Excitation energy funnelling in NIR-emissive [YbPrYb] heterometallic complexes
Salauat
Kiraev
a,
Diamantoula
Maniaki
bc,
Annika
Sickinger
a,
Leoní A.
Barrios
 bc,
David
Aguilà
bc,
David
Aguilà
 bc,
Olivier
Roubeau
bc,
Olivier
Roubeau
 d,
Yannick
Guyot
e,
Olivier
Maury
d,
Yannick
Guyot
e,
Olivier
Maury
 a,
Laura
Abad Galán
a,
Laura
Abad Galán
 *f and
Guillem
Aromí
*f and
Guillem
Aromí
 *bc
*bc
aUniv. Lyon, Institut Lumière Matière, UMR 5306 CNRS–Université Claude Bernard, Lyon 1, 10 rue Ada Byron, F-69622 Villeurbanne Cedex, France
bDepartament de Química Inorgànica i Orgànica, Universitat de Barcelona, Diagonal 645, 08028 Barcelona, Spain. E-mail: aromi@ub.edu
cInstitute of Nanoscience and Nanotechnology of the University of Barcelona (IN2UB), Barcelona, Spain
dInstituto de Nanociencia y Materiales de Aragón (INMA), CSIC and Universidad de Zaragoza, Plaza San Francisco s/n, 50009 Zaragoza, Spain
eUniv Lyon, ENS Lyon, CNRS, UMR 5182, Laboratoire de Chimie, F69342 Lyon, France
fDepartamento de Química Inorgánica, Universidad Complutense de Madrid, Avda. Complutense s/n, 28040 Madrid, Spain. E-mail: laabad03@ucm.es
First published on 2nd October 2025
Abstract
Energy transfer (ET) among different lanthanide ions is a promising avenue to enhance the numerous applications of this photophysical phenomenon, as has been demonstrated in various solid-state materials. It is far less common to observe these phenomena in discrete species containing different lanthanide ions at predetermined locations within their molecular structures. The advantages of this are many, such as the ability to investigate these phenomena with ultimate atomic control, even in solution. A site-selective molecular scaffold combining the ligands H2LA and H2LB has been developed to enable the targeted positioning of lanthanide ions based on differences in ionic radii. This architecture allows a Pr3+ ion to be stabilised in the central coordination cavity, while smaller Yb3+ ions preferentially occupy peripheral sites, giving rise to a [YbPrYb] topology within a discrete heterometallic complex. The resulting compound, [Yb2Pr(LA)2(LB)2(H2O)2(py)]NO3, has been structurally characterised and used to investigate intramolecular ET between Pr3+ and Yb3+ ions. Two structural analogues, [YbLaYb] and [LuPrLu], were employed as luminescence controls to independently probe the donor and acceptor roles of each metal ion. Photophysical studies in solution and in the solid state revealed two distinct ET processes: a highly efficient Pr3+ → Yb3+ transfer, resulting in near-infrared (NIR) emission at 980 nm, and a competitive Yb3+ → Pr3+ back-transfer, leading to non-radiative deactivation. Lifetime analyses confirmed that both transfer pathways occur with near-quantitative efficiency. These findings represent the first evidence of bidirectional Pr3+ ⇄ Yb3+ energy transfer within a molecular complex and highlight the power of ligand architecture in enabling and modulating lanthanide photophysical behaviour.
Introduction
The unique photophysical behaviour of trivalent lanthanide ions arises from their shielded 4f orbitals, which give rise to a cascade of long-lived excited states and narrow emission bands spanning from the ultraviolet to the near-infrared (NIR) region.1 These transitions are parity-forbidden, and often spin-forbidden, resulting in low absorption coefficients but long radiative lifetimes. These features render lanthanide ions central to applications in optical telecommunications, bioimaging, solid-state lighting, and solar energy conversion.2,3 As direct 4f–4f excitation is often inefficient, energy transfer (ET) processes are widely employed to sensitise their luminescence. These processes typically involve the excitation of a chromophore or another metal ion, followed by non-radiative energy transfer to the lanthanide acceptor with eventual photon emission from the lanthanide excited state.Sensitisation through ligand-to-metal ET is well established, particularly using π-conjugated organic antennae via intersystem crossing (ISC) mechanisms.4,5 A powerful alternative involves lanthanide-to-lanthanide (Ln → Ln′) energy transfer, which enables the design of up- or down-conversion systems.6 These multistep processes facilitate access to optical transitions that are otherwise difficult to achieve via direct excitation. Up-conversion (UC) is particularly attractive for generating high-energy emission from low-energy photons—relevant to NIR-to-visible imaging and photonic devices.7 Conversely, down-conversion (DC), or quantum cutting (QC), entails the redistribution of energy from one high-energy photon into two or more low-energy photons, with the potential to exceed 100% quantum yield and find application in next-generation photovoltaics.8–10
Much of the current understanding of Ln → Ln′ ET processes derives from studies on inorganic hosts such as glasses and ceramics, with only a few studies covering up-converting molecular systems, most notably those from the groups of Piguet, Nonat, and Charbonnière,11–16 including the remarkable Yb3+ cooperative luminescence.17,18 Recently, we have focused on gaining a deeper understanding of Ln → Ln′ ET in heteropolymetallic systems. In this context, we previously reported Nd → Yb ET leading to enhanced Yb3+ emission,19 as well as a detrimental Nd → Er → Nd back transfer leading to luminescence quenching.20
In this context, we decided to explore another f-element couple: Pr3+/Yb3+. Our motivation comes from a survey of the inorganic literature where cooperative sensitisation from Pr3+ to Yb3+ has often been reported in glass matrices. In particular, QC properties were described, where excitation of Pr3+ at visible wavelengths results in emission from Yb3+ in the NIR.21–24 However, the energy transfer process for this Pr3+/Yb3+ pair is still controversial with different mechanisms proposed. For example, the two-step sequential ET mechanism was first reported by Van der Ende et al. (Pr3+: 3P0 → 1G4 to Yb3+: 2F5/2 → 2F7/2 and Pr3+: 1G4 → 3H4 to Yb3+: 2F5/2 → 2F7/2),25,26 the one-step ET mechanism was reported by Van Wijngaarden et al.,26 or an experimentally evidenced cooperative ET process.27,28 Of particular relevance to this study is a process leading to one IR photon emission at 980 nm for each photon absorbed at 586 nm (resonant with the 1D2 level of Pr3+).29,30 Notably, despite the promising match of this ET pair, Pr3+ → Yb3+ energy transfer remains virtually unexplored in molecular systems, probably because of the lack of suitable systems to undergo this study. Thus, to the best of our knowledge, no reports to date have demonstrated this energy transfer pathway in discrete coordination compounds.
This gap is both striking and compelling. Molecular lanthanide complexes offer advantages over extended solids: the modularity of synthetic design, the ability to tune donor–acceptor distances, control over metal coordination environments, and the integration of additional functional units into a single scaffold. Nevertheless, the realisation of heterometallic complexes capable of selective Ln–Ln′ energy transfer presents significant synthetic challenges. The similarity in coordination chemistry across the lanthanide series often impedes site-selective metal placement. Achieving this selectivity requires precise control over ligand architecture to favour the formation of non-statistical, pure heterometallic species.
Building on our previous success in developing ligand systems capable of enforcing site-selective binding in [LnLn′]31–33 and [LnLn′Ln]20,34–36 complexes with well-defined topologies, we now explore Pr3+ to Yb3+ energy transfer within a trinuclear molecular architecture. Our strategy involves a tailored bis-diketonate/dipicolinate ligand system that yields robust, high-purity heterometallic complexes by exploiting the differences in ionic radii among the lanthanides. This enables us to construct [YbPrYb] molecules, in which the central Pr3+ ion is flanked by two Yb3+ acceptors—an arrangement that offers potential for cooperative energy transfer pathways and enhanced emission efficiency in the NIR region.
Herein, we present the synthesis, structural characterisation, and photophysical investigation of a new [Yb2Pr(LA)2(LB)2(H2O)2(py)]NO3 complex, [YbPrYb] (3) (H2LA, 2,6-bis[(3-oxo-3-naphthalene-2yl)propionyl]pyridine; H2LB, 6-(3-(naphthalene-2-yl)-3-oxopropanoyl)picolinic acid; Fig. 1). This compound serves as a rare molecular platform for studying Pr3+-to-Yb3+ energy transfer. Its analogues, [YbLaYb] (1)19 and [LuPrLu] (2), are also prepared as luminescence controls (2 reported here for the first time), permitting discrimination of donor and acceptor contributions and enabling a detailed mechanistic study of the energy transfer pathway.
Notably, the inclusion of an emissive Pr3+ complex in solution is particularly significant, as studies on the photoluminescence of praseodymium compounds in molecular environments remain scarce and are typically limited to steady-state emission analyses.37–40 It is worth noting that Pr3+ is special as a lanthanide ion, as it possesses three potentially emissive levels, namely, 3P0 (∼21![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 400 cm−1), 1D2 (∼16
400 cm−1), 1D2 (∼16![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 900 cm−1), and 1G4 (∼14
900 cm−1), and 1G4 (∼14![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 500 cm−1) (Fig. 2).41 The emission properties strongly depend on which excited state is populated. In inorganic solids, the 3P0 state is generally populated via direct f–f excitation, while in molecular systems, sensitised luminescence generally occurs from the 1D2 state.42,43 The 1G4 state, on the other hand, is often poorly emissive due to its small energy gap with the 3F4,3 states (∼1100 cm−1), making multiphonon deactivation processes highly favourable. The ability to probe both the steady-state and time-resolved behaviour of Pr3+ emission thus offers valuable insight into its sensitisation and deactivation mechanisms, expanding our understanding of lanthanide photophysics in molecular systems.
500 cm−1) (Fig. 2).41 The emission properties strongly depend on which excited state is populated. In inorganic solids, the 3P0 state is generally populated via direct f–f excitation, while in molecular systems, sensitised luminescence generally occurs from the 1D2 state.42,43 The 1G4 state, on the other hand, is often poorly emissive due to its small energy gap with the 3F4,3 states (∼1100 cm−1), making multiphonon deactivation processes highly favourable. The ability to probe both the steady-state and time-resolved behaviour of Pr3+ emission thus offers valuable insight into its sensitisation and deactivation mechanisms, expanding our understanding of lanthanide photophysics in molecular systems.
 | ||
| Fig. 2 Simplified energy levels of Pr3+ and Yb3+, where the emissive excited states are highlighted in bold along with their corresponding principal radiative transitions. | ||
Results and discussion
Synthesis and structures
The synergy between the photophysical properties of Pr3+ and Yb3+ within a molecule was studied on the new heterolanthanide coordination complex [Yb2Pr(LA)2(LB)2(H2O)2(py)]NO3, [YbPrYb] (3). It was obtained by exploiting a synthetic protocol that enables the generation of very pure and selective [LnLn′Ln] heterometallic molecules by combining stoichiometric amounts of ligands H2LA44 and H2LB34 with the corresponding amounts of specific Ln3+ and Ln′3+ salts featuring different ionic radii (r(Ln′) > r(Ln)).20,34–36 A balanced chemical equation describing the formation of 3 may be proposed (eqn (1)), albeit the yield of crystals is only about 15%. | (1) |
Compound 2, [Lu2Pr(LA)2(LB)2(H2O)2(py)]NO3, [LuPrLu] was obtained here for the first time in the same manner and with a very similar yield. Homogeneous phases of crystals of 2 or 3 were only obtained by adding one equivalent of CuCl2 to the reaction mixture, which does not participate in the reaction but plays the role of a modulator of crystallisation (see the SI for details).35 The molecular formulae and structures of 2 and 3 are equivalent as established by single crystal X-ray diffraction (see below). The atomic composition of the bulk crystalline samples of both compounds was established by C, H and N microanalysis and inductively coupled plasma (ICP) metal content determinations, the latter establishing the expected Ln/Ln′ molar ratio within the experimental error (see the SI). For both compounds, this formulation was consistent with the analysis of variable temperature magnetic susceptibility and isothermal field-dependent magnetisation measurements (see the SI and Fig. S1 for details). These experiments confirmed the presence of one Pr3+ ion (2) or two Yb3+ ions only very weakly interacting with a Pr3+ centre (3), within the respective molecules, considering their experimentally established molecular masses.
Mass spectrometry proved to be a highly valuable technique to corroborate the persistence of the triatomic [LnLn′Ln] assemblies in solution, the purity of the heterometallic 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 composition and the absence of any metal scrambling. Thus, for both compounds, all fragments composed of three metal ions exhibit the expected composition (Fig. S2–S7), while no equivalent fragments with any other metal distribution were detected. The absence of any metal scrambling is very striking, considering that it is a very common occurrence among lanthanide coordination complexes.45 This could be explained by a combination of thermodynamic reasons (as previously established through DFT calculations)36 and the presence of a kinetic barrier that results from the very compact scaffold formed by the [Ln2Ln′(LA)2(LB)2]+ assembly.
2 composition and the absence of any metal scrambling. Thus, for both compounds, all fragments composed of three metal ions exhibit the expected composition (Fig. S2–S7), while no equivalent fragments with any other metal distribution were detected. The absence of any metal scrambling is very striking, considering that it is a very common occurrence among lanthanide coordination complexes.45 This could be explained by a combination of thermodynamic reasons (as previously established through DFT calculations)36 and the presence of a kinetic barrier that results from the very compact scaffold formed by the [Ln2Ln′(LA)2(LB)2]+ assembly.
The molecular structures of 2 and 3 were determined by SCXRD at 100 K (Tables S1–S3). Both compounds are isostructural; therefore, their structures will be described jointly. The space group is triclinic P![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) . The unit cell contains two asymmetric units. The latter is composed of the main complex cation and its NO3− counterion, in addition to ten molecules of pyridine of crystallisation. The main complex consists of a trinuclear [LnLn′Ln] coordination molecular array (Fig. 1, and S8–S10; [LuPrLu], 2 and [YbPrYb], 3) where the metals are coordinated and also bridged by a total of two μ3-LA2− and two μ-LB2− ligands. The ligands engage all their β-diketonate and dipicolinate-like coordination pockets to chelate the metals, while linking them via monoatomic bridges with some of the oxygen atoms in these chelating units. In this manner, the central Pr3+ ion is bound to two O,N,O and two O,O coordination pockets and to one terminal pyridine ligand, featuring a coordination number (CN) of 11. The peripheral ions (Lu3+ or Yb3+) are surrounded by one O,N,O and two O,O pockets, in addition to one H2O ligand, exhibiting CN = 8. According to continuous symmetry measures with the program SHAPE,46 the ideal polyhedra that best describe the central and the side metal positions are a capped pentagonal antiprism and a biaugmented trigonal prism, respectively. The distances (on a 0–100 scale) of Pr3+ from its closest ideal geometry are 6.188 and 6.245 for 2 and 3, respectively, whereas for Lu3+ or Yb3+, these separations are 1.514/1.318 and 1.462/1.363, respectively (in the Ln1/Ln2 format). As explained above, the topological selectivity of the [LnLn′Ln] compounds for different lanthanides within the molecule is based on differences in atomic radii, with the central position hosting the larger metal ions. Here, the average Ln–O bond distances in 2 are 2.644 and 2.303/2.309 Å for Pr and Lu1/Lu2, respectively, while for 3, these values are 2.646 and 2.311/2.320 Å for Pr and Yb1/Yb2, respectively. Within the complex, the Ln–Pr–Ln angle is 174.45° (Ln
. The unit cell contains two asymmetric units. The latter is composed of the main complex cation and its NO3− counterion, in addition to ten molecules of pyridine of crystallisation. The main complex consists of a trinuclear [LnLn′Ln] coordination molecular array (Fig. 1, and S8–S10; [LuPrLu], 2 and [YbPrYb], 3) where the metals are coordinated and also bridged by a total of two μ3-LA2− and two μ-LB2− ligands. The ligands engage all their β-diketonate and dipicolinate-like coordination pockets to chelate the metals, while linking them via monoatomic bridges with some of the oxygen atoms in these chelating units. In this manner, the central Pr3+ ion is bound to two O,N,O and two O,O coordination pockets and to one terminal pyridine ligand, featuring a coordination number (CN) of 11. The peripheral ions (Lu3+ or Yb3+) are surrounded by one O,N,O and two O,O pockets, in addition to one H2O ligand, exhibiting CN = 8. According to continuous symmetry measures with the program SHAPE,46 the ideal polyhedra that best describe the central and the side metal positions are a capped pentagonal antiprism and a biaugmented trigonal prism, respectively. The distances (on a 0–100 scale) of Pr3+ from its closest ideal geometry are 6.188 and 6.245 for 2 and 3, respectively, whereas for Lu3+ or Yb3+, these separations are 1.514/1.318 and 1.462/1.363, respectively (in the Ln1/Ln2 format). As explained above, the topological selectivity of the [LnLn′Ln] compounds for different lanthanides within the molecule is based on differences in atomic radii, with the central position hosting the larger metal ions. Here, the average Ln–O bond distances in 2 are 2.644 and 2.303/2.309 Å for Pr and Lu1/Lu2, respectively, while for 3, these values are 2.646 and 2.311/2.320 Å for Pr and Yb1/Yb2, respectively. Within the complex, the Ln–Pr–Ln angle is 174.45° (Ln![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) Lu, 2) and 174.42° (Ln
Lu, 2) and 174.42° (Ln![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) Yb, 3); the Ln⋯Ln distance is 7.837/7.853 Å (for 2/3), while the Pr⋯Ln separations are 3.924 and 3.922 Å (for 2) and 3.932 and 3.931 Å (for 3). Interestingly, the shortest intermolecular Ln⋯Ln distances are 6.055/6.072 Å (for 2/3); thus, some of these two peripheral metals are closer when lying in two different molecules than when within the same complex. These close intermolecular contacts occur between pairs of complexes that are related by an inversion centre. Each pair is connected by two identical, complementary O–H⋯O hydrogen bonds, where the water ligand of one molecule forms a hydrogen bond with the oxygen atom of a carboxylate group on the other molecule (Fig. S9 and Table S3).
Yb, 3); the Ln⋯Ln distance is 7.837/7.853 Å (for 2/3), while the Pr⋯Ln separations are 3.924 and 3.922 Å (for 2) and 3.932 and 3.931 Å (for 3). Interestingly, the shortest intermolecular Ln⋯Ln distances are 6.055/6.072 Å (for 2/3); thus, some of these two peripheral metals are closer when lying in two different molecules than when within the same complex. These close intermolecular contacts occur between pairs of complexes that are related by an inversion centre. Each pair is connected by two identical, complementary O–H⋯O hydrogen bonds, where the water ligand of one molecule forms a hydrogen bond with the oxygen atom of a carboxylate group on the other molecule (Fig. S9 and Table S3).
Photophysical properties
To gain insight into the intramolecular Pr3+ → Yb3+ energy transfer process occurring within the [YbPrYb] (3) complex, comparative photophysical studies were carried out using two structurally related analogues: the previously reported [YbLaYb] (1)19 and the newly synthesised [LuPrLu] (2) complexes. These were selected to independently probe the roles of the donor and acceptor ions, as complex 1 displays only Yb3+ emission at 980 nm, while complex 2 is expected to exhibit luminescence solely from the excited states of Pr3+. All measurements were conducted in dilute (10−4 M) 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 mixtures of MeOH-d4 and DMSO-d6 to minimise intermolecular energy transfer events, both at room temperature (RT) and at 77 K. Solid-state measurements were also performed to confirm that the behaviour in solution mirrors the structural features identified by single-crystal X-ray diffraction analysis (Table S4). A summary of the key photophysical parameters obtained from the solution measurements is presented in Table 1.
1 mixtures of MeOH-d4 and DMSO-d6 to minimise intermolecular energy transfer events, both at room temperature (RT) and at 77 K. Solid-state measurements were also performed to confirm that the behaviour in solution mirrors the structural features identified by single-crystal X-ray diffraction analysis (Table S4). A summary of the key photophysical parameters obtained from the solution measurements is presented in Table 1.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 mixture of MeOH-d4 and DMSO-d6 (10−4 M)
1 mixture of MeOH-d4 and DMSO-d6 (10−4 M)
| Compound | λ em (nm) | τ obs (μs) | Φ ET | |||
|---|---|---|---|---|---|---|
| RT | 77 K | Type | RT | 77 K | ||
| a λ exc = 400 nm. b λ exc = 400 nm; λem = 975 nm (1, 3) and 1020 nm (2). c Calculated following eqn (2). | ||||||
| [YbLaYb] (1) | 980 | 16 | 21 | — | — | — |
| [LuPrLu] (2) | 1020 | 0.04 | 0.30 | — | — | — |
| [YbPrYb] (3) | 980 | 0.10 | 0.09 | (Yb3+ → Pr3+) | 0.99 | 0.99 |
| — | — | — | (Pr3+ → Yb3+) | 1.00 | 1.00 | |
The triplet excited states of the ligands employed in these systems, H2LA and H2LB, have previously been estimated at approximately 18![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 950 cm−1 and 19
950 cm−1 and 19![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 050 cm−1, respectively.19 These energies have been shown to be sufficiently high to enable energy transfer to Yb3+ centres, with the F5/2 state lying at approximately 10
050 cm−1, respectively.19 These energies have been shown to be sufficiently high to enable energy transfer to Yb3+ centres, with the F5/2 state lying at approximately 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 300 cm−1.19 In the case of Pr3+, these antennae are expected to sensitise primarily the 1D2 excited state, located around 16
300 cm−1.19 In the case of Pr3+, these antennae are expected to sensitise primarily the 1D2 excited state, located around 16![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 900 cm−1, as they are too low in energy to efficiently populate the higher 3P0 state (21
900 cm−1, as they are too low in energy to efficiently populate the higher 3P0 state (21![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cm−1). Based on this, we limit our consideration to a simplified mechanism of Pr3+ → Yb3+ energy transfer—unlike the multiple possible pathways proposed in ceramic materials—wherein each photon exciting the 1D2 state of Pr3+ can, through a cross-relaxation process, result in the emission of a single near-infrared photon from Yb3+ (2F5/2 → 2F7/2) at 980 nm.
000 cm−1). Based on this, we limit our consideration to a simplified mechanism of Pr3+ → Yb3+ energy transfer—unlike the multiple possible pathways proposed in ceramic materials—wherein each photon exciting the 1D2 state of Pr3+ can, through a cross-relaxation process, result in the emission of a single near-infrared photon from Yb3+ (2F5/2 → 2F7/2) at 980 nm.
This hypothesis was first examined by studying the previously reported [YbLaYb] (1) complex in deuterated solutions. Consistent with prior observations in non-deuterated conditions,19 excitation at 400 nm at RT resulted in the characteristic Yb3+ emission profile centred at 980 nm, corresponding to the 2F5/2 → 2F7/2 transition (Fig. 3a). At 77 K, the emission spectrum became significantly better resolved due to the reduced line broadening and diminished contributions from thermally populated crystal field-excited states (hot bands). Under these conditions, the four expected transitions associated with the Kramers-split 2F7/2 ground state were clearly observed, showing the same spectral features as those previously recorded in non-deuterated solution and here in the solid state (Fig. S11). These results highlight the structural robustness of complex 1 both in solution and in the solid state, where the geometry resolved by SCXRD, featuring C2v symmetry around both Yb3+ centres, is preserved in solution. Excited state lifetimes were also measured in deuterated solutions at both RT and 77 K, yielding values of 16 and 21 μs, respectively (Fig. S12). These results indicate a clear decrease in phonon-mediated non-radiative relaxation when compared with the value determined at RT in non-deuterated conditions (9 μs).47
Complex 2, [LuPrLu], was then studied in a similar manner to first assess the effectiveness of the antenna effect in populating the Pr3+ 1D2 excited state. First, the emission spectra were recorded in deuterated solution at RT and 77 K, revealing emission almost exclusively in the NIR region. The very subtle emission observed in the visible range is attributed to residual fluorescence and/or phosphorescence from the antenna along with a weak, poorly defined emission band at 607 nm that corresponds to the 1D2 → 3H4 transition of Pr3+ (Fig. S13). This is not surprising, as the most prominent visible emission of Pr3+ results from higher-lying excited states (3P0) as has been well evidenced in the literature37,39,40,48–50
Nonetheless, examples of Pr3+ complexes exhibiting NIR emission remain relatively scarce in the literature, underscoring the significance of the results presented here and demonstrating the versatility and efficiency of the antenna system employed. Upon careful analysis of the 77 K emission spectrum, three distinct emission lines can be assigned to the expected transitions originating from the 1D2 excited state, namely, 1D2 → 2F2 at 861 nm, 1D2 → 2F3,4 centred at 1022 nm, and 1D2 → 1G4 centred at 1480 nm (Fig. 3b) in agreement with the previously reported NIR-emissive Pr3+ complexes.42,51–53
As with complex 1, the emission of complex 2 was further tested in the solid state at 77 K confirming that the structure is preserved upon dissolution (Fig. S14). Finally, the excited-state lifetimes were measured and fitted monoexponentially, giving values of 0.03 μs and 0.4 μs at RT and 77 K, respectively. (Fig. S15) These lifetimes are within the typical range for Pr3+-based NIR emitters and further support the occurrence of efficient population of the 1D2 state.42,51,52
Thus, after confirming that both the Yb3+ (2F5/2) and Pr3+ (1D2) excited states are efficiently sensitised by our ligand scaffold, we proceeded to investigate the intramolecular energy transfer between the two ions in a molecular system combining both metals. To this end, the emission spectra of the [YbPrYb] (3) complex were recorded at both RT and 77 K upon excitation at 400 nm. Interestingly, upon detailed analysis, emission was observed exclusively at 980 nm (Fig. 3c), with a spectral profile identical to that of complex 1 (Fig. 3d). This observation unambiguously indicates that the emission arises from the Yb3+ 2F5/2 → 2F7/2 transition, while no detectable emission from the Pr3+ excited states was present. This behaviour suggests that the 1D2 excited state of Pr3+ is deactivated through a non-radiative pathway, most likely due to an efficient quantitative energy transfer to the neighbouring Yb3+ centres (Fig. 4). These findings are consistent with previous reports on glass matrices, where Pr3+ → Yb3+ energy transfer occurs efficiently via a cross-relaxation mechanism (Pr3+: 1D2 → 3F4; Yb3+: 2F7/2 → 2F5/2), followed by NIR photon emission from Yb3+ (2F5/2 → 2F7/2).29,30 The excited-state lifetime of complex 3 was then measured at 980 nm, yielding values of 0.10 μs at room temperature and 0.09 μs at 77 K after mono-exponential fitting (Fig. S16). When comparing these values to those previously measured for complex 1, a marked shortening of the Yb3+ excited-state lifetime is observed. Given that both the ligand scaffold and the geometry around the Yb3+ centres are identical in the two complexes, this difference strongly suggests the occurrence of an additional non-radiative deexcitation pathway for the Yb3+ excited state, typically a back energy transfer from Yb3+ to Pr3+, which is subsequently deactivated via non-radiative decay. The efficiency of the suggested Yb3+ → Pr3+ energy transfer can be estimated using the following expression:
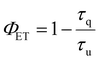 | (2) |
To further probe the occurrence of this dual-path energy transfer between the lanthanide centres, iso-absorbance diluted solutions (OD = 0.56 at 400 nm, Fig. S17) of complexes 1, 2, and 3 were studied under identical photophysical conditions at room temperature, allowing direct comparison of their emission intensities (Fig. 4). It is immediately evident that the emission of complex 1 at 980 nm is more intense than that of complexes 2 and 3 (Fig. 4a). Therefore, to better resolve the differences between complexes 2 and 3, a separate photophysical comparison was carried out (Fig. 4b). Upon moving from complex 2 to complex 3, the Pr3+ emission vanished, while a distinct Yb3+ emission at 980 nm emerged, clearly confirming efficient Pr3+ → Yb3+ energy transfer. However, when comparing this to the emission of complex 1, a marked decrease in intensity is observed, supporting the occurrence of a back-energy transfer Yb3+ → Pr3+, which is subsequently deactivated non-radiatively. We investigated the possible origin of this energy transfer by comparing the absorption spectrum of Pr3+ (here measured on the salt PrCl3·6H2O) with the Yb3+ emission in complex 1 (Fig. S18). This comparison shows that a clear spectral overlap occurs in the NIR between the Yb3+ 2F5/2 → 2F7/2 emission band and the Pr3+ 3H4 → 1G4 absorption band. In the framework of the Förster theory, this spectral overlap strongly suggests a FRET transfer from the Yb3+ 2F5/2 excited state to the Pr3+ 1G4 one, followed by non-radiative deactivation to the Pr3+ 3H4 ground state.
Conclusions
A unique combination of ligands capable of stabilising very selectively trinuclear heterometallic lanthanide complexes with a [LnLn′Ln] linear core (r(Ln′) > r(Ln)) gives access to a topologically pure [YbPrYb] complex. Photophysical investigation of this compound, assisted by comparison with the analogous [YbLaYb] and [LuPrLu] complexes, demonstrates a dual ET pathway at the molecular level: an efficient forward Pr3+ → Yb3+ process, leading to characteristic Yb3+ emission at 980 nm, and a competing back-transfer Yb3+ → Pr3+ pathway, which quenches the Yb3+ excited state through non-radiative deactivation (Fig. 5). This bidirectional energy exchange, so far never evidenced in molecular systems for this pair of lanthanide centres, underscores the dynamic nature of lanthanide–lanthanide interactions and the pivotal role of the precise coordination environment of the metals in tuning their photophysical behaviour. These findings extend current understanding of lanthanide-to-lanthanide ET into the domain of molecular chemistry and offer new strategies for developing solution-processable lanthanide materials for photonics.Author contributions
S. K.: conceptualization and photophysical data interpretation; D. M.: coordination chemistry synthesis and collection of photophysical data; A. S.: collection of photophysical data and interpretation; L. A. B.: conceptualization, ligand synthesis and coordination chemistry synthesis; D. A.: conceptualization and coordination chemistry synthesis; O. R.: crystal structure determination and magnetic properties; Y. G.: collection of photophysical data and interpretation; O. M.: conceptualization and photophysical data interpretation; L. A. G.: conceptualization, photophysical data interpretation and paper writing; and G. A.: research coordination, conceptualization and paper writing.Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article have been included as part of the supplementary information (SI). Supplementary information: crystallographic details, information on synthetic procedures, ESI-MS, absorption and emission data, lifetime measurements and fits, NMR experiments, magnetic data treatment, and DFT calculations. See DOI: https://doi.org/10.1039/d5qi01585e.CCDC 2472886 (3) and 2472887 (2) contain the supplementary crystallographic data for this paper.55a,b
Acknowledgements
G. A. thanks the NextGenerationEU/PRTR-C17.I1 (from “Plan Complementario en Comunicación Cuántica” funded by Generalitat de Catalunya and the European Union) and Generalitat de Catalunya for the ICREA Academia 2023 prize. G. A., D. M., L. A. B., D. A. and O. R. thank the Spanish Ministry of Innovation for grants PID2020-118329RB-I00, PID2022-137764OB-I00, PDC2022-133184-I00 and TED2021-129214B-I00. DM thanks the Generalitat de Catalunya for an FI-SDUR Grant (2020-FISDU-00492).References
- J.-C. G. Bünzli, On the design of highly luminescent lanthanide complexes, Coord. Chem. Rev., 2015, 293–294, 19–47 CrossRef.
- T. Jüstel, H. Nikol and C. Ronda, New developments in the field of luminescent materials for lighting and displays, Angew. Chem., Int. Ed., 1998, 37, 3084–3103 CrossRef.
- C. Alexander, Z. Guo, P. B. Glover, S. Faulkner and Z. Pikramenou, Luminescent lanthanides in biorelated applications: From molecules to nanoparticles and diagnostic probes to therapeutics, Chem. Rev., 2025, 125, 2269–2370 CrossRef.
- J.-C. G. Bünzli and S. V. Eliseeva, Basics of Lanthanide Photophysics, Springer Berlin Heidelberg, 2010, pp. 1–45. DOI:10.1007/4243_2010_3.
- A. D'Aléo, F. Pointillart, L. Ouahab, C. Andraud and O. Maury, Charge transfer excited states sensitization of lanthanide emitting from the visible to the near-infra-red, Coord. Chem. Rev., 2012, 256, 1604–1620 CrossRef.
- F. Auzel, Upconversion and anti-Stokes processes with f and d ions in solids, Chem. Rev., 2004, 104, 139–174 CrossRef PubMed.
- M. Haase and H. Schäfer, Upconverting nanoparticles, Angew. Chem., Int. Ed., 2011, 50, 5808–5829 CrossRef PubMed.
- Y. Tai, G. Zheng, H. Wang and J. Bai, Broadband down-conversion based near infrared quantum cutting in Eu2+–Yb3+ co-doped SrAl2O4 for crystalline silicon solar cells, J. Solid State Chem., 2015, 226, 250–254 CrossRef.
- F. Enrichi, C. Armellini, S. Belmokhtar, A. Bouajaj, A. Chiappini, M. Ferrari, A. Quandt, G. C. Righini, A. Vomiero and L. Zur, Visible to NIR downconversion process in Tb3+–Yb3+ codoped silica–hafnia glass and glass–ceramic sol–gel waveguides for solar cells, J. Lumin., 2018, 193, 44–50 CrossRef.
- Z. Wang and A. Meijerink, Dye-sensitized downconversion, J. Phys. Chem. Lett., 2018, 9, 1522–1526 CrossRef.
- N. Souri, P. Tian, C. Platas-Iglesias, K.-L. Wong, A. Nonat and L. J. Charbonnière, Upconverted photosensitization of Tb visible emission by NIR Yb excitation in discrete supramolecular heteropolynuclear complexes, J. Am. Chem. Soc., 2017, 139, 1456–1459 CrossRef.
- L. J. Charbonnière, A. M. Nonat, R. C. Knighton and L. Godec, Upconverting photons at the molecular scale with lanthanide complexes, Chem. Sci., 2024, 15, 3048–3059 RSC.
- A. Nonat, C. F. Chan, T. Liu, C. Platas-Iglesias, Z. Liu, W.-T. Wong, W.-K. Wong, K.-L. Wong and L. J. Charbonnière, Room temperature molecular up conversion in solution, Nat. Commun., 2016, 7, 11978 CrossRef.
- L. Aboshyan-Sorgho, C. Besnard, P. Pattison, K. R. Kittilstved, A. Aebischer, J.-C. G. Bünzli, A. Hauser and C. Piguet, Near-infrared→visible light upconversion in a molecular trinuclear d-f-d complex, Angew. Chem., Int. Ed., 2011, 50, 4108–4112 CrossRef.
- D. Zare, Y. Suffren, H. Nozary, A. Hauser and C. Piguet, Controlling lanthanide exchange in triple-stranded helicates: A way to optimize molecular light-upconversion, Angew. Chem., Int. Ed., 2017, 56, 14612–14617 CrossRef PubMed.
- N. Hamon, L. Godec, S. Sanchez, M. Beyler, L. J. Charbonnière and R. Tripier, Upconversion luminescence with bis–pyclen Yb(III) chelates: Crown vs. linear polyether linkers in discrete heteropolynuclear architectures, Angew. Chem., Int. Ed., 2025, 64, e202414608 CrossRef PubMed.
- R. C. Knighton, L. K. Soro, L. Francés-Soriano, A. Rodríguez-Rodríguez, G. Pilet, M. Lenertz, C. Platas-Iglesias, N. Hildebrandt and L. J. Charbonnière, Cooperative luminescence and cooperative sensitisation upconversion of lanthanide complexes in solution, Angew. Chem., Int. Ed., 2022, 61, e202113114 CrossRef PubMed.
- L. Godec, R. C. Knighton, N. Hamon, W. Thor, K.-L. Wong, R. Tripier and L. J. Charbonnière, Heteropolynuclear lanthanide(III) complexes for cooperative sensitization upconversion in water, J. Am. Chem. Soc., 2025, 147, 31187–31197 CrossRef PubMed.
- D. Maniaki, A. Sickinger, L. A. Barrios, D. Aguilà, O. Roubeau, N. S. Settineri, Y. Guyot, F. Riobé, O. Maury, L. A. Galán and G. Aromí, Distributive Nd-to-Yb energy transfer within pure [YbNdYb] heterometallic molecules, Inorg. Chem., 2023, 62, 3106–3115 CrossRef PubMed.
- D. Maniaki, A. Sickinger, L. A. Barrios, D. Aguilà, O. Roubeau, Y. Guyot, F. Riobé, O. Maury, L. Abad Galán and G. Aromí, Energy exchange between Nd3+ and Er3+ centers within molecular complexes, Chem. Sci., 2024, 15, 18295–18302 RSC.
- S. Tomar, N. K. Mishra, V. Chauhan, K. Kumar and C. Shivakumara, Study of multimodal light emissions from Pr3+/Yb3+ doped NaLa(MoO4)2 phosphors for optoelectronic devices and plant-growth applications, Dalton Trans., 2025, 54, 1913–1928 RSC.
- F. E. Maturi, A. Gaddam, C. D. S. Brites, J. M. M. Souza, H. Eckert, S. J. L. Ribeiro, L. D. Carlos and D. Manzani, Extending the palette of luminescent primary thermometers: Yb3+/Pr3+ co-doped fluoride phosphate Glasses, Chem. Mater., 2023, 35, 7229–7238 CrossRef PubMed.
- M. Seshadri, I. T. C. Santos, M. J. V. Bell, J. Lapointe, Y. Messaddeq and V. Anjos, Near-infrared quantum cutting luminescence in Pr3+/Yb3+ doped lead bismuth borate glass, Sci. Rep., 2022, 12, 19311 CrossRef PubMed.
- D. Ding, J. Gao, S. Zhang and L. Duo, The photoluminescence properties of Pr3+-Yb3+ co-doped gallo-germanate glasses and glass ceramics as energy converter, J. Lumin., 2020, 226, 117512 CrossRef.
- B. M. Van Der Ende, L. Aarts and A. Meijerink, Near-infrared quantum cutting for photovoltaics, Adv. Mater., 2009, 21, 3073–3077 CrossRef.
- J. T. van Wijngaarden, S. Scheidelaar, T. J. H. Vlugt, M. F. Reid and A. Meijerink, Energy transfer mechanism for downconversion in the (Pr3+, Yb3+) couple, Phys. Rev. B:Condens. Matter Mater. Phys., 2010, 81, 155112 CrossRef.
- S. Balaji, D. Ghosh, K. Biswas, G. Gupta and K. Annapurna, Experimental evidence for quantum cutting co-operative energy transfer process in Pr3+/Yb3+ ions co-doped fluorotellurite glass: dispute over energy transfer mechanism, Phys. Chem. Chem. Phys., 2016, 18, 33115–33125 RSC.
- Y. Katayama and S. Tanabe, Mechanism of quantum cutting in Pr3+–Yb3+ codoped oxyfluoride glass ceramics, J. Lumin., 2013, 134, 825–829 CrossRef.
- L. J. Borrero-González, G. Galleani, D. Manzani, L. A. O. Nunes and S. J. L. Ribeiro, Visible to infrared energy conversion in Pr3+–Yb3+ co-doped fluoroindate glasses, Opt. Mater., 2013, 35, 2085–2089 CrossRef.
- Q. J. Chen, W. J. Zhang, X. Y. Huang, G. P. Dong, M. Y. Peng and Q. Y. Zhang, Efficient down- and up-conversion of Pr3+–Yb3+ co-doped transparent oxyfluoride glass ceramics, J. Alloys Compd., 2012, 513, 139–144 CrossRef.
- D. Aguilà, L. A. Barrios, V. Velasco, O. Roubeau, A. Repollés, P. J. Alonso, J. Sesé, S. J. Teat, F. Luis and G. Aromí, Heterodimetallic [LnLn′] lanthanide complexes: Toward a chemical design of two-qubit molecular spin quantum gates, J. Am. Chem. Soc., 2014, 136, 14215–14222 CrossRef PubMed.
- J. González-Fabra, N. A. G. Bandeira, V. Velasco, L. A. Barrios, D. Aguilà, S. J. Teat, O. Roubeau, C. Bo and G. Aromí, Thermodynamic stability of heterodimetallic LnLn complexes: Synthesis and DFT studies, Chem. – Eur. J., 2017, 23, 5117–5125 CrossRef PubMed.
- D. Aguilà, V. Velasco, L. A. Barrios, J. González-Fabra, C. Bo, S. J. Teat, O. Roubeau and G. Aromí, Selective lanthanide distribution within a comprehensive series of heterometallic LnPr complexes, Inorg. Chem., 2018, 57, 8429–8439 CrossRef.
- V. Velasco, L. A. Barrios, M. Schütze, O. Roubeau, F. Luis, S. J. Teat, D. Aguilà and G. Aromí, Controlled heterometallic composition in linear trinuclear [LnCeLn] lanthanide molecular assemblies, Chem. – Eur. J., 2019, 25, 15228–15232 CrossRef PubMed.
- E. Macaluso, M. Rubín, D. Aguilà, A. Chiesa, L. A. Barrios, J. I. Martínez, P. J. Alonso, O. Roubeau, F. Luis, G. Aromí and S. Carretta, A heterometallic [LnLn′Ln] lanthanide complex as a qubit with embedded quantum error correction, Chem. Sci., 2020, 11, 10337–10343 RSC.
- D. Maniaki, D. Garay-Ruiz, L. A. Barrios, D. O. T. A. Martins, D. Aguilà, F. Tuna, D. Reta, O. Roubeau, C. Bo and G. Aromí, Unparalleled selectivity and electronic structure of heterometallic [LnLn′Ln] molecules as 3-qubit quantum gates, Chem. Sci., 2022, 13, 5574–5581 RSC.
- A. I. Voloshin, N. M. Shavaleev and V. P. Kazakov, Luminescence of praseodymium(III) chelates from two excited states (3P0 and 1D2) and its dependence on ligand triplet state energy, J. Lumin., 2001, 93, 199–204 CrossRef.
- C. Pettinari, F. Marchetti, R. Pettinari, A. Drozdov, S. Troyanov, A. I. Voloshin and N. M. Shavaleev, Synthesis, structure and luminescence properties of new rare earth metal complexes with 1-phenyl-3-methyl-4-acylpyrazol-5-ones, J. Chem. Soc., Dalton Trans., 2002, 1409, 10.1039/b108058j.
- V. M. Korshunov, M. T. Metlin, S. A. Ambrozevich, I. S. Golovanov, V. E. Gontcharenko, A. S. Selyukov and I. V. Taydakov, Impact of ligand-centered excited states on luminescence sensitization in Pr3+ complexes with β-diketones, Spectrochim. Acta, Part A, 2021, 260, 119863 CrossRef PubMed.
- N. Lalioti, E. Zygouri, V. Nastopoulos, N. Panagiotou, C. D. S. Brites, L. D. Carlos, J. Corredoira-Vázquez and V. Tangoulis, Luminescent thermometer based on a praseodymium(III) cyanide-based metal–organic framework, Inorg. Chem., 2025, 64, 192–201 CrossRef PubMed.
- W. T. Carnall, G. L. Goodman, K. Rajnak and R. S. Rana, A systematic analysis of the spectra of the lanthanides doped into single crystal LaF3, J. Chem. Phys., 1989, 90, 3443–3457 CrossRef.
- J. Scholten, G. A. Rosser, J. Wahsner, N. Alzakhem, C. Bischof, F. Stog, A. Beeby and M. Seitz, Anomalous reversal of C–H and C–D quenching efficiencies in luminescent praseodymium cryptates, J. Am. Chem. Soc., 2012, 134, 13915–13917 CrossRef PubMed.
- A. F. Martins, S. V. Eliseeva, H. F. Carvalho, J. M. C. Teixeira, C. T. B. Paula, P. Hermann, C. Platas-Iglesias, S. Petoud, É. Tóth and C. F. G. C. Geraldes, A bis(pyridine N-oxide) analogue of DOTA: Relaxometric properties of the GdIII complex and efficient sensitization of visible and NIR-emitting lanthanide(III) cations including PrIII and HoIII, Chem. – Eur. J., 2014, 20, 14834–14845 CrossRef PubMed.
- L. A. Barrios, E. Peyrecave-Lleixa, G. A. Craig, O. Roubeau, S. J. Teat and G. Aromí, Unusual crystal packing in a family of Fe{2,6-bis(pyrazol-3-yl)pyridine}(2) (2+) compounds and the effect on the occurrence of spin crossover and its cooperative character, Eur. J. Inorg. Chem., 2014, 6013–6021 CrossRef.
- M. Rancan, M. Rando, L. Bosi, A. Carlotto, R. Seraglia, J. Tessarolo, S. Carlotto, G. H. Clever and L. Armelao, Dynamic lanthanide exchange between quadruple-stranded cages: The effect of ionic radius differences on kinetics and thermodynamics, Inorg. Chem. Front., 2022, 9, 4495–4505 RSC.
- P. Alemany, D. Casanova, S. Alvarez, C. Dryzun and D. Avnir, in Reviews in Computational Chemistry, 2017, pp. 289–352. DOI:10.1002/9781119356059.ch7.
- E. Kreidt, C. Kruck and M. Seitz, in Handbook on the Physics and Chemistry of Rare Earths, ed. J.-C. G. Bünzli and V. K. Pecharsky, Elsevier, 2018, vol. 53, pp. 35–79 Search PubMed.
- H. Youssef, J. Becker, C. Pietzonka, I. V. Taydakov, F. Kraus and K. Müller-Buschbaum, Divalent europium, NIR and variable emission of trivalent Tm, Ho, Pr, Er, Nd, and Ce in 3D frameworks and 2D networks of Ln-pyridylpyrazolates, Chemistry, 2023, 5, 1006–1027 CrossRef.
- A. B. Ganaie, A. Ali and K. Iftikhar, Synthesis, structure, phase controlled colour tuning of dinuclear Pr(III) and Tb(III) complexes with fluorinated β-diketone and heterocyclic Lewis base as UV light converters, Polyhedron, 2022, 212, 115592 CrossRef.
- M. T. Metlin, S. A. Ambrozevich, D. A. Metlina, A. G. Vitukhnovsky and I. V. Taydakov, Luminescence of pyrazolic 1,3-diketone Pr3+ complex with 1,10-phenanthroline, J. Lumin., 2017, 188, 365–370 CrossRef.
- G. M. Davies, R. J. Aarons, G. R. Motson, J. C. Jeffery, H. Adams, S. Faulkner and M. D. Ward, Structural and near-IR photophysical studies on ternary lanthanide complexes containing poly(pyrazolyl)borate and 1,3-diketonate ligands, Dalton Trans., 2004, 1136–1144, 10.1039/b400992d.
- X. Feng, Y. Shang, H. Zhang, R. Li, W. Wang, D. Zhang, L. Wang and Z. Li, Enhanced luminescence and tunable magnetic properties of lanthanide coordination polymers based on fluorine substitution and phenanthroline ligand, RSC Adv., 2019, 9, 16328–16338 RSC.
- E. G. Moore, G. Szigethy, J. Xu, L.-O. Pålsson, A. Beeby and K. N. Raymond, 3-Hydroxypyridin-2-one complexes of near-infrared (NIR) emitting lanthanides: sensitization of holmium(III) and praseodymium(III) in aqueous solution, Angew. Chem., Int. Ed., 2008, 47, 9500–9503 CrossRef PubMed.
- R. Wang, C. Tan, H. Hou, H. Wang, B. Zou and R. Zeng, Multimode luminescence and highly anti-thermal quenching in Sb3/Yb3/Pr3 Co-doped Cs2NaYCl6 double perovskites, J. Mater. Chem. C, 2025, 13, 10072–10079 RSC.
- (a) CCDC 2472886: Experimental Crystal Structure Determination, 2025, DOI:10.5517/ccdc.csd.cc2p07jh; (b) CCDC 2472887: Experimental Crystal Structure Determination, 2025, DOI:10.5517/ccdc.csd.cc2p07kj.
| This journal is © the Partner Organisations 2025 |




