value is obtained by 50 times consecutive tests at 333 K. The δT values of LIR are 0.53 K. Our findings demonstrated that GMTO:Yb3+,Er3+,Tm3+ phosphors not only possessed dual-excitation capability (808/980 nm) but also enabled high-performance multimode LIR-based temperature sensing under dual-mode excitation.
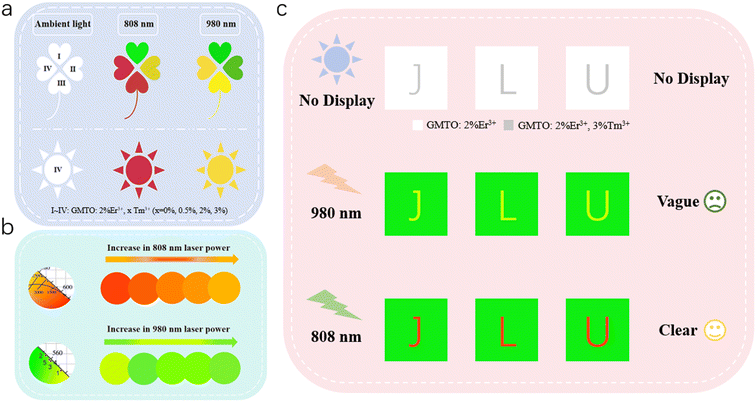
Leveraging the dual-mode excitation properties and the tunability of the UC luminescence via modulation of doping concentration and laser power (Fig. S13), we have developed strategies for advanced optical anti-counterfeiting and information storage applications. As illustrated in Fig. 5a, our design utilizes GMTO:Yb3+,Tm3+ and GMTO:Yb3+,Er3+,Tm3+ phosphors with precisely tailored doping concentrations, patterned into clover and sun motifs. These phosphors exhibit distinct chromatic responses under alternating 980 nm and 808 nm laser excitation, enabling dynamic color-switching within the encoded patterns. This functionality provides a robust platform for high-security optical encryption and multiplexed data storage. Fig. 5b schematically depicts the laser power-dependent color modulation of the UC emission in GMTO:2%Er3+,3%Tm3+, highlighting its exceptional potential for rewritable optical storage and related photonic applications. The capability to tune emission color in real time via excitation power allows for programmable spectral signatures, facilitating advanced optical encryption and establishing a new paradigm for dynamic information encoding. Furthermore, Fig. 5c demonstrates a sophisticated anti-counterfeiting application based on phosphor-mediated optical contrast. In this configuration, phosphors “I” (background) and “IV” (characters “J L U”) are selectively deposited to create a dual-responsive authentication label. Under 980 nm excitation, a faint green “J L U” pattern becomes visible, while switching to 808 nm irradiation induces a vivid red emission. This reversible high-contrast transition enables dynamic optical verification and establishes a new paradigm for secure, stimuli-responsive anti-counterfeiting technologies.
4. Conclusion
In conclusion, we have engineered a lead-free Gd2MgTiO6:Yb3+,Er3+,Tm3+ double perovskite system centered on a Tm3+-mediated energy bridge, where the Tm3+–3H5 state strategically mitigates the inefficient multiphonon relaxation of Er3+ (4I11/2 → 4I13/2). This targeted ET modulation yields a remarkable 50-fold enhancement in the R/G emission ratio of Er3+ (from 0.09 to 4.58) compared to Er3+-single-doped GMTO under 808 nm excitation, accompanied by a dramatic chromaticity shift from green (0.278, 0.709) to red (0.678, 0.320). Integrating Yb3+ further enables multidimensional optical control: by tuning excitation wavelength (808/980 nm) and laser power, we achieve programmable multicolor emission, which exhibits robust potential for laser-encoded anti-counterfeiting and rewritable optical storage. Moreover, the optimized GMTO:7%Yb3+,2%Er3+,0.5%Tm3+ phosphor demonstrates exceptional thermometric performance, with a maximum relative sensitivity of 1.03% K−1 at 313 K. This work establishes a generalizable strategy to suppress multiphonon relaxation via lanthanide energy bridges, demonstrates a single lead-free matrix integrating dynamic upconversion tuning, high-sensitivity thermal sensing, and optical encryption, and formulates design principles for phonon-engineered energy transfer in perovskite hosts. These advances that bridge spectral manipulation with multifunctional photonic applications provide a versatile platform for next-generation compact, high-efficiency optoelectronic devices in smart sensing and secure communication.Conflicts of interest
The authors declare no competing financial interest.Data availability
All data generated or analysed during this study are included in this published article and its supplementary information (SI). Supplementary information: detailed descriptions of LIR model, crystal structure, FE-SEM image and elemental mapping images, CIE chromatic coordinates and NIR emission spectrum. See DOI: https://doi.org/10.1039/d5qi02407b.Data are available from the corresponding author on reasonable request.
Acknowledgements
This work was supported by the Natural Science Foundation of Jilin Province (YDZJ202501ZYTS607) and the National Natural Science Foundation of China (Grant No. 12374374). The authors would like to acknowledge the Instrument and Equipment Sharing Platform, College of Physics (Jilin University) for testing assistance.References
- R. Wu, P. Han, D. Zheng, J. Zhang, S. Yang, Y. Zhao, X. Miao and K. Han, All-Inorganic Rare-Earth-Based Double Perovskite Nanocrystals with Near-Infrared Emission, Laser Photonics Rev., 2021, 15(11), 2100218 CrossRef CAS.
- R. Zhang, Z. Wang, X. Xu, X. Mao, J. Xiong, Y. Yang and K. Han, All-Inorganic Rare-Earth Halide Double Perovskite Single Crystals with Highly Efficient Photoluminescence, Adv. Opt. Mater., 2021, 9(19), 2100689 CrossRef CAS.
- X. Li, D. Wang, Y. Zhong, F. Jiang, D. Zhao, S. Sun, P. Lu, M. Lu, Z. Wang and Z. Wu, et al. Halide Double Perovskite Nanocrystals Doped with Rare-Earth Ions for Multifunctional Applications, Adv. Sci., 2023, 10(20), 2207571 CrossRef CAS PubMed.
- Z. Rao, X. Zhao and X. Gong, Rare-Earth-Based Lead-Free Halide Double Perovskites for Light Emission: Recent Advances and Applications, Adv. Funct. Mater., 2024, 34(44), 2406424 CrossRef CAS.
- K. Li, M. Jia, J. Zhao, G. Zhang, D. Guo, Z. Wang and Z. Fu, Simultaneous Tailoring of Robust Anti-Thermal Quenching and High Thermometric Sensitivity in Lanthanide-Doped Cs2NaYF6 Double Perovskites, Laser Photonics Rev., 2025, 19, e00662 CrossRef CAS.
- Z. Zheng, J. Zhang, X. Liu, R. Wei, F. Hu and H. Guo, Luminescence and self-referenced optical temperature sensing performance in Ca2YZr2Al3O12:Bi3+, Eu3+ phosphors, Ceram. Int., 2020, 46(5), 6154–6159 CrossRef CAS.
- F. Li, L. Tu, Y. Zhang, D. Huang, X. Liu, X. Zhang, J. Du, R. Fan, C. Yang and K. W. Krämer, et al., Size-dependent lanthanide energy transfer amplifies upconversion luminescence quantum yields, Nat. Photonics, 2024, 18(5), 440–449 Search PubMed.
- E. M. Chan, G. Han, J. D. Goldberg, D. J. Gargas, A. D. Ostrowski, P. J. Schuck, B. E. Cohen and D. J. Milliron, Combinatorial Discovery of Lanthanide-Doped Nanocrystals with Spectrally Pure Upconverted Emission, Nano Lett., 2012, 12(7), 3839–3845 Search PubMed.
- H.-L. Wang, Y.-L. Li, H.-H. Zou, F.-P. Liang and Z.-H. Zhu, Smart Lanthanide Metal–Organic Frameworks with Multicolor Luminescence Switching Induced by the Dynamic Adaptive Antenna Effect of Molecular Rotors, Adv. Mater., 2025, 37(29), 2502742 Search PubMed.
- L. Yan, J. Huang, Z. An, Q. Zhang and B. Zhou, Spatiotemporal control of photochromic upconversion through interfacial energy transfer, Nat. Commun., 2024, 15(1), 1923 CrossRef CAS PubMed.
- H. Wang, J. Yao and R. Zeng, The luminescence modulation of rare earth-doped/containing lead-free double perovskites toward multifunctional applications: a review, Nanoscale, 2024, 16(14), 6837–6852 RSC.
- N. K. Tailor, A. Listorti, S. Colella and S. Satapathi, Lead-Free Halide Double Perovskites: Fundamentals, Challenges, and Photovoltaics Applications, Adv. Mater. Technol., 2023, 8(1), 2200442 CrossRef CAS.
- V. Kumar, A. Kathiravan and M. A. Jhonsi, Beyond lead halide perovskites: Crystal structure, bandgaps, photovoltaic properties and future stance of lead-free halide double perovskites, Nano Energy, 2024, 125, 109523 CrossRef CAS.
- Z.-X. Chen, Y. Chen and Y.-S. Jiang, J. Phys. Chem. B, 2002, 106, 9986–9992 CrossRef CAS.
- J. Zhao, J. Gao and W. Li, A combinatory ferroelectric compound bridging simple ABO3 and A-site-ordered quadruple perovskite, Nat. Commun., 2021, 12, 747 CrossRef CAS PubMed.
- X. Chen, J. Xu, Y. Xu, F. Luo and Y. Du, Rare earth double perovskites: a fertile soil in the field of perovskite oxides, Inorg. Chem. Front., 2019, 6(9), 2226–2238 RSC.
- H. W. Lei, D. Hardy and F. Gao, Lead-Free Double Perovskite Cs2AgBiBr6: Fundamentals, Applications, and Perspectives, Adv. Funct. Mater., 2021, 31, 2105898 CrossRef CAS.
- Q. Liu, E. Pan, H. Deng, F. Liu and J.-F. Li, Dy3+ doped (K,Na)NbO3-based multifunctional ceramics for achieving enhanced temperature-stable piezoelectricity and non-contact optical temperature sensing performance, Inorg. Chem. Front., 2023, 10(8), 2359–2369 RSC.
- P. Du and J. S. Yu, Near-ultraviolet light induced visible emissions in Er3+-activated La2MoO6 nanoparticles for solid-state lighting and non-contact thermometry, Chem. Eng. J., 2017, 327, 109–119 CrossRef CAS.
- W. Chen, Z. Liu, L. Shen, C. Shen, L. Ding, Z. Zhang, H. Zhang, W. Xiang and X. Liang, Design and energy transfer mechanism for single-phased Gd2MgTiO6: Bi3+, Eu3+ tunable white light-emitting phosphors, J. Mater. Sci., 2018, 54, 4056–4072 CrossRef.
- M. Cao, X. Zhao and X. Gong, Ionic Liquid-Assisted One-Step Large-Scale Synthesis of Cs2ZrCl6: CDs Composites with Tunable Multicolor Afterglow Emission, Adv. Funct. Mater., 2024, 34, 2408333 CrossRef CAS.
- I. Maietta, C. Otero-Martínez, S. Fernández, L. Sánchez, Á. González-Fernández, L. Polavarapu and R. Simón-Vázquez, The Toxicity of Lead and Lead-Free Perovskite Precursors and Nanocrystals to Human Cells and Aquatic Organisms, Adv. Sci., 2025, 12, 2415574 Search PubMed.
- X. Huang, B. Li and H. Guo, Synthesis, photoluminescence, cathodoluminescence, and thermal properties of novel Tb3+-doped BiOCl green-emitting phosphors, J. Alloys Compd., 2017, 695, 2773–2780 Search PubMed.
- K. Li, H. Z. Lian, M. M. Shang and J. Lin, A novel greenish yellow-orange red Ba3Y4O9:Bi3+, Eu3+ phosphor with efficient energy transfer for UV-LEDs, Dalton Trans., 2015, 44, 20542–20550 Search PubMed.
- A. Bartkowiak, M. Runowski, A. Shyichuk, M. Majewska and T. Grzyb, Tunable yellow-green up-conversion emission and luminescence lifetimes in Yb3+-Er3+-Ho3+ multi-doped β-NaLuF4 crystals, J. Alloys Compd., 2019, 793, 96–106 CrossRef CAS.
- K. Li and D. M. Zhu, Ratiometric optical temperature sensing properties based on up-conversion luminescence of novel NaLaTi2O6:Yb3+, Er3+/Ho3+ phosphors, Mater. Res. Bull., 2025, 181, 113117 Search PubMed.
- P. X. Gao, Z. Y. Zhou, P. Dong, Q. Li, H. H. Li, J. Wang, Z. Zhou and M. Xia, Tuning the luminescence properties of blue and far-red dual emitting Gd2MgTiO6: Bi3+, Cr3+ phosphor for LED plant lamp, J. Am. Ceram. Soc., 2021, 104, 6444–6454 CrossRef CAS.
- F. K. Shang, C. H. Hu, W. Xu, L. Zhao, S. N. Zong, M. Yao, F. Z. Bai and Z. G. Zhang, Modulation of luminescence thermometry in Nd3+-Yb3+ co-activated phosphors via engineering host matrix, J. Alloys Compd., 2022, 896, 162794 Search PubMed.
- C. Mi, J. J. Zhou, F. Wang and D. Y. Jin, Thermally enhanced NIR-NIR anti-Stokes emission in rare earth doped nanocrystals, Nanoscale, 2019, 12547–12552 Search PubMed.
- F. Auzel, Multiphonon-assisted anti-Stokes and Stokes fluorescence of triply ionized rare-earth ions, J. Lumin., 1976, 12–13, 715–722 CrossRef CAS.
- H. C. Yang, S. R. Zhang, H. Y. Yang, Y. Yuan and E. Z. Li, Vibrational spectroscopic and crystal chemical analyses of double perovskite Y2MgTiO6 microwave dielectric ceramics, J. Am. Ceram. Soc., 2020, 103, 1121–1130 CrossRef CAS.
- K. Saidi, M. Dammak, K. Soler-Carracedo and I. R. Martín, Optical thermometry based on upconversion emissions in Na3Gd (VO4)2: Yb3+-Er3+/Ho3+ micro crystals, J. Alloys Compd., 2022, 891, 161993 CrossRef CAS.
- H. Y. Lu, J. S. Yang, D. C. Huang, Q. L. Zou, M. W. Yang, X. R. Zhang, Y. X. Wang and H. M. Zhu, Ultranarrow NIR bandwidth and temperature sensing of YOF: Yb3+/Tm3+phosphor in low temperature range, J. Lumin., 2019, 206, 613–617 CrossRef CAS.
- K. Li, D. Li, M. Jia, D. Guo, M. Dai, J. Zhao, G. Chen and Z. Fu, Li-Based Nanoprobes with Boosted Photoluminescence for Temperature Visualization in NIR Imaging-Guided Drug Release, Nano Lett., 2025, 25, 776–785 CrossRef CAS.
- H. X. Zhang, Y. Fan, P. Pei, C. X. Sun, L. F. Lu and F. Zhang, Tm3+-Sensitized NIR-II Fluorescent Nanocrystals for In Vivo Information Storage and Decoding, Angew. Chem., Int. Ed., 2019, 58, 10153 CrossRef CAS.
- X. F. Zhao, Z. Y. Wu, Z. N. Yang, X. Yang, Y. Y. Zhang, M. H. Yuan, K. Han, C. Q. Song, Z. F. Jiang, H. Y. Wang, S. Q. Li and X. J. Xu, Dual-Wavelength Excited Intense Red Upconversion Luminescence from Er3+-Sensitized Y2O3 Nanocrystals Fabricated by Spray Flame Synthesis, Nanomaterials, 2020, 10, 1475 CrossRef CAS.
- J. I. Eldridge, Luminescence decay-based Y2O3: Er phosphor thermometry: Temperature sensitivity governed by multiphonon emission with an effective phonon energy transition, J. Lumin., 2019, 214, 116535 CrossRef CAS.
- N. Yamada, S. Shionoya and T. Kushida, Phonon-assisted energy transfer between trivalent rare earth ions, J. Phys. Soc. Jpn., 1972, 32, 1577–1586 CrossRef CAS.
- A. Ciric, S. Stojadinovic and M. D. Dramicanin, An extension of the Judd-Ofelt theory to the field of lanthanide thermometry, J. Lumin., 2019, 216, 116749 CrossRef CAS.
- J. H. Chung, S. Y. Lee, K. B. Shim and J. H. Ryu, White Lighting Upconversion in Tm3+/Ho3+/Yb3+ Co-Doped CaWO4, Appl. Phys. Express, 2012, 5, 052602 CrossRef.
- Y. Tong, W. Zhang, R. Wei, L. Chen and H. Guo, Na2YMg2(VO4)3: Er3+, Yb3+ phosphors: Up-conversion and optical thermometry, Ceram. Int., 2021, 47, 2600–2606 Search PubMed.
- A. Dogan, M. Erdem, K. Esmer and G. Eryürek, Upconversion luminescence and temperature sensing characteristics of Ho3+/Yb3+ co-doped tellurite glasses, J. Non-Cryst. Solids, 2021, 571, 121055 CrossRef CAS.
- S. Zhou, S. Jiang, X. Wei, Y. Chen, C. Duan and M. Yin, Optical thermometry based on upconversion luminescence in Yb3+/Ho3+ co-doped NaLuF4, J. Alloys Compd., 2014, 588, 654–657 CrossRef CAS.
- G. B. Nair, S. Tamboli, S. J. Dhoble and H. C. Swart, LaOF: Yb3+, Er3+ Upconversion Nanophosphors Operating at Low Laser Powers for Nanothermometry Applications, ACS Appl, Nano Mater., 2023, 6, 15255–15265 CAS.
| This journal is © the Partner Organisations 2026 |