Synthetic approach toward the indole alkaloid TMC-205, 6,7-secoagroclavine, aurantioclavine, clavicipitic acid, and caulindoles A–D
Qianfei
Luo†
a,
Ting
Lei†
 ab,
Qian
Zhao†
c,
Binquan
Yang
a,
Xiaoyun
Pu
a,
Guidong
Lu
*a,
Minshou
Wang
*d and
Shizhi
Jiang
*abe
ab,
Qian
Zhao†
c,
Binquan
Yang
a,
Xiaoyun
Pu
a,
Guidong
Lu
*a,
Minshou
Wang
*d and
Shizhi
Jiang
*abe
aCollege of Pharmacy, Dali University, Dali 671000, PR China. E-mail: 16687325517@163.com; jiangshizhi@dali.edu.cn
bYunnan Provincial Key Laboratory of Entomological Biopharmaceutical R&D, Dali University, Dali 671000, PR China
cShaanxi Key Laboratory of Disaster Monitoring and Mechanism Simulation, College of Geography and Environment, Baoji University of Arts and Sciences, Baoji, Shaanxi 721013, PR China
dWest Yunnan University of Applied Sciences, Dali 671000, PR China. E-mail: wmshou.good@163.com
eYunnan Huapai Pharmaceutical Technology Co., Ltd, Chuxiong 675000, PR China
First published on 28th January 2026
Abstract
Herein, we report a concise and efficient one-pot total synthesis of TMC-205 in 77% overall yield (gram-scale) using only two simple and streamlined steps. In the proposed approach, a novel Heck-dehydration reaction and a practical aromatic carboxylic acid introduction strategy are reported, which are characterized by high atom economy, excellent regioselectivity and stereoselectivity (E-isomer). This synthesis protocol is protecting group-free, redox neutral, environmentally benign, and features simple operation steps. Through the implementation of this new, efficient and scalable synthesis method, the formal synthesis of a series of novel meroterpenoid natural products can be successfully realized. Moreover, the synthetic strategy and methodologies demonstrated in this paper are useful and can be easily extended to the preparation of other related biologically active molecules.
Introduction
Meroterpenoids are derived from the fusion of terpenoid and non-terpenoid moieties, endowing them with distinct biological properties and potential applications in various fields, including the pharmaceutical, medicinal and agricultural industries.1,2 Their unique structures enable interactions with specific targets in living systems, making these molecules valuable candidates for drug discovery and development.Cancer, a major global public health burden, is characterized by an abnormal growth of cells caused by a variety of genetic aberrations, leading to dysregulation of the balance between cell proliferation and cell death.3 This terrifying human disease is caused by exogenous chemical, physical or biological carcinogens, with the mechanisms of carcinogenesis often being multifactorial and complex.4,5 As a rich source, natural products play an important role in drug discovery and development.6–8 In addition, they are important as innovative molecular scaffolds for the treatment of various diseases, especially cancer.
TMC-205, which was first isolated in 2001 by Masaaki Sakurai and colleagues, is a secondary metabolite of an unidentified fungal strain, TC-1630.9 Structurally, this natural product belongs to the class of indole alkaloids featuring an unusual simplified indole-3-carboxylic acid derivative with a hemiterpene-like side chain at position 6 (Fig. 1). TMC-205 exhibits antiproliferative activity against various human cancer cell lines via activation of the SV40 promoter.10 However, early isolation attempts afforded only 3.3 mg of TMC-205 from 7.0 liters of fermentation broth of TC-1630. Such small amounts of pure compound are insufficient to conduct comprehensive and in-depth biological and SAR studies. Nevertheless, these indole alkaloids have attracted interest from both synthetic and pharmaceutical chemists because of their unique and fascinating structures and wide-ranging biological activities, most notably their antitumor activity.10 Therefore, a practical approach was followed in this study to synthesize and investigate the biological activity of these privileged indole alkaloids and their analogues.
More importantly, in connection with a research program focused on the synthesis of numerous biologically active natural products—including several promising meroterpenoids (Fig. 1), e.g., exotines A/B (2),11 flinderoles A (3),12 isoborreverine (4),13 raputindole A (5),14 conoideoxime A (6),15 yuehchkene (7),16 borreverine (8),12 borrerine (9),12 6,7-secoagroclavine (10),17 aurantioclavine (11),18 clavicipitic acid (12),19 and caulindoles A, B, C, and D (13)12—we aimed to develop a convenient and high-yield synthesis method for TMC-205, especially to access its useful structural analogs.
Thus far, only two research groups have accomplished the total synthesis of this beguilingly simple natural product. In 2014, Koide's team reported an elegant method for the first total synthesis of TMC-205.10 In their approach, TMC-205 was obtained in 5 steps via the longest linear sequence (6 steps in total) in an overall yield of 64%. In particular, Friedel–Crafts acylation, esterification (using TMSCHN2) and Suzuki–Miyaura coupling were the key steps. However, their method requires independent preparation of the isoprene fragment and involves multiple steps for the construction of the 3-carboxyl group. In addition, it also has inherent limitations, such as the need for relatively harsh reaction conditions and expensive and toxic reagents and starting materials. In 2021, our group realized a two-step, gram-scale synthesis method of TMC-205 through a well-developed diene introduction strategy and a precisely regulated Pinnick oxidation, which should be carefully performed.20 This synthesis strategy achieved excellent yields (based on recovered starting material) without any protecting groups. However, Pinnick oxidation has additional limitations, such as the requirement for a mixed solvent system (THF–tBuOH–H2O). Moreover, the required quantities of the scavenger reagent and polymerization inhibitor, the use of sensitive and explosive NaClO2, and the need for prolonged low temperature reaction conditions make the operation difficult and further scale up challenging. Hence, the strategies discussed above are not ideal synthetic methods, as their implementation could erode the simplicity, scalability and convenience of the synthesis strategy.
The synthesis of natural products involves expensive reagents, harsh reaction conditions, longer reaction times, and complex multi-step procedures, as well as cumbersome experimental/work-up procedures, which usually affect further application.21 Therefore, the development of practical synthetic strategies for the total synthesis of the bioactive natural products as well as their structural analogs remains a challenging task for synthetic organic chemists. With the aim to provide a solution, this study reveals a more effective and easily scalable one-pot procedure toward the concise synthesis of TMC-205 from inexpensive and readily available starting materials under mild conditions.
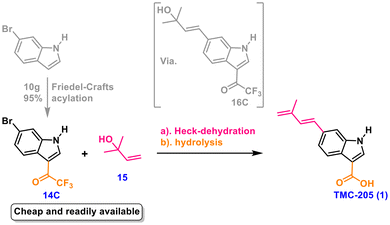
Considering the remarkable conciseness of the protecting-group-free and redox neutral strategy employed in our previous study for the simple synthesis of indiacen A,22 luteoride A and luteoride B,23 murraol and (E)-suberenol,24 and trans-dehydroosthol and citrubuntin,25 we decided to explore the possibility of preparing the target compounds via coupling of a carboxyl equivalent containing an indole moiety with an olefin side chain. In this context, a Heck coupling26 appeared to be the most attractive strategy for introducing a trans-alkene, which could undergo a simple hydrolysis reaction, ultimately leading to an “ideal” chemical synthesis process without the need for protecting groups or redox steps. Therefore, we devised a novel synthetic route employing the 6-bromoindole derivative 14 and allyl alcohol 15 as suitable starting materials for installing the desired carbon skeleton via a Heck-dehydration reaction (Scheme 1).
Results and discussion
Initially, we used 3-carboxy-6-bromindole 14A directly as the starting material with the expectation of synthesizing TMC-205 in a single step. However, when we applied the synthesis conditions of indiacen A22 for this coupling, only a trace amount of the desired product 1 was obtained, accompanied by a series of by-products. Subsequently, we screened several commonly used palladium catalysts and phosphine ligands in the reaction process, but the results were far from satisfactory. Regrettably, we concluded that early-stage installation of the 3-carboxy group was not advisable because of the difficulties in controlling the problematic acidic aryl carboxylic acid group, which presumably led to significant catalyst deactivation or decomposition of the carboxyl group during the coupling reaction. To obtain a high coupling yield, it is advisable to adjust the synthesis strategy to circumvent the tricky carboxyl group. Therefore, we changed the synthesis route by carrying out Heck coupling after introducing an ester group 14B at position 3. A direct C3 functionalization of free (N–H) indoles with methyl chloroformate under the established mild conditions could provide particularly useful indole-3-carboxylic ester intermediates in high to excellent yields.27 After many experimental attempts, it was found that the acylation yield of our 6-bromoindole substrate was low, at only 36%. Although the Heck-dehydration of the ester product could be carried out under mild conditions, the yield was still unsatisfactory (ca. 60%). The higher yield required for the coupling of the brominated substrate and 1,1-dimethylallyl alcohol is presumably due to the less reactive nature of the C–Br bond relative to the C–I bond.22,28 This unfavorable situation is exacerbated by the strong electron-donating capacities of the free nitrogen atom in the indole pentacyclic ring for the benzene ring system. As a result, the oxidative insertion of the phosphine–palladium complex into the C–Br bond is slowed, which in turn limits the entire catalytic cycle, as oxidative insertion is usually the rate-limiting step for Heck coupling.29As illustrated in Scheme 1 and based on our above experiments and analysis, our retrosynthetic analysis of TMC-205 was revised to enable late-stage introduction of the 3-carboxy group by the hydrolysis of the more suitable trifluoroacetyl group. The crucial step in our outlined synthesis was found to be the Heck-dehydration reaction of 6-bromo-3-trifluoroacetylindole 14C and 1,1-dimethylallyl alcohol 15. Moreover, the presence of the trifluoroacetyl group, as well as the unprotected enamine moiety, was found not to affect reaction progress. Notably, the introduction of trifluoroacetyl as the equivalent of carboxylic acid proved to be crucial as it can not only be easily hydrolyzed to carboxylic acid but also improve the catalytic coupling activity of brominated indole due to its strong electron-withdrawing properties. What is more, we believe that it would be a significant advantage to develop a more efficient catalyst system for Heck couplings with the low-cost but relatively less reactive bromoindole.
Our synthetic efforts commenced with 6-bromo-3-trifluoroacetyl indole 14C as the key starting material (Scheme 2). It is commercially available and can also be readily prepared from inexpensive 6-bromoindole and trifluoroacetic anhydride via the Friedel–Crafts (F–C) acylation reaction. The F–C acylation reaction of indole at position 3 proceeds rapidly under mild conditions and is generally the most direct and effective method for the preparation of trifluoroacetylindole. Furthermore, the desired product can be obtained in high purity and almost quantitative yield by conventional extraction without further purification. Besides, this reaction is scalable and could be carried out smoothly on a 10 g scale without difficulty (see the SI for details).
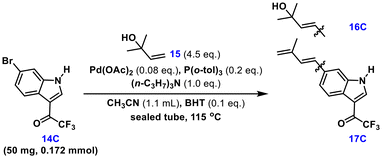
With sufficient 6-bromo-3-trifluoroacetyl indole 14C in hand, we next focused on the crucial Heck-dehydration reaction. Initially, we used acetonitrile as the solvent and treated bromide 14C and 1,1-dimethylallyl alcohol 15 with Pd(OAc)2 and P(o-tol)3 in the presence of (n-C3H7)3N as the base and BHT as the additive (polymerization inhibitor) in a sealed tube at 115 °C.20 As expected, after 4 h, the desired diene 17C was obtained in high yield (91%) with high regio- and stereoselectivity (Table 1, entry 1). The geometry of the double bond was ascertained as the E-isomer by 1H NMR spectroscopy. Encouraged by the initial experiment, we tried to prepare diene 17C at 90 °C (entry 2). Unfortunately, the desired product 17C was not formed, and the unreacted starting material 16C was recovered in 30% yield. However, the Heck cross-coupling was accomplished with good selectivity to afford allylalcohol 14C in 67% yield. We assumed that the hydroxyl elimination did not proceed smoothly at 90 °C because of the lower reaction temperature; as is well known, high temperatures are beneficial for dehydration. When 1.5 equivalents of trialkylamine were used (entry 3), the diene product 17C was obtained in low yield (21%), and “unreacted” alcohol 16C was recovered (68%). It is noteworthy that the increased dose of (n-C3H7)3N (1.5 equivalents vs. 1.0 equivalent) strongly inhibited the dehydration reaction. This result may be attributed to its alkalinity, as alkaline conditions are not conducive to hydroxyl elimination. However, further increase in temperature to 125 °C affected the yield (85%, entry 4). Surprisingly, the desired Heck cross-coupling product 16C was obtained in excellent yield in just one hour by employing an increased amount of (n-C3H7)3N (1.5 equivalents) at 90 °C (95%, entry 5).
| Entry | Variable | t [h] | Yielda (%) | ||
|---|---|---|---|---|---|
| 14C | 16C | 17C | |||
| “–” = Not detected.a Isolated yield.b Pd2(dba)3 & Pd2(dba)3·CHCl3 = 0.04 equivalents.c Palladium(II) trifluoroacetate.d Tetrakis(triphenylphosphine)palladium.e Palladium(II) hexafluoroacetylacetonate.f Bis(2,4-pentanedionato)palladium(II).g Without additional phosphorus ligand.h The (n-C3H7)3N base was 1.25 equivalents.i Bis(2-diphenylphosphinophenyl)ether.j Dimethylacetamide.k Atmospheric pressure.l N-Methylpyrrolidone.m Dichloroethane.n N,N-Diisopropylethylamine.o Gram-scale (2.0 g, 6.9 mmol). | |||||
| 1 | None | 4 | — | — | 91 |
| 2 | 90 °C | 4 | 30 | 67 | — |
| 3 | 1.5 eq. base | 4 | — | 68 | 21 |
| 4 | 125 °C | 1 | — | — | 85 |
| 5 | 90 °C, 1.5 eq. base | 1 | — | 95 | — |
| 6 | PdCl2 | 3 | 11 | — | 85 |
| 7 | Pd2(dba)3b | 5 | 12 | — | 79 |
| 8 | Pd2(dba)3·CHCl3b | 6 | 9 | — | 80 |
| 9 | Pd(TFA)2c | 2 | 18 | — | 80 |
| 10 | Pd(PPh3)4d | 13 | 2 | 25 | 54 |
| 11 | Pd(C5HF6O2)2e | 5 | 12 | — | 88 |
| 12 | Pd(acac)2f | 3 | — | 2 | 88 |
| 13 | Pd(t-Bu3P)2g | 4 | — | 4 | 91 |
| 14 | P(t-Bu)3 | 12 | — | — | 90 |
| 15 | P(t-Bu)3·HBF4 | 8/9h | 20/–h | –/8h | 73/84h |
| 16 | Dpe-Phosi | 18 | 5 | 29 | 50 |
| 17 | DMF | 7 | 11 | — | 80 |
| 18 | DMAj | 4/4k | 2/—k | 1/—k | 85/91k |
| 19 | NMPl | 4/3k | —/—k | —/—k | 88/92k |
| 20 | 1,4-Dioxane | 12 | — | 24 | 61 |
| 21 | DCEm | 6 | 10 | 4 | 80 |
| 22 | THF | 13 | 25 | — | 74 |
| 23 | Toluene | 17 | 89 | 8 | — |
| 24 | Et3N | 5 | 5 | 2 | 87 |
| 25 | DIPEAn | 4 | – | 7 | 85 |
| 26 | (n-C8H17)3N | 6 | 6 | 7 | 89 |
| 27k | DMA+Pd(t-Bu3P)2g | 4/7o | —/—o | —/—o | 92/93o |
Inspired by the results of the systematic investigation of the Heck-dehydration reaction by our group, we attempted to further improve the reaction yield to make it technically simple and less time consuming. We began our investigation by screening several readily available palladium catalysts (entries 6–12). Preliminary screening revealed that Pd(OAc)2 was the most effective palladium catalyst for this Heck-dehydration reaction, while other catalysts such as PdCl2, Pd2(dba)3, Pd2(dba)3·CHCl3, Pd(TFA)2 and Pd(C5HF6O2)2 also performed well although they were slightly less effective than Pd(OAc)2. In addition, compared with the above-mentioned catalysts, the representative Pd(0) catalyst, Pd(PPh3)4, showed lower efficiency (13 h, 54%, entry 10). More importantly, Pd(t-Bu3P)2 was also found to be another suitable catalyst, without additional phosphorus ligand, for cross-coupling reactions of 6-bromo-3-trifluoroacetylindole and 1,1-dimethylallyl alcohol (91%, entry 13).
Based on the preceding analysis, along with the fact that the nature of the ligand has a tremendous influence on the Heck coupling reaction, we assumed that Heck alkenylation of our brominated substrate required the addition of a more suitable ligand that could mediate the electronic properties and/or the steric hindrance of the Pd center. Thus, various phosphorus ligands were screened in combination with Pd(OAc)2. Nevertheless, P(o-tol)3 was observed to yield superior results (entry 1). Besides, when the electron-rich and sterically demanding P(t-Bu)3 was employed, a good yield was also observed after extending the reaction time to 12 h (90%, entry 14). Although P(t-Bu)3 (which is highly air sensitive) also affords excellent yields, P(o-tol)3 is easier to handle and the reaction time is shorter than that for P(t-Bu)3. The air stable variant of P(t-Bu)3, P(t-Bu)3·HBF4, afforded a moderate yield under the action of 1.0 eq. (n-C3H7)3N (entry 15-1). These experimental results were inconsistent with our expectations. We speculated that P(t-Bu)3·HBF430,31 forms a strong acid through hydrolysis in the reaction solution, which affects the Heck coupling and reduces the yield. In order to verify this assumption, we added an additional 0.25 equivalents of tripropylamine to the initial 1.0 equivalent, and this significantly improved the yield (73% → 84%, entry 15). When the widely known DPEphos was used instead of P(o-tol)3, the desired product was obtained in moderate yield (50%, entry 16).
After we established that the combination of Pd(OAc)2 and P(o-tol)3 was a highly effective catalyst for the coupling of our substrate, we screened various solvents and found that amide solvents such as DMA and NMP lead to the formation of diene 17C in good yield (entries 18 and 19). When other solvents such as DMF, 1,4-dioxane, dichloroethane (DCE), THF, and toluene were employed (entries 17, 20–23), a moderate or significant decrease in yield was observed. In particular, when toluene was used as a solvent, almost no reaction occurred (entry 23). Nevertheless, we were not satisfied with the use of a sealed tube, which is not ideal for large-scale synthesis. Therefore, further investigation was undertaken to develop a more practical method. Process development demonstrated that the reaction could also proceed efficiently at atmospheric pressure in DMA (91%) or NMP (92%). It is worth mentioning that this ordinary pressure reaction has greater efficacy compared to the sealed tube version and does not need higher temperature to compensate for the reduction in reaction pressure.
After examining the solvent for the reaction, we went on to investigate the role of a base. Examination of various bases (entries 24–26) indicated that trialkylamine had a positive effect on the reaction. The coupling product 17C was obtained in good yield when Et3N, DIPEA and (n-C8H17)3N were used as the base (entries 24–26). In particular, in the presence of (n-C8H17)3N, the yield of product 17C reached 89% (entry 26). Hence, these research results once again verified our previous conclusions that the presence of a tertiary amine base is necessary for an efficient Heck-dehydration reaction.22
Following the optimization of the Heck-dehydration reaction conditions, we decided to combine another suitable palladium catalyst, Pd(t-Bu3P)2, without an additional phosphorus ligand, with the best base and solvent to further enhance the synthesis efficiency of the reaction. According to the above experimental results, when DMA (entry 18) and NMP (entry 19) were used, the yield of diene 17C was relatively high. However, considering the high boiling point of NMP (m.p. = 202–205 °C), it is not suitable for recovery by vacuum distillation, so we chose DMA (m.p. = 164 °C) as the solvent to improve the yield. To our surprise, when we combined Pd(t-Bu3P)2 with (n-C3H7)3N and DMA, the yield of 17C increased to 92% (entry 27-1). Process development demonstrated that the reaction could also be performed well at the gram scale and resulted in an improved yield of up to 93% (entry 27-2) with no efficiency loss compared to the small-scale reaction.
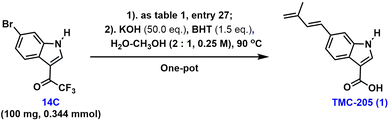
Having assembled the desired carbon skeleton, we turned our attention to the transformation of the trifluoroacetyl group to produce the carboxylic acid group for obtaining the desired TMC-205. The hydrolysis reaction was straightforward, and pure raw materials were not required. Besides, the Heck-coupling reaction in the presence of a catalytic amount of palladium complexes proceeded efficiently in high yield and did not lead to the formation of by-products. We believe that the crude concentrates of the Heck-dehydration reaction could be subjected to the described hydrolysis reaction to provide TMC-205 (1) without difficulty. Consequently, and also to achieve an efficient one-pot transformation, we decided to explore this simple hydrolysis reaction.
Indeed, when the Heck-dehydration reaction was completed, the desired crude coupling product could be obtained by simple vacuum concentration of the reaction mixture without further purification, and its purity was sufficient for further modification. Subsequently, after a few one-pot experimental trials (Table 2), we achieved the desired result using an aqueous solution of KOH and BHT as the additives in a mixed solvent system (H2O–CH3OH) (entry 1, 77% overall yield).
| Entry | Variable | t [h] | Yield (1)%a |
|---|---|---|---|
| a Isolated yield. b Gram-scale synthesis (2.0 g, 6.9 mmol). | |||
| 1 | None | 4 + 5 | 76(77b) |
| 2 | KOH (45.0 eq.) | 4 + 6 | 67 |
| 3 | KOH (55.0 eq.) | 4 + 5 | 72 |
| 4 | 80 °C | 4 + 11 | 74 |
| 5 | 100 °C | 4 + 5 | 73 |
| 6 | BHT (1.0 eq.) | 4 + 5 | 69 |
| 7 | BHT (2.0 eq.) | 4 + 4 | 71 |
| 8 | NaOH (50.0 eq.) | 4 + 3 | 55 |
| 9 | Not one-pot protocol | 4 + 6 | 56 |
It is worth noting that this one-pot procedure afforded a higher overall yield of the desired product 1, compared to the conventional “two-pot” operation [(77%, two steps in one pot, entry 1) vs. (56%, two steps, entry 9)]. Besides, the one-pot reaction provides a unique opportunity to avoid intermediate isolation and purification, offering additional advantages in terms of cost and waste reduction, thus making this strategy more sustainable and economical. To evaluate the efficacy of this reaction on a larger scale, the reaction was carried out using 2 g of substrate 14C. Fortunately, the reaction yield was not affected during scale-up (entry 1-2).
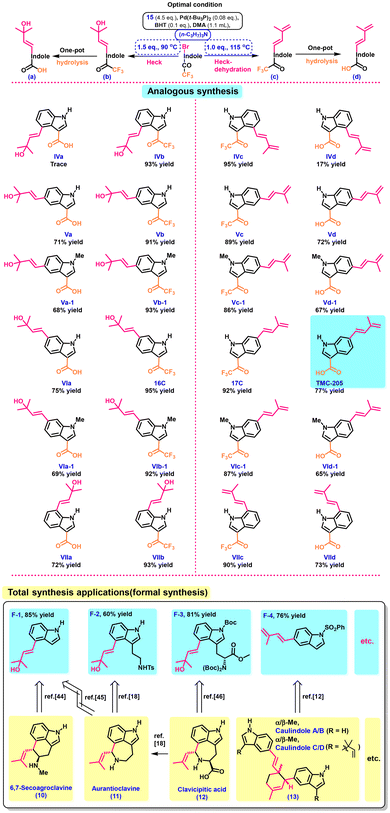
Following the optimization of the one-pot reaction conditions, we decided to proceed with the synthesis of the analogues of TMC-205, so that its pharmacological activity could be better examined and the synthetic utility of this reaction could also be demonstrated. To our satisfaction, all the selected substrates gave the desired olefin products in good to excellent yields (Table 3). In particular, this one-pot strategy can also furnish a range of desired tertiary allylic alcohols in excellent yields simply by controlling the reaction temperature and the amount of the base (Table 3, left). Although these compounds had not been isolated from natural sources prior to this study, the presence of murraol, trans-dehydroosthol, (E)-suberenol and citrubuntin in nature indicates that they may also occur naturally.32–35
We believe that the chemical versatility of TMC-205 and its analogues enables the synthesis of various complex molecules with diverse biological activities and therapeutic potential. For example, by strategically utilizing these compounds as building blocks, we can modify and introduce various dienophiles (or conjugated diolefines), functional groups, and stereochemistries, streamlining synthesis routes toward numerous complex natural products. The total synthesis of these isoprene or allylalcohol-containing molecules has attracted the attention of various research groups,36–43 highlighting their significance in the field of meroterpenoid synthesis. Moreover, their synthesis aids in the quest for novel natural products with wide-ranging applications and contributes to the development of new drugs, flavors and agricultural agents.
Based on the above rationale and leveraging the efficient synthesis method of TMC-205's analogues, to further demonstrate the usefulness of the proposed synthesis method, we next focused on the synthesis of natural products such as exotines A/B (2), flinderoles A (3), isoborreverine (4), raputindole A (5), conoideoxime A (6), yuehchkene (7), borreverine (8), borrerine (9), 6,7-secoagroclavine (10), aurantioclavine (11), clavicipitic acid (12), and caulindoles A, B, C, and D (13). We were glad to discover that the desired products 6,7-secoagroclavine (10), aurantioclavine (11), clavicipitic acid (12) and caulindoles A–D (13) could be prepared easily in a few steps from F-1, F-2, F-3 or F-4 using easily available reagents, as described in the literature.11–19 In other words, we have completed the formal synthesis of these seven compounds. Our laboratory is currently pursuing strategies to further enhance the synthesis efficiency of the abovementioned hybrid natural products, and we anticipate that the results will be reported in the future.
Conclusions
In thus study, we have developed a simple one-pot synthetic method for the naturally occurring indole alkaloid TMC-205 as well as its 28 synthetic analogues, which were obtained in excellent yields from simple and readily available starting materials. This short and efficient synthesis procedure features a successful PGF and a redox-neutral strategy. The process is also characterized by excellent stereoselectivity (E-isomer) and atom economy. The gram-scale syntheses of TMC-205 (1) and a range of its analogues will facilitate in-depth and comprehensive studies focusing on their biological properties and SAR. Importantly, this synthetic approach not only achieved the formal synthesis of a series of novel meroterpenoids, including 6,7-secoagroclavine (10), aurantioclavine (11), clavicipitic acid (12), and caulindoles A, B, C and D (13), but also facilitated their overall synthesis. Ongoing efforts are focused on refining the practical, cost-effective conditions and methodologies for these hybrid compounds. Moreover, the synthetic strategies reported in this study are being explored for application to related natural products. Further developments are underway and will be reported in due course.Experimental section
General information
NMR spectra were acquired using a Bruker AVANCE III instrument (400 MHz for 1H and 100 MHz for 13C NMR) in the specified solvent. Chemical shifts (δ) are given in parts per million (ppm) relative to tetramethylsilane, with the solvent resonance serving as an internal reference. Deuterochloroform was used as the solvent in all cases. Coupling constants (J) are provided in hertz (Hz). Multiplicity is described using the following abbreviations: s (singlet), d (doublet), t (triplet), q (quartet), dd (doublet of doublets), dt (doublet of triplets), AB q (AB-quartet), and m (multiplet). For 13C-NMR, the number of contributing carbon nuclei is indicated in parentheses following the chemical shift value. IR spectra were recorded on a Nexus FT/IR spectrometer (Thermo Nicolet, America). Only the structurally important peaks were reported as IR data, given in cm−1. High-resolution mass spectra (HRMS) were obtained using APPI or ESI ionization (positive or negative) on a Bruker Compact Quadrupole Time-of-Flight mass spectrometer, calibrated with sodium formate. Reaction progress was monitored by thin-layer chromatography on 0.2–0.25 mm silica gel plates (GF-254) using UV light (254 nm) for visualization. Phosphomolybdic acid (10 wt% in EtOH) was used as the visualizing agent. Flash chromatography was conducted using silica gel (200–300 mesh).General synthesis method for the Heck reaction
Under a nitrogen atmosphere, a double neck round-bottomed flask equipped with a magnetic stir bar and a condenser tube was loaded with 14C (or other aryl bromides, 1.0 eq.), Pd(t-Bu3P)2 (0.08 eq.), BHT (0.1 eq.), DMA (the solvent was bubbled with nitrogen for 10 min prior to use), (n-C3H7)3N (1.5 eq.) and 1,1-dimethylallyl alcohol 15 (4.5 eq.). Then the flask was placed in a 90 °C oil bath. After completion of the reaction (as monitored by TLC), the reaction mixture was cooled to room temperature, and then a saturated aqueous solution of NaHCO3 was added and stirred for another 5 min. Subsequently, the mixture was filtered through a pad of Celite and washed with ethyl acetate (EA). The filtrate was then diluted with EA and washed with water and brine. The organic layers were dried over Na2SO4, filtered and concentrated in vacuo at 30 °C to afford the crude product. After purification by flash column chromatography (PE![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) EA = 8
EA = 8![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 to 1
1 to 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1), the title compound was obtained.
1), the title compound was obtained.
General synthesis method for the Heck-dehydration reaction
Under a nitrogen atmosphere, a heavy-wall pressure vessel or a double neck round-bottomed flask equipped with a condenser tube and a magnetic stir bar was loaded with 14C (or other aryl bromides, 1.0 eq.), Pd(t-Bu3P)2 (0.08 eq.), BHT (0.1 eq.), DMA (the solvent was bubbled with nitrogen for 10 min prior to use), (n-C3H7)3N (1.0 eq.) and 1,1-dimethylallyl alcohol 15 (4.5 eq.). Then the flask was placed in a 115 °C oil bath. After completion of the reaction (as monitored by TLC, or at a given time), the reaction mixture was cooled to room temperature and a saturated aqueous solution of NaHCO3 was added and stirred for another 5 min. Afterwards, the mixture was filtered through a pad of Celite and washed with ethyl acetate (EA). The filtrate was diluted with EA and washed with water and brine. The organic layers were dried over Na2SO4, filtered and concentrated in vacuo at 30 °C to obtain the crude product. After purification by flash column chromatography (PE![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) EA = 8
EA = 8![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 to 1
1 to 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1), the title compound was obtained.
1), the title compound was obtained.
General synthesis method for the Heck-hydrolysis one-pot reaction
Under a nitrogen atmosphere, a double neck round-bottomed flask equipped with a magnetic stir bar and a condenser tube was loaded with 14C (or other aryl bromides, 1.0 eq.), Pd(t-Bu3P)2 (0.08 eq.), BHT (0.1 eq.), DMA (the solvent was bubbled with nitrogen for 10 min prior to use), (n-C3H7)3N (1.5 eq.) and 1,1-dimethylallyl alcohol 15 (4.5 eq.). Then, the flask was placed in a 90 °C oil bath. After completion of the reaction (as monitored by TLC, or at a given time), the reaction mixture was cooled to room temperature and the solvent was removed under vacuum at room temperature. Subsequently, KOH (50.0 eq.), BHT (1.5 eq.), and H2O–CH3OH (2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, 0.25 M) were added, and the flask was sealed and placed in a 90 °C oil bath. After completion of the reaction (as monitored by TLC), the reaction mixture was cooled to room temperature, then an aqueous solution of KHSO4 was added and the mixture was acidified to pH = 6–7. The resulting solution was extracted with EA, concentrated, and further purified by column chromatography on silica gel (PE
1, 0.25 M) were added, and the flask was sealed and placed in a 90 °C oil bath. After completion of the reaction (as monitored by TLC), the reaction mixture was cooled to room temperature, then an aqueous solution of KHSO4 was added and the mixture was acidified to pH = 6–7. The resulting solution was extracted with EA, concentrated, and further purified by column chromatography on silica gel (PE![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) EA = 8
EA = 8![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 to 1
1 to 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) to obtain the title compound.
1) to obtain the title compound.
General synthesis method for the Heck-dehydration-hydrolysis one-pot reaction
Under a nitrogen atmosphere, a double neck round-bottomed flask equipped with a magnetic stir bar and a condenser tube was loaded with 14C (or other aryl bromides, 1.0 eq.), Pd(t-Bu3P)2 (0.08 eq.), BHT (0.1 eq.), DMA (the solvent was bubbled with nitrogen for 10 min prior to use), (n-C3H7)3N (1.0 eq.) and 1,1-dimethylallyl alcohol 15 (4.5 eq.). Then, the flask was placed in a 115 °C oil bath. After completion of the reaction (as monitored by TLC or at a given time), the reaction mixture was cooled to room temperature and the solvent was removed under vacuum at room temperature. Subsequently, KOH (50.0 eq.), BHT (1.5 eq.), and H2O–CH3OH (2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, 0.25 M) were added and the flask was sealed and placed in a 90 °C oil bath. After completion of the reaction (as monitored by TLC), the reaction mixture was cooled to room temperature, and then an aqueous solution of KHSO4 was added and the mixture was acidified to pH = 7. The resulting solution was extracted with EA, concentrated, and further purified by column chromatography on silica gel (PE
1, 0.25 M) were added and the flask was sealed and placed in a 90 °C oil bath. After completion of the reaction (as monitored by TLC), the reaction mixture was cooled to room temperature, and then an aqueous solution of KHSO4 was added and the mixture was acidified to pH = 7. The resulting solution was extracted with EA, concentrated, and further purified by column chromatography on silica gel (PE![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) EA = 8
EA = 8![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 to 1
1 to 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) to obtain the title compound.
1) to obtain the title compound.
Application of total synthesis for the preparation of F-1− F-4
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 to 2
1 to 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1), the title compound was obtained (43.6 mg, 85% yield). 1H NMR (400 MHz, CDCl3) δ/ppm: 8.30 (s, 1H), 7.30 (dt, J = 7.9, 1.1 Hz, 1H), 7.25–7.22 (m, 2H), 7.20–7.15 (m, 1H), 7.00 (d, J = 16.2 Hz, 1H), 6.77 (ddd, J = 3.1, 2.0, 0.9 Hz, 1H), 6.55 (d, J = 16.1 Hz, 1H), 1.49 (s, 6H); 13C NMR (100 MHz, CDCl3) δ/ppm: 138.4, 136.3, 129.4, 126.2, 124.8, 124.5, 122.2, 117.5, 110.4, 101.2, 71.5, 30.1; HRMS (ESI-APPI) m/z calcd for C13H16NO [M + H]+ 202.1226, found 202.1231.
1), the title compound was obtained (43.6 mg, 85% yield). 1H NMR (400 MHz, CDCl3) δ/ppm: 8.30 (s, 1H), 7.30 (dt, J = 7.9, 1.1 Hz, 1H), 7.25–7.22 (m, 2H), 7.20–7.15 (m, 1H), 7.00 (d, J = 16.2 Hz, 1H), 6.77 (ddd, J = 3.1, 2.0, 0.9 Hz, 1H), 6.55 (d, J = 16.1 Hz, 1H), 1.49 (s, 6H); 13C NMR (100 MHz, CDCl3) δ/ppm: 138.4, 136.3, 129.4, 126.2, 124.8, 124.5, 122.2, 117.5, 110.4, 101.2, 71.5, 30.1; HRMS (ESI-APPI) m/z calcd for C13H16NO [M + H]+ 202.1226, found 202.1231.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 to 1
1 to 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1), the title compound was obtained (30.7 mg, 60% yield). 1H NMR (400 MHz, CDCl3) δ/ppm: 8.28–8.23 (m, 1H), 7.64–7.60 (m, 2H), 7.23 (q, J = 4.3 Hz, 1H), 7.19–7.15 (m, 3H), 7.13–7.10 (m, 2H), 6.94 (d, J = 2.4 Hz, 1H), 6.19 (d, J = 15.8 Hz, 1H), 5.19 (t, J = 6.3 Hz, 1H), 3.22–3.15 (m, 2H), 3.05 (t, J = 7.3 Hz, 2H), 2.35 (s, 3H), 1.42 (s, 6H); 13C NMR (100 MHz, CDCl3) δ/ppm: 143.3, 139.6, 137.3, 136.9, 131.3, 129.7, 127.1, 124.7, 124.5, 123.8, 122.4, 117.5, 112.4, 110.6, 71.3, 44.4, 30.2, 28.3, 21.6; HRMS (ESI-APPI) m/z calcd for C22H26N2O3SNa [M + Na]+ 421.1556, found 421.1550.
1), the title compound was obtained (30.7 mg, 60% yield). 1H NMR (400 MHz, CDCl3) δ/ppm: 8.28–8.23 (m, 1H), 7.64–7.60 (m, 2H), 7.23 (q, J = 4.3 Hz, 1H), 7.19–7.15 (m, 3H), 7.13–7.10 (m, 2H), 6.94 (d, J = 2.4 Hz, 1H), 6.19 (d, J = 15.8 Hz, 1H), 5.19 (t, J = 6.3 Hz, 1H), 3.22–3.15 (m, 2H), 3.05 (t, J = 7.3 Hz, 2H), 2.35 (s, 3H), 1.42 (s, 6H); 13C NMR (100 MHz, CDCl3) δ/ppm: 143.3, 139.6, 137.3, 136.9, 131.3, 129.7, 127.1, 124.7, 124.5, 123.8, 122.4, 117.5, 112.4, 110.6, 71.3, 44.4, 30.2, 28.3, 21.6; HRMS (ESI-APPI) m/z calcd for C22H26N2O3SNa [M + Na]+ 421.1556, found 421.1550.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 to 8
1 to 8![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1), the title compound was obtained (69.3 mg, 81% yield). 1H NMR (400 MHz, CDCl3) δ/ppm: 8.07 (d, J = 8.1 Hz, 1H), 7.33–7.16 (m, 4H), 6.20 (d, J = 15.7 Hz, 1H), 5.14 (dd, J = 10.9, 3.2 Hz, 1H), 3.84 (ddd, J = 15.1, 3.2, 1.1 Hz, 1H), 3.76 (s, 3H), 3.32 (dd, J = 15.1, 11.0 Hz, 1H), 1.63 (s, 9H), 1.42 (s, 3H), 1.38 (s, 3H), 1.26 (s, 18H); 13C NMR (100 MHz, CDCl3) δ/ppm: 171.6, 151.7, 149.5, 141.8, 136.4, 132.0, 127.4, 125.5, 124.6, 124.0, 121.6, 116.9, 114.5, 83.7, 83.1, 71.0, 58.9, 52.7, 29.9, 29.8, 28.3, 28.0, 27.8; HRMS (ESI-APPI) m/z calcd for C32H46O9N2Na [M + Na]+ 625.3096, found 625.3105.
1), the title compound was obtained (69.3 mg, 81% yield). 1H NMR (400 MHz, CDCl3) δ/ppm: 8.07 (d, J = 8.1 Hz, 1H), 7.33–7.16 (m, 4H), 6.20 (d, J = 15.7 Hz, 1H), 5.14 (dd, J = 10.9, 3.2 Hz, 1H), 3.84 (ddd, J = 15.1, 3.2, 1.1 Hz, 1H), 3.76 (s, 3H), 3.32 (dd, J = 15.1, 11.0 Hz, 1H), 1.63 (s, 9H), 1.42 (s, 3H), 1.38 (s, 3H), 1.26 (s, 18H); 13C NMR (100 MHz, CDCl3) δ/ppm: 171.6, 151.7, 149.5, 141.8, 136.4, 132.0, 127.4, 125.5, 124.6, 124.0, 121.6, 116.9, 114.5, 83.7, 83.1, 71.0, 58.9, 52.7, 29.9, 29.8, 28.3, 28.0, 27.8; HRMS (ESI-APPI) m/z calcd for C32H46O9N2Na [M + Na]+ 625.3096, found 625.3105.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 to 3
1 to 3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1), the title compound was obtained (36.4 mg, 76% yield). 1H NMR (400 MHz, CDCl3) δ/ppm: 7.94 (d, J = 8.6 Hz, 1H), 7.91–7.85 (m, 2H), 7.60–7.50 (m, 3H), 7.48–7.40 (m, 3H), 6.86 (d, J = 16.2 Hz, 1H), 6.63 (dd, J = 3.7, 0.8 Hz, 1H), 6.58 (d, J = 16.1 Hz, 1H), 5.15–5.03 (m, 2H), 1.97 (d, J = 1.3 Hz, 3H); 13C NMR (100 MHz, CDCl3) δ/ppm: 142.2, 138.4, 134.4, 134.0, 133.2, 131.4, 131.4, 129.4, 128.7, 127.0, 126.9, 123.4, 119.5, 117.2, 113.8, 109.6, 18.8; HRMS (ESI-APPI) m/z calcd for C19H17NO2 [M + H]+ 324.1053, found 324.1043.
1), the title compound was obtained (36.4 mg, 76% yield). 1H NMR (400 MHz, CDCl3) δ/ppm: 7.94 (d, J = 8.6 Hz, 1H), 7.91–7.85 (m, 2H), 7.60–7.50 (m, 3H), 7.48–7.40 (m, 3H), 6.86 (d, J = 16.2 Hz, 1H), 6.63 (dd, J = 3.7, 0.8 Hz, 1H), 6.58 (d, J = 16.1 Hz, 1H), 5.15–5.03 (m, 2H), 1.97 (d, J = 1.3 Hz, 3H); 13C NMR (100 MHz, CDCl3) δ/ppm: 142.2, 138.4, 134.4, 134.0, 133.2, 131.4, 131.4, 129.4, 128.7, 127.0, 126.9, 123.4, 119.5, 117.2, 113.8, 109.6, 18.8; HRMS (ESI-APPI) m/z calcd for C19H17NO2 [M + H]+ 324.1053, found 324.1043.
Conflicts of interest
There are no conflicts to declare.Data availability
The authors confirm that the data supporting this study are available within the article and its supplementary information (SI). Supplementary information: crystallographic data collection and refinement statistics. See DOI: https://doi.org/10.1039/d5ob01856k.Acknowledgements
This research was funded by the National Natural Science Foundation of China (grant no. 22361002 and 22161001); the Key Industry Science and Technology Projects for University Services in Yunnan Province (FWCY-ZNT2024023); the Joint Special Fund Project for Basic Research of Local Undergraduate Universities (part of) in Yunnan Province (no. 202101AO070315); the Basic Research Special Project of Yunnan Provincial Department of Science and Technology-General Project (202301AT070202 and 202201AT070175), the Yunnan Youth Fund Project (202401AU070048); and the Key Project of Dali Prefecture Science and Technology Plan (20242903B030009).We gratefully acknowledge the technical support provided by the Analysis and Testing Center of Dali University for nuclear magnetic resonance (NMR) spectroscopy and high-resolution mass spectrometry (HRMS) experiments.
References
- M. Nazir, M. Saleem, M. I. Tousif, M. A. Anwar, F. Surup, I. Ali, D. Wang, N. Z. Mamadalieva, E. Alshammari, M. L. Ashour, A. M. Ashour, I. Ahmed, Elizbit, I. R. Green and H. Hussain, Biomolecules, 2021, 11, 957 CrossRef CAS PubMed.
- M. H. Jiang, Z. E. Wu, L. Liu and S. H. Chen, Org. Biomol. Chem., 2021, 19, 1644 RSC.
- S. Luo, G. J. Tian, F. X. Yu and Z. D. Wen, Transl. Cancer Res., 2021, 10, 1578–1582 CrossRef CAS PubMed.
- V. Furlan and U. Bren, Int. J. Mol. Sci., 2020, 21, 695 CrossRef CAS PubMed.
- H. Sun, S. Burrola, J. Wu and W. Q. Ding, Int. J. Mol. Sci., 2020, 21, 6097 CrossRef CAS PubMed.
- F. Jamshidi-Kia, Z. Lorigooini and H. Amini-Khoei, J. Herbmed Pharmacol., 2018, 7, 1–7 CrossRef.
- F. Mahmoudi, M. H. T. Jalayeri, A. Montaseri, L. M. Khosroshahi and B. Baradaran, Int. J. Biol. Macromol., 2024, 278, 134778 CrossRef CAS PubMed.
- M. Akram, A. Rashid, R. Zainab, U. Laila, M. T. Khalil, H. Anwar, N. Thotakura and M. Riaz, J. Mod. Pharmacol. Pathol., 2023, 1, 7 Search PubMed.
- K. Sakurai, J. Kohno, M. Nishio, K. Yamamoto, T. Okuda, K. Kawano and N. Nakanishi, J. Antibiot., 2001, 54, 628–634 CrossRef PubMed.
- Y. Gao, S. Osman and K. Koide, ACS Med. Chem. Lett., 2014, 5, 863–867 CrossRef CAS PubMed.
- Y. Liu, C. Zhang, K. W. Zeng, J. Li, X. Y. Guo, M. B. Zhao, P. F. Tu and Y. Jiang, Org. Lett., 2015, 17, 4380–4383 CrossRef PubMed.
- D. H. Dethe, R. D. Erande and B. D. Dherange, Org. Lett., 2014, 16, 2764–2767 CrossRef CAS PubMed.
- D. H. Dethe, R. D. Erande and A. Ranjan, J. Org. Chem., 2013, 78, 10106–10120 CrossRef CAS PubMed.
- K. Vougogiannopoulou, N. Fokialakis, N. Aligiannis, C. Cantrell and A. L. Skaltsounis, Org. Lett., 2010, 12, 1908–1911 CrossRef CAS PubMed.
- M. Isaka, S. Palasarn, W. Choowong and S. Mongkolsamrit, Tetrahedron Lett., 2019, 60, 154–156 CrossRef CAS.
- J. H. Sheu, Y. K. Chen and Y. L. V. Hong, J. Org. Chem., 1993, 58, 5784–5787 CrossRef CAS.
- A. Regni, F. Bartoccini and G. Piersanti, Beilstein J. Org. Chem., 2023, 19, 918–927 CrossRef PubMed.
- C. B. Ji, X. Hu, S. Z. Jiang and J. H. Yang, Eur. J. Org. Chem., 2022, e202201000 CrossRef CAS.
- M. Ito, Y. Tahara and T. Shibata, Chem. – Eur. J., 2016, 22, 5468–5477 CrossRef CAS PubMed.
- T. Li, J. J. Song, L. H. Wang, T. Lei, S. Z. Jiang and F. S. Wang, Tetrahedron Lett., 2021, 74, 153137 CrossRef CAS.
- K. Rajput, V. Singh, S. Singh and V. Srivastava, RSC Adv., 2024, 14, 22480–22485 RSC.
- L. H. Wang, T. Lei, F. S. Wang, S. Z. Jiang and G. Y. Yan, Tetrahedron Lett., 2021, 66, 152822 CrossRef CAS.
- J. J. Song, G. D. Lu, B. Q. Yang, M. J. Bai, J. J. Li, F. S. Wang, T. Lei and S. Z. Jiang, Tetrahedron, 2022, 122, 132933 CrossRef CAS.
- X. S. Gong, M. J. Bai, G. D. Lu, B. Q. Yang, T. Lei and S. Z. Jiang, Tetrahedron, 2022, 126, 133061 CrossRef CAS.
- Z. W. Liu, B. Y. Ge, X. S. Gong, F. S. Wang, T. Lei and S. Z. Jiang, Molecules, 2025, 30, 1067 CrossRef CAS PubMed.
- L. J. Oxtoby, J. A. Gurak Jr., S. R. Wisniewski, M. D. Eastgate and K. M. Engle, Trends Chem., 2019, 1, 572–587 CrossRef CAS PubMed.
- Z. W. Zhang, H. Xue, H. L. Li, H. P. Kang, J. Feng, A. J. Lin and S. X. Liu, Org. Lett., 2016, 18, 3918–3921 CrossRef CAS PubMed.
- M. Somei and F. Yamada, Chem. Pharm. Bull., 1984, 32, 5064–5065 CrossRef CAS.
- M. Oestreich, The Mizoroki-Heck Reaction, Wiley, Chichester, 2009 Search PubMed.
- K. S. A. Vallin, P. Emilsson, M. Larhed and A. Hallberg, J. Org. Chem., 2002, 67, 6243–6246 CrossRef CAS PubMed.
- C. Dasaradhan and F. R. Nawaz Khan, Polycyclic Aromat. Compd., 2020, 42, 1048–1060 CrossRef.
- J. Yamahara, M. Kozuka, T. Sawada, H. Fujimura, K. Nakano, T. Tomimatsu and T. Nohara, Chem. Pharm. Bull., 1985, 33, 1676–1680 Search PubMed.
- C. Ito and H. Furukawa, Chem. Pharm. Bull., 1987, 35, 4277–4285 Search PubMed.
- G. B. Guise, E. Ritchie, R. G. Senior and W. C. Taylor, Aust. J. Chem., 1967, 20, 2429–2439 CrossRef CAS.
- T. S. Wu, Phytochemistry, 1988, 27, 3717–3718 CrossRef CAS.
- J. Reisch and A. Bathe, Liebigs Ann. Chem., 1988, 1988, 543–547 CrossRef.
- R. D. H. Murray and S. Zeghdi, Phytochemistry, 1989, 28, 227–230 CrossRef CAS.
- L. T. Lepovitz and S. F. Martin, Org. Lett., 2018, 20, 7875–7878 CrossRef CAS PubMed.
- L. T. Lepovitz and S. F. Martin, J. Org. Chem., 2021, 86, 10946–10953 CrossRef CAS PubMed.
- J. Reisch, H. M. T. B. Herath and N. S. Kumar, Liebigs Ann. Chem., 1990, 1990, 931–933 CrossRef.
- S. Bhunia, S. Chaudhuri and A. Bisai, Chem. – Eur. J., 2017, 23, 11234–11238 CrossRef CAS PubMed.
- S. Chaudhuri, S. Bhunia, A. Roy, M. K. Das and A. Bisa, Org. Lett., 2018, 20, 288–291 CrossRef CAS PubMed.
- S. Chaudhuri, S. Ghosh, S. Bhunia and A. Bisai, Chem. Commun., 2018, 54, 940–943 Search PubMed.
- S. Romanini, E. Galletti, L. Caruana, A. Mazzanti, F. Himo, S. Santoro, M. Fochi and L. Bernardi, Chem. – Eur. J., 2015, 21, 17578–17582 Search PubMed.
- S. Duan, B. Xue, H. Meng, Z. Ye, Z. F. Xu and C. Y. Li, Chin. J. Chem., 2021, 39, 1145–1152 Search PubMed.
- Z. R. Xu, Q. J. Li, L. H. Zhang and Y. X. Jia, J. Org. Chem., 2009, 74, 6859–6862 Search PubMed.
Footnote |
| † These authors contributed equally to this work and should be considered co-first authors. |
| This journal is © The Royal Society of Chemistry 2026 |