High-sensitivity lanthanide ratiometric nanothermometers in the second biological window through bidirectional thermal response engineering†
Shuohan
Sun
 a,
Dekang
Huang
a,
Dan
Li
b,
Mochen
Jia
c and
Guanying
Chen
a,
Dekang
Huang
a,
Dan
Li
b,
Mochen
Jia
c and
Guanying
Chen
 *b
*b
aSchool of Chemistry, Huazhong Agricultural University, Wuhan 430070, China
bMIIT Key Laboratory of Critical Materials Technology for New Energy Conversion and Storage, School of Chemistry and Chemical Engineering & Key Laboratory of Micro-systems and Micro-structures, Ministry of Education, Harbin Institute of Technology, Harbin 150001, China. E-mail: chenguanying@hit.edu.cn
cKey Laboratory of Materials Physics of Ministry of Education, School of Physics, Zhengzhou University, Zhengzhou 450052, China
First published on 17th July 2025
Abstract
In recent years, lanthanide-doped nanothermometers have made significant progress as non-contact temperature sensing tools in a variety of biological fields. However, limited successes have been met in high-sensitivity nanothermometers operating in the second near-infrared (NIR-II) biological window, which can enable sub-centimeter tissue penetration at micrometer-level imaging resolutions. Here, we adopted a core–shell structure to develop a high-sensitivity lanthanide ratiometric nanothermometer through a bidirectional thermal response of two emissions peaks in the NIR-II window. We showed that, under 808 nm excitation, the emission intensity at 1330 nm from neodymium ions exhibited a quenching effect, while the one at 1565 nm from erbium ions showed an enhancement at elevated temperatures. This contrasted temperature dependency endows the nanothermometer with a high relative sensitivity above 2.3% °C−1, with a maximum of 2.5% °C−1, throughout the entire physiological temperature range (30–45 °C). This high-sensitivity nanothermometer enables reliable differentiation of temperature differences in both normal and inflamed mice, highlighting its promising uses for in vivo applications.
Introduction
In living organisms, even slight temperature fluctuations can impact various biological processes.1,2 Yet, traditional contact thermometers are not suitable for in vivo temperature monitoring because their invasiveness and limited spatial resolution make it difficult to detect subtle thermal changes. This limitation has led to growing interest in advanced non-invasive nanothermometers that can provide high-resolution measurements without disturbing the biological system.3,4 Within this array of technologies, employing a luminescent nanothermometer has emerged as a highly precise remote temperature measurement method, with the potential to provide detailed temperature imaging distribution within deep tissue.5,6Among the various luminescent nanoprobes studied, lanthanide-doped nanoparticles are particularly attractive due to their narrow, rich emission peaks and inherent resistance to external environmental interference.7,8 Among them, the commonly used types are ratiometric and lifetime nanothermometers. Although lifetime nanothermometers possess the unique advantage of being unaffected by tissue coverage, they exhibit slower detection speed, necessitate more expensive experimental equipment, and require the involvement of subsequent calculations. In contrast, ratiometric nanothermometers provide a self-referencing mechanism that minimizes external perturbations and offer a rapid response time, thereby attracting extensive interest.9–11 Traditionally, ratiometric nanothermometers have relied on thermally coupled energy levels,12 such as the extensively studied 2H11/2 and 4S3/2 level pairs of the Er3+ ion.13,14 However, the limited energy gap of these levels (200–2000 cm−1) restricts further improvement of their relative sensitivity.15 In contrast, nanothermometers based on nonthermally coupled energy levels offer the potential for higher sensitivity.16–18 Specifically, when doping with different activator ions, selecting two emission peaks that vary oppositely with temperature, where one peak is thermally enhanced and the other undergoes thermal quenching, facilitates the achievement of higher relative sensitivity.
What's more, it is well known that the near-infrared (NIR) biological windows can be divided into NIR-I (650–950 nm) and NIR-II (1000–1700 nm). The weaker light scattering and molecular absorption effects within these windows help achieve deeper penetration depth, improve the accuracy of temperature measurements, and enhance imaging resolution.19,20 In particular, detection windows with wavelengths exceeding 1400 nm have been shown to achieve superior signal-to-noise ratios and deeper tissue penetration.21–25 Notably, Skripka et al. selected emission wavelengths of 1340 nm and 1550 nm, achieving a maximum relative sensitivity of 1.1% °C−1.26 Moreover, Maciejewska et al. synthesized LaPO4:1%Nd3+,20%Er3+ nanocrystals as ratiometric nanothermometers, employing the ratio of luminescence intensities at 1055 nm and 1540 nm and achieving a maximum relative sensitivity of 1.15% K−1.27 Despite these advances, strategies for the improvement of the relative sensitivity of nanothermometers operating in the NIR-II window remain elusive.28–30
In this work, we synthesized NaYF4:Er3+/Ce3+@NaYbF4@NaYF4:Nd3+/Yb3+ nanoparticles (abbreviated as NPs), whose emission peaks in the NIR-II region exhibit opposite responses to temperature under 808 nm laser excitation. As the temperature increases, the emission peak of Er3+ (1565 nm) exhibits thermal enhancement, whereas the emission peak of Nd3+ (1330 nm) undergoes thermal quenching. Based on these two emission peaks, a ratiometric nanothermometer was constructed, and its relative sensitivity was further optimized by adjusting the doping ratios of Nd3+ and Er3+. The NPs were further surface-modified with polyacrylic acid (PAA) to improve biocompatibility and were successfully employed to monitor temperature changes in a mouse model of subcutaneous inflammation. The ratiometric nanothermometer, exhibiting bidirectional temperature-dependent emission in the NIR-II region, shows great potential for monitoring temperature changes in vivo.
Results and discussion
NaYF4:Er3+/Ce3+@NaYbF4@NaYF4:Nd3+/Yb3+ was synthesized via a seed-mediated layer-by-layer method using high-temperature thermal decomposition of metal trifluoroacetates.31,32 The transmission electron microscopy (TEM) image of NaYF4:Er3+/Ce3+ (core) was obtained, confirming the successful synthesis of homogeneous spherical cores (6.9 nm) (Fig. 1a and d). Subsequently, the cores were coated with an NaYbF4 shell layer of about 4.5 nm thickness as an energy transport layer, followed by an outermost NaYF4:Nd3+/Yb3+ shell layer (about 6.5 nm in thickness) functioning as both a sensitizer layer and an activator layer (Fig. 1b, c, e and f). Both shell layers were applied using the epitaxial growth method. The measured X-ray powder diffraction (XRD) pattern compared with the standard card also proved the successful synthesis of NaYF4, which exhibited a hexagonal crystal phase (Fig. S1†). Next, to enhance the biocompatibility of the NPs, we modified the nanocrystal surfaces with PAA. This modification was confirmed using Fourier transform infrared (FTIR) spectroscopy (Fig. S2†). Initially, the surface coated with oleic acid exhibited the asymmetric and symmetric bending modes of the carboxylic group at 1465 and 1560 cm−1, along with the asymmetrical and symmetrical stretching modes of the methylene group at 2855 and 2926 cm−1. After coating with PAA, these peaks were replaced by the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O stretching mode at 1656 cm−1.33 Additionally, to evaluate the stability of the NPs in a biological environment, we dispersed the PAA-modified NPs in phosphate-buffered saline (PBS) and measured their hydrodynamic diameter using dynamic light scattering (DLS). After being stored for half a month, the DLS measurement was repeated, and no significant change in the hydrodynamic diameter was observed (Fig. S3†). Moreover, the solution remained clear without any visible aggregation, demonstrating that the NPs can remain stable in PBS. Therefore, all subsequent tests were conducted in an aqueous system.
O stretching mode at 1656 cm−1.33 Additionally, to evaluate the stability of the NPs in a biological environment, we dispersed the PAA-modified NPs in phosphate-buffered saline (PBS) and measured their hydrodynamic diameter using dynamic light scattering (DLS). After being stored for half a month, the DLS measurement was repeated, and no significant change in the hydrodynamic diameter was observed (Fig. S3†). Moreover, the solution remained clear without any visible aggregation, demonstrating that the NPs can remain stable in PBS. Therefore, all subsequent tests were conducted in an aqueous system.
For the study of luminescence performance, we first tested the absorption spectrum of NPs, NPs have a strong absorption peak at 800 nm (Fig. 2a). Next, when Nd3+ is excited by an 808 nm laser, the electrons first transition from the ground state (4I9/2) to the excited state (4F5/2). They then undergo a non-radiative relaxation process to the 4F3/2 energy level (Fig. 1g). From there, it can emit fluorescence around 1330 nm through radiative relaxation to the 4I13/2 level, or transfer energy to 2F5/2 (Yb3+) through phonon-assisted processes. Then, Yb3+ acts as a bridging ion facilitating energy transfer between the two shell regions. Then, energy migrates through the transfer layer to 4I11/2 (Er3+), followed by a non-radiative resonance energy transfer to the 4I13/2 level. Finally, 4I13/2 (Er3+) emits fluorescence at 1565 nm. In this structure, we chose the fluoride matrix due to its low phonon energy, which reduces the non-radiative relaxation process to maximize the luminescence intensity. In addition, we explored the impact of the presence of an energy migration layer on the emission peak. It can be observed that the energy migration layer facilitates energy transfer from Nd3+ to Er3+via Yb3+ ions, significantly enhancing the relative intensity of Er3+ (Fig. 2b). What's more, we measured the down-conversion quantum yield of the NPs under 808 nm laser excitation, which was determined to be 7.71% (scattering range: 770–840 nm; emission range: 850–1600 nm).
In order to evaluate the thermometric performance of the NPs as a ratiometric nanothermometer, we investigated the relationship between 1330 nm (Nd3+) and 1565 nm (Er3+) with respect to temperature (R = I1330 nm/I1565 nm). The corresponding temperature-dependent spectra of the nanothermometer are shown in Fig. 2c. With the increase of temperature, the emission at 1330 nm (Nd3+) gradually decreases, while the emission at 1565 nm (Er3+) gradually increases. Therefore, the R value exhibits a gradual decline as the temperature rises (Fig. 2d). This is because the phonon-assisted energy transfer process from Nd3+ (4F3/2) to Yb3+ (2F5/2) increases with increasing temperature, resulting in the decrease of 1330 nm (Nd3+) fluorescence.26,34,35 Moreover, the thermal enhancement of Er3+ emission arises from the increased vibrational frequency of –OH groups at elevated temperatures, which improves energy matching with the 4I11/2 → 4I13/2 (Er3+) transition, while reducing coupling with the 4I13/2 → 4I15/2 (Er3+) transition.26,36 This mechanism is corroborated by the observed prolongation of Er3+ emission lifetime with increasing temperature (Fig. S4†). What's more, many studies have shown that the doping of Ce3+ is conducive to the enhancement of Er3+ 1565 nm down-conversion emission.37,38 This is attributed to the occurrence of this process: 4I11/2 (Er3+) + 2F5/2 (Ce3+) → 4I13/2 (Er3+) + 2F7/2 (Ce3+). The increasing temperature will also further promote this process, thus promoting the thermal enhancement effect of 1565 nm (Er3+). Here, we compared the temperature-dependent 1565 nm lifetimes of Er3+ in nanocrystals with and without Ce3+ doping (Fig. S4†). We observed a more pronounced increase in lifetime with Ce3+ doping, suggesting that the thermal enhancement at 1565 nm arises from the combined effects of changes in the –OH vibrational frequency and Ce3+-induced cross-relaxation processes. In this way, the two emission peaks reverse with the change of temperature, and the ratio of the intensity of the two emission peaks can be used to construct a high-sensitivity nanothermometer.
To make a more reliable comparison, we calculated the relative sensitivity (Sr) to quantitatively evaluate the extent of R variation with temperature. The Sr is calculated using the following formula:
 | (1) |
In order to determine the optimal Nd3+ and Er3+ doping ratio for the core–shell–shell structure to obtain the maximum Sr, we synthesized a series of samples with different Nd3+ and Er3+ ion-doping ratios and measured their temperature-dependent spectra under 808 nm laser excitation (Fig. S5 and S6†). The relationship between R and temperature for each sample can be described by using the following polynomial equation (Fig. S7†):
| R = a + bT + cT2 | (2) |
Then, we fixed the Er3+ doping concentration at 5% and varied the Nd3+ doping ratios (10%, 20%, 40% and 80%). The effect on the change in the 1330 nm emission of Nd3+ and the 1565 nm emission of Er3+ is relatively small. The relative sensitivity shows a slight increase with the increase in the Nd3+ doping concentration. This is because, when the Yb3+ ion concentration is sufficiently high, the increase in the Nd3+ ion concentration may enhance the phonon-assisted energy transfer from 4F3/2 (Nd3+) to 2F5/2 (Yb3+), which facilitates the decrease in the Nd3+ emission peak intensity with increasing temperature (with a maximum decrease of 15% at the 80% Nd3+ doping ratio) (Fig. S8c†). However, this has a minimal impact on the thermal response characteristics at 1565 nm (Fig. S8d†). That is, the maximum Sr of 2.9% °C−1 at 25 °C is achieved when the Nd3+ doping concentration is optimized at 80%. However, considering the higher Sr achieved throughout the biological application range, its performance is not as good as that of the sample with an Nd3+ doping ratio of 40% (Fig. 2f). It reaches a maximum Sr of 2.5% °C−1 at 46 °C, and its Sr remains above 2.3% °C−1 throughout the physiological range (30–45 °C). Therefore, for subsequent applications, we continued to use Er3+ and Nd3+ doping ratios of 5% and 40%, respectively. Here, the Sr of the NPs is also compared with that of other nanothermometers operating in the NIR-II regions (Fig. 3).26,29,39–42 The NPs have long emission wavelengths and high relative sensitivity values, providing unique advantages for in vivo temperature sensing.
For the upcoming in vivo applications, we systematically evaluated the potential effects of probe concentration, environmental pH fluctuations, irradiation duration, and excitation power density variations on the temperature measurement parameter R. As shown in Fig. 4a and b, changing the probe concentration (0.1–1.6 mg mL−1) and adjusting the solution pH from 5 to 10 had no effect on the R value. Similarly, continuously irradiating the solution with an 808 nm laser for 30 minutes with measurements taken every 5 minutes, as well as varying the excitation power density, did not affect the R value (Fig. 4c and d). More importantly, the stability of the NPs was demonstrated through eight heating and cooling cycles (Fig. 4e). The temperature uncertainty of the NPs was assessed through 20 consecutive tests (Fig. 4f). Using the following temperature uncertainty formula, the obtained temperature uncertainty was 0.15 °C:43
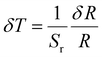 | (3) |
Before applying the NPs to the mouse model, we conducted a methyl thiazolyl tetrazolium (MTT) assay on 4T1 cells. The results show that even at a relatively high concentration (640 μg mL−1), the cell viability remains greater than 80%, confirming that the NPs exhibit no significant cytotoxicity and are suitable for biological applications (Fig. S9†). For the upcoming in vivo tests, we utilized an InGaAs camera with bandpass filters (1300/1550 nm) to capture the signals. Initially, we established a calibration curve by plotting the R values against temperature for the NPs dispersed in PBS solution (500 μg mL−1) (Fig. 5b). Then, we used 100 μL of yeast solution to induce subcutaneous inflammation in a mouse, which resulted in an increase in subcutaneous temperature. Mice from another group were injected with an equal volume of normal saline as the control group. Equal amounts of PBS solution containing the NPs (500 μg mL−1) were injected into the subcutaneous sites approximately 2 mm deep on the backs of the two mice, and the mice were then left for 12 hours to allow inflammation to develop. Under 808 nm laser excitation (0.1 W cm−2), the fluorescence intensity at the 1330 nm and 1550 nm signal channels was collected using the aforementioned InGaAs camera coupled with the corresponding optical filters (Fig. 5a). Then, the corresponding R distribution image was obtained through image processing by dividing the former by the latter. Then, based on the previously obtained calibration curve, the subcutaneous temperature distribution information of the two mice was determined from the obtained R. As a result, a clear temperature difference was observed between the groups (Fig. 5c). The subcutaneous temperature in the normal mice (left side) was 36.2 °C, whereas the inflammation group (right side) showed an elevated temperature of 39.3 °C, reflecting a 3.1 °C increase. We also measured the epidermal temperature of the mice using an infrared thermal camera (Fig. S10†), and the temperatures obtained were consistent with those measured when using the NPs. This indicates that NPs can accurately detect the increase in subcutaneous temperature caused by inflammation, demonstrating their promising potential for biomedical applications.
Conclusions
In summary, we developed a nanothermometer with two emission peaks in the NIR-II region, where one peak exhibits thermal enhancement and the other undergoes thermal quenching. As a result, it can achieve a relatively high Sr of up to 2.5% °C−1, and remains above 2.3% °C−1 throughout the entire physiological temperature range. We also demonstrated its stability and repeatability through multiple measurements and eight heating−cooling cycles between 25 and 45 °C. Furthermore, it was modified by PAA to improve its biocompatibility, and applied to a murine model of subcutaneous inflammation, demonstrating that it can effectively monitor temperature changes induced by inflammation. These results highlight the exceptional promise of Nd3+- and Er3+-doped nanocrystals with NIR-II emission peaks for delivering highly accurate temperature measurements in live mammalian systems.Experimental
Materials
Rare-earth oxides Y2O3, Nd2O3, and Yb2O3, sodium trifluoroacetate (NaTFA), and trifluoroacetic acid (TFA) were purchased from Aladdin Chemistry Co. Ltd. Oleic acid (OA, >90%), oleylamine (OM, >70%), and 1-octadecene (ODE, >90%) were obtained from Sigma-Aldrich Co. Ltd. Hexane and ethanol were purchased from Beijing Chemical Reagents, China. All materials were used as received without further purification.Synthetic procedures
Conflicts of interest
There are no conflicts to declare.Data availability
Data supporting the conclusions of this article have been included as part of the ESI.† Any additional data not included in the ESI,† which are associated with this study, are available from the authors upon reasonable request.Acknowledgements
This work was supported by grants from the National Natural Science Foundation of China (Grant No. 51972084 and 52272270), the Key Technology Research and Industrialization Demonstration Project of Qingdao (Grant No. 25-1-1-gjgg-1-gx), the Outstanding Young Scholars Project of the Natural Science Foundation of Heilongjiang Province, China (Grant No. JJ2023JQ0025), the Opening Project of State Key Laboratory of Space Power Sources (Grant No. YF07050123F2531), the Young Scientist Workshop (Harbin Institute of Technology) (Grant No. AUGA5710094420), and the Fundamental Research Funds for the Central Universities, China (Grant No. AUGA5710052614 and HIT.OCEF.2023041).References
- M. Jia, X. Chen, R. Sun, D. Wu, X. Li, Z. Shi, G. Chen and C. Shan, Lanthanide-based ratiometric luminescence nanothermometry, Nano Res., 2023, 16, 2949–2967 CrossRef CAS.
- X. Di, D. Wang, Q. P. Su, Y. Liu, J. Liao, M. Maddahfar, J. Zhou and D. Jin, Spatiotemporally mapping temperature dynamics of lysosomes and mitochondria using cascade organelle-targeting upconversion nanoparticles, Proc. Natl. Acad. Sci. U. S. A., 2022, 119, e2207402119 CrossRef CAS PubMed.
- C. D. S. Brites, R. Marin, M. Suta, A. N. Carneiro Neto, E. Ximendes, D. Jaque and L. D. Carlos, Spotlight on luminescence thermometry: basics, challenges, and cutting-edge applications, Adv. Mater., 2023, 35, 2302749 CrossRef CAS PubMed.
- Y. Shen, J. Lifante, I. Zabala-Gutierrez, M. de la Fuente-Fernández, M. Granado, N. Fernández, J. Rubio-Retama, D. Jaque, R. Marin, E. Ximendes and A. Benayas, Reliable and remote monitoring of absolute temperature during liver inflammation via luminescence-lifetime-based nanothermometry, Adv. Mater., 2022, 34, 2107764 CrossRef CAS PubMed.
- L. Pu, P. Li, J. Zhao, Y. Wang, D. Guo, L. Li, Z. Wang and H. Suo, Eu3+-activated single-band ratiometric nanothermometry by lattice negative thermal expansion, Laser Photonics Rev., 2023, 17, 2200884 CrossRef CAS.
- H. D. A. Santos, E. C. Ximendes, M. C. Iglesias-de la Cruz, I. Chaves-Coira, B. del Rosal, C. Jacinto, L. Monge, I. Rubia-Rodríguez, D. Ortega, S. Mateos, J. García Solé, D. Jaque and N. Fernández, In vivo early tumor detection and diagnosis by infrared luminescence transient nanothermometry, Adv. Funct. Mater., 2018, 28, 1803924 CrossRef.
- J. C. Martins, A. R. N. Bastos, R. A. S. Ferreira, X. Wang, G. Chen and L. D. Carlos, Primary luminescent nanothermometers for temperature measurements reliability assessment, Adv. Photonics Res., 2021, 2, 2000169 CrossRef CAS.
- A. Puccini, N. Liu and E. Hemmer, Lanthanide-based nanomaterials for temperature sensing in the near-infrared spectral region: illuminating progress and challenges, Nanoscale, 2024, 16, 10975–10993 RSC.
- M. Jia, M. Li, D. Li, X. Zhang and G. Chen, Excitation-power dependence of lanthanide-based ratiometric luminescent nanothermometry, Nano Lett., 2024, 24, 15450–15456 CrossRef CAS PubMed.
- S. Yu, J. Xu, X. Shang, W. Zheng, P. Huang, R. Li, D. Tu and X. Chen, A dual-excitation decoding strategy based on NIR hybrid nanocomposites for high-accuracy thermal sensing, Adv. Sci., 2020, 7, 2001589 CrossRef CAS PubMed.
- X. Qiu, Q. Zhou, X. Zhu, Z. Wu, W. Feng and F. Li, Ratiometric upconversion nanothermometry with dual emission at the same wavelength decoded via a time-resolved technique, Nat. Commun., 2020, 11, 4 CrossRef CAS PubMed.
- H. Huang, H. Tuxun, K. Zhong, J. Huang, H. Wei and B. Zhou, Enabling nonthermally coupled upconversion in a core–shell–shell nanoparticle for ultrasensitive nanothermometry and anticounterfeiting, ACS Appl. Nano Mater., 2024, 7, 7794–7801 CrossRef CAS.
- R. An, Y. Liang, R. Deng, P. Lei and H. Zhang, Hollow nanoparticles synthesized via Ostwald ripening and their upconversion luminescence-mediated Boltzmann thermometry over a wide temperature range, Light: Sci. Appl., 2022, 11, 217 CrossRef CAS PubMed.
- F. Vetrone, R. Naccache, A. Zamarrón, A. Juarranz de la Fuente, F. Sanz-Rodríguez, L. Martinez Maestro, E. Martín Rodriguez, D. Jaque, J. García Solé and J. A. Capobianco, Temperature sensing using fluorescent nanothermometers, ACS Nano, 2010, 4, 3254–3258 CrossRef CAS PubMed.
- Y. Wang, L. Lei, E. Liu, Y. Cheng and S. Xu, Constructing highly sensitive ratiometric nanothermometers based on indirectly thermally coupled levels, Chem. Commun., 2021, 57, 9092–9095 RSC.
- C. Mi, J. Zhou, F. Wang, G. Lin and D. Jin, Ultrasensitive ratiometric nanothermometer with large dynamic range and photostability, Chem. Mater., 2019, 31, 9480–9487 CrossRef CAS.
- L. Yan, J. Huang, Z. An, Q. Zhang and B. Zhou, Activating ultrahigh thermoresponsive upconversion in an erbium sublattice for nanothermometry and information security, Nano Lett., 2022, 22, 7042–7048 CrossRef CAS PubMed.
- D. Li, M. Jia, T. Jia and G. Chen, Ultrasensitive NIR-II ratiometric nanothermometers for 3D in vivo thermal imaging, Adv. Mater., 2024, 36, 2309452 CrossRef CAS PubMed.
- B. del Rosal, E. Carrasco, F. Ren, A. Benayas, F. Vetrone, F. Sanz-Rodríguez, D. Ma, Á. Juarranz and D. Jaque, Infrared-emitting QDs for ehermal eherapy with real-time subcutaneous temperature feedback, Adv. Funct. Mater., 2016, 26, 6060–6068 CrossRef CAS.
- M. Quintanilla, M. Henriksen-Lacey, C. Renero-Lecuna and L. M. Liz-Marzán, Challenges for optical nanothermometry in biological environments, Chem. Soc. Rev., 2022, 51, 4223–4242 RSC.
- C. Mi, X. Zhang, C. Yang, J. Wu, X. Chen, C. Ma, S. Wu, Z. Yang, P. Qiao, Y. Liu, W. Wu, Z. Guo, J. Liao, J. Zhou, M. Guan, C. Liang, C. Liu and D. Jin, Bone disease imaging through the near-infrared-II window, Nat. Commun., 2023, 14, 6287 CrossRef CAS PubMed.
- Y. Zhong, Z. Ma, F. Wang, X. Wang, Y. Yang, Y. Liu, X. Zhao, J. Li, H. Du, M. Zhang, Q. Cui, S. Zhu, Q. Sun, H. Wan, Y. Tian, Q. Liu, W. Wang, K. C. Garcia and H. Dai, In vivo molecular imaging for immunotherapy using ultra-bright near-infrared-IIb rare-earth nanoparticles, Nat. Biotechnol., 2019, 37, 1322–1331 CrossRef CAS PubMed.
- M. Zhang, J. Yue, R. Cui, Z. Ma, H. Wan, F. Wang, S. Zhu, Y. Zhou, Y. Kuang, Y. Zhong, D.-W. Pang and H. Dai, Bright quantum dots emitting at ∼1,600 nm in the NIR-IIb window for deep tissue fluorescence imaging, Proc. Natl. Acad. Sci. U. S. A., 2018, 115, 6590–6595 CrossRef CAS PubMed.
- H. D. A. Santos, I. Zabala Gutiérrez, Y. Shen, J. Lifante, E. Ximendes, M. Laurenti, D. Méndez-González, S. Melle, O. G. Calderón, E. López Cabarcos, N. Fernández, I. Chaves-Coira, D. Lucena-Agell, L. Monge, M. D. Mackenzie, J. Marqués-Hueso, C. M. S. Jones, C. Jacinto, B. del Rosal, A. K. Kar, J. Rubio-Retama and D. Jaque, Ultrafast photochemistry produces superbright short-wave infrared dots for low-dose in vivo imaging, Nat. Commun., 2020, 11, 2933 CrossRef CAS PubMed.
- E. C. Ximendes, W. Q. Santos, U. Rocha, U. K. Kagola, F. Sanz-Rodríguez, N. Fernández, A. d. S. Gouveia-Neto, D. Bravo, A. M. Domingo, B. del Rosal, C. D. S. Brites, L. D. Carlos, D. Jaque and C. Jacinto, Unveiling in vivo subcutaneous thermal dynamics by infrared luminescent nanothermometers, Nano Lett., 2016, 16, 1695–1703 CrossRef CAS PubMed.
- A. Skripka, A. Benayas, R. Marin, P. Canton, E. Hemmer and F. Vetrone, Double rare-earth nanothermometer in aqueous media: opening the third optical transparency window to temperature sensing, Nanoscale, 2017, 9, 3079–3085 RSC.
- K. Maciejewska, A. Bednarkiewicz and L. Marciniak, The influence of the Er3+ dopant concentration in LaPO4:Nd3+, Er3+ on thermometric properties of ratiometric and kinetic-based luminescent thermometers operating in NIR II and NIR III optical windows, Phys. B, 2021, 620, 413247 CrossRef CAS.
- Y. Wu, F. Li, Y. Wu, H. Wang, L. Gu, J. Zhang, Y. Qi, L. Meng, N. Kong, Y. Chai, Q. Hu, Z. Xing, W. Ren, F. Li and X. Zhu, Lanthanide luminescence nanothermometer with working wavelength beyond 1500 nm for cerebrovascular temperature imaging in vivo, Nat. Commun., 2024, 15, 2341 CrossRef CAS PubMed.
- S. Liu, Z. An, J. Huang and B. Zhou, Enabling efficient NIR-II luminescence in lithium-sublattice core—shell nanocrystals towards Stark sublevel based nanothermometry, Nano Res., 2023, 16, 1626–1633 CrossRef CAS.
- E. C. Ximendes, U. Rocha, T. O. Sales, N. Fernández, F. Sanz-Rodríguez, I. R. Martín, C. Jacinto and D. Jaque, In vivo subcutaneous thermal video recording by supersensitive infrared nanothermometers, Adv. Funct. Mater., 2017, 27, 1702249 CrossRef.
- H. Li, M. Tan, X. Wang, F. Li, Y. Zhang, L. Zhao, C. Yang and G. Chen, Temporal multiplexed in vivo upconversion imaging, J. Am. Chem. Soc., 2020, 142, 2023–2030 CrossRef CAS PubMed.
- L. Wu, M. Jia, D. Li and G. Chen, Shell engineering on thermal sensitivity of lifetime-based NIR nanothermometers for accurate temperature measurement in murine internal liver organ, Nano Lett., 2023, 23, 2862–2869 CrossRef CAS PubMed.
- D. Li, M. Jia, J. Du, Y. Li, T. Jia and G. Chen, Optomagnetic heater-thermometer nanoplatform for tumor magnetothermal therapy with operando temperature feedback, ACS Nano, 2025, 19, 8328–8337 CrossRef CAS PubMed.
- M. Kong, Y. Gu, Y. Chai, J. Ke, Y. Liu, X. Xu, Z. Li, W. Feng and F. Li, Luminescence interference-free lifetime nanothermometry pinpoints in vivo temperature, Sci. China: Chem., 2021, 64, 974–984 CrossRef CAS.
- Ł. Marciniak, A. Bednarkiewicz, M. Stefanski, R. Tomala, D. Hreniak and W. Strek, Near infrared absorbing near infrared emitting highly-sensitive luminescent nanothermometer based on Nd3+ to Yb3+ energy transfer, Phys. Chem. Chem. Phys., 2015, 17, 24315–24321 RSC.
- Y. Huang, J. Hu, Y. Guo, Z. Wang, F. Lin and H. Zhu, Effect of –OH on the thermal enhancement properties of NIR-II lanthanide-doped nanoparticles in water, Inorg. Chem. Front., 2024, 11, 3063–3072 RSC.
- Y. Sun, M. Kong, J. Ke, W. Yuan, C. Wen, Y. Gu, S. Luo and W. Feng, Nanothermometer for In Vivo Temperature detection with high spatial resolution based on core–shell rare earth nanoparticles, ACS Appl. Nano Mater., 2023, 6, 23173–23183 CrossRef CAS.
- C. Li, J. Ge, D. Song, T. Liang and Z. Li, Activatable near-infrared IIb rare-earth nanoprobe for early monitoring chemotherapy-induced liver injury, Adv. Funct. Mater., 2025, 2502077 CrossRef.
- H. D. A. Santos, D. Ruiz, G. Lifante, C. Jacinto, B. H. Juarez and D. Jaque, Time resolved spectroscopy of infrared emitting Ag2S nanocrystals for subcutaneous thermometry, Nanoscale, 2017, 9, 2505–2513 RSC.
- J. Zhao, J. Chang, N. Wang, P. Zhao, M. Zhu, Y. Liu, D. Guo, Y. Wang, P. Li, Z. Wang and H. Suo, Thermal enhancement of Er3+ NIR-II luminescence by Ho3+-mediated energy-trapping in negative thermal expansion nanocrystals, Laser Photonics Rev., 2024, 18, 2400151 CrossRef CAS.
- Q. Hu, N. Kong, Y. Chai, Z. Xing, Y. Wu, J. Zhang, F. Li and X. Zhu, A lanthanide nanocomposite with cross-relaxation enhanced near-infrared emissions as a ratiometric nanothermometer, Nanoscale Horiz., 2022, 7, 1177–1185 RSC.
- X. Yuan, E. Cui, K. Liu, Y. Jiang, X. Yang, J. Tang, L. Yang, X. Liao, Y. Zhao, W. Sun, Y. Liu and J. Liu, Lanthanide-doped NaYF4 near-infrared-II nanothermometers for deep tissue temperature sensing, Ceram. Int., 2022, 48, 35141–35149 CrossRef CAS.
- P. Cortelletti, A. Skripka, C. Facciotti, M. Pedroni, G. Caputo, N. Pinna, M. Quintanilla, A. Benayas, F. Vetrone and A. Speghini, Tuning the sensitivity of lanthanide-activated NIR nanothermometers in the biological windows, Nanoscale, 2018, 10, 2568–2576 RSC.
Footnote |
| † Electronic supplementary information (ESI) available: Characterization, XRD patterns, FT-IR spectra, temperature measurement parameters and fitting parameters and temperature-dependent spectra. See DOI: https://doi.org/10.1039/d5qi01027f |
| This journal is © the Partner Organisations 2025 |





