Open Access Article
Open Access ArticleIron oxide nanoparticles: a narrative review of in-depth analysis from neuroprotection to neurodegeneration
Ruchika
Agarwal
a,
Satadal
Adhikary
b,
Suchandra
Bhattacharya
c,
Sohini
Goswami
a,
Dipsikha
Roy
a,
Sohini
Dutta
a,
Abhratanu
Ganguly
 a,
Sayantani
Nanda
a and
Prem
Rajak
a,
Sayantani
Nanda
a and
Prem
Rajak
 *a
*a
aDepartment of Animal Science, Kazi Nazrul University, Asansol, West Bengal, India. E-mail: prem.rjk@gmail.com
bPost Graduate Department of Zoology, A. B. N. Seal College, Cooch Behar, West Bengal, India
cDepartment of Chemistry, A. B. N. Seal College, Cooch Behar, West Bengal, India
First published on 14th March 2024
Abstract
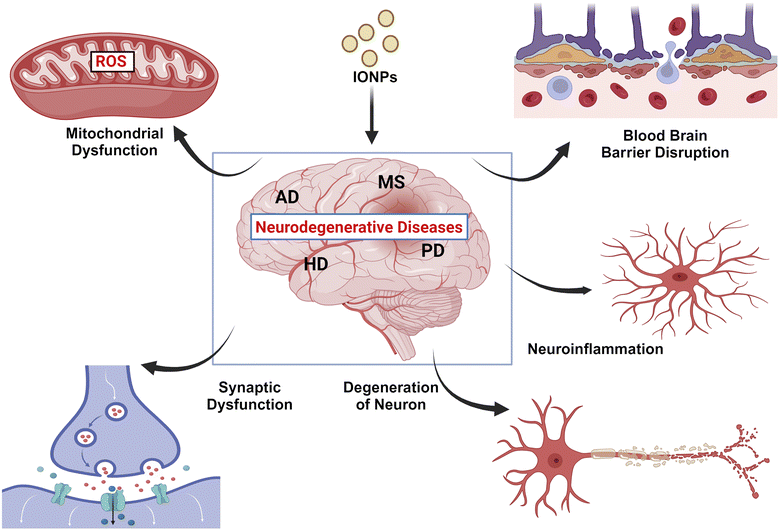
Air pollution is a major risk factor for neurological disorders. Both indoor and outdoor dusts comprise different types of iron oxides in the nano-scale range. Due to their small size and unique physico-chemical properties, iron oxide nanoparticles (IONPs) adopt the intracellular path to agglomerate inside the cell cytoplasm. Moreover, they can cross the blood–brain barrier to invade cortical tissues in the brain and impair neuronal functions. Hence, analysis of the effects of IONPs on the Central Nervous System (CNS) structure and functions is indispensable from medical perspective. A literature search was performed using three scientific databases: ScienceDirect, PubMed, and Google Scholar. Articles published till December, 2023 were screened for their relevancy. Analyses of the appropriate literature have revealed that IONPs are being employed in drug delivery systems and diagnosis of CNS-related ailments that favor neuroprotection. However, the inhalation of IONPs from air and other sources can lead to excessive accumulation of iron in the neuronal tissues, leading to a disturbance in neuronal signaling and augmenting the onset of neurodegenerative disorders. Therefore, it is essential to monitor and control the abundance of IONPs in the environment to combat adverse impacts on the human nervous system.
Environmental significanceAir pollution is a global issue that poses significant detrimental impacts on humans and other animals. Particulate matters of different shapes and sizes are the major air pollutants that harbor Iron oxide nanoparticles (IONPs). IONPs are extensively used in disease diagnosis and drug delivery to facilitate neuroprotection. However, environmental IONPs derived from vehicles and industrial processes are readily inhaled by the global population. IONPs, due to their small size and unique physico-chemical properties, can easily invade the central nervous system and accumulate inside neurons. IONP agglomerization fuels Fenton reaction that produces reactive free radicals such as hydroxyls, superoxides, and peroxides. These reactive entities injure the sub-cellular environment, leading to the induction of ferroptosis and ensuing neurodegeneration. Therefore, proper monitoring and control of the release of IONPs are essential to minimize IONP exposure. |
1. Introduction
Air pollution is a decade-old environmental issue that threatens human health and prosperity. It has been known to fuel cardiovascular ailments, endocrine disruption, breathing issues, and reproductive failure.1 Recently, air pollution has been shown to impair cortical networks of the brain implicated in depression and other mental disorders that have increased the incidence of suicides, mostly in urban areas.2 More specifically, particulate matter (PM) with aerodynamic diameters ≤10 μm (PM10) and ≤2.5 mm (PM2.5) have been reported to affect the cognitive capability of humans by disproportionately augmenting stress-related changes in brain circuitry.3 Particulate matter in the environment harbors iron oxides, even in the nanoscale range, that can enter the human body. The small size of particles enables their easy uptake into the subcellular environment and generates ROS via the Fenton reaction. The main reasons for Iron oxide nanoparticle (IONP) emission in the air are smelting and refining processes. In addition, IONPs are also released from brake wear on vehicles and various industrial sources.1 IONPs can also be released in trace quantities from the fuels and lubricants. Therefore, roadside aerosol could be one of the major sources of IONPs. Higher iron fractions might go beyond 40% of the total mass of PM2.5. Iron oxides have been detected as a dominant metal oxide in particulate matter. IONPs were detected in the range of 10–100 nm in samples collected from Tokyo.4 Indoor dust might carry magnetic IONPs that easily aggregate when coming in contact with lung fluid. It also has a tendency to produce more ROS, particularly hydroxyl radicals, leading to ferroptosis.Different types of IONPs have been detected so far. FeO, Fe3O4, α-Fe2O3, and γ-Fe2O3 have been reported as the most abundant types of IONPs.1 Therefore, it can be opined that environmental IONPs can easily be inhaled by humans where these nanosized particles make their way to sub-cellular moieties. Despite their detrimental impacts on health, IOPNs have several applications in disease diagnosis and pharmaceuticals. For instance, IONPs are widely employed as contrast agents in magnetic resonance imaging (MRI) for the detection of specific cell types. IONPs can cross the blood–brain barrier (BBB) to target carcinogenic growth and aids in treating tumors with magnetic hyperthermia.5
In the air, iron has been recognized in a variety of forms, including metallic Fe (α-Fe), maghemite (γ-Fe2O3), haematite (α-Fe2O3), magnetite (Fe3O4) and wüstite (FeO).6 A variety of anthropogenic sources contribute to the discharge of IONPs into nature. Brake fume is the major source of airborne iron-bearing NPs in roadside areas. IONPs constitute around 50% of the total particle mass released from vehicle brake systems.7 In addition, fiery coal, which is employed in producing 40% of total global electricity, is another main source of airborne IONPs.8 The extremely abundant nanosized particle fraction in metropolitan air has a high bioreactivity and the capacity to invade almost all organs. IONPs are readily taken up by the neural tissues, which aggregate and induce pathogenic alterations, leading to neurodegeneration, mainly in metropolitan cities where the risk of exposure is high due to heavy traffic and industrial prevalance. In regions with higher traffic density and frequent braking, IONPs, mostly in the magnetite form, i.e., Fe3O4 NPs are released into the nature. Fe3O4 NPs have been traced in brain autopsies and are thought to invade the neuronal tissues. Moreover, these IONPs suspended in polluted air could also reach the alveoli of the lungs following inhalation and invade the circulation to contaminate the brain by crossing BBB. This leads to the accumulation of IONPs that trigger oxidative and inflammatory reactions through the Fenton reaction to cause a variety of health issues.9 It is becoming more and more likely that excessive amount of iron in certain brain areas like the frontal lobe, temporal lobe, parietal lobe, caudate nucleus, globus pallidus amygdala, cingulate cortex, and putamen contribute significantly to the pathogenesis of AD and other neurodegenerative illnesses.10
Although dementia is more common in established nations than in developing ones, it is spreading quickly around the globe. Notably, it has been opined that dementia will affect twice as many individuals between 2020 and 2040.11 Air pollution has been associated with the onset of neurological disorders like Parkinson's disease (PD) and Alzheimer's disease (AD). Neuro-inflammation, altered immune response, BBB interruption, and amyloid plaque deposition are fuelled by long-term exposure to numerous air pollutants.12 In neural tissues, iron exhibits dual functions. It is a vital biometal that is found in a variety of metalloproteins, including cytochromes and hemoglobin. Iron is crucial for the myelination of neurons and is involved in many aspects of neural function, including the transmission of nerve impulses, production of neurotransmitters, and generation of mitochondrial energy. Redox cycling and valence changes are used to mobilize and store iron, and complex regulatory mechanisms for storage, transit, and release govern these processes. These procedures are essential because labile iron, or iron that is free and redox-active, has the potential to harm living cells. Excessive iron-induced oxidation injures surface proteins, lipids, and DNA in neural cells. Additionally, IONPs can ignite inflammatory pathways in the brain, causing the discharge of cytokines and chemokines that promote neuroinflammation.13
In light of the information above, we seek to understand the various synthesis methods of IONPs and their beneficial and detrimental effects on the human brain. We also aim to unravel how these particles are transported to the brain and accumulate in various CNS sites, fueling protein aggregation, ROS production, and apoptosis in neuronal cells, ultimately leading to neurodegenerative diseases.
2. Methodology
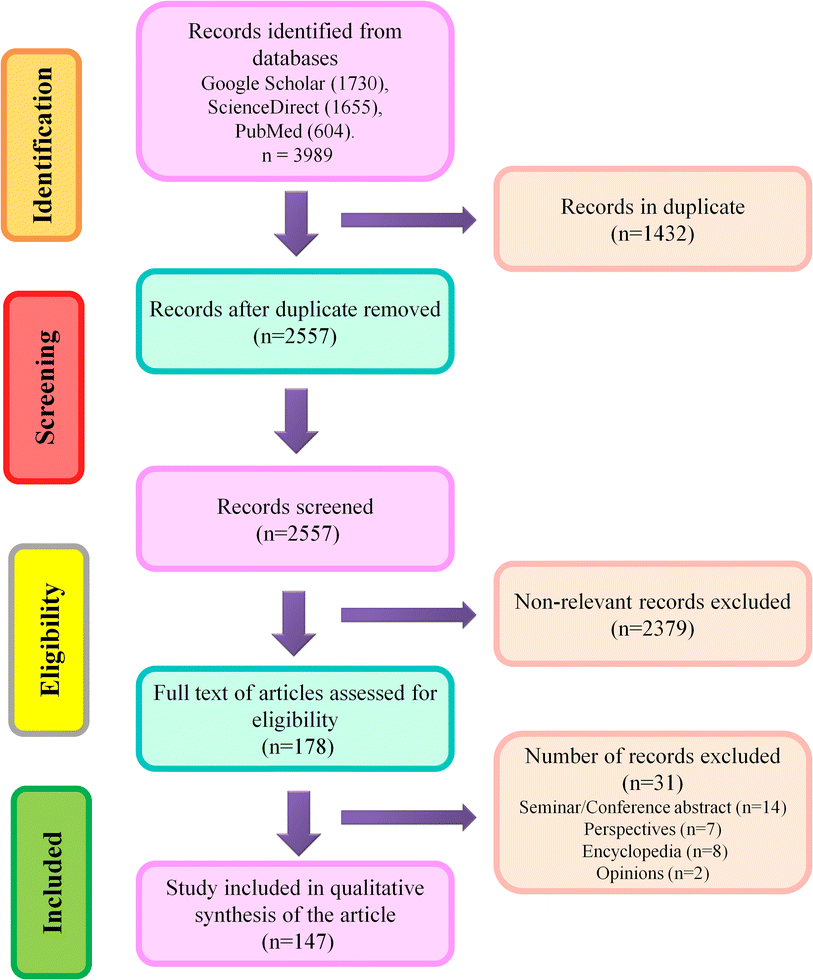
Three scientific databases such as Google Scholar, ScienceDirect, and PubMed, were used to search relevant literature using the search terms which were Iron oxide nanoparticle; Iron oxide nanoparticle toxic impacts; Alzheimer's Disease; and Parkinson's Disease. Authors have carefully screened the original articles published till December 2023, and based on titles, they eliminated identical results. Later, the title, abstract, and keywords were studied to evaluate relevant articles. Disputes were settled following discussion and joint paper revision. Non-original papers, abstracts from seminars or conferences, book chapters, viewpoints, opinions, letters, and encyclopedias were excluded from further consideration. Following an early screening, the entire texts of relevant articles were accessed for the systematic synthesis of the present article.Inclusion criteria: Articles covering the implications IONPs on neuroprotection, detrimental impacts of neurodegeneration on CNS, and alternatives to minimize IONP exposure were included in this study. Exclusion criteria: Abstracts from seminars or conferences, book chapters, viewpoints, opinions, letters, and encyclopedias were excluded from consideration. Any confusion in the selection of relevant literature was resolved through joint discussion. A schematic illustration of the screening process is given in Fig. 1.
3. Natural and artificial production of IONPs
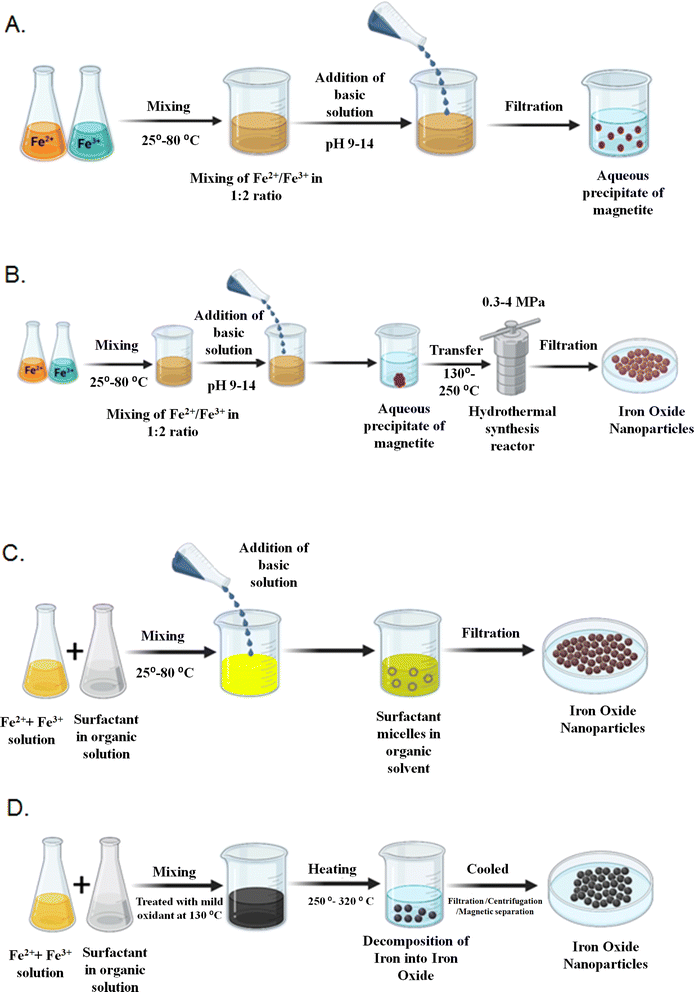
NPs are abundant in our environment. They can be produced by both natural and artificial means.14 Natural NPs are produced by natural processes, such as water mist, woodland fires, dust clouds, volcanic debris, and by biotic factors like bacteria and fungi. For instance, it was recently revealed that the bacteria Actinobacter sp. can synthesize γ-Fe2O3 when coupled with the ferric chloride precursors under aerobic surroundings.15 IONPs have been widely studied over the previous few decades. Numerous studies have detailed effective synthesis techniques that result in shape-controlled, biocompatible, stable, and monodispersed IONPs. The popular techniques which can also be used to produce excellent IONPs, include microemulsion, thermal decomposition, co-precipitation, and hydrothermal synthesis. Additionally, other approaches can be used to synthesize these NPs, including laser pyrolysis techniques,16 electrochemical syntheses,17,18 microorganism or bacterial synthesis, particularly that of iron-reducing bacteria and magnetotactic bacteria.15 Methods widely adopted for the synthesis of IONPs are discussed below.3.1 Co-precipitation
Co-precipitation is the technique used very frequently to yield Fe3O4 or γ-Fe2O3. By combining ferric and ferrous ions in an extremely basic solution at either room temperature or a higher temperature, ferric and ferrous ions are combined in a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 molar ratio. The ferric and ferrous ion ratio, ionic strength of the media, pH value, and reaction temperature change the size and shape of the IONPs. Diverse types of salts like chlorides, perchlorates, nitrates, and sulfates are used during the synthesis. This technique would significantly impact the chemical and physical appearances of the IONPs. If there is no shift in ionic configurations, the saturation magnetization (MS) values observed in nanostructured ingredients are typically lower than those found in the equivalent bulk phase.19 Furthermore, under ambient circumstances, Fe3O4 NPs are readily oxidized to Fe2O3 or dispersed into an acidic medium.
2 molar ratio. The ferric and ferrous ion ratio, ionic strength of the media, pH value, and reaction temperature change the size and shape of the IONPs. Diverse types of salts like chlorides, perchlorates, nitrates, and sulfates are used during the synthesis. This technique would significantly impact the chemical and physical appearances of the IONPs. If there is no shift in ionic configurations, the saturation magnetization (MS) values observed in nanostructured ingredients are typically lower than those found in the equivalent bulk phase.19 Furthermore, under ambient circumstances, Fe3O4 NPs are readily oxidized to Fe2O3 or dispersed into an acidic medium.
3.2 Hydrothermal synthesis
Hydrothermal synthesis encompasses a variety of wet-chemical methods for crystallizing substances in a closed receptacle from high-temperature aqueous solutions ranging from 130 to 250 °C at high vapor pressures ranging from 0.3 to 4 MPa. The method is also employed for synthesizing dislocation-free single-crystal particles. Grains produced in this procedure may have higher crystallinity than grains formed in other procedures, indicating that hydrothermal synthesis is more likely to produce extremely crystalline IONPs.20According to Sun and Zeng,22 a universal breakdown method based on the high temperature (265 °C) reaction of Fe(acac)3 in phenylether in the occurrence of oleyl amine, oleic acid, and alcohol might be used to synthesize size-controlled monodispersed Fe3O4 NPs. The larger monodispersed magnetite NPs can be synthesized and distributed in a nonpolar solvent by seed-mediated growth technique using the smaller magnetite NPs as seeds. The method is easily scaled up for bulk manufacturing and does not need a size-selection process. By heating the synthesized Fe3O4 NPs clusters at a high temperature and in the incidence of oxygen for two hours, it is simple to convert them to γ-Fe2O3 NPs.
Even though the thermal decomposition technique offers a lot of benefits for creating extremely monodispersed particles with a limited size distribution, the main drawback is that the resulting NPs are typically dissolved in nonpolar solvents. The synthesis of IONPs has been represented in Fig. 2. Some of the artificial synthesis methods are summarised in Table 1.
| Method | Solvent | Temperature (°C) | Size range (nm) | Morphology | Advantages | Disadvantages | Reference |
|---|---|---|---|---|---|---|---|
| Hydrothermal synthesis | Water–ethanol | 220 | 520 | Spherical | Temperature-dependent synthesis occurs at very high temperatures | Yield is marginal, high pressure is needed | 134 and 135 |
| Laser pyrolysis | Organic | 5–30 | 1100 | Spherical less large | The production rate is high | In a water solution, the size of the particles is uncontrolled, and extremely high temperature | 135 |
| Co-precipitation | Water | 20–90 | 15–200 | Spherical | The production rate is high and the size distribution is narrow | A lack of good shape control | 135 |
| Thermal decomposition | Organic compounds | 100–320 | 500–550 | Spherical | Size distribution is very narrow, shape control is good, and the product is highly scalable | An inert atmosphere and a high temperature are required for this process | 135 |
| Sonochemical | Water | 10–30 | 25 | Spherical and rod shaped | Small-sized particles are produced | Requirement of high pressure and temperature | 135 |
| Microemulsion | Organic | 20–50 | 4–12 | Spherical or cubic | It has a narrow size distribution and a good shape control, which makes it a good product | Produced at a low rate | 135 |
| Spray pyrolysis | Organic | 5–60 | 400–700 | Spherical but aggregated | Simple and easy production, morphology is uniform, cost-effective | Increasing the temperature (1100 °C) leads to an increase in crystallinity | 135 and 136 |
| Arc discharge | Organic | 5–30 | 1100 | Spherical or less large | Particle shapes can be easily controlled | A lot of purification is often required | 135 |
| Sol–gel | Water | 15–50 | 25–200 | Spherical | Hybrid nanoparticles can be synthesized with desired shapes and lengths | Sol–gel matrix components are usually found on the surface of products | 136 and 137 |
4. Exposure and transportation of IONPs
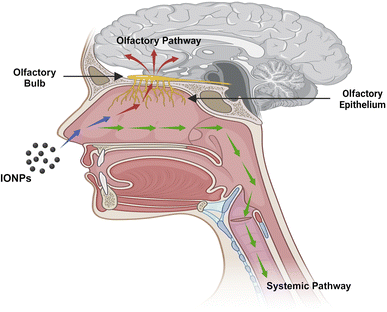
Humans are exposed to IONPs through intravenous administration, ingestion, dermal uptake, and inhalation. All of these routes ultimately employ blood circulation to cross the BBB to enter and harm CNS. The transport of IONPs from the nostrils to the brain via the olfactory epithelium is a significant route to enter the CNS. Therefore, the nasal-to-brain route is a possible channel for the non-invasive delivery of therapeutic NPs straight to the brain, thereby avoiding the BBB. The machinery of transport includes two different pathways, as represented in Fig. 3.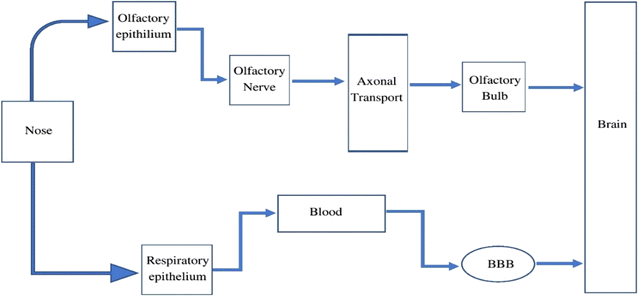 | ||
| Fig. 3 The pathway through which IONPs travel from the nasal cavity to the CNS, potentially involving olfactory nerve pathways or systemic circulation. | ||
4.1 Olfactory pathway
The inhalation of environmental particulate matter can allow IONPs to contaminate the olfactory mucosa. Neuronal receptors in olfactory mucosa are found in the olfactory epithelium and are engaged in nerve impulse transmission. The cilia at the terminal of the olfactory receptor neurons are employed in signal transduction. Paracellular or transcellular mechanisms are used to transport molecules to the odor receptor neurons. Molecules generally cross the cell junctions between cells of the nose epithelium and invade the body via paracellular transport. The neural circuit is critical to constitute the route from the nasal to the brain. The olfactory bulb, on the brain's frontal region, becomes accessible by drug molecules/NPs traveling along an axon via a nerve bundle and crossing the cribriform plate. The olfactory bulb and cerebrospinal fluid can receive the drugs or NPs from the olfactory neurons.23 By combining with the interstitial fluid in the brain, the drug or NPs can be transferred from the CSF to the brain. A substance or neuropeptide takes only a few minutes to reach the brain through olfactory transit. The olfactory neural pathway enters the brain via two distinct routes: the extra-neuronal pathway and the intraneuronal pathway.24 Axonal transport constitutes the intra-neuronal route that enables active compounds to reach the brain, and it might take from a few hours to days. Active molecules can reach the brain in just a few minutes when the transportation route involves perineural channels.25 The cortex, cerebrum, and cerebellum are among the deepest brain regions that are innervated by the olfactory neuronal route.4.2 Systemic pathway
Blood circulation also plays an essential role in the transportation of drugs or NPs to the brain from the nasal passage. Some amount of the drug or NPs are taken into the systemic blood through the nasal mucosa due to the abundant vasculature in the respiratory epithelium.26 The respiratory portion comprises both uninterrupted and fenestrated endothelium, permitting both small and large molecules to enter the bloodstream and be transported to the CNS by crossing the BBB. The BBB is a protective network of blood vessels and brain cells that effectively regulates blood flow to and from the brain. The primary physical barrier of BBB is made up of endothelial cells that are tightly placed. The 80 kDa glycoprotein transferrin (Tf) is the primary iron carrier protein in organisms. The molecule consists of two lobes and has two iron-binding sites consisting of two N- and C-terminal globular units. These sites are formed by the amino acid aspartate, histidine, and two tyrosine to create a 4-atom tetradentate ligand.27 Nearly all iron transport and exchange in the body is carried out by Tf, and healthy adults normally have about 3–4 mg of circulating iron bound to Tf. Two Fe ions, oxidized by ferroxidase and diverted into the interstitium by ferroportin, are loaded into a single Tf unit, with only about 30% of all circulating Tf units engaged at any one time. Tf saturation occurs only with iron overload. Below 1% of circulating iron is typically not bound to Tf and is referred to as non-transferrin-bound iron (NTBI).28The exact mechanism by which iron is exported from endothelial cells is unknown, but it might involve a reduction to Fe2+, transfer across the endosomal cell membrane by divalent metal transporter 1 (DMT1), and export into the extracellular space by ferroportin (FPN1). Much of the Fe2+ that crosses the BBB is oxidized by ceruloplasmin (CP), which is located in astrocytic processes that surround brain endothelial cells. Thereafter, the oxidized form is bound by Tf, which is synthesized by oligodendrocytes. Neurons internalize the Tf-TfR1 complex into endosomes, where iron is isolated from Tf after acidification, transformed to Fe2+, and transported into the cytosol via DMT1 to form part of the labile iron pool. Following that, the liberated iron may be utilized for various cellular functions, kept as ferritin cores, or expelled from the cell by FPN1. It is conceivable that cellular reductants like citrate or ascorbate, as well as circulating ferritin proteins, albumin, and ATP, reduce NTBI, which is discharged by astrocytes and later introduced by the DMT1 of oligodendrocytes. Oligodendrocytes have a lot of iron, which is necessary for the myelination of axons. However, the mechanism explaining the intake of iron by glial cells is less understood. FPN1 combines with amyloid precursor protein (APP), which may act as a ferroxidase at the intracellular membrane of neurons, astrocytes, microglia, and astrocytes. Tf is recycled after Fe2+ release to attach with the Fe3+ in the circulation. CP on astrocyte membranes oxidizes Fe2+ to Fe3+ to bind with the Tf. Neurons absorb both Tf-bound iron and NTBI. NTBI may also attach to citrate and ATP derived from astrocytes in order to serve as an iron supply for oligodendrocytes and astrocytes. Tf is synthesized by oligodendrocytes and is involved in intracellular trafficking. Ferritin, a cytosolic iron storage protein, captures and deposits NTBI in brain cells29 (Fig. 4).
5. Impact of IONPs on CNS
5.1 Beneficial effects
The ferrimagnetic family of magnetic materials, including IONPs, is used in various medicinal and bioengineering applications.30 Mixed ferrites, γ-Fe2O3, and Fe3O4 are three distinct kinds of IONPs. Superparamagnetic IONPs (SPION) synthesized after surface modification can be employed in magnetic particle imaging (MPI), targeted drug and nucleic acid delivery, MRI, biosensing, hyperthermia, biomolecule separation, and tissue repair.31 The magnetic properties of SPION enable its usage in several applications, as it can be synthesized in different shapes and sizes.32 The magnetic moments of SPION are strong when they are subjected to an external magnetic field but disappear following the removal of the magnetic field. The BBB prevents many medications from penetrating the brain at a therapeutic level, which makes imaging, drug delivery, and treatment challenging in cases of CNS-related issues like brain tumors. The primary functions of BBB include protecting the CNS from potentially harmful molecules and maintaining brain homeostasis. BBB protects CNS from hazardous substances and is also responsible for preventing many therapeutic molecules from passing through it to reach their targets in the brain. As a result, some molecules are allowed to pass through passive diffusion, while other essential substances, such as amino acids, glucose, and water, are selectively transported across the BBB to maintain the normal functions of neurons in the CNS.Due to the BBB, some tumors, such as metastatic tumors and brain gliomas, cannot be chemotherapeutically treated. A variety of methods can be employed to overcome BBB in the case of chemotherapy drugs, such as the use of intrathecal drug administration or the use of olfactrogenic agents. The drugs can also be delivered through the BBB in small doses if the BBB is disrupted biochemically, osmotically, or using vasoactive substances such as bradykinin to increase the flow to the brain.33 Certain diseases, such as cancer, are known for having poor prognoses or the failure to undertake early detection or anticipate their pathological pathways, thereby exposing patients to high morbidity and mortality rate, particularly for metastatic tumors. Therefore, there is an increased need for theranostics.34,35 Theranostic drugs are safe, tolerable, and have both therapeutic and diagnostic potential in a single substance. Due to their distinctive characteristics and simple surface manipulation, IONPs have strong prospects for theranostic uses. Theranostic IONPs can be employed as a transport mechanism for cell replacement procedures, MRI-assisted tumor detection or surgical excision, and targetable gene transfer using an external magnetic field. An integrated theranostic system consisting of IONPs in MRI contrasting agent and doxorubicin (DOX) as a chemotherapeutic agent has been developed, which are useful in diagnosing and treating diseases.
Since uncoated IONPs with a size of 16 nm tend to precipitate or aggregate, IONPs are coated with biocompatible substances.36 The process of cellular absorption and the fate of IONPs depend on the hydrophilicity and size of the surface-modifying materials. Particles having a size >100 nm are strongly phagocytosed, whereas those that are <30 nm undergo pinocytosis. Increased hydrophilicity lowers pinocytosis by hindering a particle from being transported through the hydrophobic lipid bilayer of the plasma membrane.37
Regarding toxicity, ultrasmall IONPs of size 2.3 and 4.2 nm have been demonstrated to be highly toxic. It shows toxicity at 100 mg kg−1 dosage in the mice model. On the other hand, no clear toxicity was reported for IONPs having a size of 9.3 nm. Additionally, the study also revealed that the toxicity of small nanoparticles could be diminished by splitting the dosage into four doses at five-minute intervals.38
BBB can also be breached by endogenous transport systems, including insulin receptor-mediated transcytosis, carrier-mediated transporters, such as glucose carriers and amino acid carriers, or by inhibiting active efflux transporters, such as P-glycoprotein. Vectors that target BBB transporters, for example, the transferrin receptor (TfR), are known to remain entrapped in brain capillaries rather than traveling through the BBB to the targeted location.39 Several ways to transport medications across the BBB include convection-enhanced diffusion and intracerebral insertion using needles. The beneficial effects of IONPs are illustrated schematically in Fig. 5.
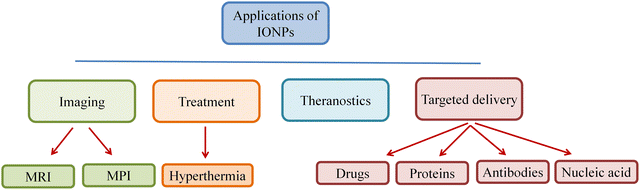 | ||
| Fig. 5 Multiple applications including targeted drug delivery and diagnostic imaging of IONPs in medical science. | ||
IONPs often exert neuroprotective roles to ameliorate the nanotoxic effects of certain compounds. For example, a natural phytochemical, sulforaphane (SF) along with Fe3O4-NPs carrier (N.SF), was found to be beneficial against the chemotherapeutic drug cisplatin (CIS), which shows severe side effects of neurotoxicity. The intranasal administration of N.SF to the CIS-treated rats were found to have neuroprotective functions on the biochemical, histopathological (striatum and cerebral cortex), and behavioral aspects.40 Other studies have shown the neuroprotective efficacy of Fe3O4-NPs in fruit fly models by executing catalase-like functions. Its neuroprotective roles have been established in PD and AD models in Drosophila. IONPs, when administered through the dietary route amongst aged Drosophila, ROS levels are diminished the flies performed better climbing performance, and their lifespan was increased. Additionally, IONPs have also been demonstrated to increase the life span of Drosophila in the AD model and alleviate the neurodegenerative condition. The neuroprotective role in the PD model was found to be exerted by means of retreating the accumulation of α-synuclein and the activation of caspase-3.41 Similar neuroprotective role of IONPs upon implantation at the lesion site and exposure to a magnetic field in an in vivo 6-OHDA rat model of PD have been established as demonstrated by the substantial improvement of mitochondrial functionality and healing of the scar.42
5.2 Detrimental effects
Several factors of IONPs, like the initial concentration, shape, size, functional group, coating, and cell type, affect the aggregation of iron inside a cell. Depending on the IONP dosage, the amount of iron in the cells increases.48 Furthermore, the amount of IONPs also influences their retention time in tissues. When IONPs are administered in high amounts, it cannot be entirely removed from the systemic circulation. However, a small amount can be eliminated within three weeks.49 As a result, the elevated dose of IONPs exposes the cells to IONPs for a long time. In the cell, comparatively small-sized IONPs may aggregate in greater concentrations than bigger ones.50 Spherical NPs degrade more slowly than cubes because they have less contact area for degradation. Compared to citrate coating, dextran coating on IONPs reduces the degradation rate.
Positively charged IONPs with amine groups are more easily absorbed by the cells than the IONPs with negatively charged carboxyl or hydroxyl groups.51 Cell type is a vital factor for IONP absorption. The brain endothelial cells absorb a smaller quantity of IONPs, whereas microglia absorb a large amount of IONPs.51 Any element that alters IONP uptake or degradation rates can impact iron deposition in tissues or cells.
In spite of its medical applications, the biomedical administration of magnetic IONPs has aroused critical concerns regarding their pharmacokinetics i.e. their distribution in various organs after absorption, eventual metabolism, and finally excretion, which as a whole contributes to IONP-induced toxicity. Adsorption of plasma proteins greatly affects the fate and distribution of IONPs after their administration. Human serum albumin when adsorbed to IONPs, showed longer circulation time in blood.52,53 Endosomes/lysosomes are the sites where the IONPs reside after their uptake by the cell and subsequently liberate into the form of free iron and add up into the cellular iron pool. Overabundance of intracellular iron oxidizes and damages the cellular proteins and nucleic acids. Up-regulation of ferritin and down-regulation of transferrin receptors in labeled cells may protect cells from possible cytotoxicity led by higher intracellular iron concentrations.54 Researchers have deployed colloidal IONPs like the superparamagnetic iron oxide nanoparticles (SPIONs) and ultrasmall superparamagnetic iron oxide (USPIO) nanoparticles as contrast agents (CA) in magnetic resonance imaging (MRI). SPIONs have their speciality in not showing magnetization without the presence of the external magnetic field. SPIONs, along with their features of high saturation magnetic moment and chemical stability also show minimized cellular toxicity.55,56
According to a study, endothelium cells exposed to 10 nm-IONPs may experience a marked rise in ROS levels.51 However, the bare NPs in the 30 nm size induced the production of ROS, while the bare IONPs in the 5 nm size did not change the level of ROS.48 Therefore, it is apparent that surface charge and coating determine the interaction of IONPs with cellular components.52 Coating IONPs with aminopropyl trimethoxysilane (APTMS) can be detrimental to cell viability and DNA content. A greater amount of IONPs is endocytosed by cells and are concentrated in the cell as a result of surface amine groups. It is more likely that the positively charged APTMS coated IONPs interact with the negatively charged DNA.44
| Organism | Method | NP type | Size (nm) | Concentration | Coating | Functional group | Explanation | Reference |
|---|---|---|---|---|---|---|---|---|
| Rat | Intranasal | Fe2O3 | 36 | 20 μg μL−1 | APTS | Amine | IONPs are delayed in being removed from the stratum and hippocampal region because of increased oxidative stress | 138 |
| Rat | Oral | Fe2O3 | 30 | 30 mg per kg per day, 300 and 1000 mg per kg per day | Bare (none) | Hydroxyl | It inhibits Mg2+, Na+–K+ and Ca2+ ATPases in the brain, reduces body weight, and inhibits acetylcholine esterase in the brain | 62 |
| Rat | Intravascular injection | Fe2O3 | 10 | 50 mg kg−1 | Bare (none) | Hydroxyl | Dopaminergic neurons were damaged and dopamine levels in the striatum were significantly decreased | 51 |
| Rat | Intratracheal instillation | Fe3O4 | < 50 | 1 and 5 mg kg−1 | Bare (none) | Hydroxyl | Body weight reduction | 139 |
| Rat | Injection into stratum | Fe3O4 | 6.5 + 3.0 | 3 μ NP 2 μL−1 in a CSF | Bare (none) | Hydroxyl | A reduction in oxidative stress within the stratum and an increase in body weight | 42 |
| Drosophila | Exposure | Fe3O4 | 20 | 200 μg mL−1 | Bare (none) | Hydroxyl | The ROS levels of six-week-old flies were significantly reduced, and their climbing ability was enhanced, and their longevity increased | 140 |
| Mouse | Intravascular injection | Fe3O4 | 5 | 0.4, 2, and 10 μg kg−1 | PEG | Hydroxyl | Heart cells are induced to undergo DNA damage and oxidative stress | 141 |
| Water fleas | Exposure | Fe2O3 | 33.3 | 10–100 mg L−1 | Bare (none) | Hydroxyl | A significant amount of toxicity is possessed by coated and uncoated IONPs when ingested and accumulated in the gastrointestinal tract | 140 |
| (Daphnia magna) | 50.4 | 1–100 mg L−1 | Dimercaptosuccinic acid | Thiol and carboxyl | ||||
| Green algae | Exposure | Fe2O3 | 33.3 | 1–100 mg L−1 | Bare (none) | Hydroxyl | IONPs coated or uncoated inhibit growth after 72 hours | 140 |
| (Raphidocelis subcapitata) | 50.4 | 1–100 mg L−1 | Dimercaptosuccinic acid | Thiol and carboxyl | ||||
| Duckweed | Exposure | Fe2O3 | 33.3 | 1–100 mg L−1 | Bare (none) | Hydroxyl | In this range of doses, NPs have no effect | 140 |
| (Lemna minor) | 50.4 | 1–100 mg L−1 | Dimercaptosuccinic acid | Thiol and carboxyl |
6 Association between IONPs and neurodegenerative diseases
6.1 Alzheimer's disease (AD)
AD is the most prevalent form of dementia worldwide. AD is a growing neurodegenerative disease that is first categorized by short-term memory loss, a stage referred to as mild cognitive impairment (MCI) and progresses to aphasia, and gradually to agnosia, i.e., the inability to recognize people or things followed by apraxia, which is the inability to perform voluntary movements along with other memory and behavioral problems.65 Clinically, the illness stage is evaluated by psychological assessments such as the mini-mental state examination (MMSE), which includes patients responding to a series of questions designed to measure their level of cognitive ability.66The expected lifespan of a person diagnosed with AD is 3 to 10 years. The disease typically manifests around the age of 65; however, it can affect much younger people in cases where a familial link has been established.67 The majority, i.e., 95% of AD cases, are sporadic (SAD), with less than 5% being genetic or “familial” (FAD). There is a clear medical demand for the treatment of AD since present therapies merely provide symptomatic relief rather than treating the underlying illness cause. Currently, two types of AD therapeutics are utilized such as glutamate antagonists and acetylcholinesterase (AChE) inhibitors, like donepezil and rivastigmine.68 Glutamate antagonists inhibit the overstimulation of glutamate in AD.69
Senile plaques, which are external Aβ deposits, and tangles of neurofibril, which are intracellular clumps of hyperphosphorylated tau protein, are the two primary neurological symptoms of AD.70 Other structural changes to the brain are also noted, including a reduction in the weight of the brain because of the loss of grey matter, a rise in ventricle size due to CSF aggregation, and a decrease in the thickness of the cortex.71,72 The medial temporal lobe, which includes the hippocampus, entorhinal cortex, and amygdala, is one of the first brain regions affected by AD.
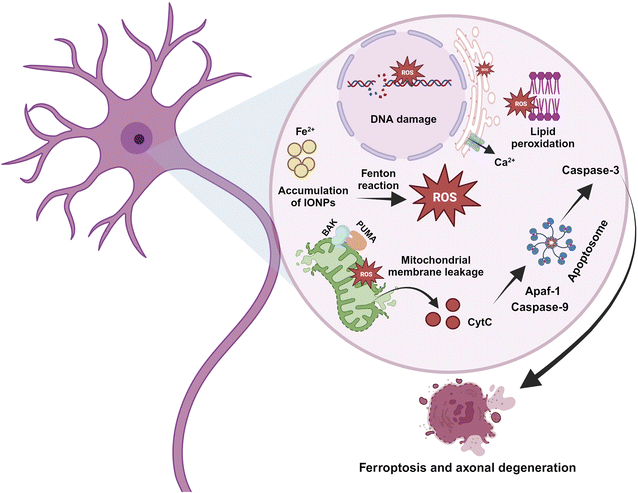
The metal hypothesis or ferroptosis hypothesis is one of the many theories proposed to cause AD. In contrast to other modes of cell death, like necrosis and apoptosis, ferroptosis is a type of programmed cell death. Iron overload is a defining feature of ferroptosis, a disorder where lipid peroxides build up to lethal quantities due to iron.73 It involves brain iron accumulation, lipid peroxide, and glutathione depletion simultaneously, which causes a cascade of events that includes neuronal communication failure, astrocyte dysregulation, neurotransmitter oxidation, myelin sheath degeneration, dementia, and cell death.
The changes in the morphology of cells undergoing ferroptosis include nuclear chromatin condensation, decreased mitochondrial membrane density, reduced or absent mitochondrial cristae, cell swelling, and ultimately, cellular as well as mitochondrial membrane disintegration.74
6.2 Parkinson's disease (PD)
PD is a neurological illness marked by a gradual degeneration of dopaminergic neurons in the substantia nigra (SN) of pars compacta. In addition to iron aggregation, increased production of inflammatory markers and ROS occur more often. These findings suggest that the dyshomeostasis of iron may be a major factor in neurodegeneration. A growing number of people are affected by PD, which affects one in ten people over the age of 60, due to the increase in life expectancy in nations with a huge number of industries.75 Aceruloplasminemia and neuroferritinopathy are conditions associated with iron accumulation in certain brain areas, like SN, leading to PD. Animal models of PD show an increased level of iron in the SN when treated with 6-hydroxydopamine (6-OHDA). These data show a connection between iron accumulation and neurodegenerative disorders.76 The accumulation of iron in the brain of PD patients correlates with a decline in Tau and APP levels. Tau interacts with APP to enhance iron export, while APP stabilizes FPN1 on the cell membrane.77 This suggests that a disturbance in the iron homeostasis in the brain may be a factor triggering iron accumulation in PD.6.3 Multiple sclerosis (MS)
MS is a degenerative and inflammatory condition that impairs the myelin sheath that surrounds axons and the oligodendrocytes that produce myelin. Several factors are identified that contribute to the neurodegeneration occurring in MS, including inflammatory molecules, protease dysfunction, mitochondrial dysfunction, and glutamate excitotoxicity. MS is increasingly associated with iron metabolism dysregulation and its cytotoxicity. MRIs are employed to examine the deposition of iron in the brains of patients suffering from MS. Increased iron concentration in deep grey matter areas has been discovered using MRI and is linked to grey matter atrophy. MRI results indicate the presence of oxidative damage and iron accumulation in deep grey matter. The mitochondrial function is further impaired in patients with MS, which is caused by frequent damage to mitochondria and the subsequent rise in ROS, which in turn leads to the loss of genomic integrity and further impairments to mitochondrial function due to iron-mediated oxidative damage.78,796.4 Huntington's disease (HD)
HD is a growing neurodegenerative disorder characterized by cognitive, psychiatric, and motor dysfunction, leading to dementia. Brain areas in HD, particularly the cerebral cortex and striatum, experience gradual degeneration beginning several years prior to clinical onset.80 In order to account for the pathogenetic mechanisms of HD, a number of factors have been implicated, including oxidative stress, dysfunction of oxidative phosphorylation, transcriptional dysregulation, and defects in axonal transport.81–83 The caudate striatum and putamen are believed to be the first structures to show signs of neuronal degeneration in HD. Once the caudate and putamen striatum structures are affected, the degeneration spreads to other myelinating regions, with the medial temporal lobe being considerably less affected.84 Due to a surge in iron deposition in the basal ganglia, several neurological disorders have been linked with injuries in the tissue. These diseases include PD, MS, AD, and other metabolic disorders.85 HD is also known to be associated with alterations in iron metabolism. Gene knockdown models have shown iron-dependent localization, oxidation, and structural organization of mutant htt inclusion bodies produced by knockdowns of the human htt gene.86 Some diseases of CNS are caused by iron overload, and the affected areas of the brain are summarised in Table 3.| Neurodegenerative diseases | Description | Brain regions affected | References |
|---|---|---|---|
| Alzheimer's disease | Alzheimer's disease leaves people unable to perform even the most basic tasks due to the slow deterioration of memory and thinking abilities | Iron elevation in cortical, hippocampal, and cerebellar neurons | 142 |
| Parkinson's disease | Parkinson's disease (PD) is a neurological disorder marked by a gradual loss of dopaminergic neurons in the pars compacta of the substantia nigra (SN) | Pars compacta of the substantia nigra | 143 |
| Huntington's disease | Huntington's disease (HD) refers to a progressive neurodegenerative disorder which is characterized by psychiatric, cognitive, and motor dysfunction that leads to dementia | Striatum and cerebral cortex | 144 |
| Neuroferritinopathy | In neuroferritinopathy (NF), abnormal excessive iron accumulation occurs primarily in the basal ganglia of the brain due to mutations in the ferritin light chain 1 (FTL1) gene | Basal ganglia | 145 |
| Amyotrophic lateral sclerosis (ALS) | Amyotrophic lateral sclerosis (ALS) is a neurodegenerative disorder characterized by motor neuron loss | Cerebral cortex, spinal cord, and brain stem | 146 |
| Pantothenate kinase-associated neurodegeneration (PKAN) | A mitochondrial pantothenate kinase 2 gene mutation, located at locus 20p13-p12.3, leads to pantothenate kinase-2-associated neurodegeneration | Globus pallidus | 147 |
| Multiple sclerosis | Multiple sclerosis (MS) is a degenerative and inflammatory condition that causes damage to the myelin sheath which surrounds axons and the oligodendrocytes that produce myelin | Elevated level of iron in deep grey matter, i.e., putamen and caudate | 14 |
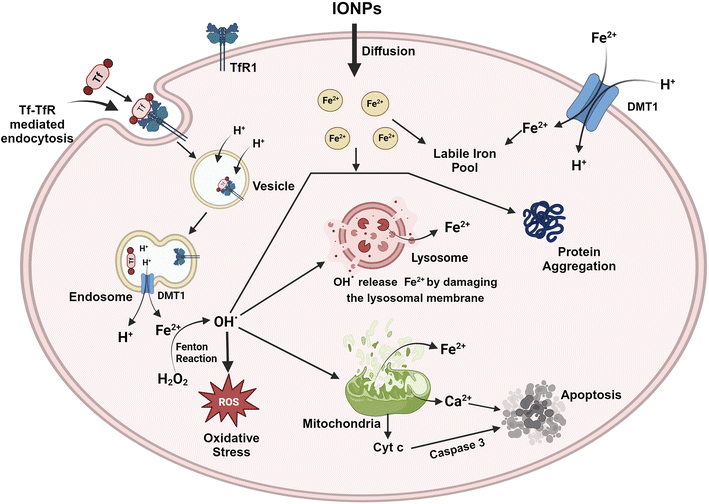
7. Molecular mechanism of IONP-induced neurodegeneration
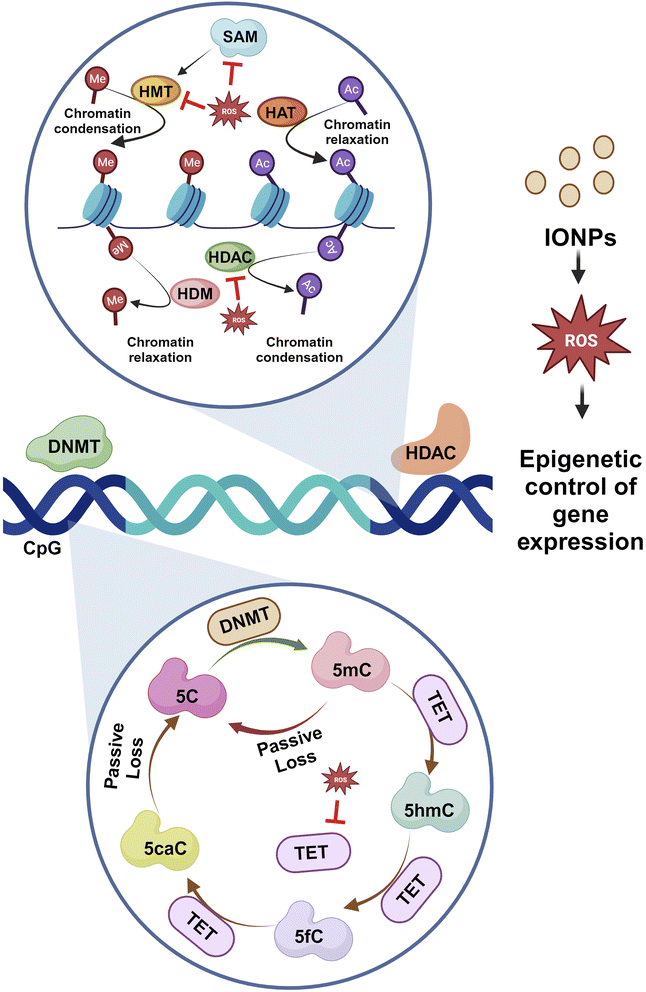
IONPs enter the BBB through the cells that line its endothelium via the TfR1-mediated endocytosis of Tf, independently as NTBI, or as a low molecular weight complex.87 As circulating diferric-transferrin binds to transferrin receptors lining the cerebral lumen, IONPs are likely to be taken up by brain vascular endothelial cells by receptor-mediated endocytosis. Astrocytes are positioned perfectly to absorb iron from the blood and circulate it to different cells present in the CNS. Astrocytes contain the iron efflux as well as the influx mechanism necessary for cell-to-cell iron transfer. The expression of DMT1 by astrocytes is thought to mediate the iron influx into the glial cells.88 Iron that is stored in astrocytes as ferritin is exported by a mechanism that employs CP as well as FPN1.89 Oligodendrocytes take up iron via the non-vesicular iron import mechanism, or NTBI via DMT1, or ferritin receptor Tim-2. Iron can be influxed into neurons and microglia via Tf-TfR-mediated uptake and effluxed via FPN1.87 The iron, exported from the endosomes via DMT1, contributes to the labile iron pool, which constitutes the iron content available in neurons. A major iron storage protein within neurons, ferritin, is regulated by iron-binding and cellular metal-sensing proteins, and then iron is recirculated by apo-Tf.13 The high incidence of iron can be toxic to the brain even though iron plays a crucial role in many metabolic processes. Thus, any change in the iron level may be detrimental to human health. IONPs can lead to oxidative stress, iron aggregation, and protein agglomeration. The surface area of IONPs is large, allowing them to participate in redox cycling. Additionally, the iron ions generated by IONPs could potentially participate in the Fenton reaction, producing OH from H2O2 and ultimately causing oxidative stress. ROS can directly impair the cell membrane, the membrane of the organelle, and DNA. ROS leads to the release of cytochrome C and Ca2+ from mitochondria and hence induces apoptosis.63 In addition, ROS leads to iron aggregation in the cytosol, and the iron is released from mitochondria as well as lysosomes by destroying the membranes of the organelles.90 ROS and free iron affect several proteins, which alter their conformation and facilitate aggregation. Several iron-sensitive brain areas can be damaged by iron dyshomeostasis. Neurodegeneration is associated with iron accumulation in the brain as a result of iron overload in the basal ganglia. PD along with AD are two sporadic neurodegenerative diseases that have been linked to high levels of iron and pathogenic triggers that are related to iron. Between oxidative stress, iron accumulation, and protein aggregation, there is a loop with positive feedback where one element encourages the other. By inducing protein agglomeration, oxidative stress, or iron aggregation, IONPs can turn on this loop. Additionally, IONPs may cause the neurons to undergo apoptotic cell death. The molecular mechanism is represented with the help of Fig. 8.IONPs tend to modulate the epigenetic activation of transcription by activating enhancers. In a study, IONPs like Fe2O3 and Fe2O3@Co NPs alter the genomic distribution of H3K27ac, which causes alterations in the activity of enhancers.91 In some cases, exposure to γ-Fe3O4 NPs could increase the cytosine methylation in the genome while disrupting genomic stability.92 Gene expression is controlled by a precise balance between methylation and acetylation events. DNA methylation is a dynamic process that covalently adds methyl groups to C5 of cytosine, mainly at CpG islands of promoters, leading to the generation of 5-methylcytosine (5-meC). Enzymes, mainly DNA methyltransferase, DNMT1, DNMT 3A, and DNA methyltransferase DNMT3B, play crucial roles in such events. Here, S-adenosyl-L-methionine acts as the methyl donor. Another enzyme referred to as ten-eleven-translocation (TET) catalyzes the conversion of 5-meC to 5-hydroxymethylcytosine (5-hmeC) and 5-carboxycytosine to assist in the demethylation process. Exposure to IONPs can trigger the methylation of histone, H3K9, and H3K27 as well as the reduced methylation of histone K3K4 and H3K36 at the 5′ end. In addition, metal NPs can contribute to the demethylation of cytosine that contributes to epigenetic changes and aberrant gene expression.93 Many of the miRNAs are essential to promote neural development. IONPs have been documented to reduce expressions of miRNAs that could significantly affect the entire epigenetic landscape.94 miRNA-124 suppresses PTBP1, leading to differentiation in neuronal cells rather than non-neuronal cells. Certain miRNAs also control the development and maturation of synapses and dendrites. Therefore, unusual alterations in DNA methylation and acetylation process through changes in enzymes and regulatory RNA activities can impact neurons, and such re-editing events contribute to neurodegenerative diseases.95
DNA methylation is crucial for memory acquisition and storage.96 The loss of function in DNMT1 and DNMT3A is associated with perturbed hippocampus-mediated memories.97 The methylation of CpG islands at promoter sites results in the formation of 5-methyl-cytosine (5mC), which is further converted into 5hmC, 5-formyl-cytosine(5fC), and 5-carboxy-cytosine (5caC) by methylcytosine dioxygenases ten-eleven translocation proteins (TET).98 The increased activity of DNMT1 is linked to the enhanced methylation of the gene for α-synuclein (SNCA). Reduced SNCA methylation at intron 1 is evident in sporadic patients.99 Histone acetylation is inhibited when SNCA binds to H3. Therefore, blocking the activity of HDAC could help prevent neurotoxicity. Studies have revealed that the inhibition of HDAC Sirtuin 2 reduces SNCA-mediated neuronal damage.100 SNCA also downregulates genes encoding for histones H1, H2B, and H4.101 Increased activities of HDAC2 and HDAC6 in the hippocampus and prefrontal cortex of AD are associated with impaired cognition. Reduced methylation, as evidenced by less 5mC, 5Fc, 5hmC, and 5caC in the entorhinal cortex and cerebellum, is linked to the onset of AD.102 Reduced methylation of cytosine residues at 207–182 of amyloid precursor protein results in elevated amyloid-β (Aβ) deposition in the brain, contributing to AD. The epigenetic modification of histones like H3 methylation, H2B ubiquitination, and H4 acetylation has been associated with Huntington's disease.95 Therefore, histone modification is critical in PD development (Fig. 9).
8 Future prospective to minimize IONP exposure
Exposure to IONPs impart negative impacts on human health, especially if it involves the CNS. As a result, it is extremely important to take the necessary precautions to lower the exposure of humans to IONPs as much as possible. Individuals can be protected against exposure to IONPs through several measures, some of which are discussed in the following subsections.8.1 Personal protective equipment
Workers dealing with high levels of iron oxide emission in industries are routinely exposed to these hazardous particles. The amount of iron oxide that enters the body through the nasal route or skin contact can be reduced using personal protective equipment, such as gloves, respirators, and protective clothing, when exposure to iron oxide is unavoidable.It may be necessary to wear a half-facepiece respirator with a particular filter with a National Institute for Occupational Safety and Health (NIOSH) type N95 or better filter if the exposure limit is expected to be breached and engineering controls are unfeasible. It is permissible to use this respirator up to ten times the maximum exposure limitation or the highest permitted concentration specified by the regulatory body or respirator manufacturer, whichever determines the lower exposure limit. In the case of particulate respirators with full-face pieces, such as NIOSH-type N100 filters, the maximum exposure limit can be 50 times the highest concentration outlined by the regulatory body or respirator manufacturer. A NIOSH-type P or R filter should be used if oil particles, such as glycerine, cutting fluids, and lubricants, are found.
It is recommended to wear a clean garment that covers the entire body and protective gloves in order to protect the skin. Safety goggles should be worn when handling chemicals. The work area should have an eye wash fountain and a quick-drill facility to add further safety.103
8.2 Electric brakes replacing conventional brakes
During the frictional braking system operation, a large volume of dust containing IONPs is emitted into the environment, which manifests in people in the form of lung diseases and several neurodegenerative diseases like Parkinson's disease (PD), Alzheimer's disease (AD), Multiple sclerosis (MS) and Huntington's disease (HD). There is no doubt that electric and hybrid vehicles need brake systems as well as their combustion engine counterparts; however, those vehicles are equipped with electric motors that facilitate the electric braking system and are known as regenerative brake systems. Unlike friction brakes, the regenerative brake system does not demand friction pads, which can lead to cleaner operation and better conversion of the braking energy into electric energy. In a handful of studies, a variety of control methodologies have been utilized to control the electric brake system, resulting in a reduction of 70–80% of mechanical brake participation. In order to minimize the contribution of frictional brakes in a conventional vehicle, the technology for braking electric automobiles can be incorporated into the automobile braking system so as to reduce brake dust emission into the air that occurs during friction braking.1048.3 Regular maintenance of braking systems, equipment and machinery
The regular maintenance of braking systems may be effective in reducing the quantity of IONPs that are released during the operation of the vehicle. The rotors, brake pads, and other components of the automobile braking system must be thoroughly inspected and replaced when necessary. When low-dust brake pads are used, fewer particles are created when they come into contact with the rotor, resulting in a reduction of IONPs that is liberated into the atmosphere.105 Keeping equipment and machinery in good working condition can help reduce the emissions of IONPs and eventually minimize the exposure. A part of this process involves cleaning and changing filters, inspecting and fixing equipment, and taking preventive maintenance measures to ensure everything runs smoothly.8.4 Biological remediation of IONPs
Biological remediation refers to the removal of pollutants and neutralizing it using living organisms. Through the process of biocorrosion, Bacillus sp. (SO-10) can facilitate the bioremediation of iron contaminants.There are several processes that occur naturally to break down iron and other metals, and biocorrosion is one of them. Using iron as a source of energy and nutrients, these bacteria produce metabolic by-products, causing metal structures to corrode faster. While this may seem like a very undesirable aspect of bioremediation, the goal of bioremediation is not to encourage biocorrosion but rather to utilize the metabolic processes of these bacteria to eliminate or neutralize the iron contamination present in the environment. An example of this is the introduction of Bacillus sp. (SO-10) into soil or water that has been contaminated with iron-based pollutants in order to degrade or transform the pollutants into less hazardous forms.
It is essential to consider that the effectiveness of bioremediation in the use of Bacillus sp. (SO-10) depends upon a lot of different factors, such as the types and concentration of the contaminants, the environment, their pH, and the availability of nutrients, i.e., a carbon source. To ensure that bioremediation is successful, it is also essential to monitor and manage the bacteria carefully to ensure that any possible risks or unforeseen repercussions are reduced to a minimum.
As a whole, bioremediation with Bacillus sp. (SO-10) can be an effective tool to address iron pollution in various settings, including wetlands, industrial sites, and ecosystems in general. However, there is a need to carefully assess the potential risks and feasibility of this approach in each specific circumstance to avoid any unforeseen consequences.106
9 Limitations
The safety of IONPs for human use is one of the major challenges preventing its clinical translation. In spite of the fact that IONPs have been approved and used in clinics, such as contrast media and iron replacements,107,108 various studies have found that factors such as size, composition, route, surface, properties, and dose of administration can potentially affect their safety. According to several studies, most IONPs do not leave the body and accumulate in vital organs such as the spleen and liver and cause toxic effects.109 Cell toxicity has been demonstrated to be significantly affected by the size and shape of IONPs. Small-sized IONPs, i.e., less than 5 nm, are responsible for generating ROS through Fenton and Haber–Weiss reactions. In addition, rod-shaped and nano-sized IONPs pose a greater risk of toxicity than micrometric and sphere-shaped particles.110 Furthermore, IONPs have a tendency to be cytotoxic or genotoxic depending on their surface charge. A positive charge on IONPs increases their intracellular accumulation and decreases the integrity of cellular membranes due to nonspecific interactions and adsorptive endocytosis.111 Creating IONPs that are capable of high load capacities while ensuring that the payload is efficiently encapsulated within them is a challenging task. In some cases, as the load capacity increases, the encapsulation efficiency decreases, which might result in an inefficient delivery of drugs.An additional challenge to IONP-targeted gene delivery is the adsorption of biomacromolecules on the surface. For example, to improve nucleic acid drug loading efficiency, IONPs are modified with a positive charge at their surfaces. In blood circulation, these positively charged nanocomplexes quickly adsorb on the negatively charged plasma proteins, creating a phenomenon known as the formation of protein corona on NPs.112–114 Apparently, protein corona negatively impacts the speed of circulation and the effectiveness of payload delivery.115–117 For example, NPs may interact with plasma proteins, altering their uptake and clearance.
Modifying IONPs might seem like a good solution; however, adding additional synthesizing steps might increase the complexity of the process and the cost of production. The cost of producing high-quality IONPs for biological applications is relatively greater because of a variety of factors, one of which is the method in which the IONPs are synthesised. Thermal decomposition and co-precipitation require controlled environments and specialized equipment, which can lead to higher production costs.118 In preclinical research, IONPs are almost always prepared in small quantities, and their large-scale production might not yield particles of the same quality. In the past decade, thermal decomposition methods have been developed for the controlled synthesis of IONPs; nevertheless, they have limited reproducibility at larger scales because of the complex synthesis process. In lab-scale systems, continuous high-temperature syntheses may reduce production costs; however, it is difficult to predict any reductions in costs at the industrial scale.119
10 Strength and recommendations
Magnetic NPs are often known as IONPs or MNPs. MNPs may be found in a variety of forms, such as ferrites (MFe2O4), where M might be Co, Cu, Ni, Mn, and Mg, metal oxides (Fe3O4, γ-Fe2O3), metal alloys (FePt, CoPt), and pure metals (Fe, Co, Ni, and Mn). Pure metals are unsuitable for in vivo application due to their high degrees of toxicity and oxidative properties. Because of these factors, IONPs like Fe3O4 and γ-Fe2O3 are utilized in a number of healthcare sectors. Aside from their biocompatibility and chemical stability, SPIONs can also significantly reduce the T1 and T2 proton relaxation times when the surface of γ-Fe2O3 and Fe3O4 are modified. Due to these characteristics, SPIONs are frequently utilized for MRI contrast enhancement, hyperthermia, targeted drug administration, nucleic acids, antibodies, tissue healing, biomolecule separation, and biosensing.120,121 A relatively large amount of MRI has been used in the diagnosis and staging of MS to assess the integrity of the BBB and to identify inflammatory lesions in the brain. In this context, IONP contrast agents are advantageous over other contrast agents based on gadolinium because they are able to detect macrophage or monocyte infiltration, which is a hallmark of the progression of MS.122–124Conventional cancer treatment has a number of drawbacks, including side effects, a lack of drug availability at the site of action, drug resistance, rapid renal clearance, and several other issues. As a result of these challenges, researchers have been able to combine hyperthermia with radiotherapy and chemotherapy. In magnetic hyperthermia, a magnetic field exposes the intratumorally injected IONPs to a heating effect, resulting in cell death near the tumor area.125,126 Due to their poor cellular architecture, cancer cells are very susceptible to damage from slight temperature changes. Furthermore, hyperthermia can also increase the temperature of the surrounding environment to 55–60 °C, which can be tolerated by healthy cells; however, cancerous cells are susceptible to increased temperature.
The unique properties of NPs enable them to be used in effective cancer therapy. Since IONPs have a number of structural advantages, they have gained global interest for nanomedicine applications, including stability in colloidal suspension, resistance to degradation in vivo, synergistic activity in improving drug sensitivity, the ability to deliver drugs at high dosages, and the ability to reverse cancer cell resistance.127,128 The cryoablation efficacy may be enhanced by Fe3O4 NPs. In cryoablation, cancerous cells are treated with extreme cold temperatures. Fe3O4 NPs can change the ability of intracellular ice formation during freezing, thawing, and recrystallization, resulting in the increased death of cancerous cells like MCF-7.129 MCF-7 is a human breast cancer cell line possessing receptors for glucocorticoids, progesterone, and estrogen.130 Therefore, IONPs may be successfully implemented to efficiently treat tumors.129
Gene delivery is a method of treating diseases utilizing nucleic acids rather than drugs or surgery. Generally, delivering biological materials like siRNA and plasmid DNA to the cells or tissues of interest without being damaged by nucleases is very challenging. Consequently, to mitigate the damage, nanocarriers involving IONPs have been employed to carry the genes and release them at the desired target sites.131 An innovative vector for the delivery of therapeutic genes to tumors has been developed. In order to deliver the desired gene to the target region, the gene carrier contained IONPs that were bound to lentiviral (LV) vectors. To check the effectiveness, this nanocarrier along with a reporter green fluorescent protein gene was injected into mice-carrying tumors, which caused sustained gene expression in areas close to the target. The results of this research open the possibility that LV-IONPs may be capable of delivering therapeutic genes to selectively inhibit tumor growth.132
11 Future prospects
Advances in the synthesis of novel nanocomposites with multifunctional modalities will enable IONPs for usage in biomedicine. IONPs hold great promise for future applications in biomedical science, including the early detection and diagnosis of diseases, delivery of drugs or genes, the imaging of surface and deep tissues, and the development of multifunctional drugs. There is a need to develop more IONP-based materials for the consumer market, and more research needs to be conducted in this area. In order to make a product sustainable in the market, there is a requirement for a positive cost-benefit balance. Therefore, different techniques should be developed to produce IONPs at a larger scale that are more cost-effective. In molecular medicine, it is essential to develop new tools for the early detection of diseases. NPs, such as IONPs, could further enhance theranostic platforms, which combine diagnostics and therapeutics approaches. The primary benefits of such efforts would be to simplify and reduce the level of invasiveness of the diagnostic process. An increasing number of groups are paying attention to personalized medicine, and it is anticipated that nanotechnology will play a crucial role in achieving a positive impact across all aspects of health care. Therefore, multifunctional IONPs might become a popular material for biomedical applications in the near future.At present, assuming an 8 hour of working period in a day, exposure of 14 mg m−3 has been set as the permissible limit for fine iron oxide by the Occupational Safety and Health Administration (OSHA). The recommended exposure limit (REL) is 5 mg m−3 for iron (in iron oxide) for a 10 hour working period in a day, while it is 5 mg m−3 for the respirable fraction of iron oxide over an 8 hour workday set by The National Institute for Occupational Safety and Health (NIOSH) and American Conference of Governmental Industrial Hygienists (ACGIH), respectively. Unfortunately, due to the unavailability of concrete hazard and toxicity assessment data for IONPs, there is no separate recommended exposure limit (REL) set for IONPs. Therefore, it is essential to set REL for IONPs in the near future since a lack of defined REL may put the workers at the verge of serious health issues.133
12 Conclusions
IONPs can either be a benediction or a curse to the human CNS depending on their physicochemical characteristics, concentrations, modes of exposure, and processes of cellular absorption. IONPs are useful in the identification and treatment of CNS disorders; nonetheless, they could also be harmful to the CNS. Studies have confirmed that IONPs can cause inflammation, oxidative stress, and ferroptosis following sub-cellular entry, potentially damaging the CNS. Moreover, the IONPs are so small that they can cross the BBB and accumulate in the brain, potentially causing neuronal death and ensuing neurodegenerative disorders. However, measures can be implemented to reduce the potential toxicity of these NPs by controlling their size, shape, and surface chemistry. Monitoring and controlling the abundance of IONPs in the air is also vital to reduce chronic exposure. Moreover, the safety of IONPs should be carefully evaluated before they are employed for clinical purposes, and strategies should be developed to minimize the potential toxicity of IONPs. Studies have proposed plausible molecular events that are engaged in IONP-induced neurodegeneration. However, many such predictions require further validation. Therefore, it would be of great importance to encourage studies for dissecting the underlying mechanism of neurotoxicity as well as to develop safer IONPs.Abbreviations
| 6-OHDA: | 6-Hydroxydopamine |
| AD: | Alzheimer's disease |
| APTMS: | Aminopropyl trimethoxysilane |
| BBB: | Blood brain barrier |
| CNS: | Central nervous system |
| CSF: | Cerebrospinal fluid |
| DMT1: | Divalent metal transporter 1 |
| DOX: | Doxorubicin |
| FAD: | Familial Alzheimer's disease |
| FPN1: | Ferroportin |
| IONP: | Iron oxide nanoparticle |
| MCI: | Mild cognitive impairment |
| MMSE: | Mini-mental state examination |
| NMDA: | N-Methyl-D-aspartate |
| NTBI: | Non-transferrin bound iron |
| PD: | Parkinson's disease |
| PM: | Particulate matter |
| ROS: | Reactive oxygen species |
| SAD: | Sporadic Alzheimer's disease |
| SPION: | Superparamagnetic IONPs |
| Tf: | Transferrin |
| TfR: | Transferrin receptor |
| αS: | α-Synuclein |
Conflicts of interest
The authors have no relevant financial or non-financial interests to disclose.Acknowledgements
Authors acknowledge Swami Vivekananda Merit-cum-Means Scholarship, Department of Higher Education, Government of West Bengal, India, which was awarded to the author, Abhratanu Ganguly (WBP211651743312) and Sayantani Nanda (WBP201638780142). Authors are thankful to https://BioRender.com which was used to prepare illustrations for this work.References
- B. A. Maher, A. González-Maciel, R. Reynoso-Robles, R. Torres-Jardón and L. Calderón-Garcidueñas, Iron-rich air pollution nanoparticles: an unrecognised environmental risk factor for myocardial mitochondrial dysfunction and cardiac oxidative stress, Environ. Res., 2020, 188, 109816 CrossRef CAS PubMed.
- S. Heo, W. Lee and M. L. Bell, Suicide and Associations with Air Pollution and Ambient Temperature: A Systematic Review and Meta-Analysis, Int. J. Environ. Res. Public Health, 2021, 18(14), 7699 CrossRef CAS PubMed.
- Z. Li, H. Yan, X. Zhang, S. Shah, G. Yang, Q. Chen, S. Han, D. Zhang, D. R. Weinberger, W. Yue and H. Y. Tan, Air pollution interacts with genetic risk to influence cortical networks implicated in depression, Proc. Natl. Acad. Sci. U. S. A., 2021, 118(46), e2109310118 CrossRef CAS PubMed.
- K. Adachi and Y. Tainosho, Characterization of heavy metal particles embedded in tire dust, Environ. Int., 2004, 30(8), 1009–1017 CrossRef CAS PubMed.
- K. Maier-Hauff, F. Ulrich, D. Nestler, H. Niehoff, P. Wust, B. Thiesen, H. Orawa, V. Budach and A. Jordan, Efficacy and safety of intratumoral thermotherapy using magnetic iron-oxide nanoparticles combined with external beam radiotherapy on patients with recurrent glioblastoma multiforme, J. Neuro-Oncol., 2011, 103(2), 317–324 CrossRef PubMed.
- T. Gonet, B. A. Maher, I. Nyirő-Kósa, M. Pósfai, M. Vaculík and J. Kukutschová, Size-resolved, quantitative evaluation of the magnetic mineralogy of airborne brake-wear particulate emissions, Environ. Pollut., 2021, 288, 117808 CrossRef CAS PubMed.
- J. Kukutschová, P. Moravec, V. Tomášek, V. Matějka, J. Smolík, J. Schwarz, J. Seidlerová, K. Safářová and P. Filip, On airborne nano/micro-sized wear particles released from low-metallic automotive brakes, Environ. Pollut., 2011, 159(4), 998–1006 CrossRef PubMed.
- C. L. Carlson and D. C. Adriano, Environmental Impacts of Coal Combustion Residues, J. Environ. Qual., 1993, 22, 227–247 CrossRef CAS.
- L. Gárate-Vélez, C. Escudero-Lourdes, D. Salado-Leza, A. González-Sánchez, I. Alvarado-Morales, D. Bahena, G. J. Labrada-Delgado and J. L. Rodríguez-López, Anthropogenic Iron Oxide Nanoparticles Induce Damage to Brain Microvascular Endothelial Cells Forming the Blood–Brain Barrier, J. Alzheimer's Dis., 2020, 76(4), 1527–1539 Search PubMed.
- J. Hammond, B. A. Maher, I. A. M. Ahmed and D. Allsop, Variation in the concentration and regional distribution of magnetic nanoparticles in human brains, with and without Alzheimer's disease, from the UK, Sci. Rep., 2021, 11(1), 9363 CrossRef CAS PubMed.
- C. P. Ferri, M. Prince, C. Brayne, H. Brodaty, L. Fratiglioni, M. Ganguli, K. Hall, K. Hasegawa, H. Hendrie, Y. Huang, A. Jorm, C. Mathers, P. R. Menezes, E. Rimmer and M. Scazufca, Alzheimer's Disease International. Global prevalence of dementia: a Delphi consensus study, Lancet, 2005, 366, 2112–2117 CrossRef PubMed.
- L. Calderón-Garcidueñas, A. C. Solt, C. Henríquez-Roldán, R. Torres-Jardón, B. Nuse, L. Herritt, R. Villarreal-Calderón, N. Osnaya, I. Stone, R. García, D. M. Brooks, A. González-Maciel, R. Reynoso-Robles, R. Delgado-Chávez and W. Reed, Long-term air pollution exposure is associated with neuroinflammation, an altered innate immune response, disruption of the blood–brain barrier, ultrafine particulate deposition, and accumulation of amyloid beta-42 and alpha-synuclein in children and young adults, Toxicol. Pathol., 2008, 36(2), 289–310 CrossRef PubMed.
- D. Hare, S. Ayton, A. Bush and P. Lei, A delicate balance: iron metabolism and diseases of the brain, Front. Aging Neurosci., 2013, 5, 34 Search PubMed.
- N. M. Hamdy, A. A. Boseila, A. Ramadan and E. B. Basalious, Iron Oxide Nanoparticles-Plant Insignia Synthesis with Favorable Biomedical Activities and Less Toxicity, in the “Era of the-Green”: A Systematic Review, Pharmaceutics, 2022, 14, 844 CrossRef CAS PubMed.
- A. A. Bharde, R. Y. Parikh, M. Baidakova, S. Jouen, B. Hannoyer, T. Enoki, B. L. Prasad, Y. S. Shouche, S. Ogale and M. Sastry, Bacteria-mediated precursor-dependent biosynthesis of superparamagnetic iron oxide and iron sulfide nanoparticles, Langmuir, 2008, 24(11), 5787–5794 CrossRef CAS.
- O. Bomatí-Miguel, L. Mazeina, A. Navrotsky and S. Veintemillas-Verdaguer, Calorimetric Study of Maghemite Nanoparticles Synthesized by Laser-Induced Pyrolysis, Chem. Mater., 2008, 20, 591–598 CrossRef.
- L. Cabrera, S. Gutierrez, N. Menendez, M. P. Morales and P. Herrasti, Magnetite nanoparticles: electrochemical synthesis and characterization, Electrochim. Acta, 2008, 53, 3436–3441 CrossRef CAS.
- C. Pascal, J. L. Pascal, F. Favier, M. L. Elidrissi Moubtassim and C. Payen, Electrochemical Synthesis for the Control of γ-Fe2O3Nanoparticle Size. Morphology, Microstructure, and Magnetic Behavior, Chem. Mater., 1999, 11(1), 141–147 CrossRef CAS.
- V. T. Binh, S. T. Purcell, V. Semet and F. Feschet, Nanotips and nano magnetism, Appl. Surf. Sci., 1998, 130, 803–814 CrossRef.
- S. Giri, S. Samanta, S. Maji, S. Ganguli and A. Bhaumik, Magnetic properties of α-Fe2O3 nanoparticle synthesized by a new hydrothermal method, J. Magn. Magn. Mater., 2005, 285, 296–302 CrossRef CAS.
- C. Solans, P. Izquierdo, J. Nolla, N. Azemar and M. J. Garcia-Celma, Nano-emulsions, Curr. Opin. Colloid Interface Sci., 2005, 10(3), 102–110 CrossRef CAS.
- S. Sun and H. Zeng, Size-controlled synthesis of magnetite nanoparticles, J. Am. Chem. Soc., 2002, 124(28), 8204–8215 CrossRef CAS PubMed.
- S. V. Dhuria, L. R. Hanson and W. H. Frey, Novel vasoconstrictor formulation to enhance intranasal targeting of neuropeptide therapeutics to the central nervous system, J. Pharmacol. Exp. Ther., 2009, 328(1), 312–320 CrossRef CAS PubMed.
- N. M. Hamdy, F. H. Shaker, X. Zhan and E. B. Basalious, Tangled quest of post-COVID-19 infection-caused neuropathology and what 3P nano-bio-medicine can solve?, EPMA J., 2022, 13(2), 261–284 CrossRef PubMed.
- X. Q. Chen, J. R. Fawcett, Y. E. Rahman, T. A. Ala and W. H. Frey II, Delivery of Nerve Growth Factor to the Brain via the Olfactory Pathway, J. Alzheimer's Dis., 1998, 1(1), 35–44 CAS.
- M. L. Schaefer, B. Böttger, W. L. Silver and T. E. Finger, Trigeminal collaterals in the nasal epithelium and olfactory bulb: a potential route for direct modulation of olfactory information by trigeminal stimuli, J. Comp. Neurol., 2002, 444(3), 221–226 CrossRef.
- B. F. Anderson, H. M. Baker, E. J. Dodson, G. E. Norris, S. V. Rumball, J. M. Waters and E. N. Baker, Structure of human lactoferrin at 3.2-A resolution, Proc. Natl. Acad. Sci. U. S. A., 1987, 84(7), 1769–1773 CrossRef CAS PubMed.
- D. Hare, S. Ayton, A. Bush and P. Lei, A delicate balance: iron metabolism and diseases of the brain, Front. Aging Neurosci., 2013, 5, 34 Search PubMed.
- S. Oshiro, M. S. Morioka and M. Kikuchi, Dysregulation of iron metabolism in Alzheimer's disease, Parkinson's disease, and amyotrophic lateral sclerosis, Adv. Pharmacol. Sci., 2011, 2011, 378278 Search PubMed.
- A. Figuerola, R. Di Corato, L. Manna and T. Pellegrino, From iron oxide nanoparticles towards advanced iron-based inorganic materials designed for biomedical applications, Pharmacol. Res., 2010, 62(2), 126–143 CrossRef CAS PubMed.
- F. Y. Cheng, C. H. Su, Y. S. Yang, C. S. Yeh, C. Y. Tsai, C. L. Wu, M. T. Wu and D. B. Shieh, Characterization of aqueous dispersions of Fe (3)O(4) nanoparticles and their biomedical applications, Biomaterials, 2005, 26(7), 729–738 CrossRef CAS PubMed.
- S. M. Dadfar, K. Roemhild, N. I. Drude, S. von Stillfried, R. Knüchel, F. Kiessling and T. Lammers, Iron oxide nanoparticles: diagnostic, therapeutic and theranostic applications, Adv. Drug Delivery Rev., 2019, 138, 302–325 CrossRef CAS PubMed.
- O. A. Marcos-Contreras, S. Martinez de Lizarrondo, I. Bardou, C. Orset, M. Pruvost, A. Anfray, Y. Frigout, Y. Hommet, L. Lebouvier, J. Montaner, D. Vivien and M. Gauberti, Hyperfibrinolysis increases blood–brain barrier permeability by a plasmin- and bradykinin-dependent mechanism, Blood, 2016, 128(20), 2423–2434 CrossRef CAS.
- V. I. Shubayev, T. R. Pisanic II and S. Jin, Magnetic nanoparticles for theragnostics, Adv. Drug Delivery Rev., 2009, 61(6), 467–477 CrossRef CAS PubMed.
- N. M. Hamdy, G. Eskander and E. B. Basalious, Insights on the Dynamic Innovative Tumor Targeted-Nanoparticles-Based Drug Delivery Systems Activation Techniques, Int. J. Nanomed., 2022, 17, 6131–6155 CrossRef PubMed.
- L. Smith, H. L. Byrne, D. Waddington and Z. Kuncic, Nanoparticles for MRI-guided radiation therapy: a review, Cancer Nanotechnol., 2022, 13, 38 CrossRef.
- J. Dulińska-Litewka, A. Łazarczyk, P. Hałubiec, O. Szafrański, K. Karnas and A. Karewicz, Superparamagnetic Iron Oxide Nanoparticles-Current and Prospective Medical Applications, Materials, 2019, 12(4), 617 CrossRef PubMed.
- L. Wu, W. Wen, X. Wang, D. Huang, J. Cao, X. Qi and S. Shen, Ultrasmall iron oxide nanoparticles cause significant toxicity by specifically inducing acute oxidative stress to multiple organs, Part. Fibre Toxicol., 2022, 19(1), 24 CrossRef CAS PubMed.
- D. T. Wiley, P. Webster, A. Gale and M. E. Davis, Transcytosis and brain uptake of transferrin-containing nanoparticles by tuning avidity to transferrin receptor, Proc. Natl. Acad. Sci. U. S. A., 2013, 110(21), 8662–8667 CrossRef CAS PubMed.
- G. I. Fouad, S. A. M. El-Sayed, M. Mabrouk, K. A. Ahmed and H. H. Beherei, Neuroprotective Potential of Intranasally Delivered Sulforaphane-Loaded Iron Oxide Nanoparticles Against Cisplatin-Induced Neurotoxicity, Neurotoxicol. Res., 2022, 40(5), 1479–1498 CrossRef PubMed.
- Y. Zhang, Z. Wang, X. Li, L. Wang, M. Yin, L. Wang, N. Chen, C. Fan and H. Song, Dietary Iron Oxide Nanoparticles Delay Aging and Ameliorate Neurodegeneration in Drosophila, Adv. Mater., 2016, 28(7), 1387–1393 CrossRef CAS PubMed.
- P. Umarao, S. Bose, S. Bhattacharyya, A. Kumar and S. Jain, Neuroprotective Potential of Superparamagnetic Iron Oxide Nanoparticles Along with Exposure to Electromagnetic Field in 6-OHDA Rat Model of Parkinson's Disease, J. Nanosci. Nanotechnol., 2016, 16(1), 261–269 CrossRef CAS PubMed.
- J. Pinkernelle, P. Calatayud, G. F. Goya, H. Fansa and G. Keilhoff, Magnetic nanoparticles in primary neural cell cultures are mainly taken up by microglia, BMC Neurosci., 2012, 13, 32 CrossRef CAS PubMed.
- W. J. Yang, J. H. Lee, S. C. Hong, J. Lee, J. Lee and D. W. Han, Difference between Toxicities of Iron Oxide Magnetic Nanoparticles with Various Surface-Functional Groups against Human Normal Fibroblasts and Fibrosarcoma Cells, Materials, 2013, 6(10), 4689–4706 CrossRef PubMed.
- F. Mazuel, A. Espinosa, N. Luciani, M. Reffay, R. Le Borgne, L. Motte, K. Desboeufs, A. Michel, T. Pellegrino, Y. Lalatonne and C. Wilhelm, Massive Intracellular Biodegradation of Iron Oxide Nanoparticles Evidenced Magnetically at Single-Endosome and Tissue Levels, ACS Nano, 2016, 10(8), 7627–7638 CrossRef CAS.
- J. Volatron, F. Carn, J. Kolosnjaj-Tabi, Y. Javed, Q. L. Vuong, Y. Gossuin, C. Ménager, N. Luciani, G. Charron, M. Hémadi, D. Alloyeau and F. Gazeau, Ferritin Protein Regulates the Degradation of Iron Oxide Nanoparticles, Small, 2017, 13(2), 1602030 CrossRef.
- A. Laskar, M. Ghosh, S. I. Khattak, W. Li and X. M. Yuan, Degradation of superparamagnetic iron oxide nanoparticle-induced ferritin by lysosomal cathepsins and related immune response, Nanomedicine, 2012, 7(5), 705–717 CrossRef CAS PubMed.
- M. Yu, S. Huang, K. J. Yu and A. M. Clyne, Dextran and polymer polyethylene glycol (PEG) coating reduce both 5 and 30 nm iron oxide nanoparticle cytotoxicity in 2D and 3D cell culture, Int. J. Mol. Sci., 2012, 13(5), 5554–5570 CrossRef CAS.
- G. Jarockyte, E. Daugelaite, M. Stasys, U. Statkute, V. Poderys, T. C. Tseng, S. H. Hsu, V. Karabanovas and R. Rotomskis, Accumulation and Toxicity of Superparamagnetic Iron Oxide Nanoparticles in Cells and Experimental Animals, Int. J. Mol. Sci., 2016, 17(8), 1193 CrossRef PubMed.
- W. J. Yang, J. H. Lee, S. C. Hong, J. Lee, J. Lee and D. W. Han, Difference between Toxicities of Iron Oxide Magnetic Nanoparticles with Various Surface-Functional Groups against Human Normal Fibroblasts and Fibrosarcoma Cells, Materials, 2013, 6(10), 4689–4706 CrossRef PubMed.
- S. Z. Imam, S. M. Lantz-McPeak, E. Cuevas, H. Rosas-Hernandez, S. Liachenko, Y. Zhang, S. Sarkar, J. Ramu, B. L. Robinson, Y. Jones, B. Gough, M. G. Paule, S. F. Ali and Z. K. Binienda, Iron Oxide Nanoparticles Induce Dopaminergic Damage: In vitro Pathways and In Vivo Imaging Reveals Mechanism of Neuronal Damage, Mol. Neurobiol., 2015, 52(2), 913–926 CrossRef CAS PubMed.
- J. P. M. Almeida, A. L. Chen, A. Foster and R. Drezek, In vivo biodistribution of nanoparticles, Nanomedicine, 2011, 6(5), 815–835 CrossRef CAS PubMed.
- J. Xie, K. Chen, J. Huang, S. Lee, J. Wang, J. Gao, X. Li and X. Chen, PET/NIRF/MRI triple functional iron oxide nanoparticles, Biomaterials, 2010, 31(11), 3016–3022 CrossRef CAS.
- G. Liu, J. Gao, H. Ai and X. Chen, Applications and Potential Toxicity of Magnetic Iron Oxide Nanoparticles, Small, 2013, 9(9–10), 1533–1545 CrossRef CAS.
- M. A. Busquets, R. Sabaté and J. Estelrich, Potential applications of magnetic particles to detect and treat Alzheimer's disease, Nanoscale Res. Lett., 2014, 9(1), 538 CrossRef PubMed.
- M. Battaglini, N. Feiner, C. Tapeinos, D. De Pasquale, C. Pucci, A. Marino, M. Bartolucci, A. Petretto, L. Albertazzi and G. Ciofani, Combining confocal microscopy, dSTORM, and mass spectroscopy to unveil the evolution of the protein corona associated with nanostructured lipid carriers during blood–brain barrier crossing, Nanoscale, 2022, 14(36), 13292–13307 RSC.
- K. Dashtipour, M. Liu, C. Kani, P. Dalaie, A. Obenaus, D. Simmons, N. M. Gatto and M. Zarifi, Iron Accumulation Is Not Homogenous among Patients with Parkinson's Disease, Parkinson's Dis., 2015, 2015, 324843 Search PubMed.
- Y. Zhang, Z. Wang, X. Li, L. Wang, M. Yin, L. Wang, N. Chen, C. Fan and H. Song, Dietary Iron Oxide Nanoparticles Delay Aging and Ameliorate Neurodegeneration in Drosophila, Adv. Mater., 2016, 28(7), 1387–1393 CrossRef CAS PubMed.
- A. L. Cortajarena, D. Ortega, S. M. Ocampo, A. Gonzalez-García, P. Couleaud, R. Miranda, C. Belda-Iniesta and A. Ayuso-Sacido, Engineering Iron Oxide Nanoparticles for Clinical Settings, Nano Biomed., 2014, 1, 2 Search PubMed.
- N. Joshi, S. Basak, S. Kundu, G. De, A. Mukhopadhyay and K. Chattopadhyay, Attenuation of the early events of α-synuclein aggregation: a fluorescence correlation spectroscopy and laser scanning microscopy study in the presence of surface-coated Fe3O4 nanoparticles, Langmuir, 2015, 31(4), 1469–1478 CrossRef CAS PubMed.
- I. M. Pongrac, I. Pavičić, M. Milić, L. Brkić Ahmed, M. Babič, D. Horák, I. Vinković Vrček and S. Gajović, Oxidative stress response in neural stem cells exposed to different superparamagnetic iron oxide nanoparticles, Int. J. Nanomed., 2016, 11, 1701–1715 CAS.
- M. Kumari, S. Rajak, S. P. Singh, S. I. Kumari, P. U. Kumar, U. S. Murty, M. Mahboob, P. Grover and M. F. Rahman, Repeated oral dose toxicity of iron oxide nanoparticles: biochemical and histopathological alterations in different tissues of rats, J. Nanosci. Nanotechnol., 2012, 12(3), 2149–2159 CrossRef CAS PubMed.
- M. JodeiriFarshbaf, K. Ghaedi, T. L. Megraw, J. Curtiss, M. Shirani Faradonbeh, P. Vaziri and M. H. Nasr-Esfahani, Does PGC1α/FNDC5/BDNF Elicit the Beneficial Effects of Exercise on Neurodegenerative Disorders?, NeuroMol. Med., 2016, 18(1), 1–15 CrossRef CAS PubMed.
- A. Sarkar and P. C. Sil, Iron oxide nanoparticles mediated cytotoxicity via PI3K/AKT pathway: role of quercetin, Food Chem. Toxicol., 2014, 71, 106–115 CrossRef CAS PubMed.
- K. Blennow, M. J. de Leon and H. Zetterberg, Alzheimer's disease, Lancet, 2006, 368(9533), 387–403 CrossRef CAS.
- M. F. Folstein, S. E. Folstein and P. R. McHugh, "Mini-mental state". A practical method for grading the cognitive state of patients for the clinician, J. Psychiatr. Res., 1975, 12(3), 189–198 CrossRef CAS PubMed.
- O. Zanetti, S. B. Solerte and F. Cantoni, Life expectancy in Alzheimer's disease (AD), Arch. Gerontol. Geriatr., 2009, 49, 237–243 CrossRef PubMed.
- B. J. Everitt and T. W. Robbins, Central cholinergic systems and cognition, Annu. Rev. Psychol., 1997, 48, 649–684 CrossRef CAS PubMed.
- R. Wang and P. H. Reddy, Role of Glutamate and NMDA Receptors in Alzheimer's Disease, J. Alzheimer's Dis., 2017, 57(4), 1041–1048 CAS.
- H. Braak and E. Braak, Neuropathological stageing of Alzheimer-related changes, Acta Neuropathol., 1991, 82(4), 239–259 CrossRef CAS PubMed.
- D. H. Salat, R. L. Buckner, A. Z. Snyder, D. N. Greve, R. S. Desikan, E. Busa, J. C. Morris, A. M. Dale and B. Fischl, Thinning of the cerebral cortex in aging, Cereb. Cortex, 2004, 14(7), 721–730 CrossRef PubMed.
- S. M. Nestor, R. Rupsingh, M. Borrie, M. Smith, V. Accomazzi, J. L. Wells, J. Fogarty and R. Bartha, Alzheimer's Disease Neuroimaging Initiative. Ventricular enlargement as a possible measure of Alzheimer's disease progression validated using the Alzheimer's disease neuroimaging initiative database, Brain, 2008, 131, 2443–2454 CrossRef PubMed.
- S. J. Dixon, K. M. Lemberg, M. R. Lamprecht, R. Skouta, E. M. Zaitsev, C. E. Gleason, D. N. Patel, A. J. Bauer, A. M. Cantley, W. S. Yang, B. Morrison and B. R. Stockwell, Ferroptosis: an iron-dependent form of nonapoptotic cell death, Cell, 2012, 149(5), 1060–1072 CrossRef CAS PubMed.
- Y. Xie, W. Hou, X. Song, Y. Yu, J. Huang, X. Sun, R. Kang and D. Tang, Ferroptosis: process and function, Cell Death Differ., 2016, 23(3), 369–379 CrossRef CAS PubMed.
- J. Parkinson, An essay on the shaking palsy. 1817, J. Neuropsychiatry Clin. Neurosci., 2002, 14(2), 223–236 CrossRef.
- S. Ayton and P. Lei, Nigral iron elevation is an invariable feature of Parkinson's disease and is a sufficient cause of neurodegeneration, BioMed Res. Int., 2014, 2014, 581256 Search PubMed.
- D. J. Hare and K. L. Double, Iron and dopamine: a toxic couple, Brain, 2016, 139, 1026–1035 CrossRef PubMed.
- D. Mahad, I. Ziabreva, H. Lassmann and D. Turnbull, Mitochondrial defects in acute multiple sclerosis lesions, Brain, 2008, 131, 1722–1735 CrossRef.
- J. W. Eaton and M. Qian, Molecular bases of cellular iron toxicity, Free Radical Biol. Med., 2002, 32(9), 833–840 CrossRef CAS PubMed.
- S. J. Tabrizi, R. I. Scahill, A. Durr, R. A. Roos, B. R. Leavitt, R. Jones, G. B. Landwehrmeyer, N. C. Fox, H. Johnson, S. L. Hicks, C. Kennard, D. Craufurd, C. Frost, D. R. Langbehn, R. Reilmann and J. C. Stout, TRACK-HD Investigators. Biological and clinical changes in premanifest and early-stage Huntington's disease in the TRACK-HD study: the 12-month longitudinal analysis, Lancet Neurol., 2011, 10(1), 31–42 CrossRef PubMed.
- L. Cui, H. Jeong, F. Borovecki, C. N. Parkhurst, N. Tanese and D. Krainc, Transcriptional repression of PGC-1alpha by mutant huntingtin leads to mitochondrial dysfunction and neurodegeneration, Cell, 2006, 127(1), 59–69 CrossRef CAS PubMed.
- F. C. Nucifora Jr., M. Sasaki, M. F. Peters, H. Huang, J. K. Cooper, M. Yamada, H. Takahashi, S. Tsuji, J. Troncoso, V. L. Dawson, T. M. Dawson and C. A. Ross, Interference by huntingtin and atrophin-1 with cbp-mediated transcription leading to cellular toxicity, Science, 2001, 291(5512), 2423–2428 CrossRef CAS PubMed.
- E. Trushina, R. B. Dyer, J. D. Badger II, D. Ure, L. Eide, D. D. Tran, B. T. Vrieze, V. Legendre-Guillemin, P. S. McPherson, B. S. Mandavilli, B. Van Houten, S. Zeitlin, M. McNiven, R. Aebersold, M. Hayden, J. E. Parisi, E. Seeberg, I. Dragatsis, I. Doyle, A. Bender, C. Chacko and C. T. McMurray, Mutant huntingtin impairs axonal trafficking in mammalian neurons in vivo and in vitro, Mol. Cell. Biol., 2004, 24(18), 8195–8209 CrossRef CAS PubMed.
- C. Landles and G. P. Bates, Huntingtin and the molecular pathogenesis of Huntington's disease, Fourth in molecular medicine review series, EMBO Rep., 2004, 5(10), 958–963 CrossRef CAS PubMed.
- L. Zecca, A. Stroppolo, A. Gatti, D. Tampellini, M. Toscani, M. Gallorini, G. Giaveri, P. Arosio, P. Santambrogio, R. G. Fariello, E. Karatekin, M. H. Kleinman, N. Turro, O. Hornykiewicz and F. A. Zucca, The role of iron and copper molecules in the neuronal vulnerability of locus coeruleus and substantia nigra during aging, Proc. Natl. Acad. Sci. U. S. A., 2004, 101(26), 9843–9848 CrossRef CAS PubMed.
- A. L. Lumsden, T. L. Henshall, S. Dayan, M. T. Lardelli and R. I. Richards, Huntingtin-deficient zebrafish exhibit defects in iron utilization and development, Hum. Mol. Genet., 2007, 16(16), 1905–1920 CrossRef CAS PubMed.
- T. Moos, T. Rosengren Nielsen, T. Skjørringe and E. H. Morgan, Iron trafficking inside the brain, J. Neurochem., 2007, 103(5), 1730–1740 CrossRef CAS.
- S. Y. Jeong and S. David, Glycosylphosphatidylinositol-anchored ceruloplasmin is required for iron efflux from cells in the central nervous system, J. Biol. Chem., 2003, 278(29), 27144–27148 CrossRef CAS.
- R. Dringen, G. M. Bishop, M. Koeppe, T. N. Dang and S. R. Robinson, The pivotal role of astrocytes in the metabolism of iron in the brain, Neurochem. Res., 2007, 32(11), 1884–1890 CrossRef CAS PubMed.
- O. Lunov, T. Syrovets, C. Röcker, K. Tron, G. U. Nienhaus, V. Rasche, V. Mailänder, K. Landfester and T. Simmet, Lysosomal degradation of the carboxydextran shell of coated superparamagnetic iron oxide nanoparticles and the fate of professional phagocytes, Biomaterials, 2010, 31(34), 9015–9022 CrossRef CAS PubMed.
- F. Gamberoni, M. Borgese, C. Pagiatakis, I. Armenia, V. Grazù, R. Gornati, S. Serio, R. Papait and G. Bernardini, Iron Oxide Nanoparticles with and without Cobalt Functionalization Provoke Changes in the Transcription Profile via Epigenetic Modulation of Enhancer Activity, Nano Lett., 2023, 23(19), 9151–9159 CrossRef CAS PubMed.
- K. Haliloğlu, A. Türkoğlu, Ö. Balpınar, H. Nadaroğlu, A. Alaylı and P. Poczai, Effects of Zinc, Copper and Iron Oxide Nanoparticles on Induced DNA Methylation, Genomic Instability and LTR Retrotransposon Polymorphism in Wheat (Triticum aestivum L.), Plants, 2022, 11(17), 2193 CrossRef PubMed.
- M. Pogribna and G. Hammons, Epigenetic Effects of Nanomaterials and Nanoparticles, J. Nanobiotechnol., 2021, 19(1), 2 CrossRef CAS PubMed.
- K. Brzóska, I. Grądzka and M. Kruszewski, Silver, Gold, and Iron Oxide Nanoparticles Alter miRNA Expression but Do Not Affect DNA Methylation in HepG2 Cells, Materials, 2019, 12(7), 1038 CrossRef PubMed.
- P. Ghosh and A. Saadat, Neurodegeneration and epigenetics: a review, Neurol., 2021, 38(6), e62–e68 CrossRef PubMed.
- C. A. Miller and J. D. Sweatt, Covalent modification of DNA regulates memory formation, Neuron, 2007, 53(6), 857–869 CrossRef CAS PubMed.
- J. Feng, Y. Zhou, S. L. Campbell, T. Le, E. Li, J. D. Sweatt, A. J. Silva and G. Fan, Dnmt1 and Dnmt3a maintain DNA methylation and regulate synaptic function in adult forebrain neurons, Nat. Neurosci., 2010, 13(4), 423–430 CrossRef CAS PubMed.
- M. Tahiliani, K. P. Koh, Y. Shen, W. A. Pastor, H. Bandukwala, Y. Brudno, S. Agarwal, L. M. Iyer, D. R. Liu, L. Aravind and A. Rao, Conversion of 5-methylcytosine to 5-hydroxymethylcytosine in mammalian DNA by MLL partner TET1, Science, 2009, 324(5929), 930–935 CrossRef CAS PubMed.
- A. Jowaed, I. Schmitt, O. Kaut and U. Wüllner, Methylation regulates alpha-synuclein expression and is decreased in Parkinson's disease patients' brains, J. Neurosci., 2010, 30(18), 6355–6359 CrossRef CAS PubMed.
- T. F. Outeiro, E. Kontopoulos, S. M. Altmann, I. Kufareva, K. E. Strathearn, A. M. Amore, C. B. Volk, M. M. Maxwell, J. C. Rochet, P. J. McLean, A. B. Young, R. Abagyan, M. B. Feany, B. T. Hyman and A. G. Kazantsev, Sirtuin 2 inhibitors rescue alpha-synuclein-mediated toxicity in models of Parkinson's disease, Science, 2007, 317(5837), 516–519 CrossRef CAS PubMed.
- S. Vartiainen, P. Pehkonen, M. Lakso, R. Nass and G. Wong, Identification of gene expression changes in transgenic C. elegans overexpressing human alpha-synuclein, Neurobiol. Dis., 2006, 22(3), 477–486 CrossRef CAS PubMed.
- D. Condliffe, A. Wong, C. Troakes, P. Proitsi, Y. Patel, L. Chouliaras, C. Fernandes, J. Cooper, S. Lovestone, L. Schalkwyk, J. Mill and K. Lunnon, Cross-region reduction in 5-hydroxymethylcytosine in Alzheimer's disease brain, Neurobiol. Aging, 2014, 35(8), 1850–1854 CrossRef CAS PubMed.
- P. I. Dolez and J. Mlynarek, Smart materials for personal protective equipment, Smart Textiles and Their Applications, 2016, pp. 497–517 Search PubMed.
- N. M. Jamadar and H. T. Jadhav, A case study on effective reduction in friction brake dust by adopting electric braking system, Life Cycle Reliability and Safety Engineering, 2020, vol. 10, pp. 91–97 Search PubMed.
- R. M. Mayer, L. D. Poulikakos, A. R. Lees, K. Heutschi, M. T. Kalivoda and P. Soltic, Reducing the environmental impact of road and rail vehicles, Environ. Impact Assess. Rev., 2012, 32, 25–32 CrossRef.
- G. Sivakami, R. Priyadarshini, V. Baby, S. Rajakumar and P. M. Ayyasamy, Bioremediation of Ferric Iron in Synthetic Metal Oxide Using Bacillus Sp. (SO-10), J. Appl. Microbiol., 2012, 2278, 1250 Search PubMed.
- M. Karimi, A. Ghasemi, P. S. Zangabad, R. Rahighi, S. M. M. Basri, H. Mirshekari, M. Amiri, Z. S. Pishabad, A. Aslani, M. Bozorgomid, D. Ghosh, A. Beyzavi, A. Vaseghi, A. R. Aref, L. Haghani, S. Bahrami and M. R. Hamblin, Smart micro/nanoparticles in stimulus-responsive drug/gene delivery systems, Chem. Soc. Rev., 2016, 45(5), 1457–1501 RSC.
- R. Landry, P. M. Jacobs, R. Davis, M. Shenouda and W. K. Bolton, Pharmacokinetic study of ferumoxytol: a new iron replacement therapy in normal subjects and hemodialysis patients, Am. J. Nephrol., 2005, 25(4), 400–410 CrossRef CAS PubMed.
- N. Schleich, F. Danhier and V. Préat, Iron oxide-loaded nanotheranostics: major obstacles to in vivo studies and clinical translation, J. Controlled Release, 2015, 198, 35–54 CrossRef CAS PubMed.
- J. H. Lee, J. E. Ju, B. I. Kim, P. J. Pak, E. K. Choi, H. S. Lee and N. Chung, Rod-shaped iron oxide nanoparticles are more toxic than sphere-shaped nanoparticles to murine macrophage cells, Environ. Toxicol. Chem., 2014, 33(12), 2759–2766 CrossRef CAS PubMed.
- S. C. Hong, J. H. Lee, J. Lee, H. Y. Kim, J. Y. Park, J. Cho, J. Lee and D. W. Han, Subtle cytotoxicity and genotoxicity differences in superparamagnetic iron oxide nanoparticles coated with various functional groups, Int. J. Nanomed., 2011, 6, 3219–3231 CAS.
- K. E. Wheeler, A. J. Chetwynd, K. M. Fahy, B. S. Hong, J. A. Tochihuitl, L. A. Foster and I. Lynch, Environmental dimensions of the protein corona, Nat. Nanotechnol., 2021, 16(6), 617–629 CrossRef CAS PubMed.
- J. Ren, N. Andrikopoulos, K. Velonia, H. Tang, R. Cai, F. Ding, P. C. Ke and C. Chen, Chemical and Biophysical Signatures of the Protein Corona in Nanomedicine, J. Am. Chem. Soc., 2022, 144(21), 9184–9205 CrossRef CAS PubMed.
- S. M. Pustulka, K. Ling, S. L. Pish and J. A. Champion, Protein Nanoparticle Charge and Hydrophobicity Govern Protein Corona and Macrophage Uptake, ACS Appl. Mater. Interfaces, 2020, 12(43), 48284–48295 CrossRef CAS PubMed.
- F. Chen, G. Wang, J. I. Griffin, B. Brenneman, N. K. Banda, V. M. Holers, D. S. Backos, L. Wu, S. M. Moghimi and D. Simberg, Complement proteins bind to nanoparticle protein corona and undergo dynamic exchange in vivo, Nat. Nanotechnol., 2017, 12(4), 387–393 CrossRef CAS PubMed.
- S. Tenzer, D. Docter, J. Kuharev, A. Musyanovych, V. Fetz, R. Hecht, F. Schlenk, D. Fischer, K. Kiouptsi, C. Reinhardt, K. Landfester, H. Schild, M. Maskos, S. K. Knauer and R. H. Stauber, Rapid formation of plasma protein corona critically affects nanoparticle pathophysiology, Nat. Nanotechnol., 2013, 8(10), 772–781 CrossRef CAS PubMed.
- Y. H. Tengjisi, Y. Fan, D. Zou, G. H. Talbo, G. Yang and C. X. Zhao, Influence of nanoparticle mechanical property on protein corona formation, J. Colloid Interface Sci., 2022, 606, 1737–1744 CrossRef CAS PubMed.
- K. A. Ross, T. M. Brenza, A. M. Binnebose, Y. Phanse, A. G. Kanthasamy, H. E. Gendelman, A. K. Salem, L. C. Bartholomay, B. H. Bellaire and B. Narasimhan, Nano-enabled delivery of diverse payloads across complex biological barriers, J. Controlled Release, 2015, 219, 548–559 CrossRef CAS PubMed.
- M. O. Besenhard, A. P. LaGrow, S. Famiani, M. Pucciarelli, P. Lettieri, N. T. K. Thanh and A. Gavriilidis, Continuous production of iron oxide nanoparticles via fast and economical high temperature synthesis, React. Chem. Eng., 2020, 5, 1474–1483 RSC.
- S. M. Dadfar, K. Roemhild, N. I. Drude, S. von Stillfried, R. Knüchel, F. Kiessling and T. Lammers, Iron Oxide Nanoparticles: Diagnostic, Therapeutic and Theranostic Applications, Adv. Drug Delivery Rev., 2019, 138, 302–325 CrossRef CAS PubMed.
- P. M. Martins, A. C. Lima, S. Ribeiro, S. Lanceros-Mendez and P. Martins, Magnetic Nanoparticles for Biomedical Applications: From the Soul of the Earth to the Deep History of Ourselves, ACS Appl. Bio Mater., 2021, 4(8), 5839–5870 CrossRef CAS PubMed.
- T. Tourdias, S. Roggerone, M. Filippi, M. Kanagaki, M. Rovaris, D. H. Miller, K. G. Petry, B. Brochet, J.-P. Pruvo, E.-W. Radüe and V. Dousset, Assessment of disease activity in multiple sclerosis phenotypes with combined gadolinium- and superparamagnetic iron oxide–enhanced MR imaging, Radiology, 2012, 264(1), 225–233 CrossRef PubMed.
- M. M. Vellinga, R. D. Oude Engberink, A. Seewann, P. J. W. Pouwels, M. P. Wattjes, S. M. A. van der Pol, C. Pering, C. H. Polman, H. E. de Vries, J. J. G. Geurts and F. Barkhof, Pluriformity of inflammation in multiple sclerosis shown by ultra-small iron oxide particle enhancement, Brain, 2008, 131, 800–807 CrossRef PubMed.
- A. Maarouf, J.-C. Ferré, W. Zaaraoui, A. Le Troter, E. Bannier, I. Berry, M. Guye, L. Pierot, C. Barillot, J. Pelletier, A. Tourbah, G. Edan, B. Audoin and J.-P. Ranjeva, Ultrasmall superparamagnetic iron oxide enhancement is associated with higher loss of brain tissue structure in clinically isolated syndrome, Mult. Scler., 2016, 22(8), 1032–1039 CrossRef CAS PubMed.
- E. Cheraghipour, S. Javadpour and A. R. Mehdizadeh, Citrate capped superparamagnetic iron oxide nanoparticles used for hyperthermia therapy, J. Biomed. Sci. Eng., 2012, 5(12), 715–719 CrossRef.
- J. Kolosnjaj-Tabi and C. Wilhelm, Magnetic nanoparticles in cancer therapy: how can thermal approaches help?, Nanomedicine, 2017, 12(6), 573–575 CrossRef CAS PubMed.
- B. Bahrami, M. Hojjat-Farsangi, H. Mohammadi, E. Anvari, G. Ghalamfarsa, M. Yousefi and F. Jadidi-Niaragh, Nanoparticles and targeted drug delivery in cancer therapy, Immunol. Lett., 2017, 190, 64–83 CrossRef CAS PubMed.
- K. Ulbrich, K. Hola, V. Subr, A. Bakandritsos, J. Tucek and R. Zboril, Targeted drug delivery with polymers and magnetic nanoparticles: covalent and noncovalent approaches, release control, and clinical studies, Chem. Rev., 2016, 116(9), 5338–5431 CrossRef CAS PubMed.
- P. Ye, Y. Kong, X. Chen, W. Li, D. Liu, Y. Xie, Y. Zhou, H. Zou, Z. Chang, H. Dai, X. Kong and P. Liu, Fe3O4 nanoparticles and cryoablation enhance ice crystal formation to improve the efficiency of killing breast cancer cells, Oncotarget, 2017, 8(7), 11389–11399 CrossRef PubMed.
- K. B. Horwitz, M. E. Costlow and W. L. McGuire, MCF-7: a human breast cancer cell line with estrogen, progesterone, and glucocorticoid receptors, Steroids, 1975, 26(6), 785–795 CrossRef CAS PubMed.
- F. M. Kievit and M. Zhang, Surface engineering of iron oxide nanoparticles for targeted cancer therapy, Acc. Chem. Res., 2011, 44(10), 853–862 CrossRef CAS PubMed.
- E. Borroni, M. Miola, S. Ferraris, G. Ricci, K. Z. Rozman, N. Kostevsek, A. Catizone, L. Rimondini, M. Prat, E. Verne and A. Follenzi, Tumor targeting by lentiviral vectors combined with magnetic nanoparticles in mice, Acta Biomater., 2017, 59, 303–316 CrossRef CAS PubMed.
- T. G. Kornberg, T. A. Stueckle, J. A. Antonini, Y. Rojanasakul, V. Castranova, Y. Yang and L. Wang, Potential Toxicity and Underlying Mechanisms Associated with Pulmonary Exposure to Iron Oxide Nanoparticles: Conflicting Literature and Unclear Risk, Nanomaterials, 2017, 7, 10, DOI:10.3390/nano7100307.
- S. Giri, S. Samanta, S. Maji, S. Ganguli and A. Bhaumik, Magnetic properties of α-Fe2O3 nanoparticle synthesized by a new hydrothermal method, J. Magn. Magn. Mater., 2005, 285, 296–302 CrossRef CAS.
- A. H. Lu, E. L. Salabas and F. Schüth, Magnetic nanoparticles: synthesis, protection, functionalization, and application, Angew Chem. Int. Ed. Engl., 2007, 46, 1222–1244 CrossRef CAS PubMed.
- S. F. Hasany, I. Ahmed, J. Rajan and A. Rehman, Systematic Review of the Preparation Techniques of Iron Oxide Magnetic Nanoparticles, Nanosci. Nanotechnol., 2013, 2, 148–158 CrossRef.
- S. Laurent, S. Dutz, U. O. Häfeli and M. Mahmoudi, Magnetic fluid hyperthermia: focus on superparamagnetic iron oxide nanoparticles, Adv. Colloid Interface Sci., 2011, 166(1–2), 8–23 CrossRef CAS PubMed.
- J. Wu, T. Ding and J. Sun, Neurotoxic potential of iron oxide nanoparticles in the rat brain striatum and hippocampus, NeuroToxicology, 2013, 34, 243–253 CrossRef CAS PubMed.
- B. Szalay, E. Tátrai, G. Nyírő, T. Vezér and G. Dura, Potential toxic effects of iron oxide nanoparticles in in vivo and in vitro experiments, J. Appl. Toxicol., 2012, 32(6), 446–453 CrossRef CAS PubMed.
- Y. Q. Zhang, R. Dringen, C. Petters, W. Rastedt, J. Köser, J. Filserd and S. Stolte, Toxicity of dimercaptosuccinate-coated and unfunctionalized magnetic iron oxide nanoparticles towards aquatic organisms, Environ. Sci.: Nano, 2016, 4, 754–767 RSC.
- A. Nemmar, S. Beegam, P. Yuvaraju, J. Yasin, S. Tariq, S. Attoub and B. H. Ali, Ultrasmall superparamagnetic iron oxide nanoparticles acutely promote thrombosis and cardiac oxidative stress and DNA damage in mice, Part. Fibre Toxicol., 2016, 13(1), 22 CrossRef PubMed.
- B. Ding, K. M. Chen, H. W. Ling, F. Sun, X. Li, T. Wan, W. M. Chai, H. Zhang, Y. Zhan and Y. J. Guan, Correlation of iron in the hippocampus with MMSE in patients with Alzheimer's disease, J. Magn. Reson. Imaging, 2009, 29(4), 793–798 CrossRef PubMed.
- J. Y. Wang, Q. Q. Zhuang, L. B. Zhu, H. Zhu, T. Li, R. Li, S. F. Chen, C. P. Huang, X. Zhang and J. H. Zhu, Meta-analysis of brain iron levels of Parkinson's disease patients determined by postmortem and MRI measurements, Sci. Rep., 2016, 6, 36669 CrossRef CAS PubMed.
- S. J. Tabrizi, R. I. Scahill, A. Durr, R. A. Roos, B. R. Leavitt, R. Jones, G. B. Landwehrmeyer, N. C. Fox, H. Johnson, S. L. Hicks, C. Kennard, D. Craufurd, C. Frost, D. R. Langbehn, R. Reilmann and J. C. Stout, TRACK-HD Investigators. Biological and clinical changes in premanifest and early-stage Huntington's disease in the TRACK-HD study: the 12-month longitudinal analysis, Lancet Neurol., 2011, 10(1), 31–42 CrossRef PubMed.
- N. Kumar, P. Rizek and M. Jog, Neuroferritinopathy: Pathophysiology, Presentation, Differential Diagnoses and Management, Tremor Other Hyperkinet. Mov., 2016, 6, 355 CrossRef.
- C. Petillon, R. Hergesheimer, H. Puy, P. Corcia, P. Vourc'h, C. Andres, Z. Karim and H. Blasco, The Relevancy of Data Regarding the Metabolism of Iron to Our Understanding of Deregulated Mechanisms in ALS; Hypotheses and Pitfalls, Front. Neurosci., 2019, 12, 1031 CrossRef PubMed.
- H. M. Schipper, Neurodegeneration with brain iron accumulation - clinical syndromes and neuroimaging, Biochim. Biophys. Acta, 2012, 1822(3), 350–360 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2024 |