Universal upconversion amplification via 4d–4f hybridization in Ln3+/MoO42− co-doped fluorapatite: mechanism and applications
Boyang
Wang
 a,
Yuankun
Ren
a,
Xiaohan
Wei
a,
Wei
Yang
a,
Yuankun
Ren
a,
Xiaohan
Wei
a,
Wei
Yang
 a,
Haoming
Su
a,
Haoming
Su
 b,
Yu
Liu
a,
Jiaqi
Liang
a and
Xiyu
Li
b,
Yu
Liu
a,
Jiaqi
Liang
a and
Xiyu
Li
 *a
*a
aState key Laboratory of Oral Diseases & National Center for Stomatology & National Clinical Research Center for Oral Diseases, West China Hospital of Stomatology, Sichuan University, Chengdu, Sichuan 610041, China
bDepartment of Chemistry, Tufts University, Medford, MA 02155, USA
First published on 4th December 2025
Abstract
Lanthanide-doped fluorapatite (FAp:Ln3+) upconversion luminescence (UCL) probes exhibit good biocompatibility but suffer from low quantum yields under near-infrared (NIR) excitation. Here, we propose a universal MoO42− doping strategy that achieves 74-fold green UCL enhancement in FAp:Yb/Er/Mo (FYEM) under 980 nm excitation (quantum yield: 0.004% at 50 W cm−2). Remarkably, stable Ln3+/MoO42− dimers induce 4d–4f orbital hybridization, as validated by density functional theory (DFT) and X-ray photoelectron spectroscopy (XPS). This mechanism extends to self-sensitized systems: FAp:Er/Mo (FEM) achieves 156-fold UCL enhancement under 980 nm excitation, while FAp:Nd/Mo (FNM) integrates 84-fold UCL amplification with photothermal synergy (ΔT = 53.5 °C). Critically, the optimized FYEM material enables dual-mode imaging, with 808/980 nm excitation UCL and NIR-II downconversion luminescence for deep-tissue imaging, besides outstanding thermal sensitivity (SA = 1.04% K−1@323 K). This work establishes a biocompatible, multi-wavelength platform for deep-tissue diagnostics, photothermal therapy, and multimodal sensing and imaging.
1. Introduction
Upconversion luminescence (UCL), which converts near-infrared (NIR) photons to higher-energy visible emissions, offers advantages for biomedical imaging, including deep-tissue penetration and minimized photodamage.1–3 Among biocompatible matrices, fluorapatite (FAp) stands out due to its intrinsic bone affinity, biodegradability, and critically lower lattice phonon energy compared to hydroxyapatite (HAp).4–6 This arises from F− substitution for the OH− groups, reducing vibrational quenching (∼1000 cm−1 for F−vs. 2700–3700 cm−1 for OH−). Nevertheless, FAp:Ln3+ suffers from low fluorescence intensity and critically low upconversion quantum yields under NIR excitation, limiting clinical translation. Conventional enhancement strategies involving transition metal (TM) cation doping (e.g., Fe3+, Mn2+) often face critical limitations,7,8 because TM ions could induce parasitic visible-light absorption, causing spectral overlap with emission bands and fluorescence quenching.9 Recent studies suggest d0-centered oxyanions (e.g., MoO42−) may circumvent these limitations. Van et al. demonstrated UCL enhancement in MoO42−-doped HAp/β-TCP composites via Ln3+/MoO42− dimers.10 But the high phonon energy of these matrices restricts efficiency, and the atomic-scale enhancement mechanism remains unearthed. Critically, FAp's lower phonon energy provides untapped potential for greater enhancement, and MoO42− doping in Ln-doped FAp has not been systematically explored.Herein, we propose a novel MoO42− doping strategy to harness its phononic advantage. By substituting PO43− with MoO42−, FAp:Yb/Er/Mo (FYEM) achieves a 74-fold green UCL enhancement under 980 nm excitation (UCQY: 0.004%), and a 287-fold amplification under 808 nm excitation. These enhancements stem from 4d–4f orbital hybridization mediated by stable Ln3+/MoO42− dimers, as validated by X-ray photoelectron spectroscopy (XPS) and density functional theory (DFT). XPS analysis reveals MoO42−-driven electron redistribution, while DFT confirms that MoO42− preferentially occupies the PO43− adjacent to Yb3+, broadening the 4f density of states near the Fermi level. This strategy could extend to self-sensitized systems. FAp:Er/Mo (FEM) exhibits a 156-fold UCL enhancement under 980 nm excitation, while FAp:Nd/Mo (FNM) integrates an 84-fold UCL amplification with synergetic photothermal conversion capability (ΔT = 53.5 °C). Critically, FAp:Nd/Yb/Er/Mo (FNYEM) achieves simultaneous excitation at both 808 nm and 980 nm, delivering strong UCL while preserving spectral purity, thereby eliminating laser-induced overheating risks. FYEM also demonstrates dual-mode imaging capabilities, combining visible UCL (510–570 nm) and NIR-II downconversion luminescence (DCL) (1450–1650 nm) for deep-tissue imaging. Additionally, green UCL of FYEM exhibits outstanding thermal sensitivity under physiological temperature (283–323 K), with maximum absolute sensitivity (SA) = 1.04% K−1; maximum relative sensitivity (SR) = 1.28% K−1, outperforming existing similar sensors including NaYF4 and Ca2MgWO6.
This work addresses three critical challenges in UCL design: (1) reconciling luminescent efficiency and versatility through MoO42− co-doping; (2) elucidating the atomic-scale mechanism of 4d–4f hybridization via XPS/DFT validation; (3) establishing a universal enhancement paradigm for diverse Ln3+ systems (Er3+, Ho3+, Tm3+). By integrating multi-wavelength excitation (808/980 nm), photothermal synergy, and multimodal sensing, our strategy enables transformative advances in deep-tissue theranostics.
2. Results
2.1. Morphology and structural characterization
All samples were synthesized via a facile one-pot hydrothermal method followed by thermal activation at 900 °C (Fig. 1a). X-ray diffraction (XRD) patterns (Fig. 1b and S1) confirm the hexagonal fluorapatite structure (P63/m space group) for all samples, with characteristic peaks at 25.9° (002), 31.9° (211), 32.3° (112), 33.1° (300), 40° (310), 46.8° (222), and 49.6° (213). Co-doping with Ln3+ (Yb3+/Er3+, Yb3+/Ho3+, Yb3+/Tm3+) and MoO42− shifts diffraction peaks toward higher 2θ values (Fig. 1b and S1), indicating lattice contraction due to the smaller ionic radii of dopants versus Ca2+ and PO43−. Aliovalent substitution (MoO42− for PO43−) also induces impurity phases in samples (minor CaMoO4 and CaCO3, denoted by symbols in Fig. 1b and S2) in MoO42−-doped samples. To quantify the relative phase content and confirm fluorapatite remains the main phase, we conducted Rietveld refinement on the XRD data of FYEM as given in Fig. S2 and Table S3. The refinement indicates that the dominant phase is the intended fluorapatite with a weight percentage of ∼93.9%, suggesting co-doped fluorapatite remains the dominant phase. Fourier transform infrared (FTIR) spectra (Fig. 1c and S3, Table S4) reveal characteristic vibrational modes of PO43− group (606–609 and 950–1100 cm−1). Mo-doped samples exhibit additional peaks at 830–850 cm−1 attributed to ν3(MoO4), corroborating successful doping of MoO42−.11 The stretching and bending bands remarkably broaden in the range of 950–1100 cm−1 after doping MoO42−, indicating local lattice distortion and symmetry breaking, and possible formation of charge-compensation defects. A slight vibration mode of CO32− from CaCO3 secondary phase can be observed in the range of 1400–1500 cm−1, possibly caused by recombination of CO2 in air upon cooling after sintering. Thermogravimetric (TG-DSC) analysis was conducted to confirm thermal stability of samples under 900 °C (Fig. S4). XPS spectra (Fig. 1d and S5) confirm successful dopant incorporation. High-resolution XPS spectra reveal electron redistribution. Notably, compared with Mo-free FYX groups, Yb 4d binding energies shift negatively by 0.3–1.1 eV in Mo-doped FYXM groups (Fig. 1e–g), indicating increased electron density at Yb3+ sites.12 Conversely, Er 4d and Nd 3d/4d peaks in self-sensitized FEM/FNM samples shift positively (Fig. S6), suggesting electron migration toward Yb3+ but away from Er3+/Nd3+. This asymmetry arises from differences in electronegativity (Yb > Mo > Er/Nd)13 and Yb3+'s propensity to stabilize its 4f configuration,14 supporting MoO42−-mediated electron modulation.Scanning and transmission electron microscopy (SEM/TEM) images (Fig. 1h–j, S7 and S8) reveal similar rod-like morphology (1–3 μm in length) with uniform elemental distribution after thermal activation. High resolution TEM (HRTEM) (Fig. S8) shows ordered lattice arrangement on the edge of rod and small particles with the interplanar spacing of 0.271 and 0.184 nm, corresponding to the (300) and (213) crystal planes of fluorapatite respectively. Dynamic Light Scattering (DLS) analysis (Fig. S9) further confirms polydispersity: primary rods (870.3 nm) with smaller spherical particles (70.7 nm), likely from high-temperature induced crystal growth. Inductively coupled plasma mass spectrometry (ICP-MS) data (Table S5) quantifies dopant incorporation efficacy. Yb![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Ln and Mo
Ln and Mo![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) P ratios align with initial stoichiometry, while Ca
P ratios align with initial stoichiometry, while Ca![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) P ratios (1.47–1.53) deviate slightly from FAp's 1.67, similar to that of hydrothermally synthesized hydroxyapatite and fluorapatite elsewhere.15
P ratios (1.47–1.53) deviate slightly from FAp's 1.67, similar to that of hydrothermally synthesized hydroxyapatite and fluorapatite elsewhere.15
2.2. Universal UCL enhancement in Yb-sensitized systems
Lifetime analysis (Fig. 2g–i, Table S6) reveals prolonged green emission decay for both doped samples: FYEM (129 μs) versus FYE (52 μs), and FYHM (137 μs) versus FYH (118 μs), reflecting inhibited relaxation towards lower energy levels. Conversely, shortened lifetimes for other emissions suggest additional decay pathways via Ln3+/MoO42− dimers. Absolute upconversion quantum yield (UCQY) for FYEM reaches 0.004% at 50 W cm−2 (Fig. S12, Table 1), comparable to state-of-the-art materials (e.g., NaYF4:Yb3+/Er3+:0.005% at 150 W cm−2 (ref. 16)), while uniquely combining high UCL efficiency with inherent biocompatibility.
To obtain a better understanding of the interaction of energy levels of Yb and Mo, we conducted partial density of states (PDOS) analysis of Yb/Mo adjacently substituted case 1, Yb/Mo distantly substituted case 3 with most negative formation energy in each case, and Yb-undoped case as shown in Fig. 3a and b and S15. It is significant that the energy levels of Yb and Mo become overlapped only if Yb occupies the Ca1 site adjacent to Mo. Also, the peak of Yb 4f around Fermi level is broadened as deduced by full width at half maximum (FWHM). These findings confirm the presence of a strong 4d–4f orbital hybridization which causes modification of the electronic structure.22,23 The redistribution of electrons between Yb and Mo is consistent with the XPS result where electrons flow from Mo towards Yb (Fig. 1e–g).
Fig. 3c–e and S16 illustrate the reflectance of FYXM and other samples. They exhibit characteristic absorption credited to transitions of doped lanthanide ions. Interestingly, the characteristic absorbance of lanthanide ions was generally elevated by one time in FYXM samples, which are probably caused by absorption of newly formed energy levels in Yb3+/MoO42− dimer. Based on the DFT study and previous analyses, we propose that Ln3+ has a tendency to interact with MoO42− and having their energy levels recombined.
It is worth mentioning that MoO42− owns intrinsic photoluminescence. Specifically, CaMoO4 itself was found to emit green emission peaking at 435 nm under 280 nm excitation, prevailingly attributed to the 1T2–1A1 allowed transition.24 We assume that 1T1, 3T2, 3T1 of MoO42− could match high energy levels of activator ions and interact with them in energy transfer. Hence, an energy transfer mechanism is proposed and illustrated in Fig. 3f, in which, the energy levels of Yb3+ and MoO42− are recombined to form new energy levels: |2F7/2, 1A1〉 (ground state), |2F5/2, 1A1〉, |2F7/2, 3T1〉, |2F7/2, 3T2〉, |2F7/2, 1T1〉 and |2F7/2, 1T2〉 (excited state). Specifically, photons are absorbed by ground state absorption (GSA) and excitation state absorption (ESA). Then the energy is transferred from excited state of Yb3+/MoO42− dimer towards activator ions, eventually resulting in UCL emissions. The mechanism could also apply to other lanthanide ions, namely Er3+ and Nd3+ which could also interact in a similar manner.
Interestingly, Yb3+/Er3+ co-doped CaMoO4 exhibited UCL with a similarly high G/R ratio as we observed in our experiment.25 Here we suggest that the Ln3+/MoO42− dimer could account for the observed UCL preference. In the case of FYEM, the red emission relies on relaxation from 2H11/2/4S3/2 of Er3+, and the larger level gap means less opportunity to get populated, resulting in relatively weak enhancement of red UCL. This preference further confirms the energy transfer mechanism in Fig. 3f, since 2H11/2/4S3/2 of Er3+ is closer to |2F7/2, 3T2〉 than 4F9/2. As the concentration further increases beyond optimal concentration, back energy transfer (BET) process probably prevails over HESET with overall UCL slumping. This could explain why the UCL performance is not in accordance with the content of CaMoO4 phase as more MoO42− was doped. In addition, the energy transfer mechanism of Ho/Mo is similar to the Er/Mo combination, both resulting in over 70 times enhancement of UCL. However, a phonon-assisted absorption process is required for the blue emission of Tm3+, which could account for the mere 8-time enhancement of blue emission in Tm/Mo combination.
In order to further examine energy transfer process between MoO42− and other lanthanides, the excitation spectra of all samples were performed. As shown in Fig. S17, all FYXM samples exhibit obvious absorption peaks which can be attributed to the transitions of activator ions. Significantly, MoO42−-doped samples exhibit a broad absorption band from 250 nm to 320 nm, which corresponds to the O → Mo charge transfer band (CTB). Then we performed emission spectra of FYE and FYXM samples excited at 280 nm (Fig. S18). Since Er3+ could not be directly excited by 280 nm excitation, FYE sample did not display any luminescence. On the contrary, the MoO42− doped FYEM samples exhibit emission composed of two parts in the region of visible emission: a wide band derived from the intrinsic emission of MoO42 and sharp green emissions attributed to Er3+. The lifetime at 505 nm is shorter than that at 530 nm or 550 nm, where emission of molybdate overlaps that of Er3+ (Fig. S19). The energy transfer process occurs from MoO42− towards Er3+ and both contribute to the decay, thereby lengthening lifetime at about 20%. And thus, we could draw a conclusion that the excited MoO42− group could transfer energy to Er3+ ions. Moreover, the green emission of Er3+ is prominent than its red emission in the range of 640–675 nm. This indicates the high G/R UCL ratio of FYEM and preference observed in other samples stem from the energy transfer process mediated by Ln3+/MoO42− dimer.
2.3. Breakthrough in self-sensitized systems
Self-sensitized upconversion systems are commonly considered limited by their relatively low efficiency. Herein, we discovered that MoO42− can also act as an intrinsic sensitizer in self-sensitized systems (FEM and FNM), eliminating the need for conventional sensitizers (e.g., Yb3+/Nd3+) through direct energy transfer mediated by Ln3+/MoO42− dimers—a mechanism analogous to Yb-sensitized systems but with enhanced versatility. Structural properties of FEM/FNM are consistent with FYXM (section 2.1), while their UCL enhancement stems from Ln3+/MoO42− dimer-mediated energy transfer (Fig. 4).Er3+ has been found to mediate self-sensitized upconversion (SSU) process under 808 and 980 nm excitation.26 Doping MoO42− brings about UCL enhancement by 23 times and 156 times under 808 nm and 980 nm excitation, respectively (Fig. 4a and b). The mechanism was further assured by luminescence lifetime analysis (Fig. 4c and S20, Tables S7 and S8). The decay time of FEM for emission at 530 nm of Er3+ was elongated by 43 times compared with FE suppose that cross relaxation process was less prone to happen in FEM sample, indicative of Er3+/MoO42− acting as sensitizer with longer lifetime. Another fact is that under either 808 nm or 980 nm excitation, UCL of FEM shares a similarly high G/R ratio with FYEM and FNYEM. It should be similar to FYEM, where electrons of Er3+/MoO42− are more likely to get pumped into 2H11/2 and 4S3/2via Er3+/MoO42− dimer. The power-dependent log–log plot indicates a two-phonon process as given in Fig. S21. Similarly, the energy transfer in FEM is proved by emission spectra under 280 nm excitation in Fig. S22 and S23.
Nd3+ is known to own a ladder-like complex 4f energy levels, which favors cross relaxation.27,28 Although Nd3+ is not an excellent SSU candidate, we successfully observed its self-sensitized UCL in Nd3+/MoO42− co-doped fluorapatite excited by 808 nm with an 84-fold enhancement in yellow emission (2P1/2–4I15/2) for the first time (Fig. 4d). A preference for yellow UCL was observed as well, as depicted in Fig. S24 by Y/G ratio. Nd3+ was found to exhibit photothermal effect under 808 nm excitation through non-radiative relaxation (e.g. from 4F5/2 to 4F3/2) and cross relaxation.29 Interestingly, after doping MoO42−, photothermal effect was enhanced as well (Fig. 4e). The decay spectra were examined and it was found that MoO42− doping leads to a shortened lifetime (Fig. 4f). These findings could be credited to Nd3+/MoO42− dimer, where more energy levels are created, thus more chances for electrons to be pumped and relaxed. The n value is slightly elevated, which is in accordance with that of FYXM and FEM samples (Fig. S25).
2.4. Multi-functional bioapplications enabled by dimer strategy
| Materials | Excitation wavelength (nm) | S A@323 K (% K−1) | Ref. |
|---|---|---|---|
| FYEM | 980 | 1.04 | This work |
| NaYF4:Yb3+/Er3+/Nd3+ | 980 | 0.23 | 33 |
| K3ZrF7:Yb3+/Er3+ | 980 | 0.23 | 34 |
| NaGdF4:Yb3+/Er3+/Fe3+ | 980 | 0.27 | 35 |
| La9.67Si6O26.5:Yb3+/Er3+/Ho3+ | 980 | 0.31 | 36 |
| Gd2Te6O15:Yb3+/Er3+ | 980 | 0.35 | 37 |
| NaYTiO4:Yb3+/Er3+/Sc3+ | 980 | 0.36 | 38 |
| Y2O3:Yb3+/Er3+ | 980 | 0.47 | 39 |
| Li2HfO3:Yb3+/Er3+ | 980 | 0.58 | 40 |
| YVO4:Yb3+/Er3+ | 980 | 0.58 | 41 |
| NaY(WO4)2:Yb3+/Er3+ | 980 | 0.66 | 42 |
| Ca2MgWO6:Yb3+/Er3+ | 980 | 0.76 | 43 |
| HAp/β-TCP:Yb/Er/Mo/Sr | 980 | 0.88 | 44 |
Then dual-mode imaging utilized 980 nm excitation for both visible UCL and NIR-II DCL, ensuring consistent high-intensity signals across modalities. The in vitro bioimaging of FYEM (Fig. 5g) shows that the porcine skin with a thickness of 1.5 mm is lit up by green UCL of FYEM under 980 nm excitation, suggesting a sufficient UCL intensity for intracutaneous bioimaging. Considering NIR-II DCL is more favorable in deep-tissue bioimaging than visible UCL, the GelMA/FYEM hydrogel is synthesized and placed subcutaneously (section 4.8). The NIR-II DCL of FYEM excited at 980 nm is successfully observed by NIR-II imaging system (Fig. 5i), indicative of remarkable deep-tissue imaging capacity. Conclusively, FYEM proves to be a promising visible/NIR-II dual-mode bioimaging material.
3. Conclusions
In this work, we have developed a universal MoO42−-doping strategy to boost UCL in lanthanide-doped fluorapatite via 4d–4f orbital hybridization. The formation of stable Ln3+/MoO42− dimer significantly enhances energy transfer efficiency, achieving remarkable UCL amplifications (UCQY = 0.004%). Crucially, this mechanism is demonstrated for the first time in self-sensitized systems (Er3+/Nd3+), overcoming traditional reliance on Yb3+ as sensitizer. The optimized material integrates multifunctionality—including dual-wavelength excitation (808/980 nm), NIR-II deep-tissue imaging (DCQY = 4.87%), and record-high temperature sensitivity (SA = 1.04% K−1@323 K). This work establishes a versatile platform for next-generation deep-tissue diagnostics, photothermal therapy, and real-time physiological monitoring.4. Methods
4.1. Materials
Ca(NO3)2·4H2O, NaOH and ethanol were obtained from Kelong Chemical Reagents Company, China. Yb(NO3)3·5H2O, Tm(NO3)3·6H2O, Ho(NO3)3·5H2O, Er(NO3)3·6H2O, Nd(NO3)3·5H2O and (NH4)6Mo7O24·4H2O were obtained from Macklin Co. Ltd, Shanghai, China. NaF and (NH4)2HPO4 were obtained from Aladdin Co. Ltd, Shanghai, China. All the chemical reagents were of analytical grade and used without further purification.4.2. Synthesis of FYX samples
The FAp:Yb/X (X = Er, Ho, or Tm) co-doped upconversion fluorapatites (FYX) were hydrothermally synthesized based on a previous method with some modification. Taking FYE as an example, an aqueous solution of (NH4)2HPO4 and NaF was added drop by drop into the mixture solution of Ca(NO3)2, Yb(NO3)3, Er(NO3)3 under vigorous stirring. The molar concentrations of Yb3+ and Er3+ ions relative to Ca2+ ions were set at 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.5
0.5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 89.5 (Table S1), and the ratio of [(Ca + Ln)/P] was kept constant at 1.67. The 1 M NaOH stock solution was added drop by drop to maintain the pH at 9–10. The solution was further stirred for 30 min to obtain a white slurry. The slurry was then transferred into a 100 mL Teflon-lined autoclave and hydrothermally treated at 180 °C for 6 h. The obtained precipitates were centrifuged (5 min at 2700g) and fully washed with deionized water and ethanol for three times. Finally, samples were fully lyophilized, ground into white powder and sintered at 900 °C for 2 h to activate the upconversion luminescence. Other Mo-free FYX samples were synthesized in a similar pattern.
89.5 (Table S1), and the ratio of [(Ca + Ln)/P] was kept constant at 1.67. The 1 M NaOH stock solution was added drop by drop to maintain the pH at 9–10. The solution was further stirred for 30 min to obtain a white slurry. The slurry was then transferred into a 100 mL Teflon-lined autoclave and hydrothermally treated at 180 °C for 6 h. The obtained precipitates were centrifuged (5 min at 2700g) and fully washed with deionized water and ethanol for three times. Finally, samples were fully lyophilized, ground into white powder and sintered at 900 °C for 2 h to activate the upconversion luminescence. Other Mo-free FYX samples were synthesized in a similar pattern.
4.3. Synthesis of Mo-doped FYXM samples
Yb/X/Mo (X = Er, Ho, or Tm) tri-doped fluorapatites were synthesized with MoO42− partially substituting PO43−, lanthanide partially substituting Ca2+ in a similar approach according to our previous research. The molar ratio of Yb3+ and Tm3+/Ho3+/Er3+ was set to be 20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.1, 10
0.1, 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.1, and 10
0.1, and 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.5, respectively,45 keeping a constant [(Ca + Ln)/(P + Mo)] molar ratio of 1.67 (Table S1). Samples were then collected and sintered as mentioned above. For the sake of brevity, FYTM8, FYHM10 and FYEM15 samples with optimal UCL properties were designated FYTM, FYHM and FYEM in the following sections if not specified.
0.5, respectively,45 keeping a constant [(Ca + Ln)/(P + Mo)] molar ratio of 1.67 (Table S1). Samples were then collected and sintered as mentioned above. For the sake of brevity, FYTM8, FYHM10 and FYEM15 samples with optimal UCL properties were designated FYTM, FYHM and FYEM in the following sections if not specified.
The self-sensitized Er3+/MoO42−, Nd3+/MoO42−, or Nd3+/Yb3+/Er3+/MoO42− co-doped fluorapatite materials were synthesized by similar means and were designated FNYEM, FEM, FNM respectively (Table S2).
4.4. Characterization
The phase composition of all samples was tested by X-ray diffraction (XRD) PANalytical Empyrean equipment (U.K.) in the 2θ range from 20° to 70° with Cu Kα radiation (λ = 1.5406 Å). TG-DSC curves were obtained in air by using Netzsch STA449 F3 Jupiter (Germany). The morphology of particles was observed by scanning electron microscopy (SEM) of Thermo Fisher Scientific Apreo 2C (U.S.) and transmission electron microscopy (TEM) using JEM-F200, JEOL (Japan). The ratio of ions was tested by inductively coupled plasma-mass spectrometry (ICP-MS) of Agilent 7800 (U.S.). The Fourier transform infrared (FTIR) spectra were conducted from Thermo Fisher Nicolet (U.S.) with a wavenumber range of 400–3000 cm−1. UV-Vis-NIR diffuse reflectance spectra (UV-Vis-NIR DRS) were conducted using Shimadzu UV 3600 (Japan). The dynamic light scattering (DLS) was tested using Litesizer 500, Anton Paar (Australia), by dispersing samples in ethanol. The X-ray photoelectron spectroscopy (XPS) was recorded by Thermo Fisher Scientific K-Alpha (U.S.). The photoluminescence (PL) excitation/emission spectra were performed by using Horiba JobinYvon Fluorologs-3 spectrofluorometer (Japan) with an external 808/980 diode laser (MDL-III-808/MDL-H-980) for upconversion, or Xenon lamp for downconversion as the excitation source with appropriate optical filter. All UCPL spectra were obtained with an excitation power ranging from 0.3–1.5 W based on relative intensity of samples and an integration time no more than 0.5 s, with a 700 nm short-pass optical filter. Diode lasers and optical filters were obtained from Changchun New Industries Optoelectronics Technology Co., Ltd, China. Fluorescence lifetime was measured with appropriate excitation source and a picosecond photon detection module (PPD-850) as the detector. Upconversion quantum yield (UCQY) was studied using ATP65, OPTOSKY (China), excited by 980 nm diode laser at ∼50 W cm−2. Downconversion quantum yield (DCQY) was tested by Fluorolog-QM, Horiba (Japan). All tests were conducted under room temperature without further clarification. The photothermal performance was recorded with a PTi120 thermal camera, Fluke (U.S.).4.5. Upconversion luminescence lifetime study
The time dependence of the UCL intensity in the decay phase could be described as | (1) |
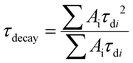 | (2) |
In order to assess the rise phase, the rise time τrise is defined as the time needed to reach up to (1 − e−1)Imax, where Imax is set 1000, 2000 or 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 according to the UCL intensity of each sample.46
000 according to the UCL intensity of each sample.46
4.6. Temperature-dependent upconversion luminescence study
According to the Boltzmann distribution law, the FIR can be expressed as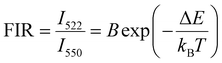 | (3) |
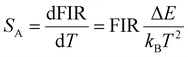 | (4) |
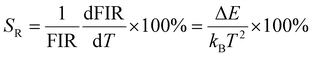 | (5) |
4.7. Cytocompatibility assay
To evaluate the cytocompatibility of lanthanide/molybdate co-doped samples, rat osteoblasts were cocultured with FYEM nanoparticles in various concentrations (0, 50, 100, and 200 μg mL−1). The cells were cultured in minimum essential medium–alpha modification (α-MEM, GIBCO, USA) supplemented with 10% newborn bovine calf serum (NBCS, GIBCO, USA) and 1% penicillin–streptomycin solution (100 U mL−1, GIBCO). The cells were incubated at 37 °C under conditions of 5% carbon dioxide and 90% humidity. Cell proliferation was then assessed by using a CCK-8 kit (APExBIO, USA), on days 1, 3, and 7.4.8. In vitro and in vivo imaging
In vitro imaging in the region of visible light was conducted by placing FYEM sample under a piece of porcine skin with a thickness of 1.5 mm. A cellphone (Xiaomi 11 Pro, China) was used with an 850 nm short-pass optical filter to record the emitted green upconversion luminescence of FYEM under 980 nm excitation. The NIR-II DC luminescence was imaged using a Series III 800 NIR-II imaging system (Suzhou NIR-Optics Technologies Co., Ltd, China), with a 1300 nm long-pass optical filter. FAp, FYE, and FYEM samples were placed in centrifuge tubes and their NIR-II luminescence was observed under 980 nm excitation with pumping power at 1 W and acquisition time at 20 ms.In order to immobilize FYEM powder during in vivo imaging experiment, the sample is mixed with GelMA hydrogel. The synthesis of GelMA hydrogel is given elsewhere.47 The GelMA/FYEM hydrogel was cured under UV lamp in a circular mold and then implanted subcutaneously in an 8-week Sprague-Dawley (SD) rat to demonstrate the ability of NIR-II bioimaging capacity in subcutaneous tissue with an average tissue thickness at about 3 mm. All animal experiments were approved by the Research Ethics Committee of West China Hospital of Stomatology (No. WCHSIRB-D-2024-744). All procedures were carried out following the ARRIVE guidelines and related principles of the care and use of laboratory animals. The NIR-II fluorescence was collected by the same NIR-II imaging system with pumping power at 3 W and acquisition time at 200 ms.
4.9. Theoretical calculation
All of the calculations are performed in the framework of the spin-polarized density functional theory with the projector augmented plane-wave method, as implemented in the Vienna ab initio simulation package (VASP).48,49 The generalized gradient approximation (GGA) proposed by Perdew, Burke, and Ernzerhof (PBE) is selected for the exchange–correlation potential.50,51 The long-range van der Waals interaction is described by the DFT-D3 approach.52 DFT+U method with the effective U value of 3.5 eV and 6 eV has been used to correct the influence of 4d electrons of Mo and Yb atoms. The cut-off energy for plane wave is set to 480 eV. The energy criterion is set to 10−5 eV in iterative solution of the Kohn–Sham equation. All the structures are relaxed until the residual forces on the atoms have declined to less than 0.02 eV Å−1. Data analysis and visualization are carried out with the help of VASPKIT53 code and VESTA.54 To avoid interlaminar interactions, a vacuum spacing of 20 Å is applied perpendicular to the slab.The formation energy Eform is expressed as
| Eform = Edefect − Eperfect − EYb + ECa | (6) |
4.10. Statistical analysis
Quantitative data were presented as mean ± standard deviation (SD) value. Data from different groups were statistically compared via one-way analysis of variance (ANOVA). The level of significance was set as *p < 0.05, **p < 0.01, ***p < 0.001, ****p < 0.0001.Author contributions
X. L. conceived and directed the research; B. W. prepared the fluorapatite nanocrystals with assistance from Y. R., W. Y., Y. L. and J. L.; B. W. collected and analyzed most of the experimental data, with contributions from X. W. and J. L.; B. W. and H. S. discussed and interpreted the DFT calculation data; all authors contributed to the data analysis; the manuscript was written by B. W. and X. L.Conflicts of interest
The authors declare that there are no competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.Data availability
The data supporting this article have been included as part of the supplementary information (SI), including supporting notes, additional experimental and calculated results, and summaries of dopant molar concentration in synthesis and luminescence rise and decay lifetimes. Supplementary information is available. See DOI: https://doi.org/10.1039/d5qi01918d.Acknowledgements
This work was supported by the National Key Research and Development Program of China (Grants No. 2021YFA1201302), the Fund from Science and Technology Department of Sichuan Province, China (Grant No. 2023NSFSC1930), and Tianfu Qingcheng Plan-Youth Science and Technology Talent Project, Sichuan, China (Grant No. 1710). We would like to express our genuine gratitude to Dr Peng Wu and Dr Yanying Wang of Analytical & Testing Center, Sichuan University, for their assistance with fluorescence testing and analysis. We acknowledge the use of BioRender for creating Fig. 1a, 5g and h (https://BioRender.com/ob4f13u).References
- S. Lamon, H. Yu, Q. Zhang and M. Gu, Lanthanide ion-doped upconversion nanoparticles for low-energy super-resolution applications, Light: Sci. Appl., 2024, 13(1), 252 CrossRef CAS PubMed.
- R. S. Ajee, P. S. Roy, S. Dey and S. Sundaresan, Upconversion nanoparticles and their potential in the realm of biomedical sciences and theranostics, J. Nanopart. Res., 2024, 26(3), 50 CrossRef CAS.
- Z. Yi, Z. Luo, X. Qin, Q. Chen and X. Liu, Lanthanide-activated nanoparticles: a toolbox for bioimaging, therapeutics, and neuromodulation, Acc. Chem. Res., 2020, 53(11), 2692–2704 CrossRef CAS PubMed.
- X. Li, Q. Zou, L. Chen and W. Li, A ternary doped single matrix material with dual functions of bone repair and multimodal tracking for applications in orthopedics and dentistry, J. Mater. Chem. B, 2018, 6(38), 6047–6056 RSC.
- X. Li and H. Chen, Yb3+/Ho3+ co-doped apatite upconversion nanoparticles to distinguish implanted material from bone tissue, ACS Appl. Mater. Interfaces, 2016, 8(41), 27458–27464 CrossRef CAS PubMed.
- B. Nasiri-Tabrizi, W. J. Basirun, C. H. Yeong and W. M. Thein, Development of the third generation of bioceramics: Doping hydroxyapatite with s-, p-, d-, and f-blocks cations and their potential applications in bone regeneration and void filling, Ceram. Int., 2023, 49(5), 7142–7179 CrossRef CAS.
- S. Mohanty and A. M. Kaczmarek, Unravelling the benefits of transition-metal-co-doping in lanthanide upconversion nanoparticles, Chem. Soc. Rev., 2022, 51(16), 6893–6908 RSC.
- M. Matakgane, T. Mokoena and M. Mhlongo, Recent trends of oxides heterostructures based upconversion phosphors for improving power efficiencies of solar cells: A review, Inorg. Chem. Commun., 2023, 156, 111202 CrossRef CAS.
- X. Li, Q. Zou, Y. Man and W. Li, Synergistic effects of novel superparamagnetic/upconversion HA material and Ti/magnet implant on biological performance and long–term in vivo tracking, Small, 2019, 15(31), 1901617 CrossRef.
- H. N. Van, N. H. Vu and V.-H. Pham, et al., A novel upconversion emission material based on Er3+-Yb3+-Mo6+ tridoped Hydroxyapatite/Tricalcium phosphate (HA/β-TCP), J. Alloys Compd., 2020, 827, 154288 CrossRef CAS.
- M. A. Goldberg, N. O. Donskaya and D. V. Valeev, et al., Mesoporous molybdate-substituted hydroxyapatite nanopowders obtained via a hydrothermal route, Ceram. Int., 2024, 50(10), 17404–17418 CrossRef CAS.
- F. Li, P. Wang and T. Zhang, et al., Efficient Removal of Antibiotic Resistance Genes through 4f–2p–3d Gradient Orbital Coupling Mediated Fenton–Like Redox Processes, Angew. Chem., Int. Ed., 2023, 62(47), e202313298 CrossRef CAS.
- M. Rahm, T. Zeng and R. Hoffmann, Electronegativity seen as the ground-state average valence electron binding energy, J. Am. Chem. Soc., 2019, 141(1), 342–351 CrossRef CAS PubMed.
- G. H. Debnath, P. Mukherjee and D. H. Waldeck, Identifying Lanthanide Energy Levels in Semiconductor Nanoparticles Enables Tailored Multicolor Emission through Rational Dopant Combinations, Acc. Chem. Res., 2025, 58(9), 1473–1483 CrossRef CAS PubMed.
- A. Wang, H. Yin and D. Liu, et al., Size-controlled synthesis of hydroxyapatite nanorods in the presence of organic modifiers, Mater. Lett., 2007, 61(10), 2084–2088 CrossRef CAS.
- J.-C. Boyer and F. C. Van Veggel, Absolute quantum yield measurements of colloidal NaYF4: Er3+, Yb3+ upconverting nanoparticles, Nanoscale, 2010, 2(8), 1417–1419 RSC.
- S. Balabhadra, M. L. Debasu, C. D. Brites, R. A. Ferreira and L. D. Carlos, Upconverting nanoparticles working as primary thermometers in different media, J. Phys. Chem. C, 2017, 121(25), 13962–13968 CrossRef CAS.
- F. H. Borges, J. C. Martins, F. J. Carlos, R. A. Ferreira and R. R. Gonçalves, Luminescent thermometry based on Er3+/Yb3+ co-doped yttrium niobate with high NIR emission and NIR-to-visible upconversion quantum yields, J. Lumin., 2022, 248, 118986 CrossRef CAS.
- F. You, S. Zheng and T. Pang, et al., Transparent Ho3+/Tm3+/Er3+/Yb3+ Codoped Nano–Glass–Ceramics for Upconversion Head–Up Display, Laser Photonics Rev., 2024, 18(12), 2400838 CrossRef CAS.
- M. L. Debasu, J. C. Riedl, J. Rocha and L. D. Carlos, et al., The role of Li+ in the upconversion emission enhancement of (YYbEr)2O3 nanoparticles, Nanoscale, 2018, 10(33), 15799–15808 RSC.
- J. Yang, P. Lin, E. Y. B. Pun, J. Yuan, X. Zhao and H. Lin, et al., Quantification of upconversion photon and thermosensitive feedback in Er3+/Yb3+ doped fluorotellurite glasses, J. Lumin., 2020, 222, 117184 CrossRef CAS.
- J. Wen, S. Hui and Q. Chang, et al., Enhancement of Electromagnetic Wave Attenuation through Polarization Loss Induced by Hybridization of Rare–Earth 4f and Mo–4d Orbitals in Liquid Plasma, Adv. Funct. Mater., 2024, 34(51), 2410447 CrossRef CAS.
- X. Wang, L. Shi and W. Ren, et al., Enhancing* CO coverage on Sm-Cu2O via 4f-3d orbital hybridization for highly efficient electrochemical CO2 reduction to C2H4, J. Energy Chem., 2024, 99, 409–416 CrossRef CAS.
- A. Phuruangrat, T. Thongtem and S. Thongtem, Preparation, characterization and photoluminescence of nanocrystalline calcium molybdate, J. Alloys Compd., 2009, 481(1–2), 568–572 CrossRef CAS.
- S. Gouraha, S. Sinha, A. Srivastava and J. Singh, Microwave assisted preparation of ternary scheelite CaMoO4: Er3+/Yb3+ nano-phosphors for up/down-conversion photoluminescence, temperature sensing and antibacterial properties, J. Photochem. Photobiol., A, 2025, 458, 115967 CrossRef CAS.
- L. Yan, B. Zhou and N. Song, et al., Self-sensitization induced upconversion of Er3+ in core–shell nanoparticles, Nanoscale, 2018, 10, 17949–17957 RSC.
- Z. Yu, W. K. Chan and T. T. Y. Tan, Neodymium–sensitized nanoconstructs for near–infrared enabled photomedicine, Small, 2020, 16(1), 1905265 CrossRef CAS.
- Z. Yu, W. Hu and H. Zhao, et al., Generating New Cross–Relaxation Pathways by Coating Prussian Blue on NaNdF4 To Fabricate Enhanced Photothermal Agents, Angew. Chem., Int. Ed., 2019, 58(25), 8536–8540 CrossRef CAS PubMed.
- L. Ma, Y. Zhou and Z. Zhang, et al., Multifunctional bioactive Nd-Ca-Si glasses for fluorescence thermometry, photothermal therapy, and burn tissue repair, Sci. Adv., 2020, 6(32), eabb1311 CrossRef CAS PubMed.
- S. Liu, Z. An and B. Zhou, Optical multiplexing of upconversion in nanoparticles towards emerging applications, Chem. Eng. J., 2023, 452, 139649 CrossRef CAS.
- P. Du, R. An, Y. Liang, P. Lei and H. Zhang, Emerging NIR-II luminescent bioprobes based on lanthanide-doped nanoparticles: from design towards diverse bioapplications, Coord. Chem. Rev., 2022, 471, 214745 CrossRef CAS.
- H. Liang, K. Yang and Y. Yang, et al., A lanthanide upconversion nanothermometer for precise temperature mapping on immune cell membrane, Nano Lett., 2022, 22(22), 9045–9053 CrossRef CAS PubMed.
- B. Cao, Y. Bao and Y. Liu, et al., Wide-range and highly-sensitive optical thermometers based on the temperature-dependent energy transfer from Er to Nd in Er/Yb/Nd codoped NaYF4 upconversion nanocrystals, Chem. Eng. J., 2020, 385, 123906 CrossRef CAS.
- H. Fu, W. Xie and Y. Qian, et al., Ultrafast Room–Temperature Synthesis of Yb3+/Er3+ Codoped K3ZrF7 Nanocrystals for Thermal Enhancement of Upconversion, Laser Photonics Rev., 2025, 19(5), 2401522 CrossRef CAS.
- J. Tang, P. Du, W. Li and L. Luo, Boosted thermometric performance in NaGdF4: Er3+/Yb3+ upconverting nanorods by Fe3+ ions doping for contactless nanothermometer based on thermally and non-thermally coupled levels, J. Lumin., 2020, 224, 117296 CrossRef CAS.
- J. Zhang, J. Chen and Y. Zhang, Temperature-sensing luminescent materials La9.67Si6O26.5: Yb3+–Er3+/Ho3+ based on pump-power-dependent upconversion luminescence, Inorg. Chem. Front., 2020, 7(24), 4892–4901 RSC.
- H. Liang, X. Lin and N. Ma, et al., Up–conversion luminescence of Er3+/Yb3+ co–doped Gd2Te6O15 tellurite glass–ceramics for optical thermometry, J. Am. Ceram. Soc., 2024, 107(12), 8191–8205 CrossRef CAS.
- Y. Wang, C. Zuo and C. Ma, et al., Effects of Sc3+ ions on local crystal structure and up-conversion luminescence of layered perovskite NaYTiO4: Yb3+/Er3+, J. Alloys Compd., 2021, 876, 160166 CrossRef CAS.
- R. Ao, L. Xing and W. Yang, A high-brightness phosphor based on Yb3+/Er3+ codoped Y2O3 micro-crystals and controllable temperature sensing sensitivity via rare earth ions, Opt. Commun., 2021, 492, 126967 CrossRef CAS.
- R. An, Y. Liang, R. Deng, P. Lei and H. Zhang, Hollow nanoparticles synthesized via Ostwald ripening and their upconversion luminescence-mediated Boltzmann thermometry over a wide temperature range, Light: Sci. Appl., 2022, 11(1), 217 CrossRef CAS PubMed.
- M. Jia, Z. Sun, M. Zhang, H. Xu and Z. Fu, What determines the performance of lanthanide-based ratiometric nanothermometers?, Nanoscale, 2020, 12(40), 20776–20785 RSC.
- M. Lin, L. Xie, Z. Wang, B. S. Richards, G. Gao and J. Zhong, Facile synthesis of mono-disperse sub-20 nm NaY (WO4)2: Er3+, Yb3+ upconversion nanoparticles: a new choice for nanothermometry, J. Mater. Chem. C, 2019, 7(10), 2971–2977 RSC.
- Y. Jiang, Y. Tong and S. Chen, et al., A three-mode self-referenced optical thermometry based on up-conversion luminescence of Ca2MgWO6: Er3+, Yb3+ phosphors, Chem. Eng. J., 2021, 413, 127470 CrossRef CAS.
- H. N. Van, D.-H. Nguyen and V.-H. Pham, Effect of Sr substituted on multifunction pure green emission of rare-earth-element doped HA/β-TCP nanocomposite for optical thermometer, Ceram. Int., 2024, 51(12), 16894–16903 CrossRef.
- J. Tang, P. Du, W. Li and L. Luo, Boosted thermometric performance in NaGdF4: Er3+/Yb3+ upconverting nanorods by Fe3+ ions doping for contactless nanothermometer based on thermally and non-thermally coupled levels, J. Lumin., 2020, 224, 117296 CrossRef CAS.
- S. J. W. Vonk, J. J. E. Maris and A. J. H. Dekker, et al., Rise and Decay of Photoluminescence in Upconverting Lanthanide-Doped Nanocrystals, ACS Nano, 2024, 18(41), 28325–28334 CrossRef CAS PubMed.
- Q. Wei, J. Su and S. Meng, et al., MiR-17-5p-engineered sEVs Encapsulated in GelMA Hydrogel Facilitated Diabetic Wound Healing by Targeting PTEN and p21, Adv. Sci., 2024, 11, 2307761 CrossRef CAS.
- G. Kresse and J. Hafner, Ab initio molecular dynamics for open-shell transition metals, Phys. Rev. B: Condens. Matter Mater. Phys., 1993, 48(17), 13115–13118 CrossRef CAS PubMed.
- G. F. L. Kresse and J. Furthmüller, Efficient iterative schemes for ab initio total-energy calculations using a plane-wave basis set, Phys. Rev. B: Condens. Matter Mater. Phys., 1996, 54(11), 169–185 Search PubMed.
- J. P. Perdew, K. Burke and M. Ernzerhof, Generalized gradient approximation made simple, Phys. Rev. Lett., 1996, 77(18), 3865–3868 CrossRef CAS PubMed.
- W. Kohn and L. J. Sham, Self-Consistent Equations Including Exchange and Correlation Effects, Phys. Rev., 1965, 140(4A), A1133–A1138 CrossRef.
- S. Grimme, J. Antony, S. Ehrlich and H. Krieg, A consistent and accurate ab initio parametrization of density functional dispersion correction (DFT-D) for the 94 elements H-Pu, J. Chem. Phys., 2010, 132(15), 154104 CrossRef PubMed.
- V. Wang, N. Xu, J.-C. Liu, G. Tang and W.-T. Geng, VASPKIT: A user-friendly interface facilitating high-throughput computing and analysis using VASP code, Comput. Phys. Commun., 2021, 267, 108033 CrossRef CAS.
- K. Momma and F. Izumi, VESTA: a three-dimensional visualization system for electronic and structural analysis, J. Appl. Crystallogr., 2008, 41(3), 653–658 CrossRef CAS.
| This journal is © the Partner Organisations 2026 |





