Recent development review of Ti3C2Tx MXene-based microsupercapacitors: a multi-dimensional analysis spanning from underlying mechanisms to integrated applications
Chuqiao
Hu
a,
Yumeng
Bai
a,
Wei
Wang
b,
Peilun
Qiu
a,
Di
Wu
a,
Jianqiao
Liu
 *a,
Ce
Fu
*a and
Guozhen
Shen
*a,
Ce
Fu
*a and
Guozhen
Shen
 *c
*c
aCollege of Information Science and Technology, Dalian Maritime University, Dlian 116026, China. E-mail: jqliu@dlmu.edu.cn; fu_ce@dlmu.edu.cn
bCollege of International Collaboration, Dalian Maritime University, Dlian 116026, China
cSchool of Integrated Circuits and Electronics, Beijing Institute of Technology, Beijing 100081, China. E-mail: gzshen@bit.edu.cn
First published on 14th April 2025
Abstract
Microsupercapacitors, as innovative energy storage devices, have garnered significant attention in microelectronics and wearable applications. The urgent demand to enhance the charge storage capacity and operational efficiency of MSCs, coupled with challenges such as limited charge-active sites and low ion transport efficiency, has driven continuous optimization of their constituent materials and structural designs. The emerging two-dimensional MXenes, with their rich surface chemical functionalities, tunable interlayer spacing, and excellent compatibility with various nanomaterials, offer unprecedented opportunities for MSCs. However, current analyses of how MXenes enhance the performance of MSCs from the energy storage mechanism perspective, their multiple applications in MSCs, and the system-level integration of MSCs are incomplete, limiting the development of this field. Herein, this review presents a comprehensive overview of the latest advancements in the energy storage mechanisms and fabrication techniques of Ti3C2Tx-based MSCs, especially emphasizes their applications across various components of MSCs, and provides a detailed summary of integrated examples of MSC-powered systems. This work offers an in-depth analysis of the key role of Ti3C2Tx in enhancing the performance of MSCs, as well as outlines the challenges and prospects for its forthcoming research, with the potential to drive continued innovation in high-performance flexible energy storage devices.
Wider impactTi3C2Tx MXene, with its tunable interlayer spacing and abundant surface functional groups, exhibits metallic-like conductivity and excellent mechanical stability, demonstrating immense potential in the energy storage field, particularly in microsupercapacitors (MSCs). Its unique two-dimensional structure and physicochemical properties lay a solid foundation for the development of high-performance, miniaturized, and sustainable energy storage devices. This article systematically reviews the latest research of Ti3C2Tx MXene-based MSCs from five aspects: mechanisms, methods, materials, structures, and applications, while thoroughly discussing key strategies and approaches for performance optimization. Looking ahead, research on Ti3C2Tx-MSCs will continue to advance toward high performance, environmental friendliness, and sustainability. This review not only discusses the strategies for enhancing the electrochemical performance of MSCs in existing literature but also provides important theoretical guidance for the design and development of next-generation high-efficiency, eco-friendly, low-cost, and scalable MSCs. With their high energy storage capacity, Ti3C2Tx-MSCs are expected to fully meet the demands of microelectronic devices and wearable technologies for high energy density, long cycle life, and flexible integration, thereby driving innovation and development in next-generation smart electronics and energy storage systems. |
1. Introduction
The rapid advancement of portable electronic devices, Internet of Things (IoT) systems, and microelectronic technologies has significantly amplified the demand for compact energy storage devices that offer high performance, flexibility, and durability.1–9 Supercapacitors (SCs), as a leading class of energy storage devices, have attracted widespread attention due to their high power density, rapid charge/discharge capabilities, and long cycle life.10–13 Modern electronic devices are increasingly diverse, requiring components to have high integration, flexibility, and shape adaptability. However, the relatively large volume of SCs limits their integration into miniaturized devices. To meet the demands of diverse application scenarios, microsupercapacitors (MSCs), with their smaller size and lighter weight, have effectively addressed the miniaturization limitations of traditional supercapacitors through planar structural designs and microfabrication techniques. These advancements enable seamless integration with other electronic components, as well as provide advantages and opportunities for their application in the integration of wearable electronics and micro-sensors.14–17 But for MSCs, factors such as low energy density, insufficient charge storage sites, and limited ion transport efficiency continue to hinder their application in high-performance energy storage devices.18,19 The relatively low energy density directly affects the duration of a single charge, making MSCs unsuitable for scenarios requiring long-term and stable energy output. Additionally, the insufficient charge storage sites limit the energy storage capacity of MSCs, reducing their competitiveness in high-capacity energy storage applications. Meanwhile, the insufficient ion transport efficiency hampers the charging and discharging speed of MSCs, adversely affecting their performance in fast-response applications.The adoption of advanced materials offers a new pathway to overcoming these challenges. As a novel family of two-dimensional transition metal carbides, nitrides, and carbonitrides, MXenes are represented by the general formula Mn+1XnTx (n = 1–4), where M denotes an early transition metal, X represents carbon and/or nitrogen, and Tx refers to surface functional groups such as –OH, –O, or –F.20,21 Among them, the –OH and –O functional groups endow MXenes with strong hydrophilicity, enhancing their dispersion in aqueous solutions and making them suitable for applications in water-based electrolytes for microsupercapacitors. Additionally, the surface charge of MXenes, influenced by the type of functional group, facilitates interactions with oppositely charged ions or molecules. In electrochemical energy storage devices, this surface charge property contributes to higher conductivity and improved electrochemical stability when they are used as electrode materials. MXenes are obtained by selectively etching their precursor MAX phases. The structural and elemental composition of MAX phases is illustrated in Fig. 1(a) and (b).22 Among MXenes, Ti3C2Tx, the most widely studied MXene,23 exhibits tremendous potential in enhancing the energy storage capacity and energy density of MSCs, owing to its unique layered structure, abundant surface chemical functionalities, and outstanding electrochemical properties. Its synthesis process is shown in Fig. 1(c);24 Ti3C2Tx prepared by this method has tunable interlayer spacing, which provides highly ordered ion transport pathways, enabling rapid diffusion of electrolyte ions to the electrode surface. Meanwhile, the diverse surface terminations actively participate in electrochemical reactions, significantly enhancing the activity of the electrode material and its charge storage capacity.25–27 The two synergistically work together to comprehensively enhance the energy density and power output of energy storage devices (Fig. 1(d)).28 In addition, Ti3C2Tx added with antioxidants such as N,N-dimethylformamide (DMF) in the synthesis stage has excellent oxidation resistance in water,29 which can improve the stability of the electrode, thus ensuring the long-term effectiveness of the device. To date, Ti3C2Tx has become a crucial material for improving the electrochemical performance of MSCs.
 | ||
| Fig. 1 (a) Structural diagram of MAX phases. (b) Periodic table of proper elements for MAX phases. The elements “M”, “A”, “X”, and “Tx” are marked with orange, green, purple, and yellow colors, respectively. Reproduced with permission.22 Copyright 2024, Elsevier. (c) Ti3C2Tx synthesized by H2SO4/LiF etchants. Reproduced with permission.24 Copyright 2020, American Chemical Society. (d) Ragone plot showing energy and power densities of state-of-the-art microsupercapacitors. The vertical shaded area shows the power consumption of different electronics (corresponding to the top axis). The power density in the Ragone plot can be directly used to estimate practical electronic applications, given the area of the energy storage device is 1 cm2. Reproduced with permission.28 Copyright 2020, Elsevier. | ||
In the device component design of MSCs, the material selection for each part is crucial; Ti3C2Tx demonstrates remarkable versatility, and can serve not only as an electrode, but also as a current collector or separator.30–34 Conventional electrode materials, such as graphene and carbon nanotubes, face challenges including a lack of surface functional groups, poor interfacial compatibility, limited charge storage sites, and inadequate structural stability.35,36 In contrast, Ti3C2Tx, with its abundant and highly active surface functional groups, significantly enhances the pseudocapacitive effect and improves interfacial compatibility with aqueous electrolytes. The synergistic effects of these multiple advantages collectively enhance the overall electrochemical performance of MSCs. Additionally, Ti3C2Tx possesses a large specific surface area and provides more effective charge storage sites, thereby increasing energy density. Moreover, the unique layered structure and strong bonding forces of Ti3C2Tx endow it with excellent mechanical stability. Ti3C2Tx-based MSCs have made significant progress in energy density. While challenges remain in improving the volumetric energy density and structural stability during cycling, these challenges provide important research directions for future technological innovations. At the same time, optimizing array manufacturing costs offers opportunities to further promote the widespread application of Ti3C2Tx-based MSCs. Thus, strategies such as optimizing the interlayer spacing of Ti3C2Tx sheets to enhance charge storage capacity, reinforcing structural stability through nanomaterial composites,37–40 and developing green and scalable manufacturing processes41–44 have been developed. With the continuous advancement of these improvement strategies, Ti3C2Tx MXene is expected to fully realize its potential in MSCs, driving the innovation and development of next-generation high-performance energy storage technologies.
Although many reviews have discussed the application of MXenes in energy storage, especially in the use of electrode materials, most of them focus primarily on the basic properties of the materials, structural design, and fabrication methods, lacking a systematic exploration of how to enhance the overall performance of MSCs by optimizing the properties of MXene materials. Therefore, this review covers various aspects of Ti3C2Tx-based MSCs, providing an in-depth analysis and systematic summary from the perspectives of both fundamental energy storage mechanisms and integrated applications of the devices. Firstly, we present an overview of the recent advancements in Ti3C2Tx MXene-based MSCs, summarize their performance evaluation metrics, and provide a detailed analysis of their energy storage mechanisms, highlighting their unique value in enhancing device performance. Subsequently, we categorize and discuss key fabrication techniques for MSCs, such as photolithography, screen printing, and inkjet printing, from the perspective of whether a mask template is utilized. We specifically elaborate on the critical role of Ti3C2Tx in enhancing MSC performance, focusing on its innovative applications in various components of MSCs, including current collectors, electrodes, and separators, emphasizing its multifunctionality in device architecture. Additionally, this review further underscores strategies for the composite integration of Ti3C2Tx with nanomaterials of different dimensions (such as 1D nanowires, 2D nanosheets, and 3D porous networks) to achieve synergistic enhancements in electrical conductivity, structural stability, and ion transport. Furthermore, we summarize the integrated applications of Ti3C2Tx MXene-based MSCs in power supply systems, with a focus on their integration with triboelectric nanogenerators, sensors, and other cutting-edge technologies. Finally, we systematically evaluate the potential directions for future improvements of Ti3C2Tx-based MSCs, addressing challenges such as scalability, cost-effectiveness, and material oxidation during long-term charge/discharge cycling. By comprehensively reviewing the latest research progress and unresolved issues, this work provides critical references for the development of next-generation MSCs and lays a theoretical foundation for the innovation and expansion of micro-energy storage technologies.
2. Development and performance evaluation
2.1. Development of Ti3C2Tx MXene-based MSCs
Since its inception, Ti3C2Tx MXene has experienced rapid development and, owing to its unique structure and exceptional properties, it has been widely applied in various fields, including microsupercapacitors,45–48 batteries,49–51 photodetectors,52–56 photocatalysis,57–59 sensors,60–62 electromagnetic interference shielding,63–65 and water purification66–68 (Fig. 2(a)). Particularly in MSC research, MXenes have gradually replaced other materials, emerging as the preferred subject of study (Fig. 2(b)). Among MSCs based on MXenes, Ti3C2Tx-based MSCs have attracted significant attention in recent years, achieving numerous groundbreaking advancements, with their development timeline illustrated in Fig. 2(c). In 2003, Kim45 constructed the first all-solid-state thin-film MSC using tungsten–ruthenium oxide (W–RuO2) electrodes and a LiPON electrolyte, marking the birth of MSCs. In 2011, Naguib et al.23 successfully synthesized two-dimensional Ti3C2Tx with abundant surface functional groups by etching Ti3AlC2 with HF, opening a new research frontier for MXene materials. In 2016, Gogotsi and colleagues33 innovatively applied Ti3C2Tx MXene to MSC electrodes and current collectors, utilizing spray-coating and laser-cutting techniques to fabricate the first MXene-based MSC (MX-MSC). Subsequently, due to their unique two-dimensional layered structure, high specific surface area, excellent conductivity, and rich surface functional groups, MXenes have rapidly gained traction in MSC applications, becoming a research hotspot. MXene-based MSCs, with their exceptional electrochemical performance, design flexibility, and broad process compatibility, have gradually established a dominant position in the field of micro-energy storage devices. | ||
| Fig. 2 The development of Ti3C2Tx-based MSCs. (a) Proportional distribution of Ti3C2Tx-related publications across various fields based on Web of Science data. (b) The number of publications on MSCs and Ti3C2Tx-based MSCs in recent years based on Web of Science data. (c) The development process of Ti3C2Tx-based MSCs. Reproduced with permission.45 Copyright 2003, American Institute of Physics. Reproduced with permission.23 Copyright 2011, Wiley-VCH. Reproduced with permission.33 Copyright 2016, Royal Society of Chemistry. Reproduced with permission.46 Copyright 2017, Royal Society of Chemistry. Reproduced with permission.47 Copyright 2018, IOP Publishing. Reproduced with permission.48 Copyright 2018, Wiley-VCH. Reproduced with permission.69 Copyright 2018, American Chemical Society. Reproduced with permission.70 Copyright 2018, Elsevier. Reproduced with permission.71 Copyright 2018, Elsevier. Reproduced with permission.72 Copyright 2019, Royal Society of Chemistry. Reproduced with permission.73 Copyright 2019, Wiley-VCH. Reproduced with permission.74 Copyright 2022, Wiley-VCH. Reproduced with permission.75 Copyright 2023, Wiley-VCH. | ||
The rapid development of advanced fabrication techniques has significantly enhanced the performance of MSCs. In 2017, Hu46 introduced laser-printed masks and vacuum filtration processes to fabricate finely patterned interdigitated MSCs, advancing the progress of high-performance micro-energy storage devices. By 2018, the fabrication methods for Ti3C2Tx-MSCs had become increasingly diverse and efficient. Li et al.47 employed a simple, low-cost scraping method to construct patterned MSC electrodes with a layered structure. By integrating these devices in series or parallel configurations, they achieved higher energy output, laying the groundwork for self-integrated devices. Additionally, Gogotsi and colleagues48 developed an automated scalpel-engraving technique, successfully manufacturing transparent and semi-transparent MSCs. In the same year, Gogotsi and his team69 conducted an in-depth investigation into the rheological behavior and viscosity properties of MXene solutions, experimentally demonstrating the feasibility of techniques such as inkjet printing, spin coating, and 3D extrusion printing in the fabrication of Ti3C2Tx-MSCs.
Alongside the continuous innovation in MSC fabrication techniques, MX-MSCs have also made remarkable progress in integrated applications. Xu et al.70 proposed a scalable two-step printing process to fabricate coplanar asymmetric MSCs, which were integrated with sensors for the first time. This not only optimized the device performance but also expanded its application fields. Furthermore, addressing the storage capacity limitations of MSCs, Jiang et al.71 designed a highly compact self-powered charging unit by combining MSCs with triboelectric nanogenerators, achieving integrated functionality for energy harvesting and storage. In 2019, Bao and colleagues72 applied the complex but highly precise photolithography technique to MX-MSC fabrication, significantly improving the structural precision and performance of the devices, marking a maturity in fabrication technologies. In the same year, K. Hota73 achieved wafer-scale fabrication of MSCs and integrated them with thin-film transistors and rectifiers, realizing the first fusion of Ti3C2Tx-MSCs with thin-film electronic devices, thereby paving the way for compact on-chip micro-power units. In recent years, with deeper exploration of Ti3C2Tx-MSCs in structural optimization and multifunctional integration, the practicality and application potential of these devices have been further enhanced. In 2022, Su et al.74 highlighted the impact of separator design on MSC performance and utilized the porous structure of Ti3C2Tx to optimize ion transport pathways, successfully fabricating MSCs with extended cycling lifespans. In 2023, Shen and his team75 integrated MSCs with photodetectors to construct a seamless all-MXene-based optoelectronic system, showcasing the immense application potential and development prospects of Ti3C2Tx-MSCs in multifunctional integrated systems.
2.2. Performance evaluation indicators
In the study of Ti3C2Tx-based MSCs, performance evaluation is a critical factor for assessing the energy storage capability and practical application potential of the devices. Compared to SCs, whose performance is evaluated based on the mass of active materials, the mass of active materials in MSCs is negligible, making mass-based performance comparisons less applicable. Therefore, substrate area and volumetric metrics play a more critical role in the performance evaluation of MSCs. Volumetric capacitance (CV, F cm−3), volumetric energy density (EV, W h cm−3), and volumetric power density (PV, W cm−3) are evaluated based on the thickness of the electrode. Based on the area, performance parameters such as areal capacitance (CA, F cm−2), areal energy density (EA, W h cm−2), and areal power density (PA, W cm−2) can also be calculated using cyclic voltammetry (CV) curves. Volumetric metrics (CV, EV, PV) are suitable for evaluating the performance of MSCs in miniaturized and compact energy storage devices, emphasizing their volumetric utilization efficiency. In contrast, areal metrics (CA, EA, PA) are more appropriate for assessing the performance of thin-film and flexible electronic devices, highlighting the energy storage capacity and power output characteristics of the devices within a limited area. The corresponding formulas are as follows: | (1) |
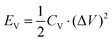 | (2) |
 | (3) |
 | (4) |
 | (5) |
 | (6) |
In addition, the efficiency (η) and cycling stability of MSCs are also critical parameters for evaluating their performance. Efficiency reflects the energy conversion capability and the degree of energy loss during the charge and discharge processes of MSCs, and cycling stability characterizes their ability to maintain electrochemical performance over prolonged and repeated cycling. High efficiency and excellent cycling stability are essential for energy storage devices to achieve high energy utilization, extended operational time, and reliable functionality in practical applications, particularly significant in scenarios requiring frequent charge and discharge, such as wireless sensors and wearable devices. The formulas are as follows:
 | (7) |
 | (8) |
 | (9) |
 | (10) |
3. Energy storage mechanism
Microsupercapacitors consist of two electrodes (anode and cathode), current collectors, a separator, and electrolyte. The synergistic interaction of these components determines the energy storage capability, which in turn influences the overall performance of the MSCs. Among these components, the electrodes serve as the core of the energy storage process by providing active sites for charge storage and transfer. The current collectors are responsible for efficiently conducting charges and enhancing the mechanical stability of devices. To prevent short circuits, separators isolate the positive and negative electrodes while facilitating ion transport channels. Meanwhile, electrolytes act as the medium for ion migration, directly influencing the conductivity and electrochemical window of MSCs.Based on differences in energy storage mechanisms, MSCs can be categorized into two major types: one is electric double-layer capacitors (EDLCs), which store energy through electrostatic adsorption and desorption, and the other is pseudocapacitors, which store charge via redox reactions and ion intercalation processes. In addition, hybrid microsupercapacitors (HMSCs) combine the advantages of both types, achieving enhanced energy density while maintaining high power density. Typically, cyclic voltammetry (CV) curves, galvanostatic charge–discharge (GCD) curves, and electrochemical impedance spectroscopy (EIS) curves are key characterization techniques for analyzing the energy storage mechanisms of MSCs. This section will provide a detailed discussion of these three types of MSCs from the perspective of energy storage mechanisms, along with their applications in optimizing MSC performance.
3.1. Electric double-layer capacitor
The core energy storage mechanisms of electric double-layer capacitors42,75–81 rely on the formation of electric double-layer structures at the electrode–electrolyte interfaces, driven by the electrostatic adsorption of ions. During the charging process, the applied electric field induces the generation of positive or negative charges on the electrode surface, attracting oppositely charged ions from the electrolyte. These ions arrange into a tightly packed double-layer structure at the interface, enabling energy storage. After the charging process, the electric double-layer structure remains stable under the influence of electrostatic attraction. During the discharging process, electrons migrate from the negative electrode to the positive electrode via an external circuit. Simultaneously, ions in the electrolyte detach from the electrode surface and diffuse back to equilibrium positions, completing the release of stored energy. This reversible and rapid process of ion adsorption and desorption ensures the long-term cycling stability and fast charge–discharge capability of EDLCs.Due to the defect sites in Ti3C2Tx providing additional electroactive sites for ion adsorption, which helps enhance the capacitance performance of the devices, Huang et al.75 designed a Ti3C2Tx-based all-solid-state flexible microsupercapacitor (FMSC) with an electric double-layer effect in a polyvinyl alcohol (PVA)/sulfuric acid (H2SO4) gel electrolyte environment (Fig. 3(a)). Fig. 3(b) illustrates the process of charge storage, where H+ and SO42− ions in the electrolyte adsorb onto the positive and negative electrodes, respectively, forming electric double layers at different electrode spacings. The ion adsorption and desorption capability can be assessed by the shape of the CV curve, as shown in Fig. 3(c). The FMSC exhibits an almost ideal symmetric rectangular CV curve, indicating that the energy storage process of the device is both fast and efficient. In addition, as the electrode spacing of the FMSC decreases (from 0.7 mm to 0.2 mm), the ion transport path during the adsorption process shortens, resulting in a reduction in transport impedance and a significant decrease in charge and discharge time, enhancing the rate performance of the device, as shown in Fig. 3(d). Moreover, the FMSC demonstrated excellent stability in long-term cycling tests, as shown in Fig. 3(e), indicating that the ion adsorption and desorption processes exhibit good reversibility.
 | ||
| Fig. 3 Electric double-layer energy storage mechanism. (a) Digital photograph of paper-based FMSCs with the finger width ranging from 0.5 to 2.5 mm. A Chinese coin was placed at the right side for size comparison. (b) Schematic diagram of the ion transport mechanism of paper-based FMSCs with different interspaces. (c) CV curves at a scan rate of 10 mV s−1 of various FMSCs. (d) GCD curves of the FMSCs at a current density of 0.1 mA cm−2. (e) The cycling performance of the paper-based FMSCs. Reproduced with permission.75 Copyright 2019, Elsevier. (f) The digital photograph of the as-fabricated flexible on-chip MSC array. (g) CV curves measured at various scan rates ranging from 20 to 500 mV s−1. (h) GCD curves obtained at current densities from 0.1 to 2.0 mA cm−2. Reproduced with permission.76 Copyright 2020, Elsevier. | ||
In similar studies that utilize the electric double-layer energy storage principle, Li et al.76 constructed an all-Ti3C2Tx MXene-based flexible microsupercapacitor array using advanced fabrication techniques, with the optical image shown in Fig. 3(f). The CV curve in Fig. 3(g), which shows no obvious redox peaks, indicates that the electrode primarily stores charge through the electric double-layer capacitance mechanism. Meanwhile, the typical triangular shape of the GCD curve in Fig. 3(h) further confirms that the charge storage capability originates from the efficient ion adsorption behavior at the electrolyte–electrode interface. In addition, Duan74 and Deshmukh78 also developed Ti3C2Tx-based MSCs with electric double-layer capacitance behavior. Owing to the rapid and reversible electrostatic ion adsorption and desorption processes, the devices exhibited extended energy storage capabilities.
3.2. Pseudocapacitor
Unlike EDLCs, the energy storage mechanism of pseudocapacitors primarily relies on rapid redox reactions82–86 at the electrode surface or within the electrode, as well as ion intercalation reactions.72,87–89 Although the electric double-layer effect is also present, it plays only a minor supporting role. As the pseudocapacitive electrode, 2D Ti3C2Tx MXene, with its abundant surface active sites, can chemically react with ions in the electrolyte, thereby enhancing the charge storage capacity of the device. Additionally, the Ti3C2Tx electrode has a large specific surface area, enabling charge storage within the crystal structure of the electrode through ion intercalation. Benefiting from this, the pseudocapacitor offers higher energy density. Moreover, both the redox reactions and ion intercalation reactions exhibit good reversibility, imparting excellent cycling stability to the MSC.Due to the ability of redox reactions to provide higher energy density, Cao et al.82 developed a symmetric zinc-ion microsupercapacitor (ZIMSC) based on a redox energy storage mechanism, using Ti3C2Tx MXene/bacterial cellulose fiber (BCF) hybrid films as electrodes (Fig. 4(a)). During charging, Zn2+ deeply infiltrate the interlayer spaces of the Ti3C2Tx-based electrode and undergo redox reactions with –O groups. Simultaneously, the BCF reacts with H2SO4 in the electrolyte to complete the energy storage process. The discharge process is the reverse of this mechanism (Fig. 4(b)). Fig. 4(c) and (d) show the CV curves and GCD curves of ZIMSCs with different electrodes, respectively. The typical rectangular and isosceles triangular shapes indicate that the addition of BCF facilitates redox reaction, enabling the device to exhibit higher areal capacitance, a broader operating voltage window, and faster charge–discharge response. This fully demonstrates the significant role of redox reactions in enhancing energy density. Furthermore, as the mass ratio of BCF increases, the MSC exhibits greater areal capacitance, confirming that the addition of BCF enhances redox reaction (Fig. 4(e)). Meanwhile, compared to pure Ti3C2Tx film electrodes, the Ti3C2Tx/BCF composite electrodes display superior cycling stability (Fig. 4(f)), indicating that the redox reaction exhibits excellent reversibility. By integrating Ti3C2Tx, MnO2 nanowires (MnONWs), and Ag nanowires (AgNWs), Li et al.84 designed a 3D-printed flexible microsupercapacitor and systematically investigated its faradaic charge storage properties and mechanical stability. As shown in Fig. 4(g), the nearly rectangular CV curves at various scan rates indicate that the device exhibits excellent redox capacitive behavior during the charge–discharge process. The GCD curves in Fig. 4(h) exhibit an asymmetrical triangular shape, further confirming the presence of a slow faradaic redox process within the nanocomposite electrodes.
 | ||
| Fig. 4 Redox reaction energy storage mechanism. (a) MD simulation model of Ti3C2Tx/BCF hybrid electrodes in the Zn(CF3SO3)2 electrolyte. A static electric field along the Z-direction was set to simulate the charging process. (b) A schematic representation of Ti3C2Tx/BCF serving as symmetrical negative and positive electrodes in ZMSCs. (c) CV curves and (d) GCD curves of the as-fabricated ZMSCs employing Ti3C2Tx/BCF host electrodes with different mass proportions of BCF under the same scanning speed and the same current density, respectively. (e) The corresponding comparison of the areal capacitance. (f) The corresponding comparison the cycling stability. Reproduced with permission.82 Copyright 2021, Elsevier. (g) CV curves of the Ti3C2Tx–AgNW–MnONW-C60 MSC at scan rates of 10–80 mV s−1. (h) GCD profiles of the Ti3C2Tx–AgNW–MnONW-C60 MSC at current densities of 0.2–0.8 mA cm−2. Reproduced with permission.84 Copyright 2020, Wiley-VCH. (i) Scheme of the as-printed Ti3C2Tx-based microsupercapacitor, indicating the pseudocapacitive energy storage mechanism induced by the protons interacting with (j) –OH group-terminated Ti. (k) Electrochemical impedance spectroscopy of a screen-printed MSC before and after the CV tests at various scan rates. (l) Normalized CV profiles of a typical screen-printed MSC (〈N〉 =3, gap = 200 μm). (m) Normalized GCD profiles of a typical screen-printed MSC (〈N〉 = 3, gap = 200 μm). Reproduced with permission.85 Copyright 2020, Wiley-VCH. | ||
Furthermore, when an acidic electrolyte is used, protons in the electrolyte react with hydroxyl groups (–OH) on the Ti3C2Tx surface, leading to changes in the valence state of Ti. Therefore, Ti3C2Tx MXene electrodes can exhibit redox-based pseudocapacitive behavior in acidic gel electrolytes. For example, Abdolhosseinzadeh et al.85 constructed an MSC based on Ti3C2Tx deposition ink using a PVA/H2SO4 gel as the electrolyte (Fig. 4(i) and (j)). The nearly vertical impedance line observed in the low-frequency region of the electrochemical impedance spectroscopy (EIS) (Fig. 4(k)) strongly confirms the pseudocapacitive behavior of the device. Furthermore, the CV curves (Fig. 4(l)) and GCD curves (Fig. 4(m)) further demonstrate that the energy storage process of the MSC primarily relies on reversible redox reaction.
 | ||
| Fig. 5 Ion intercalation energy storage mechanism. (a) Photographs of MX-MSCs in the flat state. (b) CV curves tested at 50 mV s−1. (c) Schematic diagram of parallel ion transfer between MXene nanosheet based films. Reproduced with permission.72 Copyright 2019, Royal Society of Chemistry. (d) Schematic diagram of Ti3C2Tx-MSCs. (e) Mechanism diagram of electrolyte ion migration during electrochemical charge and discharge. (f) 3D mapping from in situ Raman testing. Reproduced with permission.89 Copyright 2023, Elsevier. | ||
Furthermore, Zhao et al.89 designed an MSC based on nitrogen-doped MXene nanosheets (MXene-H) by performing surface modification of MXene using a hexaazatriphenylen (HAT) derivative (Fig. 5(d)). The surface modification effectively expanded the interlayer spacing of MXene nanosheets, significantly mitigating the self-stacking effect caused by van der Waals forces. This provided sufficient space for ion intercalation while also forming conductive pathways to enhance ionic migration. The transfer process of electrolyte ions during the charge and discharge cycles is illustrated in Fig. 5(e). During the discharge cycle, H+ ions from the electrolyte intercalate into the interlayer spaces of MXene-H and form chemical bonds with terminal –O groups, while deintercalation occurs during the charge cycle. This reversible intercalation and deintercalation behavior alters the valence state of Ti in MXene-H, thereby generating faradaic capacitance. Furthermore, intercalation weakens the interlayer interactions in MXene-H. The changes in the A1g peak observed in Fig. 5(f) can be attributed to variations in the valence state of Ti and interlayer interactions caused by ion intercalation/deintercalation during the charge and discharge process. The final designed MSC exhibits outstanding energy storage performance, fully demonstrating the critical role of the ion intercalation mechanism in optimizing electrochemical performance.
3.3. Hybrid microsupercapacitor
Hybrid MSCs (HMSCs)43,80,90–96 combine the properties of EDLCs and pseudocapacitors, incorporating chemical reactions through the pseudocapacitive mechanism to significantly enhance energy density while retaining the fast charge–discharge capability of EDLCs. The two mechanisms complement each other under different current densities, leveraging the advantage of high energy density at low current densities while maintaining excellent power performance at high current densities. The intrinsic energy storage process involves ions in the electrolyte migrating toward the electrode surface under an applied electric field and forming an electric double layer through adsorption. Simultaneously, some ions undergo redox reactions with active sites on the electrode or intercalate into the interlayer structure of the electrode, enabling charge storage during the charging process. During discharge, these ions release charges through deintercalation or reverse redox reactions, while the electrostatically adsorbed ions quickly return to the electrolyte, completing the charge release process.HMSCs have been widely studied due to the advantages brought by the dual-effect mechanism. For example, Gao and colleagues92 designed a flexible zinc-ion hybrid microsupercapacitor (ZIHMSC) based on an Ti3C2Tx MXene anode and a V2O5 cathode, using a polyacrylamide (PAM) hydrogel containing dissolved ZnSO4 as the electrolyte (Fig. 6(a)). The structure of V2O5 is similar to that of MXenes (Fig. 6(b) and (c)), and their relatively large interlayer spacing enables them to accommodate various small-sized ions. Especially in zinc-ion batteries, the intercalation and deintercalation process of Zn2+ exhibits high reversibility. Fig. 6(d) illustrates the electrochemical reaction mechanism of the ZIHMSC during the charge and discharge cycles. During charge, Zn2+ ions deintercalate from the V2O5 electrode and are subsequently adsorbed onto the surface of the Ti3C2Tx electrode through the electrolyte, accompanied by the intercalation of a small number of ions. In this process, the valence state of the V element increases. During discharge, Zn2+ ions desorb from the surface of the Ti3C2Tx electrode and then intercalate into the interlayer spaces of V2O5 through the electrolyte. This energy storage mechanism, based on Zn2+ adsorption/desorption (Ti3C2Tx) and intercalation/deintercalation (V2O5), endows the ZIHMSC with excellent electrochemical performance (Fig. 6(e) and (f)). Fig. 6(g) presents the charge–discharge cycling stability of the HMSC, confirming the excellent cycling durability of the MSC under the dual-effect mechanism.
 | ||
| Fig. 6 Hybrid MSCs’ energy storage mechanism. (a) The preparation and assembly process of the ZIHMSC. (b) The sectional view of the V2O5 film. (c) The sectional view of the Ti3C2Tx MXene film. (d) The schematic of the energy storage mechanism of the ZIHMSC in the charge and discharge states. (e) The CV profiles of the ZIHMSC in the hydrogel electrolyte at 10–100 mV s−1. (f) The GCD profiles of the ZIHMSC in the hydrogel electrolyte at 0.34–3.45 mA cm−2. (g) The cycle stability performances and coulombic efficiency of the ZIHMSC in the hydrogel electrolyte under 2.30 mA cm−2, and the inset exhibits the first ten and the last ten cycles of the ZIHMSC. Reproduced with permission.92 Copyright 2022, Elsevier. (h) Schematic Diagram of the Ti3C2Tx-DMAC MXene-based Zn-ion MSC integrated with a photodetector. (i) Schematic diagram of the charge conversion during the charging and discharging process. (j) Cell transformation diagram of Fe(CN)64+ and Fe(CN)63+. Reproduced with permission.93 Copyright 2023, American Chemical Society. | ||
Furthermore, Liu et al.93 investigated the effects of different electrolytes on device performance and proposed the use of K4Fe(CN)6 (KFCN) as a redox additive to enhance redox reactions, thereby improving the volumetric capacitance of the Ti3C2Tx-N,N-dimethylacetamide (DMAC)-based ZIHMSC (Fig. 6(h)). Fig. 6(i) illustrates the energy storage mechanism of the ZIHMSC. During discharge, Zn is oxidized to Zn2+, which either adsorbs onto the surface of the Ti3C2Tx-DMAC electrode or intercalates into its interlayer spaces. Simultaneously, Fe(CN)63− is reduced to Fe(CN)64− on the cathode surface (Fig. 6(j)), providing additional faradaic pseudocapacitance to the system. In addition, free K+ ions can enhance the conductivity of the electrolyte within the solid-state structure, facilitating the formation of the electric double layer. The combination of multiple effects enables the MSC to exhibit a high volumetric capacitance of 2107.4 F cm−3 at a scan rate of 5 mV s−1. Moreover, the teams of Jin38 and Yang94 also independently constructed HMSCs. Their research demonstrated that, through the synergistic interaction between redox reactions and electric double-layer effects, the HMSC exhibited exceptional electrochemical performance.
Therefore, HMSCs ingeniously integrate the advantages of both EDLCs and pseudocapacitors, creating an energy storage system that combines rapid response with high capacity. In this system, the device achieves rapid charge storage through the formation of an electric double layer via ion adsorption, while redox reactions and ion intercalation provide additional capacitance for the HMSCs, significantly enhancing its energy storage performance. This design overcomes the limitations of a single mechanism in terms of cycling stability and energy storage efficiency, enabling HMSCs to exhibit excellent performance across various application scenarios. Additionally, the highly reversible charge transfer process ensures the stability and reliability of devices during long-term operation, further demonstrating its research significance in the field of high-performance energy storage devices.
4. Fabrication procedure
The increasing demand for device operating environments and precision conditions has driven the rapid development of MSC fabrication techniques. Existing fabrication techniques can be broadly categorized into mask-assisted and mask-free methods based on the precision requirements of the devices. Mask-assisted methods, such as photolithography, screen printing, and spray coating, are typically employed for large-scale production and applications with lower precision requirements, as they enable simultaneous processing of multiple devices. In contrast, mask-free methods, including inkjet printing, laser scribing, vacuum filtration, and 3D printing, offer higher precision and greater adaptability, making them widely used for applications involving complex structures, high resolution, cost efficiency, and rapid prototyping. These various fabrications have differing impacts on the electrochemical performance of MSCs, including energy density, power density, and cycling stability. This section will systematically summarize the latest advancements in each fabrication method and analyze their respective advantages.4.1. Mask-assisted methods
Mask-assisted electrode patterning techniques, such as photolithography, screen printing, and spray coating, have become critical technologies for fabricating microsupercapacitors due to their rapid large-area processing capability and broad compatibility with various substrates. Furthermore, these methods, enabled by precise mask design and processing workflows, allow for micron-scale and even nanoscale control over electrode shapes and dimensions. This level of precision optimizes ion transport pathways within the electrodes, enhancing charge storage efficiency and speed, thereby meeting the demands of high-precision microstructured devices.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 bending cycles. This improvement, with the volumetric capacitance increasing by approximately 140%, is attributed to the gradual wetting of the electrodes by the electrolyte, which enhances the ion transport pathways. These results highlight the immense potential of flexible MSCs for applications in wearable devices.
000 bending cycles. This improvement, with the volumetric capacitance increasing by approximately 140%, is attributed to the gradual wetting of the electrodes by the electrolyte, which enhances the ion transport pathways. These results highlight the immense potential of flexible MSCs for applications in wearable devices.
 | ||
| Fig. 7 Photolithography and screen printing. (a) Schematic of the fabrication of a flexible microsupercapacitor device. Reproduced with permission.98 Copyright 2022, Elsevier. (b) Schematic illustration of the fabrication process of Ti3C2Tx-based microsupercapacitors. Reproduced with permission.80 Copyright 2019, Wiley-VCH. (c) Schematic illustration of the fabrication process of the Ti3C2Tx–CNTs LbL assembly based interdigital microsupercapacitor. Reproduced with permission.97 Copyright 2021, Elsevier. (d) Schematic of the fabrication of printable MX-MSCs. (e) Optical images of aqueous H-MXene ink in the normal and inverted states (left) and the fabricated MX-MSCs on various substrates (right). (f) Optical images of L-MXene ink in the normal and inverted states (left), the prepared MX-MSCs on a PET substrate and the used screen mesh after screen printing (right). (g) Apparent viscosity of H-MXene and L-MXene inks versus shear rate. Reproduced with permission.99 Copyright 2021, Wiley-VCH. (h) Schematic illustration of direct screen printing of MXene sediments. (i) Areal capacitance of MSCs with different number of printed passes (〈N〉). (j) Width distribution (top) and variation distribution (bottom) of MXene printed lines with 〈N〉 = 3. Reproduced with permission.85 Copyright 2020, Wiley-VCH. (k) The fabrication processes of screen-printed flexible MSC devices. Reproduced with permission.44 Copyright 2019, Wiley-VCH. | ||
In addition, Jiang et al.80 fabricated MX-MSCs using a photolithography-spray coating process (Fig. 7(b)). Benefiting from the high precision of the photolithography process, the electrode spacing of the device is only 10 μm, which provides shorter ion transport pathways and reduces ion transport resistance. As a result, the device exhibits a high volumetric capacitance (30 F cm−3), excellent rate capability (300 V s−1), and an extremely short relaxation time constant (τ0 = 0.45 ms), surpassing traditional capacitors. Moreover, using Ti3C2Tx–carbon nanotube (CNT) composites as electrodes, Wang et al.97 developed a novel on-chip microsupercapacitor through the combination of photolithography and vacuum filtration techniques. The fabrication process of the device is shown in Fig. 7(c). Overall, MSCs fabricated via photolithography exhibit high volumetric capacitance and excellent cycling stability, highlighting the significant advantages of photolithography in constructing high-precision energy storage devices.
Additionally, the linewidth and the number of printing passes (〈N〉) during the printing process also influence the performance of the MSCs. As an illustration, Abdolhosseinzadeh et al.85 utilized an ink composed of notch precursors and multilayer Ti3C2Tx deposits to fabricate MSCs via screen printing with different numbers of printing passes, and subsequently compared their performance. Fig. 7(h) depicts the process of collecting and adjusting waste material for printing devices. As shown in Fig. 7(i), during the screen printing process, the areal capacitance increases from 27 to 158 mF cm−2 as 〈N〉 increases from 1 to 5. This demonstrates that optimizing the number of printing passes can effectively regulate the charge storage performance of the devices. Fig. 7(j) indicates that the final printed line (width ≈ 235 μm, 〈N〉 = 3) achieves higher printing resolution and excellent spatial uniformity (as low as 6%) compared to lines with other widths and numbers of printing passes, thereby enabling precise pattern printing. In a similar study, Li et al.44 utilized Ti3C2Tx composite ink to fabricate MSCs with high volumetric energy density and power density via screen printing (Fig. 7(k)).
 | ||
| Fig. 8 Spray coating. (a) Schematic illustration of the assembly process for the in-plane device. (b) Top-view optical microscope image of the in-plane device. (c) XRD patterns of the Ti3AlC2 MAX, Ti3C2Tx, PET, and Ti3C2Tx-coated PET films. Reproduced with permission.32 Copyright 2024, Springer Nature. (d) Schematic diagram of the preparation of an interdigital Zn-ion microsupercapacitor by a facile spraying method and microphotography of the interdigital Zn-ion MSCs. Reproduced with permission.91 Copyright 2023, Royal Society of Chemistry. (e) Schematic of the fabrication process of the μcup electrodes by screen-printing or blade coating and subsequent laser scribing, followed by spray coating of the electrolyte layer and the top electrode. (f) and (g) Dimensions of the μcups and the different layers for thin walled samples. (h) Close-up of the electrolyte–Ti3C2Tx interface. (i) Volumetric capacitance (C/V) at different scan rates for an increasing thickness of the MXene top electrode. (j) Ragone plot comparison of this work to other MSC systems, showing the advantages of employing the μcup-MSCs in achieving both high energy density and power density. Reproduced with permission.83 Copyright 2023, Wiley-VCH. | ||
In addition to constructing planar electrodes, the spray coating method can also be used to fabricate MSCs with 3D electrode structures. Recently, Zhang et al.83 successfully fabricated a Ti3C2Tx-graphene-based “μcup” MSC by spraying the electrolyte and the top-layer Ti3C2Tx electrode. The specific fabrication process is illustrated in Fig. 8(e). First, the μcup shaped graphene bottom electrode is prepared via screen printing or laser engraving. Next, the electrolyte is sprayed onto the cup walls, and finally, Ti3C2Tx ink is sprayed to fill the cup structure. The SEM images clearly reveal the 3D structure of MSCs with different thicknesses and the layered characteristics of Ti3C2Tx nanosheets (Fig. 8(f) and (g)). A close-up of the electrolyte–Ti3C2Tx interface further reveals the horizontally aligned Ti3C2Tx layers (Fig. 8(h)). Fig. 8(i) shows that at low scan rates, the volumetric capacitance of the devices significantly improves with the increase in electrode thickness, fully demonstrating the advantages of the spray coating method in constructing 3D MSCs. The final device achieved an energy density of 3.99 μW h cm−2 and maintained 2.82 μW h cm−2 at a maximum power density of 588.9 μW cm−2 (Fig. 8(j)), providing a new perspective for fabricating high-performance 3D MSCs using the spray coating method.
4.2. Mask-free methods
Mask-free electrode patterning methods such as inkjet printing, laser scribing, vacuum filtration, and 3D printing have shown tremendous potential in MSC fabrication due to their flexibility and efficiency. Compared to mask-assisted methods, these techniques do not require additional masks or complex procedures, allowing for greater flexibility to accommodate various design requirements. They support the fabrication of devices on flexible substrates and complex curved surfaces, making them particularly suitable for small-scale customized production. In addition, mask-free methods significantly reduce production costs by minimizing the reliance on customized masks. They also decrease the waste and resource consumption associated with mask fabrication, thereby reducing the environmental impact and demonstrating greater sustainability and cost-effectiveness. | ||
| Fig. 9 Inkjet printing. (a) Schematic illustration of the inkjet-printing assisted fabrication process of MSCs. (b) Conductivity study of inkjet-printed p-Ti3C2Tx and Ti3C2Tx@C-180 MSCs after 60 days in a H2SO4/PVA electrolyte at ambient temperature. (c) and (d) Stability study of inkjet-printed p-Ti3C2Tx and Ti3C2Tx@C-180 MSCs for various periods up to 60 days in a H2SO4/PVA electrolyte at ambient temperature. Scale bar: 10 mm. Reproduced with permission.106 Copyright 2022, Elsevier. (e) Schematic illustration of an all-inkjet-printing-based heterostructure SSC (top) and MSC (bottom). (f) CA of a 30L SSC with different electrolytes. (g) CA of a 30L MSC with excess H2O and 0.5 M H2SO4 at a current density of 50 μA cm−2. Reproduced with permission.107 Copyright 2021, Elsevier. (h) Schematic illustration of direct Ti3C2Tx ink printing. (i) Areal capacitance of inkjet-printed MSCs with different 〈N〉. Reproduced with permission.86 Copyright 2019, Springer Nature. | ||
Apart from the ink, the electrode structure also significantly influences the performance of MSCs. For example, Wang et al.107 fabricated microsupercapacitors and sandwiched microsupercapacitors (SSCs) based on 2D heterostructures by inkjet printing Ti3C2Tx MXene nanosheets as electrodes (Fig. 9(e)). Fig. 9(f) and (g) show the areal capacitance measurements of SSCs and MSCs, respectively, in different electrolyte environments. It can be observed that under the same electrolyte conditions, the areal capacitance of SSCs is significantly higher than that of MSCs. This result underscores the essential role of electrode design in enhancing the performance of MSCs. Additionally, other factors, such as the number of printing passes during the printing process, can also influence the performance of MSCs. Zhang et al.86 fabricated MSCs using Ti3C2Tx MXene ink via inkjet printing without any additives or binary solvent systems in the organic electrolyte (Fig. 9(h)). Fig. 9(i) shows the areal capacitance of MSCs printed with different 〈N〉, where 〈N〉 = 2 results in an areal capacitance of 1.3 mF cm−2, while 〈N〉 = 25 achieves a significantly higher areal capacitance of 12 mF cm−2. This indicates that, to a certain extent, an increase in 〈N〉 results in a corresponding increase in areal capacitance, highlighting the significant impact of the number of printing passes on the electrochemical performance of MSCs.
 | ||
| Fig. 10 Laser scribing and vacuum filtration. (a) Optical images of laser written “USTB, CAS, Flextronics, Institute of semiconductor” words, cartoon MSC, butterfly-shaped Zn-ion MSC, and MSC array. (b) Schematic diagram exhibiting the fabrication process of the patterned MSC array via laser writing technology. (c) Ragone plot of areal energy density vs. power density for the fabricated in situ annealed MSC in comparison with the reported Zn-ion devices. (d) Variation of areal capacitance with various scan rates. (e) CV curves of the designed MSC under different bending states. Reproduced with permission.90 Copyright 2021, Springer. (f) Schematic illustration of the fabrication of flexible Ti3C2Tx//LIG AMSCs. Reproduced with permission.30 Copyright 2024, Elsevier. (g) Schematic diagram of the preparation process of the Ti3C2Tx/CMC/CNT film. (h) Illustration of components of the MSC device. Reproduced with permission.38 Copyright 2023, Elsevier. (i) Fabrication schematic of Ti3C2Tx-MSCs in a 20 M LiCl-gel electrolyte. (j) CV curves at different potential windows of Ti3C2Tx-MSCs. Reproduced with permission.87 Copyright 2022, Oxford University Press. (k) Schematic diagram of an all-flexible self-powered integrated system combining MXene-H-MSCs and an MXene hydrogel pressure sensor. Reproduced with permission.89 Copyright 2023, Elsevier. | ||
Moreover, patterned electrodes can also be directly fabricated using masks during the vacuum filtration process, simplifying subsequent processing steps. For example, Wu and colleagues87 innovatively designed a water-in-LiCl (WIL, 20 M LiCl) salt gel electrolyte and fabricated Ti3C2Tx-MSCs using high-pressure vacuum filtration. As shown in Fig. 10(i), Ti3C2Tx dispersions were uniformly filtered through a mask to form interdigitated microelectrodes on a polyvinylidene fluoride (PVDF) membrane. The mask was then removed, and the prepared microelectrodes were fully transferred onto a flexible PET substrate. Finally, Ti3C2Tx-MSCs were fabricated by drop-casting the LiCl-gel electrolyte. The cyclic voltammetry curves in Fig. 10(j) show that the stable voltage window of the MSC can be extended to 1.6 V, significantly enhancing the energy storage performance of the MSC. In a similar study, Zhao et al.89 also fabricated MSCs using a mask-assisted vacuum filtration method (Fig. 10(k)). The MSC exhibited an areal capacitance of 210.1 mF cm−2, an energy density of 29.18 mW h cm−2, as well as excellent cycling stability and flexibility.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 charge–discharge cycles (Fig. 11(d)). In addition, Huang et al.43 developed a high-performance Ti3C2Tx hydrogel supercapacitor electrode and further extended it to the fabrication of fully 3D-printed Ti3C2Tx microsupercapacitors, as shown in Fig. 11(e). These studies fully demonstrate the potential of 3D printing in the fabrication of high-performance energy storage devices.
000 charge–discharge cycles (Fig. 11(d)). In addition, Huang et al.43 developed a high-performance Ti3C2Tx hydrogel supercapacitor electrode and further extended it to the fabrication of fully 3D-printed Ti3C2Tx microsupercapacitors, as shown in Fig. 11(e). These studies fully demonstrate the potential of 3D printing in the fabrication of high-performance energy storage devices.
 | ||
Fig. 11 3D printing and supplementary fabrications. (a) Schematic illustration of 3D printing a symmetric MSC with interdigitated electrodes using the Ti3C2Tx MXene sediment. (b) CV curves collected at different scan rates from 2 to 100 mV s−1. (c) Ragone plots of 3D printed MSCs in this study compared with other reported MSCs. (d) Long-cycle stability test of MSCs with 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cycles at a current density of 10 mA m−2 with a capacity retention of 93.1%. Reproduced with permission.117 Copyright 2023, Elsevier. (e) The fabrication process of the 3D printed all-MXene MSC via MSES. Reproduced with permission.43 Copyright 2021, Wiley-VCH. (f) Schematic showing the preparation procedure of binder-free ink and 3D printing of designed patterns. Reproduced with permission.101 Copyright 2019, Wiley-VCH. (g) Schematic of fabrication of MXene/CNT microsupercapacitor (MSC) devices. Reproduced with permission.120 Copyright 2021, Elsevier. (h) Schematic illustration of the fabrication process of planar microsupercapacitors. Reproduced with permission.47 Copyright 2018, IOP Publishing. (i) and (j) Versatile patterning including sculpting text, symbols, intricate designs and scalable scalpel patterning of spray coated Ti3C2Tx thin films (thickness, 150 nm), scale bar 5 mm. (k) Direct automated scalpel engraving of Ti3C2Tx thin films to fabricate microsupercapacitors; geometric parameters marked in an orange frame. (l) SEM images showing the edge view of thin films with a thickness of 20, 50, and 150 nm after scalpel cut. Reproduced with permission.48 Copyright 2018, Wiley-VCH. 000 cycles at a current density of 10 mA m−2 with a capacity retention of 93.1%. Reproduced with permission.117 Copyright 2023, Elsevier. (e) The fabrication process of the 3D printed all-MXene MSC via MSES. Reproduced with permission.43 Copyright 2021, Wiley-VCH. (f) Schematic showing the preparation procedure of binder-free ink and 3D printing of designed patterns. Reproduced with permission.101 Copyright 2019, Wiley-VCH. (g) Schematic of fabrication of MXene/CNT microsupercapacitor (MSC) devices. Reproduced with permission.120 Copyright 2021, Elsevier. (h) Schematic illustration of the fabrication process of planar microsupercapacitors. Reproduced with permission.47 Copyright 2018, IOP Publishing. (i) and (j) Versatile patterning including sculpting text, symbols, intricate designs and scalable scalpel patterning of spray coated Ti3C2Tx thin films (thickness, 150 nm), scale bar 5 mm. (k) Direct automated scalpel engraving of Ti3C2Tx thin films to fabricate microsupercapacitors; geometric parameters marked in an orange frame. (l) SEM images showing the edge view of thin films with a thickness of 20, 50, and 150 nm after scalpel cut. Reproduced with permission.48 Copyright 2018, Wiley-VCH. | ||
Moreover, during the 3D printing process, the electrode thickness can be adjusted by precisely controlling the number of printed layers, enabling the fabrication of MSCs with varying energy storage capacities. For example, Yu et al.101 modified MXene through nitrogen doping (MXene-N) and applied it to 3D extrusion printing technology, fabricating MSC electrodes with varying numbers of layers to optimize their electrochemical performance. The process of ink optimization and pattern printing is illustrated in Fig. 11(f). The study found that during the 3D printing process, the areal capacitance significantly increased with the number of printed layers. Benefiting from the high-quality three-layer electrode structure, the MSC achieved a high areal energy density of 0.42 mW h cm−2 with an areal capacitance of 8.2 F m−2.
Additionally, the team led by Gogotsi48 innovatively proposed a method for fabricating MSCs using the automated scalpel technique. This method achieves high-precision engraving of planar functional devices by precisely controlling the motion trajectory of the blade on the surface (Fig. 11(i) and (j)). Fig. 11(k) illustrates the process of fabricating microsupercapacitors by engraving Ti3C2Tx films using the automated scalpel technique. This technique can be used to engrave films of different thicknesses (Fig. 11(l)); what's more, compared to laser scribing, which causes edge thermal oxidation when removing excess parts, it has no impact on the untouched areas. Additionally, Li et al.121 also utilized the automated scalpel technique to engrave their developed Ti3C2Tx-poly(3,4-ethylenedioxythiophene) (PEDOT) composite films, in order to fabricate MSCs. Compared to Ti3C2Tx-based MSCs, this device exhibits significantly improved performance, with the areal capacitance increasing by fivefold and the rate capability enhanced, achieving 2.4 mF cm−2 at a scan rate of 10 mV s−1. The mentioned methods are suitable for the design and fabrication of various functional films, offering a new approach for the controllable manufacturing of complex electrode structures.
In most fabrication methods, both symmetric and asymmetric microsupercapacitors can be effectively constructed by introducing design variations. Therefore, most of the above preparation methods are suitable not only for symmetric MSCs, but also for asymmetric structures of hybrid MSCs. For example, mask-assisted techniques are applicable to both types of MSCs by employing different stencil shapes during the fabrication of the positive and negative electrodes. Liu demonstrated this by using distinct masks to spray-coat the electrodes,91 successfully fabricating a Zn/Ti3C2Tx-DMSO-based asymmetric MSC. Moreover, approaches that allow for precise structural and material control, such as laser scribing and 3D printing, are also suitable for both symmetric and asymmetric MSCs. For instance, Wang et al.30 designed impedance-matched Ti3C2Tx/LIG-based asymmetric MSCs via laser scribing.
However, compared to symmetric MSCs, the fabrication of asymmetric MSCs presents significantly greater challenges. Since asymmetric devices are typically composed of two different electrode materials, the fabrication of such devices makes the optimization of charge balance and electrochemical compatibility between the electrodes a critical design concern. Additionally, methods like spin coating and vacuum filtration are less suitable for fabricating asymmetric MSCs. These methods are effective when the same material is used for both electrodes, but in asymmetric configurations, they lack the ability to precisely control the spatial distribution and layering of dissimilar materials, which is essential for achieving optimal performance.
5. Application strategies of Ti3C2Tx in MSCs
In MX-MSCs, the electrodes, as the most crucial component, play a decisive role in determining the performance of the devices. By providing efficient charge storage and rapid charge transport, they directly impact the energy density and power density of the devices. The current collectors, separators and other functional components play important roles in charge transport, ion isolation, and structural stability of the devices. Yu122 pointed out that MXenes can be utilized in various components of MSCs based on their specific working mechanisms, including electrode materials, current collectors, and separators. As the most extensively studied and widely applied MXene, Ti3C2Tx is considered an outstanding candidate for the electrodes of MSCs due to its large specific surface area, tunable layered structure, and abundant surface functional groups. In addition, the excellent electrical conductivity and mechanical stability of Ti3C2Tx make it highly suitable for fabricating current collectors. Meanwhile, the abundant porous structure of Ti3C2Tx allows ions to migrate freely between electrodes, laying the foundation for it to serve as a separator. This section will focus on the multifunctional applications of Ti3C2Tx in MSCs and its impact on the performance of devices.5.1. Core component: electrode
The structure and materials of the electrodes significantly influence the energy storage performance of MSCs. Among these factors, the electrode material directly affects the electrochemical performance of capacitors, including cycling stability, capacitance, and energy density. Therefore, the selection of electrode materials is crucial in MSCs. For Ti3C2Tx, owing to its metallic-like conductivity, it can directly serve as the conductive electrodes of MSCs. Combining Ti3C2Tx with materials of different dimensions can expand its interlayer spacing, thereby weakening its self-stacking effect and further enhancing the energy storage capacity of devices. This section will elaborate on the versatile applications of Ti3C2Tx in the fabrication of electrodes from the perspectives of pure Ti3C2Tx-based electrodes and composite electrodes.| Material | Electrolyte | Areal capacitance (mF cm−2) | Energy density | Power density | Cycling stability | Ref. |
|---|---|---|---|---|---|---|
| Ti3C2Tx | PVA/H2SO4 gel | 2000 (1.2 mA cm−2) | 0.1 mW h cm−2 | 0.38 mW cm−2 | 90% (10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cycles) 000 cycles) |
43 |
| Ti3C2Tx | LiCl gel | 19.9 (5 mV s−1) | 31.7 mW h cm−3 | — | 103% (10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cycles) 000 cycles) |
87 |
| Ti3C2Tx | EMIMBF4/PVDF-HFP | 44 (0.1 mA cm−2) | 13.9 μW h cm−2 | — | 92% (1000 cycles) | 72 |
| 43.7 mW h cm−3 | 19.2 mW cm−3 | |||||
| Ti3C2Tx | PVA/H2SO4 gel | — | 0.32 μW h cm−2 | 158 μW cm−2 | 97% (10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cycles) 000 cycles) |
86 |
| Ti3C2Tx | PVA/H2SO4 gel | 28.5 (0.75 mA cm−2) | 8.84 μW h cm−2 | 0.11 mW cm−2 | 92% (10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cycles) 000 cycles) |
70 |
| Ti3C2Tx | PAM/ZnSO4 gel | 129 (0.34 mA cm−2) | 48.9 μW h cm−2 | 673 μW cm−2 | 77% (10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cycles) 000 cycles) |
92 |
| Ti3C2Tx | PVA/H2SO4 gel | — | 41.9 mW h cm−3 | 1.2 W cm−3 | 140% (10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cycles) 000 cycles) |
98 |
| 29.7 mW h cm−3 | 26.8 W cm−3 | 95% (10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cycles) 000 cycles) |
89 | |||
| Ti3C2Tx | PVA/H2SO4 gel | 210.1 (0.1 mA cm−2) | 29.18 mW h cm−2 | 5.0 mW cm−2 | ||
| Ti3C2Tx | Zn(CF3SO3)2-K3Co(CN)6 | — | 178.3 μW h cm−3 | 9.9 mW cm−3 | 70% (5000 cycles) | 91 |
| Ti3C2Tx | LiCl gel | 43 (0.2 mA cm−2) | 35 μW h cm−2 | 240 μW cm−2 | — | 31 |
| 53 mW h cm−3 | 364 mW cm−3 | |||||
| Ti3C2Tx | PVA/H2SO4/EBB gel | 12.5 (5 mV s−1) | 2.42 μW h cm−2 | 0.23 mW cm−2 | 95% (1000 cycles) | 32 |
| Ti3C2Tx | PVA/ZnCl2 gel | 72.02 (10 mV s−1) | 0.02 mW h cm−2 | 0.50 mW cm−2 | 80% (5000 cycles) | 90 |
| 0.18 mW h cm−3 | 24.63 μW cm−3 | |||||
| Ti3C2Tx | PVA/H2SO4 gel | 46.24 (20 mV s−1) | 68.51 mW h cm−3 | 6.16 W cm−3 | 98.5% (10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cycles) 000 cycles) |
77 |
| Ti3C2Tx | PVA/H3PO4 gel | 25.5 (5 mV s−1) | 2.3 mW h cm−3 | 159.6 mW cm−3 | 90% (8000 cycles) | 47 |
| Ti3C2Tx | PVA/H2SO4 gel | 5.4 (10 mV s−1) | — | — | 84% (10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cycles) 000 cycles) |
30 |
| Ti3C2Tx | PVA/Zn(CF3SO3)2 | — | 439.7 mW h cm−3 | 6.1 W cm−3 | 100% (6000 cycles) | 41 |
| Ti3C2Tx | PVA/ZnCl2 | 84.07 (20 mV s−1) | 16.814 μW h cm−2 | 252.21 μW cm−2 | 95% (10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cycles) 000 cycles) |
75 |
| Ti3C2Tx | PVA/H2SO4 gel | 1100 (20 mV s−1) | 13.8 μW h cm−2 | 2.5 mW cm−2 | 100% (10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cycles) 000 cycles) |
99 |
| Ti3C2Tx | PVA/H2SO4 gel | 158 (10 mV s−1) | 1.64 μW h cm−2 | 778.3 μW cm−2 | 95.8% (17![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cycles) 000 cycles) |
85 |
| Ti3C2Tx | PVA/H2SO4 gel | 2337 (2 mV s−1) | 207.81 μW h cm−2 | 3.74 mW cm−2 | 93.1% (10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cycles) 000 cycles) |
117 |
| Ti3C2Tx | PVA/H2SO4 gel | 23.4 (5 mV s−1) | 1.48 mW h cm−3 | 189.9 mW cm−3 | 92.4% (5000 cycles) | 76 |
| Ti3C2Tx | PVA/H3PO4 gel | 87 (2 mV s−1) | 11.8 mW h cm−3 | 100.8 mW cm−3 | 82% (5000 cycles) | 116 |
| Ti3C2Tx | PVA/H3PO4 gel | — | 2.8 mW h cm−3 | 225 mW cm−3 | 95% (10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cycles) 000 cycles) |
71 |
| Ti3C2Tx | PVA/H2SO4 gel | 61.5 (2 mV s−1) | 6.73 μW h cm−2 | — | 80.7% (5000 cycles) | 123 |
| Ti3C2Tx | PVA/H2SO4 gel | 5.89 (3.47 μA cm−2) | — | — | 90% (500 cycles) | 124 |
| Ti3C2Tx | PVA/H3PO4 gel | 0.7 (1 mV s−1) | — | — | — | 80 |
| Ti3C2Tx | PVA/H2SO4 gel | 25 (200 mV s−1) | 0.77 μW h cm−2 | 46.6 mW cm−2 | 92% (10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cycles) 000 cycles) |
125 |
 | ||
| Fig. 12 Pure Ti3C2Tx or composite materials serving as electrodes. (a) Schematic drawing of the AMSCs. Reproduced with permission.30 Copyright 2024, Elsevier. (b) Working mechanism of the fabricated MSC. Reproduced with permission.91 Copyright 2022, Elsevier. (c) SEM image of the cross-sectional area of the MSC. (d) SEM images of the Ti3C2Tx–CNTs composite. Reproduced with permission.97 Copyright 2021, Elsevier. (e) Schematic diagram of the electrochemical mechanism in MPM. Reproduced with permission.37 Copyright 2022, IOP Publishing. (f) Schematic illustration of the RGO/CNF/Ti3C2Tx composite paper. (g) Cross-sectional SEM images of the RGO/CNFs/Ti3C2Tx composite paper. Reproduced with permission.126 Copyright 2020, Royal Society of Chemistry. (h) Proposed capacitive mechanism of the Ti3C2Tx/BCN compound materials in MSCs for galvanostatic charge/discharge. (i)–(k) FE-SEM pictures of Ti3C2Tx powder, BCN microspheres and Ti3C2Tx/BCN-3 microflowers. Reproduced with permission.40 Copyright 2021, Royal Society of Chemistry. | ||
Although Ti3C2Tx as an electrode enables the device to exhibit favorable electrochemical performance, the presence of hydrophilic surface functional groups (such as –OH) in aqueous electrolytes may lead to excessive interlayer adsorption of water molecules, thereby hindering the effective intercalation and diffusion of electrolyte ions. Moreover, in acidic or alkaline electrolytes, the highly reactive functional groups may trigger side reactions, causing structural degradation of Ti3C2Tx or changes in its interlayer interactions, thereby affecting the cycling stability and energy storage capacity of the device. The intercalation strategy can effectively reduce ion diffusion resistance caused by the interlayer stacking of Ti3C2Tx, while surface modification can regulate the type and distribution of functional groups, minimizing the likelihood of side reactions and optimizing ion transport pathways. These approaches can collectively enhance the electrochemical performance of the electrodes. For example, Liu et al.91 constructed a ZIHMSC using Ti3C2Tx intercalated with dimethyl sulfoxide (DMSO) as the cathode (Fig. 12(b)). DMSO, with its relatively large molecular size, effectively expanded the interlayer spacing of Ti3C2Tx, significantly facilitating the rapid intercalation and deintercalation of ions. Moreover, the presence of sulfinyl groups (![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) S
S![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O) provides additional active sites for the electrode, further increasing the capacitance of the MSC. The device exhibits a higher areal capacitance compared to unmodified MX-MSCs. Similarly, Feng et al.116 utilized Mn2+ intercalation to effectively enhance the energy density of Ti3C2Tx-based MSCs. Moreover, Zhao et al.89 modified the surface of Ti3C2Tx using a hexaazatriphenylene (HAT) derivative and applied the resulting N-doped Ti3C2Tx nanosheets to MSCs, significantly enhancing the energy storage performance of the device.
O) provides additional active sites for the electrode, further increasing the capacitance of the MSC. The device exhibits a higher areal capacitance compared to unmodified MX-MSCs. Similarly, Feng et al.116 utilized Mn2+ intercalation to effectively enhance the energy density of Ti3C2Tx-based MSCs. Moreover, Zhao et al.89 modified the surface of Ti3C2Tx using a hexaazatriphenylene (HAT) derivative and applied the resulting N-doped Ti3C2Tx nanosheets to MSCs, significantly enhancing the energy storage performance of the device.
In addition, while diversely modifying and designing the electrode materials and structures enhance their interfacial properties and electrochemical activity for a significant improvement in the performance of MSCs, the multi-dimensional modification and adjustment of the electrolyte, as a crucial medium for charge transfer, are related to the migration rate of ions during the charging and discharging processes, the reversibility of electrode reactions, and the working stability of the whole device. Therefore, to solve the problems such as the low energy density of MSCs, an ionic liquid (IL) electrolyte with an interface layer of large electrochemical window can be employed as a solution. For example, Zhu et al.127 designed a stable ionic liquid (IL)–Ti3C2Tx microemulsion system using tween-20 (TW20), which achieved the uniform distribution of the IL within the in-plane electrodes. This increased the ion-accessible surface area of the Ti3C2Tx electrode. As a result, the Ti3C2Tx-IL-based MSC has a wide voltage window ranging from 0 to 3 V, as well as high areal and volumetric energy densities of 50.7 μW h cm−2 and 39 mW h cm−3. Moreover, the aqueous electrolyte also has similar functions. For example, the MXene interdigital electrode is encapsulated with a chemically crosslinked poly-vinyl alcohol (PVA) aqueous hydrogel electrolyte (GPAH) thin film combined with graphene oxide (GO),128 which can achieve a single cell areal capacitance of up to 40.8 mF cm−2. The hydroxylated MXene MSCs using an acidic polyacrylamide (PAM) hydrogel electrolyte have achieved excellent power density (16.50 mW cm−2, 110.91 μW h cm−2) and energy density (394.59 μW h cm−2 at 0.825 mW cm−2),129 which benefit from the adsorption mechanism of NH4+/H+. However, aqueous electrolytes limit the voltage window of MXenes. To address this issue, a high-voltage scanning strategy can be further employed.130 For instance, Wu and his team used a high-concentration “water-in-LiBr” gel electrolyte, which enabled the MXene symmetric MSC to have a high voltage window of 1.8 V.131 They also utilized a water-in-LiCl salt gel electrolyte to effectively regulate the reaction kinetics in Ti3C2Tx MXene microelectrodes, thus preparing high-voltage MXene MSCs.87
1D Nanostructure. 1D nanostructures, such as nanowires and nanotubes, when combined with Ti3C2Tx, can form support and bridging effects between layers, effectively reducing layer stacking. This optimizes charge transport pathways, enhances conductivity, and improves electrochemical activity, making them excellent composite electrode materials (Table 2). For example, Fang et al.97 assembled CNTs and Ti3C2Tx layer by layer (LbL) to fabricate MSCs based on Ti3C2Tx–CNT composite electrodes (Fig. 12(c)). As observed in Fig. 12(d), CNTs act as spacers for Ti3C2Tx nanosheets, preventing their stacking and aggregation during the assembly process. The resulting electrodes provide rapid diffusion pathways for electrolyte ions and enhance interfacial electron reaction efficiency, thereby improving the capacitive performance. The final device exhibited an excellent areal capacitance of 61.38 mF cm−2 at a current density of 0.5 mA cm−2. Similarly, Wang et al.37 combined Ti3C2Tx MXene with polyaniline (PANI) and multi-walled carbon nanotubes (MWCNTs) to form a composite (MPM) (Fig. 12(e)). Due to the synergistic enhancement effect among the components of the MPM composite, the device achieved an excellent specific capacitance of 414 F g−1 at 1 A g−1. In addition, MnO2 nanowires,114 MnO2 mixed with Ag nanowires,84 and bacterial cellulose (BC)82,88 have also been utilized to form composite electrodes with Ti3C2Tx. These 1D materials are inserted between Ti3C2Tx layers and act as structural supports, effectively expanding its interlayer spacing. This allows a sufficient number of ions to freely intercalate and deintercalate within the Ti3C2Tx structure, enabling the constructed MSCs to exhibit improved charge storage capabilities.
| Material | Electrolyte | Areal capacitance (mF cm−2) | Energy density | Power density | Cycling stability | Ref. |
|---|---|---|---|---|---|---|
| Ti3C2Tx/BC-KOH | PAM/Zn(CF3SO3)2 gel | 223 | 42 μW h cm−2 | — | 72% (1000 cycles) | 88 |
| Ti3C2Tx/MnO2 | EMIMBF4/PVA gel | 24.7 (5 mV s−1) | 21.5 μW h cm−2 | 0.154 mW cm−2 | 83.6% (3000 cycles) | 114 |
| Ni(OH)2-Ni-Ti3C2Tx@CW | NaOH aqueous solution | 19.81 (4 mA cm−2); 10.33 (20 mA cm−2) | 206 μW h cm−2 | 1.94 mW cm−2 | 89.3% (7000 cycles) | 132 |
| AgNW-Ti3C2Tx-MnO2-C60 | PVA-KOH gel polymer | 216.2 (10 mV s−1) | 9.2 μW h cm−2 | 58.3 mW cm−2 | 75% (1000 cycles) | 84 |
| Ti3C2Tx/BC | Zn(CF3SO3)2/PAM | — | 34.0 μW h cm−2 | — | 72.3% (3000 cycles) | 82 |
| Ti3C2Tx-PANI@MWCNTs | PVA/H2SO4 gel | 30.2 (0.1 mA cm−2) | — | — | 70.2% (10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cycles) 000 cycles) |
37 |
| Ti3C2Tx-CNTs | PVA/H2SO4 gel | 61.38 (0.5 mA cm−2) | 5.46 μW h cm−3 | 200 mW cm−3 | 85.5% (50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cycles) 000 cycles) |
97 |
| Ti3C2Tx-BC | PVA/H2SO4 gel | 111.5 (2 mA cm−2) | 5.52 μW h cm−2 | — | 72.2% (5000 cycles) | 110 |
| Ti3C2Tx-PANI@CNTs | PVA/H2SO4 gel | 78.2 | 2.72 μW h cm−2 | 50 μW cm−2 | 93.3% (10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cycles) 000 cycles) |
115 |
2D Nanostructure. 2D nanosheets offer a large surface area while possessing excellent conductivity and outstanding mechanical properties. As a 2D material, Ti3C2Tx can enhance overall electrochemical performance through interlayer interactions when combined with other 2D nanosheets (Table 3). As an illustration, Wei et al.126 fabricated MSCs by combining 2D reduced graphene oxide (rGO) and cellulose nanofibers (CNFs) with Ti3C2Tx. As shown in Fig. 12(f), in the composite paper, rGO and Ti3C2Tx serve as the cuticle layer, while CNFs act as the nacre layer. The two components are stacked to form a 2D/2D composite electrode. Fig. 12(g) shows the SEM image of the composite paper. The multi-component interconnected structure exhibits high conductivity, enabling effective interfacial regulation and charge transfer under complex environmental stimuli, while simultaneously creating mutually linked functional responses. The final MSC exhibited a volumetric capacitance of 148.25 F cm−3 and a volumetric energy density of 20.5 mW h cm−3. In addition, Yue et al.39 also fabricated MSCs based on Ti3C2Tx–rGO composite aerogel electrodes. At a scan rate of 1 mV s−1, the MSC exhibited an areal capacitance of 34.6 mF cm−2 and demonstrated excellent long cycling life. In similar studies, 2D materials such as poly(3,4-ethylenedioxythiophene)-poly(styrene sulfonate) (PEDOT:PSS),121 laser-induced graphene (LIG),78 boron-modified MoS2,111 V2CTx133 and carboxymethyl cellulose/carbon nanotube (CMC/CNT)38 have also been combined with Ti3C2Tx to fabricate MSC electrodes. The combination of 2D materials with Ti3C2Tx not only effectively increases the specific surface area of the electrode but also regulates its surface chemical environment through interlayer synergistic interactions. This optimizes the interfacial structure and charge transfer pathways, thereby significantly enhancing the electrochemical performance and environmental adaptability of MSCs.
| Material | Electrolyte | Areal capacitance (mF cm−2) | Energy density | Power density | Cycling stability | Ref. |
|---|---|---|---|---|---|---|
| Ti3C2Tx/rGO/CNFs | PVA/KOH gel | — | 20.5 mW h cm−3 | 250 mW cm−3 | 93.75% (6000 cycles) | 126 |
| Ti3C2Tx/rGO | PVA/H2SO4 gel | 34.6 (1 mV s−1) | — | — | 91% (15![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cycles) 000 cycles) |
39 |
| Ti3C2Tx/CMC/CNT | PVA/H2SO4 gel | 1431.4 (1 mA cm−2) | 258.8 μW h cm−2 | 0.75 mW cm−2 | 93.2% (15![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cycles) 000 cycles) |
38 |
| Ti3C2Tx-LIG | PVA/H2SO4 gel | 100 (10 mV s−1) | 21.16 μW h cm−2 | 0.075 mW cm−2 | 91% (10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cycles) 000 cycles) |
78 |
| B-1 T MoS2/Ti3C2Tx | PVA/K2SO4 gel | 72.31 (75 μA cm−2) | 5.7 μW h cm−2 | 77.1 μW cm−2 | 100% (10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cycles) 000 cycles) |
111 |
| V2CTx/Ti3C2Tx | H2SO4 gel | 390 (10 mV s−1) | 5.1 mW h g−1 | 350 mW g−1 | 95% (10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cycles) 000 cycles) |
133 |
| Ti3C2Tx/PEDOT:PSS | PVA/H2SO4 gel | — | 1.49 μW h cm−2 | 1.25 mW cm−2 | 96.9% (10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cycles) 000 cycles) |
134 |
| Ti3C2Tx/PEDOT | PVA/H2SO4 gel | 2.4 (10 mV s−1) | 8.7 mW h cm−3 | 0.55 W cm−3 | 90% (10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cycles) 000 cycles) |
121 |
3D Nanostructure. The combination of 3D nanostructures with Ti3C2Tx can provide larger energy storage space and superior mechanical strength, enhance the diffusion efficiency of electrolyte ions, and improve the overall stability of the electrode, thereby increasing the power density and cycling stability of the MSCs. As an example, Tu et al.40 were the first to propose using a tube furnace to anneal and dry traditional 2D boron carbon nitride (BCN) nanosheets in an N2 atmosphere, converting them into 3D-BCN microspheres. These were then combined with Ti3C2Tx to form Ti3C2Tx–BCN microflowers, which were applied as electrodes in all-solid-state flexible microsupercapacitors (Fig. 12(h)). Fig. 12(i)–(k) show the FE-SEM images of Ti3C2Tx powder, BCN microspheres, and Ti3C2Tx/BCN microflowers, respectively. From Fig. 12(k), it can be observed that the microflowers possess rough surfaces with numerous ridge-like structures, a characteristic that significantly increases the specific surface area of the electrode. Additionally, during the compositing process, the BCN microspheres effectively prevented the restacking of Ti3C2Tx nanosheets by releasing gases, thereby further enhancing the electrochemical performance of the resulting MSCs.
Additionally, Wu et al.135 utilized aerosol jet printing (AJP) technology to synthesize hybrid Ti3C2Tx/C nanospheres by binding Ti3C2Tx nanosheets with C nanoparticles, which were then used to construct planar microsupercapacitors. The C nanospheres increased the interlayer spacing of Ti3C2Tx nanosheets and effectively prevented their restacking, thereby enhancing the areal capacitance of the device. This concept provides a straightforward and efficient approach for fabricating 3D-structured MXenes with suppressed self-stacking, paving the way for diverse high-performance micro energy storage devices.
Therefore, combining Ti3C2Tx with materials of different dimensions according to specific application scenarios and requirements can effectively overcome the limitations of pure Ti3C2Tx electrodes. The reasons are as follows: firstly, introducing materials with excellent conductivity and electrochemical activity (such as graphene, carbon nanotubes, and conductive polymers) can optimize the ion intercalation and deintercalation processes, accelerate charge transport, and enhance the conductivity of Ti3C2Tx. Secondly, the diversified structures formed after the combination can provide a larger specific surface area and more electrochemical active sites, thereby enhancing the capacitive performance. Meanwhile, it can also improve the structural stability and corrosion resistance of Ti3C2Tx, thereby enhancing the cycling stability of the devices.
Modifying and compositing electrode materials can significantly enhance the performance of MSCs, and changes in electrode structure also have a profound impact on MSC performance and applications. The interdigitated electrode can provide a large contact area between the electrode and the electrolyte within a small area, promote the rapid diffusion of electrolyte ions across the electrode surface, and the fabrication process is simple, making it the most common electrode structure. Compared with the interdigitated structure, the sandwich structure can enhance the interaction between the electrodes and the electrolyte (Fig. 9(e) and (f)). Under the same electrolyte conditions, this structure can improve the wettability of the electrolyte, so that the MSC based on it has a higher area capacitance than the device based on the conventional interdigital structure.107 Besides, changing the radian of the interdigital electrode, such as arch design, can improve the flexibility of the electrode and make it possible to develop high-performance MSCs suitable for curved surfaces such as the eyeball.109 Moreover, a novel planar electrode structure based on Ti3C2Tx, resembling a “skeletal connection” (Fig. 10(a), lower right), has been developed to enhance device performance. This unique structure can significantly increase the contact area between the electrode and the electrolyte, providing more surface sites for ion adsorption, thus enhancing the energy storage capacity of the device.90 Different from the planar electrodes mentioned, linear fiber electrodes with structures similar to textiles or wires can easily achieve functions such as bending, winding, and laying, making it more convenient to integrate with substrates of different shapes or other electronic components. This is especially true in applications that require flexible or three-dimensional structures. Moreover, the layered structure of linear fiber electrodes contributes to a higher surface area, more electroactive sites, and optimized ion diffusion pathways, enabling excellent capacitance and relatively high rate capability.132 These studies collectively emphasize the crucial role of the electrode structure in determining the electrochemical performance of MSCs. Thoughtful design of electrode structures can enhance the electrode–electrolyte interaction, improve overall device performance, and allow them to adapt to different energy storage scenarios.
5.2. Functional components: current collector and separator
In addition to the electrodes, which serve as the core components, MSCs also include current collectors to enhance conductivity and separators to isolate the two electrodes. Current collectors and separators, as functional components, also play a critical role in enhancing the performance of MSCs. With its excellent conductivity, outstanding electrochemical stability, and superior mechanical strength, Ti3C2Tx is widely regarded as an ideal choice for fabricating current collectors. Moreover, when Ti3C2Tx serves as a separator, its highly ordered layered structure provides efficient pathways for ion transport, while its porous structure facilitates the diffusion of electrolyte ions between the electrodes. Therefore, Ti3C2Tx, as a current collector and separator, not only enhances the electrochemical performance of MSCs but also extends the lifespan of the device.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 charge–discharge cycles, the capacitance retention is 98.5%, demonstrating good cycling stability. Moreover, the use of modified Ti3C2Tx current collectors brings about further improvements in the performance of devices. For example, Liu et al.91 intercalated ions into Ti3C2Tx and fabricated a flexible ZIHMSC (Fig. 8(d)) composed of a conductive Ti3C2Tx-LiCl current collector, a Ti3C2Tx-DMSO cathode, and a Zn anode. The Ti3C2Tx-LiCl current collector contributes to the formation of a three-dimensional conductive network, making it an ideal substrate for the rapid transfer of ions and electrons.
000 charge–discharge cycles, the capacitance retention is 98.5%, demonstrating good cycling stability. Moreover, the use of modified Ti3C2Tx current collectors brings about further improvements in the performance of devices. For example, Liu et al.91 intercalated ions into Ti3C2Tx and fabricated a flexible ZIHMSC (Fig. 8(d)) composed of a conductive Ti3C2Tx-LiCl current collector, a Ti3C2Tx-DMSO cathode, and a Zn anode. The Ti3C2Tx-LiCl current collector contributes to the formation of a three-dimensional conductive network, making it an ideal substrate for the rapid transfer of ions and electrons.
 | ||
Fig. 13 Ti3C2Tx serving as a current collector and separator. (a) Schematic diagram showing the fabrication process of the Ti3C2Tx-based MSC array via laser cutting technology. (b) SEM image of the monolayer Ti3C2Tx flakes with a large size. (c) TEM image of the monolayer Ti3C2Tx flakes with a large size. (d) I–V curves of the large-sized and small-sized Ti3C2Tx film. (e) Areal capacitance with various scan rates from 20 to 2000 mV s−1. Reproduced with permission.77 Copyright 2020, Elsevier. (f) Preparation of Ti3C2Tx separators. (g) Cycling stability of the microsupercapacitors assembled with GFM and Ti3C2Tx-coated GFM separators after 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cycles at current densities of 1 and 5 A g−1. Reproduced with permission.34 Copyright 2023, Royal Society of Chemistry. (h) Schematic diagram depicting the preparation and function of the Ti3C2Tx-GF separator. (i) Digital photos of bare GF and Ti3C2Tx-GF. The inks used for printing with different concentrations are displayed as the insets. The marked number represents x mg mL−1, x = 0, 1, 3, and 5. (j) Schematic illustration of the polarized charge distribution within different separators. (k) Polarization–electric field (P–E) loops of GF and Ti3C2Tx-GF. Reproduced with permission.74 Copyright 2022, Wiley-VCH. 000 cycles at current densities of 1 and 5 A g−1. Reproduced with permission.34 Copyright 2023, Royal Society of Chemistry. (h) Schematic diagram depicting the preparation and function of the Ti3C2Tx-GF separator. (i) Digital photos of bare GF and Ti3C2Tx-GF. The inks used for printing with different concentrations are displayed as the insets. The marked number represents x mg mL−1, x = 0, 1, 3, and 5. (j) Schematic illustration of the polarized charge distribution within different separators. (k) Polarization–electric field (P–E) loops of GF and Ti3C2Tx-GF. Reproduced with permission.74 Copyright 2022, Wiley-VCH. | ||
6. Integrated application
The development of energy storage devices demands power supply modules that combine miniaturization with efficient energy delivery. Benefiting from the exceptional energy density, power density, miniaturized structural design, and highly flexible tunability of Ti3C2Tx-based MSCs, they exhibit unique competitive advantages in the field of integrated energy storage. Connecting multiple MSC units in series or parallel enables the self-integration of MSCs, resulting in integrated arrays with enhanced energy storage capacity. These arrays can flexibly adjust operating voltage and capacity to meet the diverse requirements of various electronic devices, while also simplifying the overall design and manufacturing process. This approach improves the integration and performance of systems. In addition, MSCs, with their compact size, exceptional flexibility, and outstanding cycling stability, can not only meet the structural adaptation requirements of complex devices but also seamlessly integrate with flexible electronic devices, providing stable and reliable energy storage support. These characteristics endow MSCs with unique advantages when integrated with various functional devices, such as sensors, photodetectors, triboelectric nanogenerators, and thin-film electronic devices. MSCs can provide integrated systems with long-lasting, stable energy support or efficient control functionality. This section will explore the roles and advantages of MSCs in integrated systems in detail.6.1. Self-integration
When multiple MSCs are connected in series or parallel to form self-integrated arrays,47,77,85,94,104,106,110,113,125 they not only significantly enhance energy storage and output capabilities but also improve system reliability and stability, providing critical support for the design of multifunctional integrated energy storage devices. For instance, Hu and colleagues110 fabricated all-solid-state microsupercapacitor arrays (MSCAs) consisting of four MSC units connected in series and parallel on an elastic substrate, as shown in Fig. 14(a) and (d). From the CV curves (Fig. 14(b)) and GCD curves (Fig. 14(c)) of a single MSC and the series-connected MSCA, it can be observed that the output voltage of the series-connected MSCA is four times that of a single MSC under the same discharge duration. For the parallel-connected MSCA, the output current is nearly four times that of a single MSC, while the charge–discharge time is extended to four times that of a single MSC (Fig. 14(e) and (f)). Similarly, Abdolhosseinzadeh85 and Sangili106 also investigated the self-integration performance of their designed MSCs. As shown in Fig. 14(g)–(i), when n MSC units are connected in series, the total operating voltage of the array becomes n times that of a single MSC, while the total capacitance remains unchanged. In contrast, under the parallel integration mode, the total operating voltage of the array remains the same as that of a single MSC, while the capacitance is significantly increased. These results are consistent with those obtained by Jiao. The above studies fully demonstrate that the self-integration strategy enables precise regulation of voltage or current by connecting individual MSC units in series or parallel, thereby significantly enhancing the overall energy storage capacity of the array. This highlights the promising application potential of MSC self-integration in high-performance energy storage systems. | ||
| Fig. 14 Self-integration and integrated with sensors. (a) Optical photographs of an as-fabricated MSCA consisting of 4 MSC units connected in series. (b) CV curves for the MSCA and a single MSC unit at a scan rate of 50 mV s−1. (c) GCD curves for the MSCA and a single MSC unit at a high current density of 2.0 mA cm−2. (d) Optical photographs of an as-fabricated MSCA consisting of 4 MSC units connected in parallel. (e) CV curves for the MSCA and a single MSC unit at a scan rate of 50 mV s−1. (f) GCD curves for the MSCA and a single MSC unit at a high current density of 2.0 mA cm−2. Reproduced with permission.110 Copyright 2019, Wiley-VCH. (g) CV profiles of different tandem devices at 50 mV s−1. Reproduced with permission.85 Copyright 2020, Wiley-VCH. (h) CV profiles at 20 mV s−1 (inset: schematic circuit diagram of three single MSCs connected in series and parallel configurations). (i) GCD profiles at 0.3 mA cm−2 of three MSC devices (Ti3C2Tx@C-180 MSCs) connected in parallel and in series. Reproduced with permission.106 Copyright 2022, Elsevier. (j) Schematic illustration and structure of the hemispherical integrated device. (k) CV curves of MSCs at different scan rates. (l) Ragone plot of areal energy density vs. power density for Ti3C2Tx-MSCs in comparison with the reported MSCs. Reproduced with permission.109 Copyright 2024, Royal Society of Chemistry. (m) Schematic illustration of the Ti3C2Tx/BP-based self-powered smart sensor system. Reproduced with permission.136 Copyright 2021, Wiley-VCH. | ||
6.2. Sensor
Sensors typically consume a significant amount of energy during operation, necessitating the integration of efficient power supply modules to ensure continuous energy support. Compared to traditional power sources, MSCs, with their high energy density and miniaturized design, have emerged as an ideal choice to meet this demand. Recently, Shen and colleagues109 reported a hemispherical integrated system based on Ti3C2Tx MXene, which connects microsupercapacitors with strain sensors for intraocular pressure (IOP) monitoring. Fig. 14(j) shows a schematic diagram of the integrated system, where the strain sensor provides accurate and real-time IOP data, while the MSC supplies the necessary energy to power the sensor. At a scan rate of 5 mV s−1, the hemispherical Ti3C2Tx-based MSC exhibits a high areal capacitance of 32 mF cm−2 (Fig. 14(k)) and an energy density of 10 mW h cm−2 (Fig. 14(l)), providing robust energy support for the strain sensor. By integrating the MSC and the strain sensor onto the same deformable substrate, this self-powered system successfully achieves independent, continuous, and stable IOP monitoring functionality.In addition, to expand the application of wearable electronics in the biomedical field, efforts should focus on enabling accurate and continuous detection of physiological signals without relying on external power sources. For example, by integrating a flexible pressure sensor, a microsupercapacitor, and flexible solar cells, Zhang136 designed a self-powered smart sensing system based on Ti3C2Tx/black phosphorus (BP) (Fig. 14(m)), enabling real-time monitoring of human cardiac activity under physiological conditions. In the entire system, the flexible solar cell array harvests energy by converting light into electricity, directly powering the sensor under illuminated conditions. Moreover, the generated photocurrent can also be used to charge the micro-supercapacitor, ensuring continuous operation of the system in dark environments. In a similar integration study, Yang and his team89 developed a miniature integrated system composed of a solar cell panel, Ti3C2Tx-HMSC, and a Ti3C2Tx hydrogel pressure sensor. This system was connected in series, designed to independently and effectively respond to subtle changes in body movement. Additionally, Gao et al. have also demonstrated that MXene-based MSCs are compatible with sensors and other devices, ensuring long-term stable operation.137 Therefore, the integration of the Ti3C2Tx-based MSC with sensors not only addresses the energy supply challenges of sensors, demonstrating excellent suitability for long-term continuous monitoring, but also exhibits broad application prospects in the field of wearable devices.
6.3. Photodetector
The integration of Ti3C2Tx-based MSCs with photodetectors enables efficient energy storage and management, allowing the photodetectors to achieve continuous and stable responses. For example, Liu et al.93 integrated MX-MSCs with a Ti3C2Tx-dicyandiamide and thiourea etched solution (DCTES)-based photodetector, designing a fully MXene-based flexible self-powered integrated photodetection system. As shown in Fig. 6h, the cathode of the MSC serves as one electrode of the photodetector, while the external electrode functions as the other electrode of the photodetector. In the integrated system, the MSC, with its outstanding performance, is capable of providing the integrated photodetector with a stable and continuous voltage exceeding 0.9 V (Fig. 15(a)), ensuring stable response (Fig. 15(b)). This provides strong technical support for other power-supplied integrated sensing systems. | ||
| Fig. 15 Integration with a photodetector, a triboelectric nanogenerator and thin-film electronic devices. (a) Self-discharge curve of MSCs (the inset is a digital image taken during the integrated system test). (b) I–V curves of the Ti3C2Tx-DCTES MXene photodetector driven by the MSCs. Reproduced with permission.93 Copyright 2023, American Chemical Society. (c) Schematic of the all-MXene-based integrated system. The system contains a WCC, a Zn-ion MSC, and a photodetector, all of which are composed of Ti3C2Tx. (d) Schematic diagram of the integrated wireless power sensor system. Reproduced with permission.75 Copyright 2023, Wiley-VCH. (e) Working mechanism of the TENG. (f) Schematic illustration of the mechanism for generating electricity to charge the microsupercapacitor. Reproduced with permission.71 Copyright 2018, Elsevier. (g) Schematic illustration of the flexible multifunctional self-powered system with Cu-EGaIn electrodes. Reproduced with permission.79 Copyright 2022, Elsevier. (h) Schematic illustration of the integrated microsupercapacitor-thin film rectifier circuits fabricated on a glass substrate. (i) CV curves of microsupercapacitors sharing the same RuO2 electrodes with thin film rectifiers. (j) GCD curves of microsupercapacitors sharing the same RuO2 electrodes with thin film rectifiers. Reproduced with permission.73 Copyright 2022, Elsevier. (k) Schematic diagram of the AC-line filtering circuit. (l) Output voltage signal after filtering using the Ti3C2Tx microsupercapacitor, in comparison with the output voltage signal produced using commercial AEC. Reproduced with permission.80 Copyright 2019, Wiley-VCH. | ||
Although the direct integration of MSCs with photodetectors supports their normal operation, the integrated systems require external circuit connections to recharge the MSCs. To address this issue, Duan et al.75 developed a seamlessly integrated all-MXene-based system through a simple one-step laser scribing method. As shown in Fig. 15(c), the system integrated a wireless charging coil (WCC), microsupercapacitor, and photodetector; all these three modules were entirely composed of Ti3C2Tx and connected by highly conductive MXene wires, eliminating the need for additional welding or assembly processes. Fig. 15(d) shows the circuit diagram of the system in operation. According to Faraday's law of electromagnetic induction, an alternating current in the primary coil (L1) generates a varying magnetic field, inducing a current in the secondary coil (L2), thereby enabling non-contact energy transfer. The received energy is stored in the MSC and used as an internal power source to drive the photodetector or other sensors. The introduction of the WCC eliminates the need for external power wiring and facilitates device integration, achieving a more streamlined and efficient system.
6.4. Triboelectric nanogenerator
Aside from charging MSCs through external circuits or non-contact energy transfer, Ti3C2Tx-based MSCs can also be integrated with triboelectric nanogenerators (TENGs) to achieve both energy generation and storage. Jiang et al.71 proposed a highly compact self-charging power unit composed of an MX-MSC and a TENG. The process of energy harvesting by the TENG and charging the MSC is illustrated in Fig. 15(e) and (f). This unit can harvest and store energy generated by human activities in standby mode and provide continuous power to electronic devices during operation. This approach cleverly converts ambient mechanical energy generated by human motion into electrical energy for utilization, effectively addressing the limitation of storage device capacity and offering a new strategy for sustainable energy supply.In a similar study, Sun and his team79 developed a flexible multifunctional self-powered system. As shown in Fig. 15(g), the system consists of a liquid-metal-based TENG, a micro-supercapacitor with Cu particle mixed liquid metal (Cu-EGaIn)/Ti3C2Tx composite electrodes, a liquid-metal-based power management module (PMM), and functional circuits. During the entire operation process, the TENG first generates energy. However, the output current of the TENG is alternating current (AC) and typically has high output impedance, characterized by high voltage and low current, making it difficult to directly charge the MSC. The incorporation of the PMM, integrated with a low-loss full-wave bridge rectifier and a high-efficiency buck inverter, not only converts the AC output to DC output but also regulates the voltage and current to meet the operating requirements of the MSC. This enables effective energy harvesting from high-output-impedance energy sources and completes the efficient process from energy collection to storage. The above studies fully demonstrate that integrating MSCs with triboelectric nanogenerators and power management modules can efficiently achieve energy generation, management, and storage, providing important insights for the development of compact, multifunctional, and sustainable self-powered systems.
6.5. Thin-film electronic
In previous studies, the development of self-powered electronic systems required the integration of on-chip energy storage units with various types of energy harvesters. However, most studies involving MSCs rely on bulky rectifiers for energy harvesting interfaces, which are challenging to integrate. To overcome this issue, K. Hota et al.73 proposed a novel approach combining on-chip microsupercapacitors with thin-film transistors (TFTs) and thin-film rectifiers to fabricate a highly integrated monolithic rectifier-storage unit. In this design, all three components utilize Ti3C2Tx electrodes, providing an efficient integrated solution for electrochemical on-chip energy storage. Fig. 15(h) illustrates the circuit diagram of the integrated microsupercapacitor thin-film rectifier fabricated on a glass substrate, showing, from left to right, the MX-MSC, the field-effect rectifier, and a typical single microsupercapacitor/rectifier unit. In the integrated unit, the MSC exhibits a wide operating voltage window, rapid charge–discharge response, and excellent cycling stability (Fig. 15(i) and (j)), providing an efficient and reliable solution for the integration of self-powered systems.Additionally, on-chip fabricated MSCs can be integrated into thin-film electronic devices in a compatible manner, serving as ripple filtering units for high-frequency sources or as energy harvesters. For example, Alshareef and his colleagues80 constructed a thin-film filtering circuit integrated with a function generator, a full-wave rectifier, and a Ti3C2Tx-based MSC. The circuit diagram is shown in Fig. 15(k). The sinusoidal AC input signal is first generated by the function generator and then converted into a DC signal through the full-wave bridge rectifier. Subsequently, the signal is passed through the microsupercapacitor, resulting in a significantly smoothed output signal (Fig. 15(l)). Benefiting from the high areal capacitance and rapid response of the microsupercapacitor, the AC signal in the integrated circuit is successfully converted into a constant output voltage. Moreover, Wang et al.138 developed a wafer-scale fabrication method for Ti3C2Tx MXene-based MSCs, featuring a high-potential device structure with integrated fractal microelectrodes. The novel structure increased the number of adsorption sites and enhanced the electric field, thereby improving the ion transport rate. As a result, the constructed FMSCs successfully filtered DC from various AC currents at a high frequency of 20 kHz, demonstrating outstanding filtering performance in AC line-filtering circuits.
7. Challenges and prospects
7.1. Challenges and innovative strategies
In recent years, Ti3C2Tx has made significant progress as an energy storage material in microsupercapacitor applications. However, Ti3C2Tx-based MSCs still face several key challenges in performance optimization, large-scale fabrication, and integrated applications. To enhance the overall electrochemical performance of the devices and expand their application potential, the following challenges need to be addressed.Additionally, compared to pure Ti3C2Tx, its composites with materials of different dimensions (such as one-dimensional nanowires, two-dimensional nanosheets, and three-dimensional porous structures) exhibit superior electrochemical performance in MSC electrodes. However, current research on Ti3C2Tx-based composite materials for MSCs remains limited, and the underlying mechanisms and design strategies have yet to be fully explored. In the future, researchers can focus more on the development of Ti3C2Tx composite materials to unlock their potential in high-performance MSCs.
7.2. Summary and future perspectives
In summary, Ti3C2Tx MXene, with its tunable interlayer spacing and abundant surface functional groups, exhibits metallic-like conductivity and excellent mechanical stability, demonstrating immense potential in the energy storage field, particularly in microsupercapacitors. Its unique two-dimensional structure and physicochemical properties lay a solid foundation for the development of high-performance, miniaturized, and sustainable energy storage devices. This article systematically reviews the latest research of Ti3C2Tx MXene-based MSCs from five aspects: mechanisms, methods, materials, structures, and applications, while thoroughly discussing key strategies and approaches for performance optimization.Specifically, we first introduced the composition of Ti3C2Tx-based MSCs, with a focus on explaining the energy storage mechanisms of different types of MSCs, including electric double-layer capacitors, pseudocapacitors, and hybrid MSCs. In addition, considering the significant impact of fabrication processes on the performance of MSCs, this article reviews the existing patterning techniques for MSCs and systematically summarizes the performance enhancement achieved by mask-assisted methods including photolithography, screen printing, and spray coating, as well as mask-free methods like inkjet printing, laser scribing, vacuum filtration, and 3D printing. Meanwhile, we also discussed the application of Ti3C2Tx in electrode design, current collector optimization, and separator improvement, with a focus on the synergistic effects of its composites with nanomaterials of different dimensions. Furthermore, we highlighted the unique value of these composite strategies in enhancing the areal capacitance, rate performance, and energy density of MSCs. Additionally, to showcase the application prospects of MX-MSCs, we compiled practical examples of their use in self-powered systems. We elaborated on their immense potential for self-integration or integration with devices such as sensors, photodetectors, and triboelectric nanogenerators, and conducted an in-depth analysis of their critical role in ensuring the long-term stable operation of such systems.
Looking ahead, research on Ti3C2Tx-MSCs will continue to advance toward high performance, environmental friendliness, and sustainability. This review not only discusses the strategies for enhancing the electrochemical performance of MSCs in existing literature but also provides important theoretical guidance for the design and development of next-generation high-efficiency, eco-friendly, low-cost, and scalable MSCs. With their high energy storage capacity, Ti3C2Tx-MSCs are expected to fully meet the demands of microelectronic devices and wearable technologies for high energy density, long cycle life, and flexible integration, thereby driving innovation and development in next-generation smart electronics and energy storage systems.
Author contributions
C. H. and Y. B. wrote the original draft. W. W., P. Q., and D. W. managed the data. J. L., C. F., and G. S. edited the article and provided the funding support.Data availability
No primary research results, software or code have been included and no new data were generated or analysed as part of this review.Conflicts of interest
Chuqiao Hu and Yumeng Bai contributed equally to this work. And the authors declare that they have no conflict of interest. They have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.Acknowledgements
This work is financially supported by the Liaoning Applied Fundamental Research Project (Grant No. 2022JH2/101300158 and 2023JH2/101300014), the Scientific Research Funding Project of Educational Department of Liaoning Province (Grant No. LJKZ0060) and the Fundamental Research Funds for the Central Universities (Grant No. 3132023235).Notes and references
- A. Ahmed, S. Sharma, B. Adak, M. M. Hossain, A. M. LaChance, S. Mukhopadhyay and L. Sun, InfoMat, 2022, 4, e12295 CrossRef CAS.
- A. Rafique, I. Ferreira, G. Abbas and A. C. Baptista, Nano-Micro Lett., 2023, 15, 40 CrossRef CAS PubMed.
- S. Bairagi, I. Shahid ul, M. Shahadat, D. M. Mulvihill and W. Ali, Nano Energy, 2023, 111, 108414 CrossRef CAS.
- J. Zhang, S. Ricote, P. V. Hendriksen and Y. Chen, Adv. Funct. Mater., 2022, 32, 2111205 CrossRef CAS.
- J. Xu, X. Cai, S. Cai, Y. Shao, C. Hu, S. Lu and S. Ding, Energy Environ. Mater., 2023, 6, e12450 CrossRef CAS.
- H. Liu, J. Song, Z. Zhao, S. Zhao, Z. Tian and F. Yan, Adv. Sci., 2024, 11, 2305347 CrossRef CAS PubMed.
- D. C. Nguyen, M. Ding, P. N. Pathirana, A. Seneviratne, J. Li, D. Niyato, O. Dobre and H. V. Poor, IEEE Internet of Things J., 2022, 9, 359–383 Search PubMed.
- N. Cheng, S. Wu, X. Wang, Z. Yin, C. Li, W. Chen and F. Chen, IEEE Internet of Things J., 2023, 10, 14438–14461 Search PubMed.
- A. Sahasrabudhe, L. E. Rupprecht, S. Orguc, T. Khudiyev, T. Tanaka, J. Sands, W. Zhu, A. Tabet, M. Manthey, H. Allen, G. Loke, M.-J. Antonini, D. Rosenfeld, J. Park, I. C. Garwood, W. Yan, F. Niroui, Y. Fink, A. Chandrakasan, D. V. Bohórquez and P. Anikeeva, Nat. Biotechnol., 2023, 42, 892–904 CrossRef PubMed.
- D. Gao, Z. Luo, C. Liu and S. Fan, Green Energy Environ., 2023, 8, 972–988 CrossRef CAS.
- R. Akhter and S. S. Maktedar, J. Materiomics, 2023, 9, 1196–1241 CrossRef.
- M. Şahin, F. Blaabjerg and A. Sangwongwanich, Energies, 2022, 15, 674 CrossRef.
- M. Zhong, M. Zhang and X. Li, Carbon Energy, 2022, 4, 950–985 CrossRef CAS.
- C. Zhang, Z. Peng, C. Huang, B. Zhang, C. Xing, H. Chen, H. Cheng, J. Wang and S. Tang, Nano Energy, 2021, 81, 105609 CrossRef CAS.
- L. B. Karroubi, S. G. Patnaik, B. D. Assresahegn, B. Bounor, C. C. H. Tran, S. H. Choudhury, D. Bourrier, D. Guay and D. Pech, Energy Storage Mater., 2022, 47, 134–140 CrossRef.
- M. Tahir, L. Li, L. He, Z. Xiang, Z. Ma, W. A. Haider, X. Liao and Y. Song, Energy Environ. Mater., 2022, 7, e12546 CrossRef.
- W. Wang, W. Zou and Q. Fan, Int. J. Electrochem. Sci., 2022, 17, 22062 CrossRef CAS.
- Z. Niu, L. Zhang, L. Liu, B. Zhu, H. Dong and X. Chen, Adv. Mater., 2013, 25, 4035–4042 CrossRef CAS PubMed.
- F. Bu, W. Zhou, Y. Xu, Y. Du, C. Guan and W. Huang, npj Flex. Electron., 2020, 4, 31 CrossRef.
- A. M. Patil, A. A. Jadhav, N. R. Chodankar, A. T. Avatare, J. Hong, S. D. Dhas, U. M. Patil and S. C. Jun, Coord. Chem. Rev., 2024, 517, 216020 CrossRef CAS.
- C. Zhang, J. Energy Chem., 2021, 60, 417–434 CrossRef CAS.
- M. Gao, F. Wang, S. Yang, A. Gaetano Ricciardulli, F. Yu, J. Li, J. Sun, R. Wang, Y. Huang, P. Zhang and X. Lu, Mater. Today, 2024, 72, 318–358 CrossRef CAS.
- M. Naguib, M. Kurtoglu, V. Presser, J. Lu, J. Niu, M. Heon, L. Hultman, Y. Gogotsi and M. W. Barsoum, Adv. Mater., 2011, 23, 4248–4253 CrossRef CAS PubMed.
- M. Guo, W.-C. Geng, C. Liu, J. Gu, Z. Zhang and Y. Tang, Chem. Mater., 2020, 32, 8257–8265 CrossRef CAS.
- J. Chen, Y. Ding, D. Yan, J. Huang and S. Peng, SusMat, 2022, 2, 293–318 CrossRef CAS.
- D. N. Ampong, E. Agyekum, F. O. Agyemang, K. Mensah-Darkwa, A. Andrews, A. Kumar and R. K. Gupta, Discover Nano, 2023, 18, 3 CrossRef CAS PubMed.
- A. Sikdar, F. Héraly, H. Zhang, S. Hall, K. Pang, M. Zhang and J. Yuan, ACS Nano, 2024, 18, 3707–3719 CrossRef CAS PubMed.
- Q. Jiang, Y. Lei, H. Liang, K. Xi, C. Xia and H. N. Alshareef, Energy Storage Mater., 2020, 27, 78–95 CrossRef.
- J. Wang, G. Li, G. Xie, Z. Huang, P. Zhang, B. Xu, X. Xie and N. Zhang, SusMat, 2024, 4, e202 CrossRef CAS.
- L. Wang, Y. Ding, Z. Xu, J. Li, Y. Guan, L. Yang, H. Gu and H. Fang, Energy Storage Mater., 2024, 65, 103132 CrossRef.
- Y. Zhu, S. Zheng, J. Qin, J. Ma, P. Das, F. Zhou and Z.-S. Wu, Fundam. Res., 2024, 4, 307–314 CrossRef CAS PubMed.
- S. Guo, R. Zhu, J. Chen, W. Liu, Y. Zhang, J. Li and H. Li, Microsyst. Nanoeng., 2024, 10, 89 CrossRef CAS PubMed.
- Y.-Y. Peng, B. Akuzum, N. Kurra, M.-Q. Zhao, M. Alhabeb, B. Anasori, E. C. Kumbur, H. N. Alshareef, M.-D. Ger and Y. Gogotsi, Energy Environ. Sci., 2016, 9, 2847–2854 RSC.
- Q. Han, W. Yang, W. Li, M. Wu, J. Yao, M. Zhao and X. Lu, New J. Chem., 2023, 47, 3516–3523 RSC.
- M. Horn, B. Gupta, J. MacLeod, J. Liu and N. Motta, Curr. Opin. Green Sustainable Chem., 2019, 17, 42–48 CrossRef.
- L. Sun, X. Wang, Y. Wang and Q. Zhang, Carbon, 2017, 122, 462–474 CrossRef CAS.
- Q. Wang, Y. Fang and M. Cao, Nanotechnology, 2022, 33, 295401 CrossRef CAS PubMed.
- H. Xu, J. Zhu, M. Xu, Z. Lei, Q. Hu and X. Jin, J. Colloid Interface Sci., 2023, 645, 974–984 CrossRef CAS PubMed.
- Y. Yue, N. Liu, Y. Ma, S. Wang, W. Liu, C. Luo, H. Zhang, F. Cheng, J. Rao, X. Hu, J. Su and Y. Gao, ACS Nano, 2018, 12, 4224–4232 CrossRef CAS PubMed.
- D. Tu, W. Yang, Y. Li, Y. Zhou, L. Shi, J. Xu and Y. Yang, J. Mater. Chem. C, 2021, 9, 11104–11114 RSC.
- W. Liu, L. Li, C. Hu, D. Chen and G. Shen, Adv. Mater. Technol., 2022, 7, 2200158 CrossRef CAS.
- G. Wang, R. Zhang, H. Zhang and K. Cheng, J. Colloid Interface Sci., 2023, 645, 359–370 CrossRef CAS PubMed.
- X. Huang, J. Huang, D. Yang and P. Wu, Adv. Sci., 2021, 8, 2101664 CrossRef CAS PubMed.
- H. Li, X. Li, J. Liang and Y. Chen, Adv. Energy Mater., 2019, 9, 1803987 CrossRef.
- H.-K. Kim, S.-H. Cho, Y.-W. Ok, T.-Y. Seong and Y. S. Yoon, J. Vac. Sci. Technol., B, 2003, 21, 949–952 CrossRef CAS.
- H. Hu and T. Hua, J. Mater. Chem. A, 2017, 5, 19639–19648 RSC.
- P. Li, W. Shi, W. Liu, Y. Chen, X. Xu, S. Ye, R. Yin, L. Zhang, L. Xu and X. Cao, Nanotechnology, 2018, 29, 445401 CrossRef PubMed.
- P. Salles, E. Quain, N. Kurra, A. Sarycheva and Y. Gogotsi, Small, 2018, 14, 1802864 CrossRef PubMed.
- S. H. Yang, Y. J. Lee, H. Kang, S.-K. Park and Y. C. Kang, Nano-Micro Lett., 2021, 14, 17 CrossRef PubMed.
- W. Kou, Y. Zhang, W. Wu, Z. Guo, Q. Hua and J. Wang, Green Energy Environ., 2024, 9, 71–80 CrossRef CAS.
- Z. Chang, W. Liu, J. Feng, Z. Lin, C. Shi, T. Wang, Y. Lei, X. Zhao, J. Song and G. Wang, Batteries Supercaps, 2024, 7, e202300516 CrossRef CAS.
- C. Hu, Z. Du, Z. Wei, L. Li and G. Shen, Appl. Phys. Rev., 2023, 10, 021402 CAS.
- C. Hu, Z. Wei, L. Li and G. Shen, Adv. Funct. Mater., 2023, 33, 2302188 CrossRef CAS.
- C. Hu, H. Chen, L. Li, H. Huang and G. Shen, Adv. Mater. Technol., 2022, 7, 2101639 CrossRef CAS.
- C. Hu, L. Li and G. Shen, Chin. J. Chem., 2021, 39, 2141–2146 CrossRef CAS.
- C. Hu, R. Chai, Z. Wei, L. Li and G. Shen, J. Semicond., 2024, 45, 052601 CrossRef CAS.
- L. Wang, T. Yang, L. Peng, Q. Zhang, X. She, H. Tang and Q. Liu, Chin. J. Catal., 2022, 43, 2720–2731 CrossRef CAS.
- M. Xu, X. Ruan, D. Meng, G. Fang, D. Jiao, S. Zhao, Z. Liu, Z. Jiang, K. Ba, T. Xie, W. Zhang, J. Leng, S. Jin, S. K. Ravi and X. Cui, Adv. Funct. Mater., 2024, 34, 2402330 CrossRef CAS.
- H. Li, B. Sun, T. Gao, H. Li, Y. Ren and G. Zhou, Chin. J. Catal., 2022, 43, 461–471 CrossRef CAS.
- L. Zhao, H. Xu, L. Liu, Y. Zheng, W. Han and L. Wang, Adv. Sci., 2023, 10, 2303922 Search PubMed.
- Y. Ma, Y. Cheng, J. Wang, S. Fu, M. Zhou, Y. Yang, B. Li, X. Zhang and C. W. Nan, InfoMat, 2022, 4, e12328 Search PubMed.
- A. Y. S. Tan, H. T. A. Awan, F. Cheng, M. Zhang, M. T. T. Tan, S. Manickam, M. Khalid and K. Muthoosamy, Chem. Eng. J., 2024, 482, 148774 CrossRef CAS.
- Y. Yu, P. Yi, W. Xu, X. Sun, G. Deng, X. Liu, J. Shui and R. Yu, Nano-Micro Lett., 2022, 14, 77 CrossRef CAS PubMed.
- H. Wang, Y. Jiang, Z. Ma, Y. Shi, Y. Zhu, R. Huang, Y. Feng, Z. Wang, M. Hong, J. Gao, L. C. Tang and P. Song, Adv. Funct. Mater., 2023, 33, 2306884 CrossRef CAS.
- L. Liu, W. Chen, H. Zhang, L. Ye, Z. Wang, Y. Zhang, P. Min and Z. Yu, Nano-Micro Lett., 2022, 14, 111 CrossRef CAS PubMed.
- F. A. Janjhi, I. Ihsanullah, M. Bilal, R. Castro-Muñoz, G. Boczkaj and F. Gallucci, Water Resour. Ind., 2023, 29, 100202 CrossRef CAS.
- Z. Lei, X. Sun, S. Zhu, K. Dong, X. Liu, L. Wang, X. Zhang, L. Qu and X. Zhang, Nano-Micro Lett., 2021, 14, 10 CrossRef PubMed.
- C. Xing, L. Liu, X. Guo, M. Zhang, M. Zhou, S. Zhang and C. Liu, Sep. Purif. Technol., 2023, 317, 123774 CrossRef CAS.
- B. Akuzum, K. Maleski, B. Anasori, P. Lelyukh, N. J. Alvarez, E. C. Kumbur and Y. Gogotsi, ACS Nano, 2018, 12, 2685–2694 CrossRef CAS PubMed.
- S. Xu, Y. Dall’Agnese, G. Wei, C. Zhang, Y. Gogotsi and W. Han, Nano Energy, 2018, 50, 479–488 CrossRef CAS.
- Q. Jiang, C. Wu, Z. Wang, A. C. Wang, J.-H. He, Z. L. Wang and H. N. Alshareef, Nano Energy, 2018, 45, 266–272 CrossRef CAS.
- S. Zheng, C. Zhang, F. Zhou, Y. Dong, X. Shi, V. Nicolosi, Z.-S. Wu and X. Bao, J. Mater. Chem. A, 2019, 7, 9478–9485 RSC.
- M. K. Hota, Q. Jiang, Z. Wang, Z. L. Wang, K. N. Salama and H. N. Alshareef, Adv. Mater., 2019, 31, 1807450 CrossRef PubMed.
- Y. Su, B. Liu, Q. Zhang, J. Peng, C. Wei, S. Li, W. Li, Z. Xue, X. Yang and J. Sun, Adv. Funct. Mater., 2022, 32, 2204306 CrossRef CAS.
- Z. Duan, C. Hu, W. Liu, J. Liu, Z. Chu, W. Yang, L. Li and G. Shen, Adv. Mater. Technol., 2023, 8, 2300157 CrossRef CAS.
- H. Huang, X. Chu, H. Su, H. Zhang, Y. Xie, W. Deng, N. Chen, F. Liu, H. Zhang, B. Gu, W. Deng and W. Yang, J. Power Sources, 2019, 415, 1–7 CrossRef CAS.
- L. Li, D. Chen and G. Shen, Chem. Res. Chin. Univ., 2020, 36, 694–698 CrossRef CAS.
- S. Deshmukh, K. Ghosh, M. Pykal, M. Otyepka and M. Pumera, ACS Nano, 2023, 17, 20537–20550 CrossRef CAS PubMed.
- J. Yang, J. Cao, J. Han, Y. Xiong, L. Luo, X. Dan, Y. Yang, L. Li, J. Sun and Q. Sun, Nano Energy, 2022, 101, 107582 CrossRef CAS.
- Q. Jiang, N. Kurra, K. Maleski, Y. Lei, H. Liang, Y. Zhang, Y. Gogotsi and H. N. Alshareef, Adv. Energy Mater., 2019, 9, 1901061 CrossRef.
- D. Lee, E. Kim, C. W. Ahn, Y. Lee, U. H. Choi and J. Kim, ACS Appl. Mater. Interfaces, 2024, 16, 26004–26014 CrossRef CAS PubMed.
- Z. Cao, J. Fu, M. Wu, T. Hua and H. Hu, Energy Storage Mater., 2021, 40, 10–21 CrossRef.
- C. Zhang, R. Schneider, M. Jafarpour, F. Nüesch, S. Abdolhosseinzadeh and J. Heier, Small, 2023, 19, 2300357 CrossRef CAS PubMed.
- X. Li, H. Li, X. Fan, X. Shi and J. Liang, Adv. Energy Mater., 2020, 10, 1903794 CrossRef CAS.
- S. Abdolhosseinzadeh, R. Schneider, A. Verma, J. Heier, F. Nüesch and C. Zhang, Adv. Mater., 2020, 32, 2000716 CrossRef CAS PubMed.
- C. Zhang, L. McKeon, M. P. Kremer, S.-H. Park, O. Ronan, A. Seral-Ascaso, S. Barwich, C. Ó. Coileáin, N. McEvoy, H. C. Nerl, B. Anasori, J. N. Coleman, Y. Gogotsi and V. Nicolosi, Nat. Commun., 2019, 10, 1795 CrossRef PubMed.
- Y. Zhu, S. Zheng, P. Lu, J. Ma, P. Das, F. Su, H.-M. Cheng and Z.-S. Wu, Nat. Sci. Rev., 2022, 9, 7 Search PubMed.
- R. Peng, K. Han and K. Tang, J. Electrochem. Soc., 2022, 169 Search PubMed.
- D. Zhao, Z. Chen, N. Fu and Z. Yang, Mater. Today Sustain., 2023, 24, 100523 Search PubMed.
- L. Li, W. Liu, K. Jiang, D. Chen, F. Qu and G. Shen, Nano-Micro Lett., 2021, 13, 100 CrossRef CAS PubMed.
- W. Liu, L. Li and G. Shen, Nanoscale, 2023, 15, 2624–2632 RSC.
- X. Li, Y. Ma, Y. Yue, G. Li, C. Zhang, M. Cao, Y. Xiong, J. Zou, Y. Zhou and Y. Gao, Chem. Eng. J., 2022, 428, 15 CrossRef.
- W. Liu, C. Hu, L. Li and G. Shen, ACS Appl. Energy Mater., 2023, 6, 7387–7394 CrossRef CAS.
- Y. Xie, H. Zhang, H. Huang, Z. Wang, Z. Xu, H. Zhao, Y. Wang, N. Chen and W. Yang, Nano Energy, 2020, 74, 104928 CrossRef CAS.
- W. Chen, Y. Zhou, M. Cao, Z. Shi, Y. Liu, C. Zhang, X. Li and Y. Ma, J. Alloys Compd., 2023, 965, 171488 CrossRef CAS.
- C. Zhang, R. Guo, H. Wang, X. Xie and C. Du, Appl. Surf. Sci., 2022, 598, 153796 CrossRef CAS.
- R. Wang, S. Luo, C. Xiao, Z. Chen, H. Li, M. Asif, V. Chan, K. Liao and Y. Sun, Electrochim. Acta, 2021, 386, 138420 CrossRef CAS.
- E. Kim, J. Song, T.-E. Song, H. Kim, Y.-J. Kim, Y.-W. Oh, S. Jung, I.-S. Kang, Y. Gogotsi, H. Han, C. W. Ahn and Y. Lee, Chem. Eng. J., 2022, 450, 138456 CrossRef CAS.
- S. Zheng, H. Wang, P. Das, Y. Zhang, Y. Cao, J. Ma, S. Liu and Z. S. Wu, Adv. Mater., 2021, 33, 2005449 CrossRef CAS PubMed.
- J. Azadmanjiri, T. N. Reddy, B. Khezri, L. Děkanovský, A. K. Parameswaran, B. Pal, S. Ashtiani, S. Wei and Z. Sofer, J. Mater. Chem. A, 2022, 10, 4533–4557 RSC.
- L. Yu, Z. Fan, Y. Shao, Z. Tian, J. Sun and Z. Liu, Adv. Energy Mater., 2019, 9, 1901839 CrossRef.
- B. Balan, S. Ramasamy, S. P. Rajendra, M. S. AlSalhi and S. Angaiah, Adv. Funct. Mater., 2024, 34, 2408639 CrossRef CAS.
- B. Balan, M. Narayanasamy, S. P. Rajendra, M. S. AlSalhi and S. Angaiah, Adv. Mater. Technol., 2023, 8, 2301329 CrossRef CAS.
- X. Li, S. Xiong, G. Li, S. Xiao, C. Zhang and Y. Ma, Mater. Lett., 2023, 346, 134481 CrossRef CAS.
- C. Couly, M. Alhabeb, K. L. Van Aken, N. Kurra, L. Gomes, A. M. Navarro-Suárez, B. Anasori, H. N. Alshareef and Y. Gogotsi, Adv. Electron. Mater., 2017, 12, 12762–12776 Search PubMed.
- A. Sangili, B. Unnikrishnan, A. Nain, Y.-J. Hsu, R.-S. Wu, C.-C. Huang and H.-T. Chang, Energy Storage Mater., 2022, 53, 51–61 CrossRef.
- Y. Wang, M. Mehrali, Y.-Z. Zhang, M. A. Timmerman, B. A. Boukamp, P.-Y. Xu and J. E. ten Elshof, Energy Storage Mater., 2021, 36, 318–325 CrossRef.
- C. Wu, B. Unnikrishnan, I. W. P. Chen, S. G. Harroun, H. Chang and C. Huang, Energy Storage Mater., 2020, 25, 563–571 CrossRef.
- W. Liu, Z. Du, Z. Duan, H. Liu, L. Li, K. Jiang, D. Chen and G. Shen, J. Mater. Chem. A, 2024, 12, 16457–16465 RSC.
- S. Jiao, A. Zhou, M. Wu and H. Hu, Adv. Sci., 2019, 6, 1900529 CrossRef PubMed.
- S. Radhakrishnan, S. Lakshmy, K. A. Sree Raj, B. Chakraborty, J. S. Cho, S. M. Jeong and C. S. Rout, J. Energy Storage, 2024, 99, 113478 CrossRef.
- J. Tang, W. Yi, X. Zhong, C. Zhang, X. Xiao, F. Pan and B. Xu, Energy Storage Mater., 2020, 32, 418–424 CrossRef.
- C. Zhang, J. Chen, J. Gao, G. Tan, S. Bai, K. Weng, H. M. Chen, X. Ding, H. Cheng, Y. Yang and J. Wang, Nano Lett., 2023, 23, 3435–3443 CrossRef CAS PubMed.
- X. Feng, J. Ning, M. Xia, H. Guo, Y. Zhou, D. Wang, J. Zhang and Y. Hao, Nanotechnology, 2020, 32, 035402 CrossRef PubMed.
- Q. Wang, Y. Fang and M. Cao, Ceram. Int., 2022, 48, 21935–21944 CrossRef.
- X. Feng, J. Ning, B. Wang, H. Guo, M. Xia, D. Wang, J. Zhang, Z.-S. Wu and Y. Hao, Nano Energy, 2020, 72, 104741 CrossRef CAS.
- M. Yuan, L. Wang, X. Liu, X. Du, G. Zhang, Y. Chang, Q. Xia, Q. Hu and A. Zhou, Chem. Eng. J., 2023, 451, 138686 CrossRef CAS.
- A. Levitt, D. Hegh, P. Phillips, S. Uzun, M. Anayee, J. M. Razal, Y. Gogotsi and G. Dion, Mater. Today, 2020, 34, 17–29 CrossRef CAS.
- Y. Zhu, Q. Zhang, J. Ma, P. Das, L. Zhang, H. Liu, S. Wang, H. Li and Z. S. Wu, Carbon Energy, 2024, 6, e481 CrossRef CAS.
- E. Kim, B.-J. Lee, K. Maleski, Y. Chae, Y. Lee, Y. Gogotsi and C. W. Ahn, Nano Energy, 2021, 81, 105616 CrossRef CAS.
- J. Li, A. Levitt, N. Kurra, K. Juan, N. Noriega, X. Xiao, X. Wang, H. Wang, H. N. Alshareef and Y. Gogotsi, Energy Storage Mater., 2019, 20, 455–461 CrossRef.
- L. Yu, L. Lu, X. Zhou, L. Xu, Z. Alhalili and F. Wang, ChemElectroChem, 2021, 8, 1948–1987 CrossRef CAS.
- X. Li, Y. Ma, P. Shen, C. Zhang, M. Cao, S. Xiao, J. Yan, S. Luo and Y. Gao, Adv. Mater. Technol., 2020, 5, 2000272 CrossRef CAS.
- L. Fan, P. Wen, X. Zhao, J. Zou and F. Kim, ACS Appl. Nano Mater., 2022, 5, 4170–4179 CrossRef CAS.
- N. Kurra, B. Ahmed, Y. Gogotsi and H. N. Alshareef, Adv. Energy Mater., 2016, 6, 1601372 CrossRef.
- J. Wei, Y. Teng, T. Meng, X. Bu, H. Du and D. Li, J. Mater. Chem. A, 2020, 8, 24620–24634 RSC.
- C. Ren, S. Qiu, J. Zhai, K. Zhang, J. Lu, J. Gao, C. Wang, Y. Zhang and X.-D. Zhu, Nano Res., 2022, 16, 4926–4932 CrossRef.
- P. Das, X. Shi, Q. Fu and Z. Wu, Adv. Mater., 2020, 30, 1908758 CAS.
- J. Shi, K. Niu, L. Zhang, Q. Chen, M. Deng, L. Sun, S. Cheng and Y. Gao, ACS Energy Lett., 2024, 9, 2000–2006 CrossRef CAS.
- X. Li, X. Ma, Y. Hou, Z. Zhang, Y. Lu, Z. Huang, G. Liang, M. Li, Q. Yang, J. Ma, N. Li, B. Dong, Q. Huang, F. Chen, J. Fan and C. Zhi, Joule, 2021, 5, 2993–3005 CrossRef CAS.
- Y. Zhu, Q. Zhang, J. Ma, P. Das, L. Zhang, H. Liu, S. Wang, H. Li and Z. S. Wu, Carbon Energy, 2024, 6, e481 CrossRef CAS.
- N. Darmiani, A. Irajizad, A. Esfandiar and P. Asen, Energy Fuels, 2022, 36, 2140–2148 CrossRef CAS.
- S. Xu, H. Yang, Y. Li, H. Huang, X. Liang, Y. Wang, Q. Hu, G. Wei and Y. Yang, Chem. Eng. J., 2023, 464, 142645 CrossRef CAS.
- H. Xue, P. H. Huang, L. L. Lai, Y. Su, A. Strömberg, G. Cao, Y. Fan, S. Khartsev, M. Göthelid, Y. T. Sun, J. Weissenrieder, K. B. Gylfason, F. Niklaus and J. Li, Carbon Energy, 2024, 6, e442 CrossRef CAS.
- Y. Wu, A. Lin, J. Zhang, D. Zhao, L. Fan, C. Lu, S. Wang, L. Cao and F. Gu, Front. Chem., 2022, 10, 933319 CrossRef CAS PubMed.
- Y. Zhang, L. Wang, L. Zhao, K. Wang, Y. Zheng, Z. Yuan, D. Wang, X. Fu, G. Shen and W. Han, Adv. Mater., 2021, 33, 2007890 CrossRef CAS PubMed.
- C. Gao, Y. Gu, Y. Zhao and L. Qu, Energy Mater. Adv., 2022, 2022, 9804891 Search PubMed.
- D. Wang, L. Qiao, M. Xia, J. Huang, C. Zhang, J. Ning, X. Feng, J. Zhang and Y. Hao, Chem. Eng. J., 2023, 476, 146567 CrossRef CAS.
| This journal is © The Royal Society of Chemistry 2025 |




