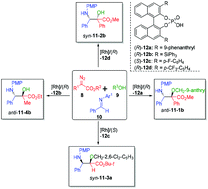
Norstatine derivatives are of important value in pharmaceutical science. However, their catalytic asymmetric synthesis is rare. We developed a sustainable method via chiral phosphoric acid (CPA)-[Rh(OAc)2]2 co-catalyzed multi-component reactions (MCR) of diazoacetates with alcohol/water and imines. This method allows us to synthesize a library of 45 norstatines with excellent enanotioselectivites and broad substrate scope which includes anti-α-aryl-norstatines 11-1, anti-α-alkyl-norstatines 11-2, syn-α-hydro-norstatines 11-3 and syn-α-aryl-norstatines 11-4. The sustainability of this method lies in the reliable scalability, improved safety, and reusable [Rh(OAc)2]2 catalyst. The synthetic value of norstatine derivatives was demonstrated by preparing oxazolinone 14, ezetimibe analogue 15, and Taxol C-13 chain 16. Mechanistic study reveals that the synergetic catalysis of CPA and [Rh(OAc)2]2 is essential to maintain chemo- and enantioselectivity. Control experiments support the mechanism where the reactions proceed through the trapping of hyper-reactive oxonium ylides with imines. Shortly, we report herein the sustainable catalytic enantioselective synthesis of both syn- and anti-norstatine derivatives. We believe that this method might shed light on the sustainable synthesis of norstatine derivative-based drug candidates.