CoOx electro-catalysts anchored on nitrogen-doped carbon nanotubes for the oxygen evolution reaction†
Abstract
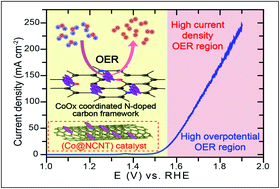
The development of high-performance, low-cost transition metal oxide nanoparticle-supported carbon catalysts for the oxygen evolution reaction (OER) is one of the biggest challenges faced in the process of commercializing water electrolyzers and rechargeable metal–air batteries. Cobalt oxides (CoOx) have been explored owing to their high availability and high activity when applied in reactions involving oxygen, including the oxygen evolution reaction (OER) and oxygen reduction reaction (ORR). Effective anchoring between the metal oxide active sites and the conducting carbon support is another puzzling task to consider when enhancing the overall catalytic activity of the materials. To investigate the role of anchoring sites on the carbon support, we grew catalytically active cobalt oxide nanoparticles on pristine carbon nanotubes (CNTs), oxy-group functionalized CNTs (fCNTs), and nitrogen-doped (N-doped) CNTs (NCNTs). The anchoring sites on CNTs not only hold the CoOx nanoparticles but they are also involved in controlling the size and distribution of the CoOx during the synthesis process by creating nucleating sites. The distribution and interaction of CoOx on CNT, fCNT, and NCNT was confirmed by employing structural and spectroscopic characterization techniques such as TEM, XRD, XPS, and Raman analysis. The electrochemical OER activity screening of the prepared catalysts showed the highest performance of CoOx-supported NCNT (Co@NCNT) with the lowest overpotential (η) of 310 mV @ 10 mA cm−2 and a lower kinetic Tafel slope of ∼74.67 mV dec−1.
- This article is part of the themed collection: Organic Materials for Energy Conversion and Storage


 Please wait while we load your content...
Please wait while we load your content...