 Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Current mixture toxicity assessments on soil organisms and applied risk assessments
Fabian Balk
 *a,
Eva Laubera,
Benoît J. D. Ferrari
*a,
Eva Laubera,
Benoît J. D. Ferrari
 ab,
Mathieu Renaud
ab,
Mathieu Renaud
 b and
Marion Junghans
b and
Marion Junghans
 *a
*a
aSwiss Centre for Applied Ecotoxicology, Ueberlandstrasse 133, 8600 Duebendorf, Switzerland. E-mail: fabian.balk@oekotoxzentrum.ch; marion.junghans@oekotoxzentrum.ch
bSwiss Centre for Applied Ecotoxicology, Station 2, 1015 Lausanne, Switzerland
First published on 21st April 2026
Abstract
There is increasing evidence that the majority of agricultural and off-field soils contain multiple plant protection product (PPP) residues that persist in soils for extended periods after application. Such PPP residues in soil were found to affect the soil organisms and their communities with little knowledge on the implications for the functioning of soil ecosystems. A growing body of scientific studies points to the increased risk from the co-occurrence of diverse PPP residues in soils, and international efforts recently resulted in the endorsement of the first directive to protect soil health in the EU to ensure an EU-wide soil health monitoring system. In order to inform such developments, the scientific literature and environmental regulations were reviewed here for currently practiced mixture risk assessment (MRA) methods in soil. Further, relevant aspects for a soil MRA were scrutinised to identify knowledge gaps in soil MRA and to evaluate novel emerging assessment concepts for their usability in soil MRA. From these insights, key conclusions are drawn for soil MRA to inform the next steps for a more realistic and accurate MRA.
Environmental significanceSoil monitoring surveys revealed that pesticide residues are omnipresent in European soils, where the co-presence of several substances at sample sites was rather the rule than the exception. Yet, current environmental risk assessments (ERAs) in soil still evaluate pesticides or product formulations in isolation, overlooking potential mixture effects on soil organisms and soil ecosystem functions. This review synthesises the scientific evidence for mixture toxicity in soils and identifies major data gaps and conceptual shortcomings in current scientific and regulatory ERA practice in soil. On this basis, a trophic-level-specific approach to mixture risk assessment is proposed to assess mixture risks in soils. These insights may support international developments such as the new EU Soil Monitoring Law and experimental soil studies. |
1. Introduction
Soils have fundamental functions on which our civilization depends.1,2 Among numerous factors that have the potential to degrade soil quality are PPP residues, which can persist in soils much longer than intended after the application of plant protection products (PPP) to crops, and they may exert undesired effects on non-target soil-dwelling organisms, i.e. soil organisms.3–8 Several regional as well as international soil surveys have evidenced the presence of a multitude of PPP residues in European soils, where the co-presence of PPP residues at the same site appeared as the rule rather than the exception.4,6,7,9–16 This implies that soil organisms are exposed to a cocktail of substances, which may pose risks to individual soil organisms, higher organisational levels, and potentially soil ecosystem functions.5,17–20 Exposure to several PPP residues at once or in sequence can be particularly problematic since they may cause effects on soil organisms at low and regulatorily deemed safe concentrations, where single exposures would have no effect.21,22 Currently, mixture effects of PPP residues in soil, which are applied as PPP in sequence or as tank mixtures in the field, are not considered in environmental risk assessments (ERA) that aim to protect non-target soil organisms from harm through pesticide exposure. Furthermore, PPP residues already present from previous growing seasons are not considered in ERA.23 Nevertheless, the increasing evidence for the widespread presence of PPP residues in agricultural and non-agricultural lands implies that the regulatory common single substance exposure is a rather theoretical consideration and that ERA should include co-exposure to multiple substances that may occur in soil. The issue of substance mixtures has been acknowledged by European regulatory bodies, which provide guidance on how the risk of environmental mixtures can be assessed,24 and some regulatory authorities do address mixture assessments in contaminated sites or in the risk assessment (RA) for higher animals.25 The evidence of co-occurring PPP residues in soil and the largely absent consideration of mixture effects in the environment in European regulatory ERA calls for a RA that goes beyond the single substance assessment and focusses on the risk that is caused by the presence of multiple substances in soil.21,26 In fact, many other stressors, such as environmental and climatic conditions or agronomic practises, should be considered for a more holistic ERA.26–28 In environmental research, the majority of mixture toxicity and mixture risk research has focused on aquatic organisms and ecosystems.29 Only in the last decade has the study of mixture toxicity to soil organisms gained momentum; however, mixture risk assessment (MRA) methods tailored to the soil ecosystem are still scarce.Historically, environmental MRA were first developed to protect the aquatic environment from harm through accumulating and co-occurring chemical exposure. Therefore, MRA for soil organisms were derived from risk characterisation methods addressing the aquatic environment. Yet, despite the different constitutions of soil and water as exposure media and the contrasting ecological traits of the inhabiting organisms, the assumption of bioavailability in soil and water is similar in the corresponding ERA. Also, pesticide residue concentrations in soil may show less dynamic fluctuations compared to waterbodies, but can show much more heterogeneous contamination patterns on larger geographical scales.12 As a result, PPP residues of varying persistence can form complex mixture exposures with unknown consequences for the soil ecosystem.6 These aspects call for an ERA that is more tailored toward the soil ecosystem.
In this review, the current scientific and regulatory literature on mixture toxicity and RA in soil is evaluated to identify critical aspects for the design of a RA framework for PPP residue mixtures in soil. First, the general key concepts that are used to predict mixture toxicity and risk, and how they are applied in soil MRA, are presented. Second, studies that deal with experimental mixture toxicity and concurrent risk assessment in soils are summarised and evaluated for their limitations and strengths. Third, key insights from the regulatory literature that address mixture risks in the terrestrial environment are outlined. Fourth, novel concepts in MRA are discussed for their applicability in soil. Finally, the conclusions draw key learnings from the scientific literature and regulation on currently practised risk assessments of PPP residue mixtures in soil.
2. Study evaluation
The scientific literature was searched using the search engines Web of Science (https://www.webofscience.com/wos/woscc/basic-search) and PubMed (https://pubmed.ncbi.nlm.nih.gov/). Two sets of keyword combinations were used in both search engines: (1) risk* AND soil AND mixture* AND pesticide* and (2) mixture AND soil AND toxic* AND risk. The articles (last searched on 27th January 2026) were screened for their relevance by their title, abstract and keywords. The studies were evaluated as relevant based on whether the title/abstract/keyword list addressed mixture experimentation or MRA with or of non-target soil organisms or assessed the impact on soil functions due to exposure to PPP mixtures or mixtures of PPP with other contaminants. The use of a RA is defined here as a framework that categorises exposure concentrations in terms of their potential risk to soil. Studies dealing exclusively with plastic particles or metal exposure were disregarded in this review. In addition to the scientific literature search, relevant regulatory guidelines that deal with RA of chemical mixtures were searched. It should be noted that, for the sake of completeness, the combined exposure of pesticides with other soil contaminants (metals, microplastics, industrial chemicals, pharmaceuticals, antimicrobials, etc.) is also reviewed, and where relevant RA methods were only applied to non-pesticidal substances (e.g. industrial substances), the study was included here as well. The search was completed using expert knowledge and by additional manual searches.The total number of search hits from the search engines were 1388 (Web of Science) and 741 (PubMed) for the date range 1900 to 27.01.2026. After removal of duplicates, a total of 1320 studies remained, which were screened for their relevance as noted above. The study evaluation resulted in 108 studies identified as relevant for this review. A complete list of all 1320 studies with indication of the relevant studies is available in the SI Table S1.
The current literature on mixture studies in soil shows divergent perspectives on mixture effects and MRA in soil organisms. With the review's intention in mind, two main categories were made out to distinguish the studies: (i) Studies that assessed the risk of non-target soil organisms exposed to mixtures using experimental toxicity data from their own experiments or toxicity data from literature or databases, and (ii) studies that strive to assess or predict the measured mixture toxicity effects as synergistic, antagonistic or as null model conform.
3. Prediction of mixture toxicity
The study of mixtures can be approached from two perspectives, termed the bottom-up approach and the top-down approach. In the bottom-up approach, substance identities, concentrations and single-exposure effect concentrations of the relevant mixture constituents in the studied mixture are known. This enables the prediction of mixture effects and assessment of mixture risks, which can be experimentally verified. The top-down approach assumes no previous knowledge of the mixture constitution and focuses on the effects of a given mixture. Typically, the top-down approach is an effect-directed analysis, where an extract of an environmental sample goes through several cycles of toxicity testing, fractionation and chemical quantification to identify the toxicants that cause the observed effects.30 This approach was not used in any of the reviewed studies and is therefore not further scrutinised here. The bottom-up approach is commonly used in soil mixture studies and is further detailed below.Mixture effects at no-effect concentrations, such as no-observed effect concentrations from single PPP exposure studies (NOEC = highest non-toxic concentration in a chronic dose–response study), have been demonstrated for similarly as well as dissimilarly acting substances in terms of their mode of action.31,32 The effect size has been shown to be predictable by the two main concepts of concentration addition (CA)33 and independent action (IA).34 CA postulates that similarly acting substances (sharing the same mode of action and leading to a common toxicological effect) behave in a mixture as if they were dilutions of one another, i.e. their normalised effect concentrations can be added up for effect prediction. IA predicts that mixture effects of substances with different modes of action that share a common toxicological endpoint29 equal the sum of the individual effects of the mixture components, while considering the fraction of the test population already affected and thus ensuring that the overall effect cannot exceed 100%. Both concepts assume complete knowledge of the substances present in a mixture and the toxicities of each individual substance (effect concentration x for CA, whole concentration–response curve for IA), which classifies them as bottom-up approaches. Other commonly applied methods for mixture toxicity predictions and effect classification for detection of synergism/antagonism are summarised in the SI (SI, Section 1 and Table S2).
In general, deviations of measured effects from these prediction methods are classified as antagonism (lower observed mixture effect than predicted) or synergism (higher observed mixture effect than predicted). Synergistic effects can occur as a result of direct substance–substance interactions, from toxicokinetic and/or toxicodynamic interactions,35 as well as through the combination with environmental stressors, such as elevated temperatures or humidity.36 They are regularly quantified by the model deviation ratio (model over observation), where ratios <0.5 and >2 indicate antagonism and synergism, respectively, but other thresholds and methods are also used.37 A detailed summary of the applied methods to detect synergism and antagonism is presented in the SI (SI, Section 1). From this summary, as well as from previous work,38 it can be concluded that there is no standard for synergism and antagonism classification. Methods for the detection of synergism and antagonism vary widely: they partly consider experimental uncertainty through statistical testing or define a range for null model conformity, as the model deviation ratio does. The relevance of interaction and synergistic/antagonistic effects at environmentally relevant concentrations, however, is at least questionable, since deviations from additivity have rarely been found to date at such concentrations.38–40 In realistic exposure scenarios and experimentation, it is extremely difficult, and often computationally infeasible, to account for all possible combinations of mixtures that may occur in the field. This calls for more pragmatic approaches in terms of the MRA in soils. In the context of MRA, the determination of specifically synergistic effects would become relevant if a lower-tier RA identified a mixture risk, which could be experimentally verified in a refining higher-tier RA step to reduce uncertainty in the mixture risk prediction. The current experimental knowledge on synergistic mixtures proves useful to identify potential mixture risks, but experimental coverage of all mixtures becomes very difficult if one considers the nearly infinite substance combinations that may be encountered in the field. Therefore, the focus of low-tier MRA should be on enabling the regulatory handling of mixtures in soil to reduce the overall risk of the co-occurrence of substances.
4. Main findings from the literature review
Studies that fall into the first category (conducted an MRA, see Section 3) are relevant for the evaluation of soil MRA, while studies in the second category (mixture effect prediction, see Section 3) give valuable insights into experimental aspects of mixture studies in soil. Studies falling into the second category are frequently exposure studies in soil using standard test species along with selected mixture constituents that are often binary mixtures of pesticides or pesticides with other contaminants. The methods to detect mixture-induced antagonisms or synergisms in these studies have a wide spectrum that ranges from statistical significance testing to additivity-based effect classification or hybrid versions (SI, Section 1).The studies falling under the first category are scrutinised under the aspects of mixture effect analysis with concurrent MRA in Sections 5.1 and 5.2, while experimental aspects of mixture studies with no concurrent MRA are discussed in Section 5.3.
4.1. Findings of PPP residue surveys and mixture risk assessments in soil
Based on European soil surveys conducted on different geographical scales, it is without question that the majority of agricultural soils contain one to several PPP residues. The main findings of the studies are the following:(i) Irrespective of whether farmed or not-farmed (and in case of farmed soil, the farming practice: conventional or organic), in most cases, several PPP residues were present in the soils.4,5,7,9–16,41–48
(ii) In a majority of the reviewed studies, the PPP residue mixtures with realistic exposure concentrations (Table S3) had negative impacts on collembola, earthworms and plants,44,49–61 while the effects on the soil microorganism community were much more variable and transient.17,19,36,49,53,59,62–65 The assessed endpoints were mostly mortality, reproduction and biomass for earthworms and collembola, while it was seed germination, shoot/root growth and biomass for plants. All of these are meaningful endpoints currently used in the ERA of non-target soil organisms. However, most of the studies on soil microorganism communities assessed non-standardised effects on abundance and diversity of bacteria or fungi, mycorrhization and abundance of genes related to nitrogen-fixation. While these microbial biomarkers are very sensitive sublethal endpoints, current guidelines do not recognise them as usable endpoints for ERA (with the exception of exo-enzymes), likely due to their dynamic character. At these different and non-comparable organisational levels (e.g. diversity vs. functioning), ecological variability has much more influence and the impact of other environmental stressors increasingly hampers the effect prediction.62,66
(iii) In a majority of the studies with MRA (Table S4), a mixture risk to soil organisms was indicated in parts of the surveyed fields or assessed mixtures.4,5,7,12,13,15,44,47,67–70 The MRA frequently relied on mortality or reproduction effects in earthworms and collembola, which were collected from literature and databases. They often represented the most sensitive organism group for which data were collected.
Comparability among the studies for their most frequently detected PPP residues and associated toxicities and mixture risks is limited. Sampling times vary among studies: in some studies, samples were taken during the vegetative period,11,13,14,44 while in others, samples were taken outside the vegetative period,4,10 and in one study, the sampling time points were not reported.12 Only the sample timing can influence the outcome of an MRA regarding mixture risks as well as the mixture composition. For example, soil sampling during the vegetative period likely results in higher mixture risk of PPP residues than soil sampling outside the vegetative period. Further, the scope of the monitoring and the scientific inquiry differed among the studies. For example, Brühl et al. (2024)11 focussed on currently used pesticides according to the location and altitude of the surveyed region, while Hagner et al. (2025)13 measured currently used and banned pesticides on a national scale, which were dependent on the agricultural field type and agroecological practices at the sample sites. The studies scrutinise different scientific hypotheses and, therefore, a comparison of the most frequently detected substances is hampered.
The major issue for MRA that occurs with the heterogeneous study aims in monitoring studies is that the differing numbers and substance identities likely create contrasting mixture risk indications, which hamper comparability across studies. Since the monitoring datasets would most likely be used to sum up risk quotients,12,23 it is crucial to identify missing substances to explain potential differences in the observed mixture risks. For a holistic MRA, it is advisable to include legacy substances, whose PPP application has been forbidden, as well as currently used substances in PPP. The inclusion of both substance groups would enable the simulation of various scenarios of substance exclusion to evaluate management actions for mixture risk reduction. To make use of the heterogeneous monitoring datasets for MRA, various strategies could be used to improve comparability. Options are to focus on those substances that occur frequently in all datasets or prioritise the substance selection according to a weighing of detection frequency, ecotoxicity, and PPP approval status. If important substances are lacking for certain datasets, modelling approaches that make soil concentration predictions based on substance application rates and other factors, such as physicochemical properties of the substances, could fill those gaps, but require an uncertainty analysis.14,71
4.2. Current risk assessment methods for mixtures in soil
Table 1 summarises the major MRA methods that are applied in the reviewed studies. The majority of studies attempted to quantify the mixture risk by using the sum of risk quotients, RQsum, to identify potential harm to soil organisms. Only a few other methods were applied in a small number of studies: among them, the integral risk index and the hazardous concentration determination via species sensitivity distribution (SSD; Table 1). All these methods are bottom-up approaches, where the concentrations of all components or component groups (see below) in the mixture and their toxicities need to be known.| Method | Formula | Description |
|---|---|---|
| a Vašíčková, Hvězdová,4 Pelosi, Bertrand,5 Froger, Jolivet,7 Franco, Vieira,12 Hagner, Rämö,13 Honert, Mauser,15 Panico, van Gestel,44 Mu, Yang,47,48 Jiang, Wang,50 Tourinho, Hochmanová,60 Fekadu Demsie,68 Sánchez-del Cid,69 Roani,70 Zhao, Yang,91 González-Naranjo and Boltes,134 González-Naranjo, Boltes,135 Wang, Zhang,136 Gallego, Shipley,137 Urbaniak, Baran.138b Nadal, Kumar.83c Gainer, Bresee.86 | ||
| Sum of risk quotients (RQsum) | 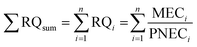 |
Sum of individual risk quotients of pesticides (RQi) to calculate aggregate risk, also termed the hazard index. RQi is calculated as a ratio of measured or predicted environmental concentration (MEC or PEC) over predicted no-effect concentration (PNEC) from toxicity data on soil organisms for each substance in the mixture. In experiments with filter paper and aqueous exposure, the PNECsoil is calculated from PNECwater with an equilibrium partitioning approach (EC, 2022)a |
| Integral risk index | 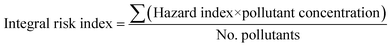 |
Hazard index (HI) is based on USEPA definition (on P = persistent, B = bioaccumulative and T = toxic). To build the HI, the variables are grouped according to the PBT category by summing the values. The self-organising map normalises the values to set a range between 0 and 1 and uses weightings for each evaluated criterion. No. pollutants = number of pollutantsb |
| Species sensitivity distribution (SSD) |  |
SSD is used to determine the hazard concentration that affects x% of the population (HCX). Typically, a log-logistic model is fitted to data of one type of endpoint (e.g. EC10 or NOEC of reproduction endpoint) across different species. The best fit model is used to obtain hazard concentrations affecting x% of the assessed community (F(x, α, β)). Note that the parameterisation of the model may vary from that presented here (cumulative distribution function represented if α > 0, β > 0, and x > 0)c |
RQsum is obtained by the sum of individual quotients of measured or predicted substance concentrations in soil over their predicted no-effect concentration (PNEC). The PNEC is typically derived from the lowest NOEC available from bioassays with soil organisms exposed to a single substance (experimental or literature data) and divided by an assessment factor (AF).4,12,47 This commonly applied method has the intention of protecting the in-soil organisms and plants as a first-tier approach for mixture risk screening and therefore attempts to account for various uncertainties.4,12,29,47 Such uncertainties arise, for example, from the extrapolation of laboratory-based exposure scenarios to field conditions and to account for species sensitivities of soil organisms, which are not testable due to lacking standard or well-established test methods, but still may be exposed in the field. In the relevant studies, the choice of the AF varied from 5 to 1000 to account for uncertainty, and depended on the quality and availability of toxicity data. For example, an acute toxicity endpoint (50% effective or lethal concentration from an acute dose–response study) was multiplied by an AF of 1000 to transform it into its chronic equivalent when no other acceptable chronic NOEC or EC10 was available.4 EU regulation guides the choice of the AF size dependent on the toxicity data availability for different trophic levels,72 which has been followed in the studies that apply PNEC in the RQsum method: The highest AF of 1000 is chosen when only short-term toxicity endpoints are available, an AF of 100 is chosen when one chronic NOEC is available, an AF of 50 is chosen when two long-term NOEC are available for two different trophic levels and an AF of 10 is chosen when three long-term NOEC are available for three different trophic levels. The lowest AF from 5–1 are selected on a case-by-case basis for higher-tier field studies/data of model ecosystems or where sufficient data are available to construct an SSD. The RQsum method relies not just on the quality of the data but also is constrained by the availability and representativeness of single-substance toxicity data for soil organisms, which may introduce additional uncertainty into mixture risk estimates. Currently, there is no other option than to use AF to consider the uncertainties noted above since the direct link between chemical exposure and the integrity of an ecosystem is difficult to establish. Therefore, the AF values are used in line with the precautionary principle and the resulting mixture risks represent a lower- or intermediate-tier screening for potential risks.
Data scarcity on non-target soil organisms has been reported in concurrent risk assessment studies that use the RQsum method.4,5,12,44 For example, Franco et al. (2024)12 found a median of 2 for the available number of effect concentrations for each of the monitored PPP residues, which implies that for a considerable number of the PPP, only one data point was available from their sources of toxicity data, which comprised the Pesticide Properties Database (PPDB),73 the EFSA OpenFoodTox database,74 and the USEPA ECOTOX Knowledge database.75 The use of PPDB and the USEPA ECOTOX Knowledge database for the extraction of toxicity values, however, requires a careful evaluation of the studies and reported endpoints.76,77 It occurs frequently in these databases that entries fail to state whether a NOEC was the highest tested exposure concentration or whether it was derived from a qualitatively acceptable concentration–response curve. Further, it is often not clear whether the reported concentration was corrected for bioavailability or not.
It was also noted that the majority of the available effect concentrations for MRA in the studies were measured in earthworms4,12 that were, for a long time, implicitly assumed to be representative of the soil ecosystem in the prospective RA. Regulatory requirements changed in 2009![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 78 and additional toxicity data on collembola, predatory mites, and microbial nitrogen fixation are now, under specific exposure circumstances, required to increase the coverage of the different trophic levels in soil. These data are publicly available to EU citizens through the EU prospective RA dossiers of PPP, and partly through databases such as IUCLID (https://iuclid6.echa.europa.eu/).
78 and additional toxicity data on collembola, predatory mites, and microbial nitrogen fixation are now, under specific exposure circumstances, required to increase the coverage of the different trophic levels in soil. These data are publicly available to EU citizens through the EU prospective RA dossiers of PPP, and partly through databases such as IUCLID (https://iuclid6.echa.europa.eu/).
However, toxicity data are largely limited to a few standard laboratory species, which do not fully represent field populations. For example, earthworms, collembola, and mites encompass multiple species with distinct ecological traits, exposure pathways, and life histories, which will result in different sensitivities and exposure patterns. This has led to calls for including more options for intermediate- or higher-tier testing (e.g. effects on communities) in soil ecological risk assessment.79 Only one survey study on European soils attempted to fill gaps in the toxicity data with modelled values using a quantitative structure–activity relationship model (QSAR), where low reliability predictions were excluded, and it was found that the mixture toxicity driving substances were generally not affected by a lack of experimental toxicity data.12 The data availability on toxicity values limits any bottom-up MRA – that is, the usual regulatory approach – and may result in underestimation of the risk posed to soil organisms. Here, the data scarcity calls for more ecotoxicity testing on a diverse range of soil organisms, which goes beyond earthworms and robust, reliable modelling methods for interpolation. However, since many renewal assessment reports of active substances (which form the bases of the databases PPDB and Openfoodtox) are pending to date, the missing data for some substances might become accessible in the coming years. In the past, only earthworms (acute, later chronic test) and microorganisms were obligatory data requirements in RA dossiers of active substances. In addition to the number of available toxicity data, it is important to note that apical endpoints, such as mortality or reproduction, are more prevalent for single-exposure studies relative to molecular toxicity endpoints (e.g. protein and gene expression levels).
Several studies calculated the PNECsoil not from classic toxicity studies in soil but from PNECwater using the equilibrium partitioning approach.50,80,81 The equilibrium partitioning approach is an accepted method for use in RA for certain substances and is implemented in European regulation; it assumes a partitioning between the aqueous and solid fractions in soil until substance equilibrium is reached between the two compartments.72 It makes the basic assumption that the chemical partitioning in soil follows the substance's hydrophobicity, described by its KOW. A major drawback of this approach is the common use of toxicity endpoints from aquatic organisms for PNECwater derivation, which is then translated via partitioning to a PNECsoil without the consideration of toxicity endpoints from soil organisms. Soil is a three-phase system (i.e., it consists of solid particles, liquid and gas), and the use of PNECsoil derived by the equilibrium partitioning method from PNECwater considers effects on soil organisms as if they were exposed exclusively through the soil pore water and not the soil particles.72 In addition, the equilibrium partitioning approach relies on simplified and often default soil parameters (e.g. bulk soil density, organic carbon content, and pore water fraction), which may not reflect site-specific conditions. Since these parameters directly influence the distribution of chemicals between solid and aqueous phases, their generic treatment may introduce additional uncertainty when translating PNECwater to PNECsoil. Hence, this method is considered only as a “screening approach” for the environmental RA of biocides.82
The two lesser applied risk assessment methods differ considerably in their approaches to soil risk assessment (Table 1). The integral risk index is a machine learning-based approach that applies neural networks and self-organising maps to derive a risk scale and relate all sample sites with their relative risk to each other.83 The method is innovative, but has only been applied to human toxicity data with the protection goal of human health. To date, this approach has not been followed up on to evaluate its applicability in soil mixture risk case studies.
The other less commonly applied RA uses SSD. This method has been widely integrated and applied as a probabilistic approach in regulatory RA.84,85 Briefly, for an SSD, all available toxicity data on comparable effect concentrations (e.g. EC50 or LC50) across a range of species are selected and arranged from high to low toxicity. The resulting cumulative curve is then used to derive the so-called hazardous concentration (HCX), at which x percent (commonly 5%) of the selected species community is affected and defined as a threshold value. However, the use of SSD in soil MRA has been very sparse. Only one study concerning MRA applied SSD on mixture experiments with polycyclic aromatic hydrocarbons on different organisms, including plants.86 The major challenge that an SSD approach for MRA faces is the availability of effect sizes for one mixture at different concentrations for at least 8–10 (ideally >15) species to fulfil the SSD quality criteria.35,72,79,87 It is recommended that soil RA use the SSD method when at least 10 equal to – data points (as opposed to ≥ or ≤ – values) from different species and at least 3 taxonomic groups are available.88 However, these already data-intensive requirements would only cover one specific mixture, leaving out the infinite number of possible mixtures that may occur in the field. Therefore, the SSD approach does not appear to be pragmatically feasible as a standard method in MRA.
One option to include functional elements and address functional redundancies in the SSD approach is the recently introduced eco-indicator sensitivity distribution (EcoSD) approach.89 The approach is currently employed for single-substance testing, and its implementation for mixtures would entail considerably higher data requirements. Two options may be applicable if mixtures were to be assessed with an EcoSD. Either the mixtures would have to be experimentally tested over a range of concentrations for many different soil organisms, or the assessment would have to rely on the availability of single-exposure data and assume conformity to a mixture model to predict mixture effects for the SSD derivation. Both options deserve further scientific inquiry and promise valuable insights for EcoSD as a probabilistic MRA.
4.3. Experiments in soil with mixtures
The scientific literature on experimental mixture studies in soil was reviewed to extract relevant data, including the identity of tested substances, organisms and endpoints, as summarised in Fig. 1, with complementary information in Tables S5–S7. With regard to Fig. 1A, most authors of the screened studies chose the test substances because of their frequent detection in the environment, their high sales numbers in the (regional) commercial market, and the general availability of knowledge on their mode of action and other toxic properties, such as endocrine-disruptive potential. From the surveyed experimental studies, more than half worked with binary mixtures, while a small number of studies used field-sampled soils and the contained realistic mixtures of residues and exposure concentrations (Table S8, binary: 40 studies, binary/ternary: 1 study, several substances: 21 studies, no applicable classification: 26 studies). Interestingly, the pesticide classes of the most frequently studied substances in the experimental studies (Fig. 1A) do not reflect the most frequently detected substances in monitoring studies. Clearly, insecticides are assumed, due to their often known mode of action on target organisms, to have stronger effects on non-target organisms such as earthworms and collembola. Therefore, they may have been preferentially studied in experimental mixture studies, although they are a less frequently detected substance class in soil monitoring studies.4,5,9–16,41,63 Fungicides are underrepresented in the experimental mixture studies, while they are more frequently detected in monitoring studies. To determine whether pesticide residue mixtures pose a risk to soil organisms in the field and assess potential effects, it would be informative to prioritise mixtures that are frequently detected in the field, as well as consider the PPP's current approval status and ecotoxicity.In studies that used field-sampled soils contaminated with PPP residues and metals without additional substance spiking, it can be assumed that exposure concentrations to the soil organisms were realistic. Further, those studies that applied PPP formulations at manufacturer-recommended doses or simply tested contaminated field-sampled soils give more realistic indications of the consequences on soil organisms in the field.17,19,36,44,50–54,60,86,90–96 Studies that used equipotent exposure concentrations for their mixture design, the resulting exposure levels may resemble an acute exposure scenario, since the concentrations were mostly related to the 50% lethal or effect concentrations to soil organisms.97–99 These concentrations are commonly well above the corresponding detected environmental concentrations in monitoring studies. Concentrations promoting stronger effects (50% effect concentration) may eventually occur immediately after PPP application in the field and thus may provide information on the short-term effects on the exposed organisms. This short-term exposure is rarely captured in monitoring studies; rather, it occurs by chance during sampling in the vegetative season.15 The studies that used equipotent exposure concentrations for their mixture design are often laboratory-based experiments (Table S8) and may introduce more uncertainties in the extrapolation to field conditions. Further, it is unclear whether PPP mixtures of equipotent exposure concentrations represent a realistic composition of environmentally relevant mixtures. Therefore, in the first line, mixture studies with equipotent exposure concentrations make valuable contributions to the testing of experimental mixtures for potential interaction or predictability of CA and IA for a range of substance mixtures.
With regard to the tested soil organisms, the earthworm Eisenia fetida was by far the most often used species for experiments, while the next two most often tested organisms were the springtail Folsomia candida and the soil microbial community (Fig. 1B). Soil organisms for which established culture methods and standard guidelines for testing exist were preferentially used for experimentation. It stands out that there was no clearly preferred plant model species. The main reason for preferred experimentation with certain plant species appeared to be their regional economic relevance and sensitivity to pesticide exposure.
Some studies addressed the effect of PPP residue mixtures on fungi, the soil fungi community, or the overarching soil microbial community.17,19,36,49,59,62,95,100,101 Most often, these studies assessed fungal taxa abundance and community structure, which are more dynamic ecosystem-level effect assessments. A less frequently assessed endpoint was the arbuscular mycorrhizal fungi colonisation through Glomeromycota, beneficial plant symbionts,49,63,95,102 which links directly to nutrient cycling as an ecosystem service.89 Fungicides are regularly detected in European soils, which undoubtedly target undesired fungal growth on crops.5,6,12,13,15 Yet, the impact of PPP residues on non-target fungi such as the ecologically important Glomeromycota103 remains largely unassessed in experimental mixture studies or in regulatory ERA in the EU. The currently single soil microorganism endpoint of N-mineralisation required in European pesticide regulation implicitly addresses fungi activity in soil, but the inclusion of more direct toxicity endpoints in ERA, such as spore germination of Glomus mosseae,104 is desirable.
Regarding quantified endpoints (Fig. 1C), mortality, diverse biomarkers, reproduction and seed germination rank among the most frequently measured in the experimental mixture studies. For each of the test organisms and measured endpoints, official test guidelines can be found.105–112 Where microorganism communities were studied (mostly biomarkers such as enzyme activity or gene expression to quantify species richness or diversity of fungi and bacteria), no standard test guideline or uniform common method was followed,6,17,19,49,53,63,91,93–96,101 except for one study.113 Beside common gene analyses (16S rRNA gene for bacteria and Internal Transcribed Spacer regions for fungi) to derive diversity indices, such as the Shannon or Simpson index,17,49,91,96,101 many studies characterised the microbial community structure by diverse methods, for example, mycorrhization of plant roots, soil microbial biomass63 or, less often, soil aggregate stability.19 Qualitative effect analysis may be possible through these methods; however, for quantitative RA, standard methods103 should be followed to enable regulatory scrutiny and comparability among the studies. Yet microbial life in soil is currently only assessed in regulatory ERA of PPP through nitrogen transformation,114 and an update to recommend more diverse methods is advisable.115
From the above observations, it can be concluded that there is a strong reliance in experimentation on well-established test animals and plants with corresponding apical endpoints. In RA, these organisms and endpoints are assumed to represent the ecosystem health due to their ecological function in the soil ecosystem.116,117 However, molecular toxicity endpoints, summarised here as biomarkers (Fig. 1C), were the second most frequently tested endpoints, although their relationship to effects at the population level or even the soil-ecosystem is uncertain, less sensitive compared to functional endpoints,65 or simply unknown. Currently, only exo-enzymes can be used as biomarkers for the ecological functioning of soil and are usable for terrestrial RA.118–120
In general, ecological effects are currently not well integrated in regulatory RA, although scientific evidence would justify an integration of ecological markers for ERA.28 However, standardisation and effect prediction of ecological effects for ERA have rarely been attempted,19,62,66 and their usability for regulatory ERA remains to be evaluated, provided that sufficient data are available for such an assessment.
Only a small fraction of the reviewed experimental studies addressed bioavailability as an influencing factor for exposure concentrations and thus toxicity to soil organisms, although the majority reported the organic carbon fraction, the assumed main adsorbing soil constituent, in the tested soil. Some experimental studies addressed the influence of bioavailability either by quantification of tissue concentrations in exposed organisms,50,92,99,121–126 correction of quantified concentrations in soil to consider the soil organic carbon bound fraction,86,121 or by consideration of a substance's hydrophobicity and soil organic content for literature-retrieved toxicity values according to regulatory guidelines.60
In comparison to the RA of substances in the aquatic environment, the RA in soil faces challenges that are associated with the soil matrix. Soil is a complex aggregation that consists of large parts of mineral particles, but also of organic matter, air pockets, pore water and living organisms. Bioavailability is influenced by a host of variables, which are more or less well quantified. To estimate the bioavailable substance fraction in soil and sediment, Kf, the partitioning coefficient that defines the ratio between concentration in bulk soil and pore water, is normalised to the soil organic carbon fraction, fOC,127 to result in KOC, as is implemented in the EU evaluation of industrial substances.128 However, this relation is based on neutral organic compounds and is not applicable to ionisable organic compounds.129,130 In the case of ionisable organic compounds, at least the local pH and water hardness should be additionally considered in the estimation of bioavailability in soil.129 Since soil is largely negatively charged, the mobility and thus bioavailability of negatively and positively charged substances can vary considerably. In order to account for bioavailability, an overly simplistic method is still used for pesticide authorisation in the EU. In this method the effect concentrations for earthworms and other soil macro-organisms are divided by 2 in case the log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) KOW (decadic logarithm of the octanol–water partition coefficient) of the substance is >2, unless it can be demonstrated that the toxicity is independent of fOC.131 In addition to bioavailability, knowledge of exposure pathways to many soil organisms is not well known. Therefore, it remains questionable how the estimated bioavailable fraction in soil corresponds to the actual dose that soil organisms are potentially exposed to and may take up. To date, the study of toxicokinetics of substances in soil organisms is mainly focussed on earthworms,123–125,132,133 whilst other soil organisms have rarely been investigated.122 If the data availability improved on bioavailability and concurrent uptake pathways concerning different soil-dwelling species, these could be readily incorporated into the current RA approaches to reduce uncertainty.
KOW (decadic logarithm of the octanol–water partition coefficient) of the substance is >2, unless it can be demonstrated that the toxicity is independent of fOC.131 In addition to bioavailability, knowledge of exposure pathways to many soil organisms is not well known. Therefore, it remains questionable how the estimated bioavailable fraction in soil corresponds to the actual dose that soil organisms are potentially exposed to and may take up. To date, the study of toxicokinetics of substances in soil organisms is mainly focussed on earthworms,123–125,132,133 whilst other soil organisms have rarely been investigated.122 If the data availability improved on bioavailability and concurrent uptake pathways concerning different soil-dwelling species, these could be readily incorporated into the current RA approaches to reduce uncertainty.
4.4. Most commonly applied mixture risk assessment in soil
The summation of substance-specific risk quotients, RQsum, is the common indicator for soil mixture risk estimation (Tables 1 and S4).4,5,7,12,13,15,44,47,48,50,60,68–70,81,91,134–138 Notably, the majority of the studies used earthworm toxicity endpoints (preferably chronic over acute) for derivation of PNEC values and RQsum calculations. About half the studies exclusively assessed the mixture risk for earthworms, whilst others intended the RA of the soil ecosystem and used earthworms as the representative soil organism (Table S8). The latter approach has potential for underestimation of mixture risks, due to the varying sensitivities of the soil organism groups to the different pesticide groups. For example, non-target plants are likely to be more sensitive to herbicidal mixture exposure than earthworms or springtails.139 Therefore, the corresponding RQsum based on earthworm toxicities to herbicides would underestimate the mixture risk for non-target plants. Ecotoxicity data of several trophic levels in the soil ecosystem are usually available in the EU registration dossiers of PPP, but their extraction for MRA is laborious and spray application rates require non-standardised unit transformations (g ha−1 application to mg g−1 of a certain soil depth). Therefore, most studies presumably relied on already extracted ecotoxicity datasets from earlier studies, e.g. Vašíčková et al. (2019),4 or from databases such as PPDB (Table S8). Yet, data gaps in the MRA are unavoidable due to missing ecotoxicity data, especially if larger numbers of screened PPP residues were monitored in the studies. Such data gaps and the potential underestimation of mixture risks were often not discussed (Table S8). A few studies attempted to fill the ecotoxicity data gaps through the use of either ecotoxicity data from structurally related substances4,67 or quantitative structure–activity relationships,12,23 but the discussion and uncertainty analysis were rudimentary. Besides these methods, more consistent data gap filling could be achieved through organism-specific models,140 but these need careful assessment with regard to the assessed soil organism and ecotoxicity endpoint. Such models are currently limited to earthworms, and adaptation of the model to other soil organisms would be desirable to avoid cross-species extrapolation (see above). Many studies additionally categorised the RQsum into risk classes, as first proposed by Sánchez-Bayo et al. (2002),1414,5,7,13,15,44,48,70 who derived the categories based on toxicity data that did not include soil organisms. The bases of aquatic and terrestrial toxicity data increase the uncertainty of these risk classes for MRA in soil. This raises the question of whether a soil-tailored classification system is required to classify mixture risk sizes and which soil organisms are sensitive enough to reflect mixture toxicity effects. So far, very few studies have attempted to examine the correlation between observed experimental mixture effects and the predicted mixture risks and their numerical sizes; namely, Panico et al. 2022![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 44 and Tourinho et al. 2025.60 From these studies, it can be concluded that a more accurate correlation between mixture effects and the size/height of the mixture risk depends on two aspects: First, availability of high-quality ecotoxicity data for the assessed soil organism and the relevant effect endpoint, and, second, their bioavailability correction to the properties of the assessed soils. The correction of ecotoxicity data to the bioavailable fraction was only considered in one of the assessed studies (that applied RQsum) by consideration of the EU-guidance-recommended correction for soil organic matter.60 None of the other studies considered this aspect, which likely resulted in a collection of ecotoxicity data that are not standardised to the same soil organic matter content, which reduces the informative value of the corresponding MRA. As noted above, if more knowledge on the bioavailability aspect for the different soil organisms became available, it could be implemented in ERA, but, for now at least, the correction for soil organic matter should be performed as the minimum requirement for cross-soil comparability and to include an aspect of bioavailability. Further, in the context of the European soil monitoring law,142 it would be desirable to have a central database that transparently provides all relevant information on soil ecotoxicity data across multiple trophic levels to enable robust and comparable MRA of monitoring programmes.
44 and Tourinho et al. 2025.60 From these studies, it can be concluded that a more accurate correlation between mixture effects and the size/height of the mixture risk depends on two aspects: First, availability of high-quality ecotoxicity data for the assessed soil organism and the relevant effect endpoint, and, second, their bioavailability correction to the properties of the assessed soils. The correction of ecotoxicity data to the bioavailable fraction was only considered in one of the assessed studies (that applied RQsum) by consideration of the EU-guidance-recommended correction for soil organic matter.60 None of the other studies considered this aspect, which likely resulted in a collection of ecotoxicity data that are not standardised to the same soil organic matter content, which reduces the informative value of the corresponding MRA. As noted above, if more knowledge on the bioavailability aspect for the different soil organisms became available, it could be implemented in ERA, but, for now at least, the correction for soil organic matter should be performed as the minimum requirement for cross-soil comparability and to include an aspect of bioavailability. Further, in the context of the European soil monitoring law,142 it would be desirable to have a central database that transparently provides all relevant information on soil ecotoxicity data across multiple trophic levels to enable robust and comparable MRA of monitoring programmes.
4.5. Mixture toxicity in current regulation
In general, the challenge of mixture toxicity is recognised, but the current legislation and related guidelines provide only partial instructions for MRA85,143–151 and tend to focus on the protection of human health from mixture effects. Principally, CA is the default approach for MRA for substance groups with a similar mode of action, and special cases, such as synergistic effects, require in vivo evidence. Mixture types are differentiated into intentional (e.g. pesticide or biocidal product formulations), unintentional (e.g. discharges from production, transport or disposal) and coincidental mixtures (e.g. several sources over space and time).151Coincidental mixtures, as they occur typically in the environment, are partly addressed by substance grouping and relative potency factor methods in monitoring directives (Directive 2000/60/EC, Directive 2013/39/EU) or in the context of contaminated sites and the derivation of soil standards for soil contaminants to protect human health143,145,160,161,166 or non-target organisms in soil.85,146,149 For substance groups that align with the assumption of toxic additivity, such as petroleum hydrocarbons, dioxins/furans and polychlorinated biphenyls, fractions of petroleum hydrocarbons (carbon chain length-dependent), or relative potency factors, may be used for mixture assessment.143,145,147–149,152 Another option is to determine the affected fractions of species from SSD per substance at a contaminated site, which are subsequently incorporated into the CA or IA model depending on the substances' mode of action.85,146
Besides the agreement on CA as the default method for MRA, current guidelines have little coherence in the applied methods and protection goals from mixture effects. Common methodologies are only coherent for well-studied legacy substances, where the CA concept would apply or where relative potency factors were established. Other substance groups are not explicitly addressed and an MRA is made dependent on available data and where a mixture effect is evident and relevant to the formulated protection goal. The regulations leave it largely open of how a mixture effect may be detected and does not require the generation of data or fill data gaps with model approaches to inform the MRA. Intentional mixtures are the only mixture types where clear guidance on a MRA is available,150,151 and the regulations on pesticidal78,153–156 and biocidal products157 are the most prominent examples of the operationalisation of the assessment. These products are mixtures containing one or more active ingredients along with co-formulants (safeners, synergists, adjuvants and other additives). The use of tank mixtures of products, the sequential application of the different PPPs over time, as well as the potential formation of biotransformation and degradation products, which may prolong or increase the product's overall toxicity, have been acknowledged as mixture toxicity problems.158,159 However, given that these aspects are not yet considered in an MRA,155,158,159 this may result in further tightening of the safety limits of individual substances.
Mixture effects are deemed relevant in ecotoxicological testing and RA if the toxicity of the PPP cannot be predicted based on the toxicity of the active ingredients.154,160,161 This can be the case when: (i) the PPP contains more than one active substance or (ii) the toxicity of the PPP differs from that of the active ingredient. For PPP authorisation, the applicant (i.e. the owner of the PPP) has to demonstrate the safe use of the product containing one or more active substances. Detailed qualitative and quantitative information on the composition of the PPP must be provided that includes the active ingredient(s), any relevant impurities, safeners, synergists and co-formulants.154 The exact composition – beyond the amount of active ingredient(s) – is, however, confidential and not disclosed publicly. As a result, soil-ecotoxicological testing and ERA of the co-formulants themselves for the soil ecosystem are not systematically performed, which may have consequences for the accuracy of any MRA.162–164 This problem has been recognised in the scientific community, but data on the mixture effects of certain active ingredients and co-formulants on non-target soil organisms is scarce in experimental mixture studies. In our review, only experiments with toluene, xylene, and trichloroethylene were found.136 The reasoning for testing these substances was because of their function as co-formulants in PPP.136
Overall, current regulations and guidelines give little incentive to generate new data on toxicity to soil organisms, but rather rely on currently available data and concepts. With the upcoming soil monitoring law142 in the EU, the situation may be changed (1) by obtaining information on the general contamination with various substance classes in various land uses and landscapes through long-term monitoring programmes and (2) by setting soil standards to protect soil health from single substances as well as substance mixtures. Both aspects would encourage the generation of toxicity data on observed mixtures, enable the refinement of model approaches, and ultimately expand the knowledge on mixture effects in the soil ecosystem.
4.6. Novel concepts
A small number of studies applied unconventional concepts for effect prediction in experimental mixture studies (for details, see SI, Section 3) or the estimation of mixture risks. Most of these studies focussed on mixture effect prediction rather than presenting a new framework for MRA. The largest part of the unconventional experimental mixture studies attempted to provide mixture effect predictions of realistic substance exposures, partly in combination with abiotic stressors such as salinity, humidity or temperature.19,36,52,62,124,165–167 The following subsections outline and discuss the usability of novel MRA methods, while the SI provides more details on the experimental studies.In a proof-of-concept study, the landscape-based ERA principles were applied, where reported toxicity data from RA dossiers informed population–relevant parameters to feed a simplified population dynamics model.169 Overall, this population model simulated the application of folpet and pirimicarb in a defined agricultural area over time and space to quantify the effects on mammal and bird populations. This exercise enabled the identification of risk drivers for population effects, but not yet the risk interpretation needed for a regulatory context. Similar exercises could be realised in the context of the soil ecosystem with toxicokinetic-toxicodynamic models of selected soil organisms in combination with landscape-level exposure models.170,171
For risk quotient calculation, the lowest overall chronic toxicity endpoint across the trophic levels per substance is selected to represent the substance's toxicity (marked in bold in Fig. 2: 0.56, 29 and 23 mg active substance per kg soil) in the PNEC derivation (“Quality Criterion”, Fig. 2). Then, any trophic level with toxicity values within a factor of 10 of the lowest toxicity endpoint is considered sensitive enough to be included in the mixture risk assessment (in Fig. 2 column “Considered?”) for the assessed substance and is assigned the quality criterion/PNEC of the assessed substance. In this example, the Quality Criterion is represented through fictive PNEC, which were derived based on the lowest and most reliable NOEC or ECx across the different trophic levels. The AF size is selected dependent on the ecotoxicity data availability per substance, in line with EU guidance (https://op.europa.eu/s/y4d4). Note that, in line with the precautionary principle, missing toxicity data in the trophic levels always trigger a consideration of the corresponding trophic level (e.g. “Substance 3”, Fig. 2). Trophic levels with more than 10 times higher toxicity values than the sensitive level are not considered relevant for the assessed substance and are set as 0 for risk quotient derivation for that trophic level. Once all substances in a mixture are assessed, the risk quotients are calculated by division of MEC over the corresponding quality criterion (shown in italic in Fig. 2) and summed per trophic level. The example in Fig. 2 shows that the highest concern for mixture risk is identified for trophic level 1 and exceeds the commonly accepted mixture risk threshold of 1, while the mixture risk is lower for the other two trophic levels. The overall risk of the mixture is equal to the risk for the most sensitive trophic level.
The resulting mixture risk of RQsum and RQtaxa cannot be translated directly into predicted effects, since they combine various toxicity endpoints, species and uncertainty factors in the summation of risk quotients.24 Therefore, they qualify as an intermediate-tier mixture risk assessment, between RQsum and species-specific mixture risk assessment. The application of the RQtaxa or RQsum method could stop the refinement of an MRA in case the overall resulting mixture risk is ≤1, meaning there is no concern for mixture risks. RQsum and RQtaxa recognise that often the high-quality ecotoxicity data for various soil trophic levels are missing for the assessed substances in mixture, and therefore represent a conservative MRA approach that makes the maximum use of available data. Higher-tier assessments that build on the mixture risk identification of RQtaxa could further refine the MRA by focusing on the more sensitive organisms. For example, the grouping of the available data could be more strictly enforced according to the principles of CA, and, in case of missing experimental values, data generation in new experiments could also be guided.
As indicated in Fig. 2, the RQtaxa concept can be adapted to the number of tropic levels considered relevant for MRA and can be tailored to regulatory protection goals. Further, RQtaxa can also guide the generation of new toxicity data and point to knowledge gaps on taxa sensitivity to pesticides.
To date, this approach has not been applied in soil MRA, but it is regularly being applied for assessing surface water quality by Swiss cantonal laboratories.175,177 The main uncertainties of risk indication by RQtaxa are similar to RQsum and concern the origin of toxicity values (study quality, acute or chronic endpoint) and the use of assessment factors to cover uncertainty (e.g. extrapolation from tested to untested species, extrapolation of laboratory data to field conditions) and differences in sensitivity (e.g. acute to chronic endpoint extrapolation). The trophic levels in RQtaxa could be associated with soil functions to obtain risk estimates to address overarching soil protection goals. The evaluation of RQtaxa for its utility in soil MRA requires further scrutiny, and the comparison with other soil MRA methods, such as RQsum, would provide valuable insights.
Other concepts that use the risk quotients for mixture risk indication but have not been applied in a soil context are the mixture allocation factor (MAF) and maximum cumulative ratio (MCR). Both are currently under consideration for inclusion in the chemical registration under REACH.178 The MAF guides the determination of reduction factors, by which a single substance concentration in a mixture needs to be reduced to meet protection goals.179 It would be a simple but effective method to solve the above noted mixture problem in prospective soil RA of PPP (Section 5.5). Empirical evidence of MAF size distributions from soil monitoring campaigns would be readily available and could provide information on how big an additional assessment factor would need to be for sufficient protection from mixture effects in soil organisms.
The MCR classifies whether the risk from a given mixture is caused by a single substance, a small number of substances, or multiple substances in the mixture;180 thus it characterises the excess risk caused by the co-presence of multiple substances. The MCR indicates whether a given sample contains a mixture problem when MCR > 2 and RQsum > 1. Both indicators, MAF and MCR, again rely on the concentration additivity of the mixture constituents. One of the reasons why these methods have not yet been applied in soil ecotoxicology may be that the specific protection goals for flora and soil fauna have not been agreed upon in the European Union, which would give clear guidance on risk reduction aims. However, it would be a valuable exercise to obtain MAF and MCR distributions from soil contamination surveys to inform soil MRA and get a measure of the present excess risk in soils due to the co-presence of substances.
5. Conclusions for soil mixture risk assessments and recommendations
The surveyed scientific studies used mainly toxicity endpoints for earthworms, presumably the most sensitive species group, and concurrent data were often extracted from other studies or common repositories such as the PPDB. This practice may overlook other important non-target soil organisms, which may also play substantial roles in maintaining soil health, with concurrent underestimations by the MRA. For PPP residues, toxicity endpoints of the different trophic levels may be obtained through the assessment reports made publicly available by EFSA (https://open.efsa.europa.eu/), which usually contain toxicity data from some soil macro-organisms and microorganisms. In general, there was a noticeable data gap in MRA studies on certain soil organism species, such as predatory mites or fungi. Especially, beneficial fungi were not represented by a single model species, and chronic effects on these organisms through PPP residues remained largely unknown. Therefore, the generation of more toxicity data across all possible soil organism species will be important to create an even representation. The data generation would also benefit the establishment of mixture risk classes based on soil organism toxicities and not on that of aquatic organisms.Bottom-up risk assessments, which assume CA, are the most frequently applied concepts for soil MRA of PPP residues in the current scientific literature. In terms of assessment method, the summation of risk quotients, RQsum, is the most commonly applied method to estimate mixture risks in soil. RQtaxa, the refinement of RQsum, avoids taxa-specific risk overestimations and points to where toxicity data for specific trophic levels are missing, thereby guiding the generation of new toxicity data. Novel MRA methods lack available toxicity data (landscape-based ERA) or give a more general risk trend (TAT). In fact, with currently available toxicity data on soil organisms, RQsum is the most pragmatic approach for MRA, with the RQtaxa offering a potential for refinement while maintaining the same conceptual principles of the well-established RQsum.
Improvements in mixture risk assessments have to deal with data gaps on certain less-represented taxonomic groups and better consider bioavailability to allow general concepts and conclusions to be more broadly applicable across soils. Ideally, the goal for a more holistic assessment of soil ecosystem health would be the combination of diverse stressors, such as temperature increase, fertilisation, farming practises, and PPP exposure. Therefore, it is an encouraging development that these factors are increasingly being considered in experimental studies. More realistic exposure scenarios may uncover the extent of PPP residue effects relative to other factors and are one step closer to a landscape-based risk assessment.
Author contributions
Fabia Balk: conceptualisation, investigation, methodology, visualisation, writing – original draft, writing – review & editing. Eva Lauber: writing – review & editing. Benoit Ferrari: funding acquisition, project administration, writing – review & editing, Mathieu Renaud: funding acquisition, project administration, writing – review & editing, Marion Junghans: conceptualisation, methodology, funding acquisition, writing – review & editing.Conflicts of interest
The views and opinions expressed in this study are those of the authors and are not of their affiliated institutions or the funding agency.Data availability
All data for image generation are made available through the supplementary information (SI) via a data package (https://doi.org/10.25678/000FJH) and github (https://github.com/fabigpb/soil_mixture_risks; the figures were produced with the code and data version v1.3_review_publication: https://github.com/fabigpb/soil_mixture_risks/releases/tag/v1.3_review_publication). Supplementary information: tabular overviews to the various aspects detailed in the main text: overview on study search hits from the literature search (Table S1), descriptive methods for mixture effect classification and prediction (Table S2), overview on experimental mixture studies with realistic exposure concentrations (Table S3), overview of the case studies that apply the sum of risk quotients for their mixture risk assessment (Table S4), frequency of tested substances, organisms and measured endpoints in the reviewed experimental studies (Tables S5–S7), experimental studies with mixtures and exposed soil organisms (Table S8). See DOI: https://doi.org/10.1039/d5va00405e.Acknowledgements
The study was funded by the Swiss Federal Office for the Environment (FOEN). We thank Micha Wehrli for reviewing Fig. 2 to ensure they are accessible to readers with colour vision deficiencies.References
- P. M. Haygarth and K. Ritz, The future of soils and land use in the UK: Soil systems for the provision of land-based ecosystem services, Land Use Policy, 2009, 26, S187–S197 CrossRef.
- P. Pereira, I. Bogunovic, M. Muñoz-Rojas and E. C. Brevik, Soil ecosystem services, sustainability, valuation and management, Curr. Opin. Environ. Sci. Health, 2018, 5, 7–13 CrossRef.
- J. Stolte, M. Tesfai, L. Øygarden, S. Kværnø, J. Keizer, F. Verheijen, et al., Soil Threats in Europe, Publications Office of the European Union, 2015 Search PubMed.
- J. Vašíčková, M. Hvězdová, P. Kosubová and J. Hofman, Ecological risk assessment of pesticide residues in arable soils of the Czech Republic, Chemosphere, 2019, 216, 479–487 CrossRef.
- C. Pelosi, C. Bertrand, G. Daniele, M. Coeurdassier, P. Benoit and S. Nélieu, et al., Residues of currently used pesticides in soils and earthworms: A silent threat? Agric, Ecosyst. Environ., 2021, 305, 107167 CrossRef.
- T. D. Bucheli, E. Barmettler, N. Bartolomé, I. Hilber, K. Hornak and R. G. Meuli, et al., Pesticides in Agricultural Soils: Major Findings from Various Monitoring Campaigns in Switzerland, CHIMIA, 2023, 77(11), 750–757 CrossRef CAS PubMed.
- C. Froger, C. Jolivet, H. Budzinski, M. Pierdet, G. Caria and N. P. A. Saby, et al., Pesticide Residues in French Soils: Occurrence, Risks, and Persistence, Environ. Sci. Technol., 2023, 57(20), 7818–7827 CrossRef CAS.
- H. Tan, X. Hu, Y. Zhang, L. Zhang, Y. Chen and X. Zhang, et al., Binary combined toxicity of neonicotinoids and co-existing pollutants to non-target invertebrates: A meta-analysis, Environ. Res., 2025, 285, 122419 CrossRef CAS.
- P. Kosubová, L. Škulcová, Š. Poláková, J. Hofman and L. Bielská, Spatial and temporal distribution of the currently-used and recently-banned pesticides in arable soils of the Czech Republic, Chemosphere, 2020, 254, 126902 CrossRef.
- V. Geissen, V. Silva, E. H. Lwanga, N. Beriot, K. Oostindie and Z. Bin, et al., Cocktails of pesticide residues in conventional and organic farming systems in Europe – Legacy of the past and turning point for the future, Environ. Pollut., 2021, 278, 116827 CrossRef CAS.
- C. A. Brühl, N. Engelhard, N. Bakanov, J. Wolfram, K. Hertoge and J. G. Zaller, Widespread contamination of soils and vegetation with current use pesticide residues along altitudinal gradients in a European Alpine valley, Commun. Earth Environ., 2024, 5(1), 72 CrossRef.
- A. Franco, D. Vieira, L.-A. Clerbaux, A. Orgiazzi, M. Labouyrie and J. Köninger, et al., Evaluation of the ecological risk of pesticide residues from the European LUCAS Soil monitoring 2018 survey, Integrated Environ. Assess. Manag., 2024, 20(5), 1639–1653 CrossRef CAS.
- M. Hagner, S. Rämö, H. Soinne, V. Nuutinen, R. Muilu-Mäkelä and J. Heikkinen, et al., Pesticide residues in boreal arable soils: Countrywide study of occurrence and risks, Environ. Pollut., 2024, 357, 124430 CrossRef CAS.
- D. Knuth, L. Gai, V. Silva, P. Harkes, J. Hofman and M. Šudoma, et al., Pesticide Residues in Organic and Conventional Agricultural Soils across Europe: Measured and Predicted Concentrations, Environ. Sci. Technol., 2024, 58(15), 6744–6752 CrossRef CAS PubMed.
- C. Honert, K. Mauser, U. Jäger and C. A. Brühl, Exposure of insects to current use pesticide residues in soil and vegetation along spatial and temporal distribution in agricultural sites, Sci. Rep., 2025, 15(1), 1817 CrossRef CAS PubMed.
- K. M. Mauser, J. Wolfram, J. W. Spaak, C. Honert and C. A. Brühl, Current-use pesticides in vegetation, topsoil and water reveal contaminated landscapes of the Upper Rhine Valley, Germany, Commun. Earth Environ., 2025, 6(1), 166 CrossRef.
- F. Walder, M. W. Schmid, J. Riedo, A. Y. Valzano-Held, S. Banerjee and L. Büchi, et al., Soil microbiome signatures are associated with pesticide residues in arable landscapes, Soil Biol. Biochem., 2022, 174, 108830 CrossRef CAS.
- C. Bertrand, S. Aviron, C. Pelosi, J. Faburé, S. Le Perchec and L. Mamy, et al., Effects of plant protection products on ecosystem functions provided by terrestrial invertebrates, Environ. Sci. Pollut. Res., 2024, 32, 2956–2974 CrossRef PubMed.
- P. Meidl, A. Lehmann, M. Bi, C. Breitenreiter, J. Benkrama and E. Li, et al., Combined application of up to ten pesticides decreases key soil processes, Environ. Sci. Pollut. Res., 2024, 31, 11995–12004 CrossRef CAS PubMed.
- J. Riedo, M. C. Rillig and F. Walder, Beyond Dosage: The Need for More Realistic Research Scenarios to Understand Pesticide Impacts on Agricultural Soils, J. Agric. Food Chem., 2025, 73, 10093–10100 CrossRef CAS.
- C. A. Brühl and J. G. Zaller, Biodiversity Decline as a Consequence of an Inappropriate Environmental Risk Assessment of Pesticides, Front. Environ. Sci., 2019, 7, 1–4 Search PubMed.
- N.-F. Wan, L. Fu, M. Dainese, L. P. Kiær, Y.-Q. Hu and F. Xin, et al., Pesticides have negative effects on non-target organisms, Nat. Commun., 2025, 16(1), 1360 CrossRef CAS PubMed.
- D. Vieira, A. Franco, D. De Medici, J. Martin Jimenez, P. Wojda and A. Jones, Pesticides Residues in European Agricultural Soils - Results from LUCAS 2018 Soil Module, In: J. R. Centre, Publications Office of the European Union, Luxembourg, 2023 Search PubMed.
- S. J. More, V. Bampidis, D. Benford, S. H. Bennekou, C. Bragard and T. I. Halldorsson, et al., (European Food Saftey Authority Scientific Committee) Guidance on harmonised methodologies for human health, animal health and ecological risk assessment of combined exposure to multiple chemicals, EFSA J., 2019, 17(3), e05634 Search PubMed.
- A. Aagaard, P. Berny, P.-F. Chaton, A. L. Antia, E. McVey and M. Arena, et al., EFSA (European Food Safety Authority) Risk assessment for Birds and Mammals, EFSA J., 2023, 21(2), e07790 Search PubMed.
- P. Kotschik, M. Renaud, J. Princz, I. Rijk, U. Menke and B. Brooks, et al., Challenges and opportunities for the environmental risk assessment of chemicals in soils: a recap and follow-up of a SETAC webinar, Integrated Environ. Assess. Manag., 2025, vjaf102 CrossRef.
- C. J. Topping, A. Aldrich and P. Berny, Overhaul environmental risk assessment for pesticides, Science, 2020, 367(6476), 360–363 CrossRef CAS.
- J. V. Tarazona, M. de Alba-Gonzalez, C. Bedos, P. Benoit, C. Bertrand and O. Crouzet, et al., A conceptual framework for landscape-based environmental risk assessment (ERA) of pesticides, Environ. Int., 2024, 191, 108999 CrossRef PubMed.
- A. Kortenkamp, T. Backhaus and M. Faust, State of the Art Report on Mixture Toxicity - Final Report, Executive Summary, European Commission, Brussels, Belgium, 2009 Search PubMed.
- W. Brack, S. Ait-Aissa, R. M. Burgess, W. Busch, N. Creusot and C. Di Paolo, et al., Effect-directed analysis supporting monitoring of aquatic environments — An in-depth overview, Sci. Total Environ., 2016, 544, 1073–1118 CrossRef CAS.
- M. Faust, R. Altenburger, T. Backhaus, H. Blanck, W. Boedeker and P. Gramatica, et al., Joint algal toxicity of 16 dissimilarly acting chemicals is predictable by the concept of independent action, Aquat. Toxicol., 2003, 63(1), 43–63 CrossRef CAS PubMed.
- M. Faust, R. Altenburger, T. Backhaus, H. Blanck, W. Boedeker and P. Gramatica, et al., Predicting the joint algal toxicity of multi-component s-triazine mixtures at low-effect concentrations of individual toxicants, Aquat. Toxicol., 2001, 56(1), 13–32 CrossRef CAS PubMed.
- S. Loewe and H. Muischnek, Über Kombinationswirkungen, Naunyn Schmiedebergs Arch. Exp. Pathol. Pharmakol., 1926, 114(5), 313–326 CrossRef CAS.
- C. I. Bliss, The Toxicology of Poisons Applied Jointly, Ann. Appl. Biol., 1939, 26(3), 585–615 CrossRef CAS.
- ECHA, Guidance on Biocidal Products Regulation: Volume IV Environment - Assessment and Evaluation (Parts B+C), 2017, ECHA-17-G-23-EN; Cat. Number: ED-01-17-897-EN-N; ISBN: 978-92-9020-151-9 Search PubMed.
- C. Drocco, A. Coors, M. Devers, F. Martin-Laurent, N. Rouard and A. Spor, Evaluating the effects of environmental disturbances and pesticide mixtures on N-cycle related soil microbial endpoints, Peer Community J., 2025, 5, e33 CrossRef.
- J. B. Belden, R. J. Gilliom and M. J. Lydy, How well can we predict the toxicity of pesticide mixtures to aquatic life?, Integrated Environ. Assess. Manag., 2007, 3(3), 364–372 CrossRef CAS.
- O. Martin, M. Scholze, S. Ermler, J. McPhie, S. K. Bopp and A. Kienzler, et al., Ten years of research on synergisms and antagonisms in chemical mixtures: A systematic review and quantitative reappraisal of mixture studies, Environ. Int., 2021, 146, 106206 CrossRef CAS PubMed.
- A. Boobis, B. Robert, C. Shanna, C. Kevin, E. Michelle and F. Susan, et al., Critical analysis of literature on low-dose synergy for use in screening chemical mixtures for risk assessment, Crit. Rev. Toxicol., 2011, 41(5), 369–383 CrossRef CAS.
- N. Cedergreen, Quantifying Synergy: A Systematic Review of Mixture Toxicity Studies within Environmental Toxicology, PLoS One, 2014, 9(5), e96580 CrossRef PubMed.
- M. Hvězdová, P. Kosubová, M. Košíková, K. E. Scherr, Z. Šimek and L. Brodský, et al., Currently and recently used pesticides in Central European arable soils, Sci. Total Environ., 2018, 613–614, 361–370 CrossRef PubMed.
- V. Silva, H. G. J. Mol, P. Zomer, M. Tienstra, C. J. Ritsema and V. Geissen, Pesticide residues in European agricultural soils – A hidden reality unfolded, Sci. Total Environ., 2019, 653, 1532–1545 CrossRef CAS PubMed.
- A. Ukalska-Jaruga, B. Smreczak and G. Siebielec, Assessment of Pesticide Residue Content in Polish Agricultural Soils, Molecules, 2020, 25(3), 587 CrossRef CAS PubMed.
- S. C. Panico, C. A. M. van Gestel, R. A. Verweij, M. Rault, C. Bertrand and B. C. A. Menacho, et al., Field mixtures of currently used pesticides in agricultural soil pose a risk to soil invertebrates, Environ. Pollut., 2022, 305, 119290 CrossRef CAS PubMed.
- C. Pelosi, C. Bertrand, V. Bretagnolle, M. Coeurdassier, O. Delhomme and M. Deschamps, et al., Glyphosate, AMPA and glufosinate in soils and earthworms in a French arable landscape, Chemosphere, 2022, 301, 134672 CrossRef CAS PubMed.
- V. Silva, L. Gai, P. Harkes, G. Tan, C. J. Ritsema and F. Alcon, et al., Pesticide residues with hazard classifications relevant to non-target species including humans are omnipresent in the environment and farmer residences, Environ. Int., 2023, 181, 108280 CrossRef CAS PubMed.
- H. Mu, X. Yang, K. Wang, D. Tang, W. Xu and X. Liu, et al., Ecological risk assessment of pesticides on soil biota: An integrated field-modelling approach, Chemosphere, 2023, 326, 138428 CrossRef CAS PubMed.
- A. Rodríguez-Seijo, P. Pérez-Rodríguez, M. Arias-Estévez, A. Gómez-Armesto, M. Conde-Cid and V. Santás-Miguel, et al., Occurrence, persistence and risk assessment of pesticide residues in European wheat fields: A continental scale approach, J. Hazard. Mater., 2025, 494, 138291 CrossRef.
- F. Rivera-Becerril, D. van Tuinen, O. Chatagnier, N. Rouard, J. Béguet and C. Kuszala, et al., Impact of a pesticide cocktail (fenhexamid, folpel, deltamethrin) on the abundance of Glomeromycota in two agricultural soils, Sci. Total Environ., 2017, 577, 84–93 CrossRef CAS PubMed.
- R. Jiang, M. Wang, W. Chen and X. Li, Ecological risk evaluation of combined pollution of herbicide siduron and heavy metals in soils, Sci. Total Environ., 2018, 626, 1047–1056 CrossRef CAS PubMed.
- J. Amossé, S. Bart, F. Brulle, C. Tebby, R. Beaudouin and S. Nélieu, et al., A two years field experiment to assess the impact of two fungicides on earthworm communities and their recovery, Ecotoxicol. Environ. Saf., 2020, 203, 110979 CrossRef.
- L. Pitombeira de Figueirêdo, D. B. Athayde, M. A. Daam, C. A. M. van Gestel, GdS. Guerra and P. J. Duarte-Neto, et al., Impact of temperature on the toxicity of Kraft 36 EC® (a.s. abamectin) and Score 250 EC® (a.s. difenoconazole) to soil organisms under realistic environmental exposure scenarios, Ecotoxicol. Environ. Saf., 2020, 194, 110446 CrossRef PubMed.
- S. Nistala and A. Kumar, Effect of toxicological interaction of chlorpyrifos, cypermethrin, and arsenic on soil dehydrogenase activity in the terrestrial environment, Ecotoxicology, 2023, 32(5), 606–617 CrossRef CAS PubMed.
- I. Boughattas, F. Vaccari, L. Zhang, F. Bandini, B. Miras-Moreno and O. Missawi, et al., Co-exposure to environmental microplastic and the pesticide 2,4-dichlorophenoxyacetic acid (2,4-D) induce distinctive alterations in the metabolome and microbial community structure in the gut of the earthworm Eisenia andrei, Environ. Pollut., 2024, 344, 123213 CrossRef CAS PubMed.
- L. L. Caldas, D. S. Alexandre, A. Pretti Ogura, T. JdS. Pinto, R. A. Moreira and TdO. Zacharias, et al., Field application doses of fipronil and 2,4-D and toxic effects on non-target soil organisms: a microcosm-level approach, Environ. Toxicol. Chem., 2025, 1965–1974 CrossRef CAS.
- M. dos Santos Lima, J. A. de Paula Silva, M. T. Firmino, S. R. L. Pontes, M. Benvindo-Souza and S. DMe, et al., Genotoxic, Mutagenic and Behavioral Effects of Flumioxazin, S-Metolachlor, and Their Mixture: A Multilevel Ecotoxicological Assessment Using Eisenia Fetida, Water, Air, Soil Pollut., 2025, 237(6), 341 CrossRef.
- I. Gruss, P. Bączek, I. Ćwieląg-Piasecka, S. Jędrzejewski, J. Magiera-Dulewicz and K. Twardowska, Assessing the ecotoxicological effects of pesticides on non-target plant species, Environ. Monit. Assess., 2025, 197(9), 1047 CrossRef CAS PubMed.
- I. Gruss, I. Ćwieląg-Piasecka, P. Bączek, J. Twardowski, S. Jędrzejewski and K. Twardowska, et al., Reproduction, growth, and survival responses of Eisenia fetida and Folsomia candida to individual and combined pesticide exposures, Environ. Toxicol. Pharmacol., 2025, 120, 104844 CrossRef CAS PubMed.
- Q. Ma, Y. Zhou, R. E. Parales, S. Jiao, Z. Ruan and L. Li, Effects of herbicide mixtures on the diversity and composition of microbial community and nitrogen cycling function on agricultural soil: A field experiment in Northeast China, Environ. Pollut., 2025, 372, 125965 CrossRef CAS.
- P. S. Tourinho, Z. Hochmanová, P. Kukučka, O. Jegede, V. Silva and V. Aparicio, et al., Effects of realistic pesticide mixtures on the springtail Folsomia candida, Environ. Toxicol. Chem., 2025, 44, 1347–1356 CrossRef CAS PubMed.
- S. van Loon, G. Xie, C. Svendsen, M. H. S. Kraak, L. de Jeu and N. C. Schut, et al., Microplastics and PFAS as ubiquitous pollutants affect potencies of highly toxic chemicals in mixtures, J. Hazard. Mater., 2025, 500, 140493 CrossRef CAS PubMed.
- M. C. Rillig, M. Ryo, A. Lehmann, C. A. Aguilar-Trigueros, S. Buchert and A. Wulf, et al., The role of multiple global change factors in driving soil functions and microbial biodiversity, Science, 2019, 366(6467), 886–890 CrossRef CAS PubMed.
- J. Riedo, F. E. Wettstein, A. Rösch, C. Herzog, S. Banerjee and L. Büchi, et al., Widespread Occurrence of Pesticides in Organically Managed Agricultural Soils—the Ghost of a Conventional Agricultural Past?, Environ. Sci. Technol., 2021, 55(5), 2919–2928 CrossRef CAS PubMed.
- P. Mäder, A. Listl, Z. Hochmanová, W. Armbruster, P. Harkes and C. Poll, et al., Transfer of prosulfocarb and boscalid residues from maize leaves to soil and their effects on soil microorganisms, Environ. Pollut., 2025, 383, 126862 CrossRef PubMed.
- L. Å. Medici, P. A. Niklaus, F. Walder and M. Langer, Impact of plant protection product applications on soil microbial nitrogen cycle function not fully captured by gene quantification, Appl. Soil Ecol., 2025, 213, 106297 CrossRef.
- B. A. Woodcock, M. Gibbs, H. Hesketh, S. Short, A. Robinson and E. Eagles, et al., Synergistic Pesticide Effects on Complex Arthropod Trophic Interactions in Arable Systems, Environ. Sci. Technol., 2025, 59(35), 18594–18604 CrossRef CAS PubMed.
- D. J. Pérez, F. G. Iturburu, G. Calderon, L. A. E. Oyesqui, E. De Gerónimo and V. C. Aparicio, Ecological risk assessment of current-use pesticides and biocides in soils, sediments and surface water of a mixed land-use basin of the Pampas region, Argentina, Chemosphere, 2021, 263, 128061 CrossRef PubMed.
- A. Fekadu Demsie, G. T. Yimer, S. S. Sota and M. A. Argaye, Concentrations and ecological risk assessment of pesticide residues on soil biota in irrigated soil of Ziway, Ethiopia, Integrated Environ. Assess. Manag., 2025, 473–483 Search PubMed.
- E. L. Sánchez-del Cid, J. Rendón-von Osten, MdC. Á. González-Chávez, A. Torres-Dosal and E. Huerta-Lwanga, Pesticide residues in tropical agricultural soils: Distribution, seasonality, and earthworm ecological risk, Emerging Contam., 2025, 11(1), 100436 CrossRef.
- R. Roani, R. T. Dudas, W. C. Demetrio, F. M. O. Lourenço, G. A. Ramos and J. C. Niemeyer, et al., Pesticide residues and earthworm reproduction in eighteen Brazilian soils, Environ. Toxicol. Chem., 2026, 613–631 CrossRef CAS PubMed.
- M. Gibbs, F. Mancini, C. Carvell, G. Powney, C. A. Harrower and G. Skinner, et al., Two decades of spatial and temporal pesticide risk to non-target invertebrates in England farmlands, Environ. Int., 2025, 204, 109856 CrossRef CAS PubMed.
- EC. (European Comission), Technical Guidance Document on Risk Assessment (Part II), Joint Research Centre, 2003, EUR 20418 EN/2, https://op.europa.eu/s/y4d4 Search PubMed.
- K. A. Lewis and T. John, J. WD, and Green A. An international database for pesticide risk assessments and management, Hum. Ecol. Risk Assess., 2016, 22(4), 1050–1064 CrossRef CAS.
- EFSA. (European Food Safety Authority), Chemical hazards data— OpenFoodTox, 2023.
- USEPA. (United States Environmental Protection Agency), ECOTOX Knowledge Base, 2023 Search PubMed.
- C. Casado-Martinez, G. Dell'Ambrogio, S. Campiche, A. Kroll, E. Lauber and M. Marti-Roura, et al., Incorporation of sediment- and soil-specific aspects in the Criteria for Reporting and Evaluating Ecotoxicity Data (CRED), Integrated Environ. Assess. Manag., 2024, 20(6), 2162–2174 CrossRef PubMed.
- C. T. A. Moermond, A. Franco, M. C. Casado-Martinez, K. Kidd, M. Korkaric and M. Ågerstrand, Improving the reporting of ecotoxicity data: will SETAC lead the way?, Integrated Environ. Assess. Manag., 2026, 22(2), 335–337 CrossRef PubMed.
- Consolidated Text (21.11.2022), Regulation (EC) No 1107/2009 of the European Parliament and of the Council of 21 October 2009 Concerning the Placing of Plant Protection Products on the Market and Repealing Council Directives 79/117/EEC and 91/414/EEC Search PubMed.
- EFSA. (European Food Safety Authority), Scientific Opinion addressing the state of the science on risk assessment of plant protection products for in-soil organisms, EFSA J., 2017, 15(2), e04690 Search PubMed.
- E. Patyra, C. Nebot, R. E. Gavilán, K. Kwiatek and A. Cepeda, Prevalence of veterinary antibiotics in natural and organic fertilizers from animal food production and assessment of their potential ecological risk, J. Sci. Food Agric., 2023, 103(7), 3638–3644 CrossRef CAS PubMed.
- H. Liu, R. Li, W. Hu, L. Jian, B. Huang and Yn Fan, et al., Multi-medium residues and ecological risk of herbicides in a typical agricultural watershed of the Mollisols region, Northeast China, Sci. Total Environ., 2024, 937, 173507 CrossRef CAS PubMed.
- ECHA, Guidance on the Biocidal Products Regulation. Volume IV Environment - Assessment and Evaluation (Parts B + C), European Chemicals Agency, 2017, Version 2.0 Search PubMed.
- M. Nadal, V. Kumar, M. Schuhmacher and J. L. Domingo, Definition and GIS-based characterization of an integral risk index applied to a chemical/petrochemical area, Chemosphere, 2006, 64(9), 1526–1535 CrossRef CAS PubMed.
- L. Posthuma, G. W. S. II and T. P. Traas, Species Sensitivity Distributions in Ecotoxicology, CRC Press, 2001 Search PubMed.
- L. Posthuma and G. W. Suter, Ecological Risk Assessment of Diffuse and Local Soil Contamination Using Species Sensitivity Distributions, Dealing with Contaminated Sites: from Theory towards Practical Application, F. A. Swartjes, Springer Netherlands, Dordrecht, 2011, p. 625–691 Search PubMed.
- A. Gainer, K. Bresee, N. Hogan and S. D. Siciliano, Advancing soil ecological risk assessments for petroleum hydrocarbon contaminated soils in Canada: Persistence, organic carbon normalization and relevance of species assemblages, Sci. Total Environ., 2019, 668, 400–410 CrossRef CAS PubMed.
- EFSA PPR Panel. (European Food Safety Authority Panel on Plant Protection Products and their Residues), Guidance on tiered risk assessment for plant protection products for aquatic organisms in edge-of-field surface waters, EFSA J., 2013, 11(7), 3290 Search PubMed.
- M. Marti-Roura, G. Dell'Ambrogio, S. Campiche, J. Wong, M. Junghans, M. Renaud, et al., Methodology proposal for the derivation of Soil Guideline Values for Plant Protection Product residues. Part 2 - Recommendations for the derivation of Soil Guideline Values, Swiss Centre for Applied Ecotoxicology, Swiss Centre for Applied Ecotoxicology, Dübendorf, 2023, p. 57 pp Search PubMed.
- H. O. Fajana, E. G. Lamb and S. D. Siciliano, A shift from individual species to ecosystem services effect: Introducing the Eco-indicator Sensitivity Distribution (EcoSD) as an ecosystem services approach to redefining the species sensitivity distribution (SSD) for soil ecological risk assessment, Integrated Environ. Assess. Manag., 2023, 20, 1166–1179 CrossRef PubMed.
- A. Gainer, M. Cousins, N. Hogan and S. D. Siciliano, Petroleum hydrocarbon mixture toxicity and a trait-based approach to soil invertebrate species for site-specific risk assessments, Environ. Toxicol. Chem., 2018, 37(8), 2222–2234 CrossRef CAS PubMed.
- F. Zhao, L. Yang, L. Chen, Q. Xiang, S. Li and L. Sun, et al., Soil contamination with antibiotics in a typical peri-urban area in eastern China: Seasonal variation, risk assessment, and microbial responses, J. Environ. Sci., 2019, 79, 200–212 CrossRef CAS PubMed.
- X. Li, M. Wang, R. Jiang, L. Zheng and W. Chen, Evaluation of joint toxicity of heavy metals and herbicide mixtures in soils to earthworms (Eisenia fetida), J. Environ. Sci., 2020, 94, 137–146 CrossRef CAS PubMed.
- P. Chakravarty, D. Chowdhury and H. Deka, Ecological risk assessment of priority PAHs pollutants in crude oil contaminated soil and its impacts on soil biological properties, J. Hazard. Mater., 2022, 437, 129325 CrossRef CAS PubMed.
- M. M. Bahar, S. V. A. C. Samarasinghe, D. Bekele and R. Naidu, Residual hydrocarbons in long-term contaminated soils: implications to risk-based management, Environ. Sci. Pollut. Res., 2024, 31(15), 22759–22773 CrossRef CAS PubMed.
- I. García Carriquiry, V. Silva, F. Raevel, P. Harkes, R. Osman and O. Bentancur, et al., Effects of mixtures of herbicides on nutrient cycling and plant support considering current agriculture practices, Chemosphere, 2024, 349, 140925 CrossRef PubMed.
- X. Li, S. Zhang, R. Guo, X. Xiao, B. Liu and R. K. Mahmoud, et al., Transformation and Degradation of PAH Mixture in Contaminated Sites: Clarifying Their Interactions with Native Soil Organisms, Toxics, 2024, 12(5), 361 CrossRef CAS PubMed.
- J. Jensen and L. E. Sverdrup, Joint Toxicity of Linear Alkylbenzene Sulfonates and Pyrene on Folsomia fimetaria, Ecotoxicol. Environ. Saf., 2002, 52(1), 75–81 CrossRef CAS PubMed.
- M. J. B. Amorim, C. Pereira, V. B. Menezes-Oliveira, B. Campos, A. M. V. M. Soares and S. Loureiro, Assessing single and joint effects of chemicals on the survival and reproduction of Folsomia candida (Collembola) in soil, Environ. Pollut., 2012, 160, 145–152 CrossRef CAS PubMed.
- T. Pescatore, V. Di Nica, A. Finizio, N. Ademollo, F. Spataro and J. Rauseo, et al., Sub-lethal effects of soil multiple contamination on the avoidance behaviour of Eisenia fetida, Ecotoxicol. Environ. Saf., 2021, 226, 112861 CrossRef CAS PubMed.
- S.-X. Li, X.-R. Gao, J. Yi, L.-Y. Jia and J. Ren, A new strategy of using periphyton to simultaneously promote remediation of PAHs-contaminated soil and production of safer crops, Environ. Res., 2024, 246, 118149 CrossRef CAS PubMed.
- Y. Zhu, M. Ke, Z. Yu, C. Lei, M. Liu and Y. Yang, et al., Combined effects of azoxystrobin and oxytetracycline on rhizosphere microbiota of Arabidopsis thaliana, Environ. Int., 2024, 186, 108655 CrossRef CAS PubMed.
- C. I. García, V. Silva, P. Inchausti, S. Niell, A. Berro Pizzarossa and M. Medina, et al., The effects of sequential herbicide applications on phosphorus cycling and mycorrhization in soybean: A two-year field study, Agric. Ecosyst. Environ., 2025, 392, 109754 CrossRef.
- D. G. Karpouzas, Z. Vryzas and F. Martin-Laurent, Pesticide soil microbial toxicity: setting the scene for a new pesticide risk assessment for soil microorganisms (IUPAC Technical Report), Pure Appl. Chem., 2022, 94(10), 1161–1194 CrossRef CAS.
- ISO, ISO Technical Specification 10832 — Soil Quality — Effects of Pollutants on Mycorrhizal Fungi — Spore Germination Test, ISO, Geneva, Switzerland, 2009 Search PubMed.
- OECD, Test No. 207: Earthworm, Acute Toxicity Tests, 1984 Search PubMed.
- EC. (European Comission), Biological test method. Tests for toxicity of contaminated soil to earthworms (Eisenia andrei, Eisenia fetida, or Lumbricus terrestris) In: Section MDaA, editor. Report: EPS 1/RM/43. Ottawa, Onatrio, Canada: Environment Canada, 2004.
- EC. (European Comission), Biological test method - test for measuring emergence and growth of terrestrial plants exposed to contaminants in soil, Section MDaA, Environment Canada, Ottawa, Ontario, Canada, 2005 Search PubMed.
- OECD, Test No. 208: Terrestrial Plant Test: Seedling Emergence and Seedling Growth Test, 2006 Search PubMed.
- ISO. 11269-2:2012 Soil quality—Determination of the effects of pollutants on soil flora In: 4 TCITS, Part 2: Effects of Contaminated Soil on the Emergence and Early Growth of Higher Plants, Geneva, Switzerland, International Organization for Standardization, 2012 Search PubMed.
- EC. (European Comission), Biological test method - test for growth in contaminated soil using terrestrial plants native to the boreal region, Environment Canada EPS 1/RM/56 Environmental Protection Series, 2013.
- ISO. 11267:2014, Soil quality — Inhibition of reproduction of Collembola (Folsomia candida) by soil contaminants, 4 TcITS, International Organization for Standardization, Geneva, Switzerland, 2014 Search PubMed.
- OECD, Test No. 232: Collembolan Reproduction Test in Soil, 2016 Search PubMed.
- P. A. Karas, C. Baguelin, G. Pertile, E. S. Papadopoulou, S. Nikolaki and V. Storck, et al., Assessment of the impact of three pesticides on microbial dynamics and functions in a lab-to-field experimental approach, Sci. Total Environ., 2018, 637–638, 636–646 CrossRef CAS PubMed.
- OECD, Test No. 216: Soil Microorganisms: Nitrogen Transformation Test, 2000 Search PubMed.
- K. Schlich, C. Diaz, K. Derz, U. Hommen, V. Reiermann, M. Winter, et al., Correctly Assessing the Performance and Threats of Microorganisms in Agricultural Soils – Identifying Meaningful Endpoints under Field-Relevant Pesticide, Biocide and Pharmaceutical Exposure, German Environment Agency, 2025 Search PubMed.
- A. M. Keith and D. A. Robinson, Earthworms as Natural Capital: Ecosystem Service Providers in Agricultural Soils, Econ. J., 2012, 2, 91–99 Search PubMed.
- A. M. Potapov, F. Beaulieu, K. Birkhofer, S. L. Bluhm, M. I. Degtyarev and M. Devetter, et al., Feeding habits and multifunctional classification of soil-associated consumers from protists to vertebrates, Biol. Rev., 2022, 97(3), 1057–1117 CrossRef PubMed.
- M. N. Filimon, S. O. Voia, D. L. Vladoiu, A. Isvoran and V. Ostafe, Temperature dependent effect of difenoconazole on enzymatic activity from soil, J. Serb. Chem. Soc., 2015, 80(9), 1127–1137 CrossRef CAS.
- NEPC. (National Environment Protection Council), Draft for public consultation: Schedule B5b - Guideline on Methodology to Derive Ecological Investigation Levels in Contaminated Soils, 2011, https://www.nepc.gov.au/sites/default/files/2022-09/schedule-b5b-guideline-methodology-derive-eils-sep10.pdf.
- RIVM, (National Institute for Public Health and the Environment) RIVM report 601782001/2007 - Guidance for the derivation of environmental risk limits within the framework of ‘International and national environmental quality standards for substances in the Netherlands’ (INS), 2007, https://www.rivm.nl/bibliotheek/rapporten/601782001.pdf.
- T. Jager, F. A. A. Sánchez, B. Muijs, E. G. van der Velde and L. Posthuma, Toxicokinetics of polycyclic aromatic hydrocarbons in Eisenia andrei (Oligochaeta) using spiked soil, Environ. Toxicol. Chem., 2009, 19(4), 953–961 CrossRef.
- M. Broerse, H. Oorsprong and C. A. M. van Gestel, Cadmium affects toxicokinetics of pyrene in the collembolan Folsomia candida, Ecotoxicology, 2012, 21(3), 795–802 CrossRef CAS PubMed.
- I. Giska, C. A. M. van Gestel, B. Skip and R. Laskowski, Toxicokinetics of metals in the earthworm Lumbricus rubellus exposed to natural polluted soils – relevance of laboratory tests to the field situation, Environ. Pollut., 2014, 190, 123–132 CrossRef CAS PubMed.
- A. J. Bednarska, M. Choczyński, R. Laskowski and M. Walczak, Combined effects of chlorpyriphos, copper and temperature on acetylcholinesterase activity and toxicokinetics of the chemicals in the earthworm Eisenia fetida, Environ. Pollut., 2017, 220, 567–576 CrossRef CAS PubMed.
- Z. M. Świątek, C. A. M. van Gestel and A. J. Bednarska, Toxicokinetics of zinc-oxide nanoparticles and zinc ions in the earthworm Eisenia andrei, Ecotoxicol. Environ. Saf., 2017, 143, 151–158 CrossRef PubMed.
- J. Li, M. E. Hodson, C. D. Brown, M. J. Bottoms, R. Ashauer and T. Alvarez, Earthworm lipid content and size help account for differences in pesticide bioconcentration between species, J. Hazard. Mater., 2024, 468, 133744 CrossRef CAS PubMed.
- S. W. Karickhoff, Semi-empirical estimation of sorption of hydrophobic pollutants on natural sediments and soils, Chemosphere, 1981, 10(8), 833–846 CrossRef CAS.
- ECHA. Guidance on the implementation of REACH, Guidance on Information Requirements and Chemical Safety Assessment Chapter R 8: Characterisation of Dose [concentration]-Response for Human Health: ECHA, 2008 Search PubMed.
- G. Sigmund, H. P. H. Arp, B. M. Aumeier, T. D. Bucheli, B. Chefetz and W. Chen, et al., Sorption and Mobility of Charged Organic Compounds: How to Confront and Overcome Limitations in Their Assessment, Environ. Sci. Technol., 2022, 56(8), 4702–4710 CrossRef CAS PubMed.
- B. G. van Hall, C. Sweeney, M. Bottoms and C. A. M. van Gestel, Influence of soil organic matter content on the toxicity of pesticides to soil invertebrates: A review, Integrated Environ. Assess. Manag., 2023, 19(6), 1457–1472 CrossRef CAS PubMed.
- EC. (European Commission), Draft Guidance Document on Terrestrial Ecotoxicology under Council Directive 91/414/EEC. SANCO/10329/2002 Rev 2 Final, 2002 Search PubMed.
- L. J. Lister, C. Svendsen, J. Wright, H. L. Hooper and D. J. Spurgeon, Modelling the joint effects of a metal and a pesticide on reproduction and toxicokinetics in Lumbricid earthworms, Environ. Int., 2011, 37(4), 663–670 CrossRef CAS PubMed.
- S. Bart, S. Short, T. Jager, E. J. Eagles, A. Robinson and C. Badder, et al., How to analyse and account for interactions in mixture toxicity with toxicokinetic-toxicodynamic models, Sci. Total Environ., 2022, 843, 157048 CrossRef CAS PubMed.
- V. González-Naranjo and K. Boltes, Toxicity of ibuprofen and perfluorooctanoic acid for risk assessment of mixtures in aquatic and terrestrial environments, Int. J. Environ. Sci. Technol., 2014, 11(6), 1743–1750 CrossRef.
- V. González-Naranjo, K. Boltes, I. de Bustamante and P. Palacios-Diaz, Environmental risk of combined emerging pollutants in terrestrial environments: chlorophyll a fluorescence analysis, Environ. Sci. Pollut. Res., 2015, 22(9), 6920–6931 CrossRef PubMed.
- W. Wang, J. Zhang, J. Wu, R. Yu, Y. Zhang and L. Sun, et al., Acute Toxicity and Ecotoxicological Risk Assessment of Three Volatile Pesticide Additives on the Earthworm—Eisenia fetida, Int. Res. J. Publ. Environ. Health, 2021, 18(21), 11232 CrossRef CAS PubMed.
- J. L. Gallego, E. R. Shipley, P. Vlahos and J. Olivero-Verbel, Occurrence and toxicological relevance of pesticides and trace metals in agricultural soils, sediments, and water of the Sogamoso River basin, Colombia, Chemosphere, 2024, 354, 141713 CrossRef CAS PubMed.
- M. Urbaniak, A. Baran, J. Giebułtowicz, A. Bednarek and L. Serwecińska, The occurrence of heavy metals and antimicrobials in sewage sludge and their predicted risk to soil — Is there anything to fear?, Sci. Total Environ., 2024, 912, 168856 CrossRef CAS PubMed.
- O. Jegede, P. S. Tourinho, V. Geissen and J. Hofman, Prioritization of currently used pesticides in soils of main European cropping systems and an Argentinian cropping system for assessment of mixture toxicity and risk on terrestrial biota, J. Hazard. Mater., 2025, 495, 138942 CrossRef CAS PubMed.
- M. Kotli, G. Piir and U. Maran, Predictive Modeling of Pesticides Reproductive Toxicity in Earthworms Using Interpretable Machine-Learning Techniques on Imbalanced Data, ACS Omega, 2025, 10(5), 4732–4744 CrossRef CAS PubMed.
- F. Sánchez-Bayo, S. Baskaran and I. R. Kennedy, Ecological relative risk (EcoRR): another approach for risk assessment of pesticides in agriculture, Agric. Ecosyst. Environ., 2002, 91(1), 37–57 CrossRef.
- EC. (European Commission), Directive of the European Parliament and of the Council on Soil Monitoring and Resilience (Soil Monitoring Law), 2023, https://environment.ec.europa.eu/publications/proposal-directive-soil-monitoring-and-resilience_en.
- USEPA. (United States Environmental Protection Agency), Guidelines for the Health Risk Assessment of Chemical Mixtures, 1986, EPA/630/R-98/002 Search PubMed.
- CCME. (Canadian Council of Ministers of the Environment), A Protocol for the Derivation of Environmental and Human Health Soil Quality Guidelines, Sites CCoMotESoEQCfC, Canada, Winnipeg, Manitoba, Canada, 1996 Search PubMed.
- USEPA. (United States Environmental Protection Agency), Supplementary Guidance for Conducting Health Risk Assessment of Chemical Mixtures, EPA, 2000, EPA/630/R-00/002 Search PubMed.
- D. de Zwart and L. Posthuma, Complex mixture toxicity for single and multiple species: Proposed methodologies, Environ. Toxicol. Chem., 2005, 24(10), 2665–2676 CrossRef CAS PubMed.
- MfE. (Ministry for the Environment), Guidelines for Assessing and Managing Petroleum Hydrocarbon Contaminated Sites in New Zealand, Ministry for the Environment, Wellington, 2011, https://environment.govt.nz/assets/publications/Files/modules-final-jun99.pdf Search PubMed.
- MfE. (Ministry for the Environment), Methodology for Deriving Standards for Contaminants in Soil to Protect Human Health, Ministry for the Environment, Wellington, 2011, https://environment.govt.nz/assets/Publications/Files/methodology-for-deriving-standards-for-contaminants-in-soil.pdf Search PubMed.
- NEPC. (National Environment Protection Council), Draft for public consultation: Schedule B5a - Guideline on ecological Risk Assessment, 2013, https://www.nepc.gov.au/sites/default/files/2022-09/schedule-b5a-guideline-ecological-risk-assessment-sep10-0.pdf.
- A. Kienzler, E. Berggren, J. Bessems, S. Bopp, S. van der Linden and A. Worth, Assessment of Mixtures - Review of Regulatory Requirements and Guidance, Publications Office of the European Union, 2017 Search PubMed.
- A. Kienzler, S. K. Bopp, S. van der Linden, E. Berggren and A. Worth, Regulatory assessment of chemical mixtures: Requirements, current approaches and future perspectives, Regul. Toxicol. Pharmacol., 2016, 80, 321–334 CrossRef CAS PubMed.
- (Canadian Council of Ministers of the Environment), A Protocol for the Derivation of Environmental and Human Health Soil Quality Guidelines, ISBN-10 1-896997-45-7, 2006 Search PubMed.
- Consolidated text (21.11.2022), Commission Regulation (EU) No 283/2013 of 1 March 2013 setting out the data requirements for active substances, in Accordance with Regulation (EC) No 1107/2009 of the European Parliament and of the Council Concerning the Placing of Plant Protection Products on the Market Text with EEA Relevance Search PubMed.
- Consolidated text (21.11.2022): Commission Regulation (EU) No 284/2013 of 1 March 2013 setting out the data requirements for plant protection products, in Accordance with Regulation (EC) No 1107/2009 of the European Parliament and of the Council Concerning the Placing of Plant Protection Products on the Market Text with EEA Relevance Search PubMed.
- HC. (Health Canada), 2024. Pest Management Regulatory Agency (PMRA). A Framework for Risk Assessment and Risk Management of Pest Control Products, PMRA Guidance Document.
- USEPA. (United States Environmental Protection Agency), Pesticide Registration, About Pesticide Registration, 2026, https://www.epa.gov/pesticide-registration/about-pesticide-registration Search PubMed.
- Consolidated text (11.06.2024), Commission Regulation (EU) No 528/2012 of the European Parliament and of the Council of 22 May 2012 Concerning the Making Available on the Market and Use of Biocidal Products (Text with EEA relevance)Text with EEA Relevance Search PubMed.
- EFSA. (European Food Safety Authority), Scientific Opinion addressing the state of the science on risk assessment of plant protection products for non-target terrestrial plants, EFSA J., 2014, 12(7), 3800 CrossRef.
- EFSA. (European Food Safety Authority), EFSA Guidance Document for predicting environmental concentrations of active substances of plant protection products and transformation products of these active substances in soil, EFSA J., 2017, 15(10), e04982 Search PubMed.
- C. Lee-Steere, Environmental Risk Assessment Guidance Manual for Industrial Chemicals, Australian Environment Agency, 2009, https://www.nepc.gov.au/sites/default/files/2022-09/cmgt-nchem-eragm-industrial-chemicals-200902.pdf Search PubMed.
- C. Lee-Steere, Environmental Risk Assessment Guidance Manual for Agricultural and Veterinary Chemicals, Australian Environment Agency, 2009, https://www.nepc.gov.au/sites/default/files/2022-09/cmgt-nchem-eragm-agricultural-and-veterinary-chemicals-200902.pdf Search PubMed.
- EFSA. (European Food Safety Authority), Technical Workshop on Risk Assessment of the Plant Protection Products, 9.2 Topic 2 : Exposure and Risk Assessment Parma, Italy: European Food Safety Authority, 2023, https://www.efsa.europa.eu/sites/default/files/2023-07/9.2-breakout-session-topic-2.pdf Search PubMed.
- G. E. Seralini, Pesticides in Formulations: New Revolutionary Findings, Toxics, 2024, 12(2), 151 CrossRef PubMed.
- E. A. Straw, End co-formulant secrecy, Environ. Sci. Pol., 2024, 154, 103701 CrossRef CAS.
- S. M. Long, F. Reichenberg, L. J. Lister, P. K. Hankard, J. Townsend and P. Mayer, et al., Combined chemical (Fluoranthene) and drought effects on Lumbricus rubellus demonstrate the applicability of the independent action model for multiple stressor assessment, Environ. Toxicol. Chem., 2009, 28(3), 629–636 CrossRef CAS PubMed.
- M. Liess, K. Foit, S. Knillmann, R. B. Schäfer and H.-D. Liess, Predicting the synergy of multiple stress effects, Sci. Rep., 2016, 6(1), 32965 CrossRef CAS PubMed.
- M. Wehrli, S. Slotsbo, S. Fomsgaard Inge, B. B. Laursen, J. Gröning and M. Liess, et al., A Dirt(y) World in a Changing Climate: Importance of Heat Stress in the Risk Assessment of Pesticides for Soil Arthropods, Glob. Change Biol., 2024, 30(10), e17542 CrossRef CAS PubMed.
- J. van Dijk, M. Gustavsson, S. C. Dekker and A. P. van Wezel, Towards ‘one substance – one assessment’: An analysis of EU chemical registration and aquatic risk assessment frameworks, J. Environ. Manage., 2021, 280, 111692 CrossRef CAS PubMed.
- A. G. Ukbamichael, T. Sagredo, E. Urionabarrenetxea, M. Soto and J. V. Tarazona, A simplified landscape-based approach for including agronomical and ecological characteristic in pesticide risk assessments for terrestrial vertebrates, Environ. Pollut., 2025, 385, 127142 CrossRef CAS PubMed.
- T. Schad, S. Bub, M. Wang, C. M. Holmes, J. Kleinmann and K. Hammel, et al., A spatiotemporally explicit modeling approach for more realistic exposure and risk assessment of off-field soil organisms, Integrated Environ. Assess. Manag., 2023, 20(1), 263–278 CrossRef PubMed.
- K. Rakel, V. Roeben, G. Ernst and A. Gergs, Advancing Soil Risk Assessment: A Novel Earthworm Cocoon Test with a Complementary Toxicokinetic–Toxicodynamic Modeling Approach, Environ. Toxicol. Chem., 2024, 43(11), 2377–2386 CrossRef CAS PubMed.
- R. Schulz, S. Bub, L. L. Petschick, S. Stehle and J. Wolfram, Applied pesticide toxicity shifts toward plants and invertebrates, even in GM crops, Science, 2021, 372(6537), 81–84 CrossRef CAS PubMed.
- S. Bub, J. Wolfram, L. L. Petschick, S. Stehle and R. Schulz, Trends of Total Applied Pesticide Toxicity in German Agriculture, Environ. Sci. Technol., 2023, 57(1), 852–861 CrossRef CAS PubMed.
- J. Wolfram, D. Bussen, S. Bub, L. L. Petschick, L. Z. Herrmann and R. Schulz, Increasing applied pesticide toxicity trends counteract the global reduction target to safeguard biodiversity, Science, 2026, 391(6785), 616–621 CrossRef CAS PubMed.
- M. Junghans, P. Kunz and I. Werner, Toxizität von Mischungen. Aktuelle, praxisorientierte Ansätze für die Beurteilung von Gewässerproben, Aqua Gas, 2013, 94(5), 54–61 Search PubMed.
- S. Spycher, S. Mangold, T. Doppler, M. Junghans, I. Wittmer and C. Stamm, et al., Pesticide Risks in Small Streams—How to Get as Close as Possible to the Stress Imposed on Aquatic Organisms, Environ. Sci. Technol., 2018, 52(8), 4526–4535 CrossRef CAS PubMed.
- P. Borer, K. Guthruf, V. Maurer, C. Minkowski, K. Rehberger, M. Ruff, et al., Zustand der Gewässer Gewässerbericht 2019–2022, Amt für Wasser und Abfall AWA. Gewässer- und Bodenschutzlabor GBL, Amt für Wasser und Abfall AWA, Bern, 2023 Search PubMed.
- EC. (European and Commission), COMMISSION STAFF WORKING DOCUMENT Progress report on the assessment and management of combined exposures to multiple chemicals (chemical mixtures) and associated risks Accompanying the document Communication From The Commission To The European Parliament, The Council, The European Economic And Social Committee And The Committee Of The Regions Chemicals Strategy for Sustainability Towards a Toxic-Free Environment, 2020.
- T. Backhaus, The mixture assessment or allocation factor: conceptual background, estimation algorithms and a case study example, Environ. Sci. Eur., 2023, 35(1), 55 CrossRef.
- P. S. Price and X. Han, Maximum cumulative ratio (MCR) as a tool for assessing the value of performing a cumulative risk assessment, Int. J. Environ. Res. Publ. Health, 2011, 8(6), 2212–2225 CrossRef PubMed.
| This journal is © The Royal Society of Chemistry 2026 |






