Design and assessment of dimeric trehalose glycolipids as a strategy to enhance Mincle-mediated vaccine adjuvanticity
Emma M.
Dangerfield†
 ab,
M. A. Thathsaranie P.
Manthrirathna†
a,
James C.
Robinson
a,
Brenda S.
Luong
ab,
M. A. Thathsaranie P.
Manthrirathna†
a,
James C.
Robinson
a,
Brenda S.
Luong
 ab,
Olga R.
Palmer
c,
Lisa M.
Connor
c,
Mattie S. M.
Timmer
ab,
Olga R.
Palmer
c,
Lisa M.
Connor
c,
Mattie S. M.
Timmer
 *ab and
Bridget L.
Stocker
*ab
*ab and
Bridget L.
Stocker
*ab
aSchool of Chemical and Physical Sciences, Victoria University of Wellington, PO Box 600, Wellington, New Zealand. E-mail: mattie.timmer@vuw.ac.nz; bridget.stocker@vuw.ac.nz
bCentre for Biodiscovery, Victoria University of Wellington, PO Box 600, Wellington, New Zealand
cMalaghan Institute of Medical Research, Wellington 6012, New Zealand
First published on 2nd February 2026
Abstract
It has been suggested that the clustering of the macrophage inducible C-type lectin (Mincle), either with itself or with the related macrophage C-type lectin (MCL), can lead to improved Mincle-mediated signalling and adjuvant activity. To this end, we synthesised dimeric ligands that contained Mincle agonists trehalose dibehenate (TDB) or C18-brartemicin (C18Brar), and which were linked by C5, C10 or C14 acyl chains. Using mMincle NFAT-GFP reporter cells and murine WT and Mincle−/− bone marrow derived macrophages (BMDMs), we demonstrated that the dimeric ligands activate Mincle signalling, with the C10-linked TDB dimer 3b leading to high levels of IL-1β, IL-6 and, in particular, TNF-α in vitro. Dimer 3b was then tested for its adjuvant properties in vivo using OVA as a model antigen and was found to induce significantly more germinal centre B cells compared to OVA alone and TDB + OVA (unconjugated); however neither TDB nor dimer 3b led to an increase in T cell numbers relative to OVA alone. Taken together, these data provide proof of concept that dimeric Mincle ligands are a new class of potential Mincle-mediated vaccine adjuvants.
Introduction
The macrophage inducible C-type lectin (Mincle, Clec4e, or Clecsf9) is a pattern-recognition receptor (PRR) that has shown much promise as a molecular target for the development of T helper (TH)-1/TH-17-skewing vaccine adjuvants.1 CAF (Cationic Adjuvant Formulation) liposomal adjuvant systems, which contain trehalose dibehenate (TDB, 1, Fig. 1) in combination with dimethyldioctadecylammonium bromide (DDA) and, at times, other immunostimulants, have shown exceptional potential as clinically relevant vaccine adjuvants for both subcutaneous and mucosally-delivered vaccines.1–3 Other 6,6′-acylated trehalose glycolipids, including branched trehalose glycolipids1,4,5 and lipidated brartemicin derivatives6–10 such as C18Brar (2),6 have also demonstrated promising adjuvant activity in several studies, including in combination with TB antigens11,12 and in the veterinary health space.13,14 To elicit their immunomodulatory effects, Mincle ligands, such as TDB (1), bind to the receptor and initiate the FcRγ-Syk-Card9-Bcl10-Malt1 signalling axis.15–18 This ultimately leads to the NFκB-mediated transcription of cytokines, chemokines, and small molecule mediators, such as NO. More recently, a C18Brar-muramyl dipeptide (MDP)-conjugate that was developed to target two PRRs, Mincle and NOD2, respectively, was found to lead to a synergistic improvement in the immune response and a reduction in adjuvant loading.19 Mincle and the related C-Type lectin receptor (CLR) macrophage C-type lectin (MCL) are mutually regulated and act as heterodimers for ligand binding.20–22 Mincle has also been found to exist as a pre-formed disulfide-linked dimer.23 While much remains to be elucidated about the role of Mincle clustering in immune responses, it has been suggested that Mincle clustering enhances the immune response via a variety of mechanisms that include enhanced phagocytosis and inflammatory gene responses,20,21,24 as well as inflammasome activation.25 These responses may in turn augment Mincle-mediated adjuvant activity.1 Clustering may be achieved via the 3D aggregation of individual Mincle ligands using a delivery vehicle1,26 or via a single Mincle agonist that can bridge multiple receptors, as evidenced by crystallographic data which indicated that the highly immunostimulatory Mincle agonist phenolic glycolipid III (PGL-III) bridged two hMincle proteins.27The Mincle binding site can accommodate a trehalose glycolipid acyl chain of approximately eight carbon atoms from the trehalose to the edge of the carbohydrate recognition domain (CRD).28–30 With this in mind, we envisioned preparing two series of dimeric Mincle ligands: those based on TDB (3a–c) and those based on C18Brar (4a–c) (Fig. 2). The individual monomeric units within each dimer would be joined by acyl chains of varying lengths (n = 1, 6, 10). We proposed that these dimers could bind to Mincle heterodimers or Mincle/MCL dimers, with the non-conjugated acyl chain on each trehalose monomer being accommodated by the major hydrophobic groove in Mincle.28,29 If successful, these ligands would be the first examples of dimeric Mincle agonists. For comparison, we were also interested in determining whether the constrained ring-closed product 5 would activate Mincle. Potentially, such a compound may be a Mincle antagonist; however, to date, Mincle-binding DNA aptamers, rather than glycolipids,1 have shown the most promise as Mincle-mediated antagonists.31
Results and discussion
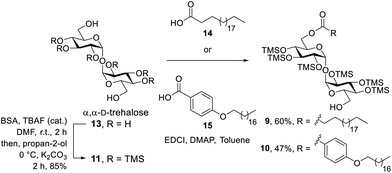
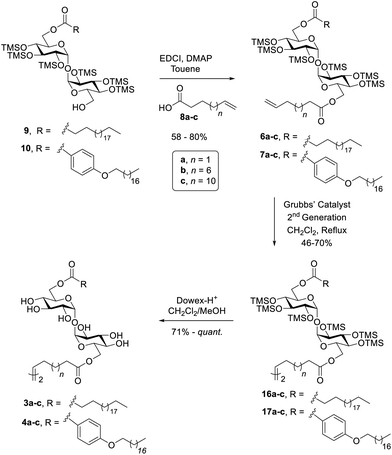
To prepare the target compounds, a retrosynthetic strategy was proposed whereby dimers 3a–c and 4a–c could be accessed via a metathesis reaction of protected asymmetric diesters (6a–c and 7a–c) with alkene functionalities at the end of one lipid tail, followed by desilylation using Dowex-H+ (Scheme 1). The synthesis of the TMS-protected asymmetric diesters 6a–c and 7a–c could in turn be achieved via an EDCI/DMAP mediated coupling between the commercially available alkene functionalised carboxylic acids 8a–c and the corresponding monoesters 9 or 10. Behenic acid monoester 9 was prepared from readily accessible TMS-protected trehalose 11 by controlling the reaction conditions to obtain the mono-esterified product, as previously described.32 A similar methodology was proposed for the synthesis of benzoyl monoester 10. The preparation of the constrained glycolipid 5 was envisaged to proceed under RCM conditions employing the TMS-protected diester of undecenoic acid 12 followed by global deprotection, with undecenoic acid diester 12 being prepared via an esterification reaction between TMS-protected trehalose 11 and undecenoic acid (8b). | ||
| Scheme 1 Retrosynthetic analysis for the synthesis of dimeric glycolipids, 3a–c, and 4a–c, and ring-closed product 5. | ||
With the retrosynthetic strategy in place, α,α′-D-trehalose (13) was persilyated using bis(trimethylsilyl)acetamide (BSA) and catalytic tetrabutylammonium fluoride (TBAF), with the more labile primary TMS groups subsequently being removed by the addition of K2CO3 to afford the required TMS-product 11 according to literature procedures32 (Scheme 2). By carefully controlling the ratio of behenic acid (14), TMS-trehalose 11, EDCI and DMAP (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1.8
1.8![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2
2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4.4, respectively), and by performing the reaction under dilute conditions (30–40 mL dry toluene per mmol of 11), monoester 9 was synthesised in 60% yield, which was an improvement on the previously reported literature yield.32 In an analogous manner, benzoyl monoester 10 was synthesised in 47% yield from TMS-protected trehalose 11 and p-(octadecyloxy)benzoic acid (15), whereby the latter was prepared according to our previously published protocols.14
4.4, respectively), and by performing the reaction under dilute conditions (30–40 mL dry toluene per mmol of 11), monoester 9 was synthesised in 60% yield, which was an improvement on the previously reported literature yield.32 In an analogous manner, benzoyl monoester 10 was synthesised in 47% yield from TMS-protected trehalose 11 and p-(octadecyloxy)benzoic acid (15), whereby the latter was prepared according to our previously published protocols.14
With the target monoesters 9 and 10 in hand, these were then conjugated with the alkene-functionalised carboxylic acids 8a–c (where n = 1, 6, and 10) under the mediation of EDCI and DMAP to give the asymmetric diesters 6a–c and 7a–c in 60–80% yield (Scheme 3). The asymmetric diesters were treated with Grubb's second-generation catalyst in CH2Cl2 under reflux to form the cross-coupled dimeric ligands. This led to TMS-protected dimers 16a–c and 17a–c in 46–72% yield. Evidence for the desired dimerisation was observed in the 1H NMR spectra by the presence of vinylic proton resonances at ca. δ 5.30–5.40 ppm and the absence of proton resonances at ca. δ 5.80 and 5.03–4.89 ppm, which were assigned to terminal alkene protons in the starting diesters. In addition, HRMS data corresponding to the dimeric products (e.g., m/z 1327.8482 for 16c) matched the calculated values (m/z 1327.8489 for [C132H272O26Si12 + 2Na]2+). The protected dimers 16a–c and 17a–c were then subjected to desilylation reactions using Dowex-H+ to generate the final target glycolipids 3a–c and 4a–c in good to excellent yields.
To synthesise the constrained trehalose glycolipid 5, TMS-protected trehalose 11 was reacted with undecenoic acid 8b in toluene in the presence of EDCI and DMAP to produce diester 18 in 86% yield (Scheme 4). Diester 18 was then subjected to RCM using Grubbs’ second-generation catalyst. This led to the cyclic glycolipid 19 in 28% yield. Use of Hoveyda–Grubbs’ second generation catalyst did not improve the outcome, with the desired ring-closed product being isolated in only 11%. This low yield observed for the olefin RCM was proposed to be, in part, due to the loss of TMS-protecting groups from the starting material/product, as observed by TLC, whereby multiple products with lower Rf values were observed, and by HRMS data consistent with partially deprotected by-products. We also speculated that intermolecular reactions between two or more diester units occurred, which additionally reduced the yield of the target product. Nevertheless, as protected glycolipid 19 was obtained in sufficient yield, it was subjected to desilylation using Dowex-H+ to generate the constrained glycolipid 5 in quantitative yield.
To establish whether the target ligands could signal via Mincle, nuclear factor activated T cells (NFAT)–green fluorescent protein (GFP) reporter cells expressing mMincle and coupled to FcRγ were stimulated with the synthesised dimers (3a–c, 4a–c), cyclic ligand (5), as well as TDB (1) and C18Brar (2), and the production of GFP was measured via flow cytometry (Fig. 3). All dimeric compounds (3a–c, 4a–c) activated mMincle at both 0.1 and 1 nmol per well concentrations, while the cyclic TDB ligand 5 showed a modest ability to signal through Mincle at lower ligand concentrations and exhibited similar activity to TDB (2) when tested at 1 nm per well. For the dimeric ligands, the effect of concentration on reporter cell activation was modest, although a slight trend towards increasing GFP production with increasing linker length was observed.
Having demonstrated that the ligands can signal through mMincle, we then tested the ability of compounds to activate both wild-type and Mincle−/− bone marrow-derived macrophages (BMDMs), as determined by measuring the release of the proinflammatory cytokines IL-1β, IL-6 and TNF-α (Fig. 4). From these studies, it was observed that all dimers induced the Mincle-dependent production of IL-1β, except dimer 4b for which Mincle-independent IL-1β production was also observed (Fig. 4A and B). We previously observed a Mincle-independent increase in IL-1β for a C18Brar derivative with a C18 alkyl chain at the ortho-, rather than the para-position, and determined that this was due to caspase-1 dependent NLRP3 inflammasome-mediated cell death.7,33 It remains to be seen whether a similar Mincle-independent mechanism of action occurs for 4b. All dimeric ligands also led to a significant increase in IL-6 and TNF-α production by the BMDMs in a Mincle-dependent manner (Fig. 4A), with dimer 3b (n = 6) leading to TNF-α production by the BMDMs that was significantly greater than that elicited by the ‘gold-standard’ Mincle agonist, TDB. Apart from very modest levels of IL-6, the cyclic compound 5 did not lead to cytokine production in these assays. As with the NFAT-GFP assays, there was no consistent effect of concentration on the cellular responses. This phenomenon has been observed for several promising Mincle-mediated adjuvants6,7,10,11,34 and is poorly understood, although it may be due to immunological feedback mechanisms25,35 or, perhaps more likely, the complex way in which the ligands aggregate in solution.1,26 In the BMDM assays, there was no correlation between linker length and cytokine production.
The ability of the C10-linked TDB dimer 3b to induce good cytokine production in vitro was promising, particularly since IL-6 and IL-1β are thought to be important for the activation of Th-1 immunity,36–38 while TNF-α has been shown to play a critical role in dendritic cell (DC) maturation36,37,39 and in host protection against a variety of infectious diseases, including those caused by viruses (e.g., foot-and-mouth disease37) and intracellular (e.g., Mycobacterium tuberculosis)40 and extracellular (e.g., Klebsiella pneumoniae)41 bacteria, and can activate macrophages to phagocytose and kill mycobacteria and other pathogens.42 To this end, we were interested in exploring the adjuvant potential of the C10-linked TDB dimer 3b using OVA (which contains the immunogenic peptide sequence SIINFEKL as a model antigen).
To this end, C57BL/6 mice (n = 5/group) were vaccinated using a routine vaccination schedule.43 This involved intramuscular (i.m.) vaccination on day 0 with an oil-in-water emulsion containing the C10-linked TDB dimer 3b + OVA, TDB + OVA, PBS (negative control) or OVA only (negative control) and an i.m. boost 3 weeks later and, after a further 7 days, the mice were sacrificed and the inguinal lymph node (iLN) was harvested and B and T cell populations were analysed by flow cytometry (Fig. 5A–F) (see the SI for the flow gating strategies). Following this vaccination protocol, it was observed that only 3b induced significantly more SIINFEKL+CD8+T cells compared to PBS (Fig. 5D), and significantly more OVA+ germinal centre B cells compared to OVA alone and TDB + OVA (Fig. 5F). This illustrates the potential of 3b to expand B cell populations and augment the immune response. However, no significant increase in the T follicular CD4+ helper cells was observed in response to either TDB or 3b (Fig. 3B), which is an immune response that is essential for supporting germinal centre (GC) B cells activation, and survival within GC, as well as for the production of high-affinity antigen-specific antibodies and isotype switching.44 Although TDB is a potent Mincle agonist,1,2,4 it exhibited only modest adjuvant activity in this assay. The adjuvant activity of 3b could perhaps be improved by ensuring better co-delivery of the antigen and multiple copies of the adjuvant to the same antigen presenting cell (APC) either through adsorption of both antigen and adjuvant to a particulate delivery vehicle or via covalent conjugation of the antigen to the adjuvant.1,12 Such studies will be explored in due course.
Conclusions
In this study, the first dimeric Mincle ligands were synthesised and shown to signal and activate immune cells via Mincle. Notably, the dimeric ligand 3b, which contains two units of TDB linked by a C10 acyl group, led to IL-1β, IL-6 and TNF-α production, with the level of TNF-α being significantly greater than that shown by TDB alone. The potential of dimer 3b as a vaccine adjuvant was further demonstrated in an in vivo i.m. vaccination protocol using OVA as the model antigen. Here, 3b led to a significant increase in OVA-specific CD8+ T cells and GC B cells, with the B cell response to 3b being greater than that shown by TDB. Unfortunately, neither TDB nor dimer 3b led to increased CD4+ T cell numbers when formulated as the o.i.w. emulsion. This may be due to poor co-delivery of antigen and adjuvant to the same APC, which could be explored in future vaccination studies by using particulate carrier systems that can adsorb both antigen and adjuvant or via the chemical conjugation of antigen to adjuvant. In summary, we provide the first example to illustrate how two Mincle ligands can be conjugated to provide Mincle-mediated agonist activity, thereby highlighting a potential new approach for the development of improved Mincle-mediated adjuvants.Experimental
General methods
Unless otherwise stated, all reactions were performed under an argon atmosphere. Methanol, EtOAc, and light petroleum ether (pet. ether) were distilled prior to use. Dichloromethane was distilled over phosphorous pentoxide, under an Ar atmosphere. All other chemicals were purchased from commercial suppliers and used as received. All solvents were evaporated under reduced pressure. Reactions were monitored by TLC-analysis using Macherey-Nagel silica gel pre-coated plastic sheets (0.20 mm, fluorescent indicator UV-254) and visualised under UV light (254 nm) or by dipping in 10% H2SO4 in EtOH followed by charring, dipping in ceric ammonium molybdate solution or dipping in KMnO4 solution (2% in H2O). Column chromatography was performed using silica gel (40–63 µm). High resolution mass spectroscopy data were obtained on an Agilent 6530 Q-TOF mass spectrometer with a JetStream™ electrospray ionisation source in positive or negative mode. Optical rotations were recorded on an Autopol II polarimeter (Rudolph Research Analytical) at 589 nm (sodium D-line). Infrared spectra were obtained for the compounds (as a thin film) using a Bruker Platinum ATR instrument and are reported in wave numbers (cm−1). Nuclear magnetic resonance spectra were recorded at 20 °C in CDCl3, C5D5N or D2O as indicated, using JEOL JNM-ECZ spectrometers operating at 500 or 600 MHz. Chemical shifts (δ) are reported in ppm relative to residual solvent peaks. Peak assignments in NMR spectra were made using 2D-NMR experiments (COSY, HSQC, and HMBC). TDB (1),45 C18Brar (2),46 2,2′,3,3′,4,4′-hexa-O-trimethylsislyl-α,α′-D-trehalose (11)45 and p-(octadecyloxy)benzoic acid (15)46 were prepared according to previously published procedures.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) EtOAc, 1
EtOAc, 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0 → 19
0 → 19![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, v/v) to obtain behenic acid monoester 9 as a colourless viscous oil (620 mg, 0.320 mmol, 60%). The data obtained for this compound matched literature values.47
1, v/v) to obtain behenic acid monoester 9 as a colourless viscous oil (620 mg, 0.320 mmol, 60%). The data obtained for this compound matched literature values.47
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) EtOAc, 1
EtOAc, 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0 → 47
0 → 47![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3, v/v) to obtain benzoic acid monoester 10 as a colourless viscous oil (347 mg, 0.302 mmol, 47%). Rf = 0.63 (pet. ether
3, v/v) to obtain benzoic acid monoester 10 as a colourless viscous oil (347 mg, 0.302 mmol, 47%). Rf = 0.63 (pet. ether![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) EtOAc, 17
EtOAc, 17![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3, v/v); [α]22.9D = +80 (c = 0.1, CH2Cl2); 1H NMR (500 MHz, CDCl3) δ 7.98 (d, J3″,4″ = 9.0 Hz, 2H, H-3″), 6.90 (d, J3″,4″ = 9.0 Hz, 2H, H-4″), 4.93 (d, J1/1′,2/2′ = 3.1 Hz, 2H, H-1 and H-1′), 4.54 (dd, J6a,b = 12.0 Hz, J5,6a 2.4 Hz, 1H, H-6a), 4.24 (dd, J6a,b = 12.0 Hz, J5,6b 2.4 Hz, 1H H-6b), 4.13–4.07 (m, 1H, H-5), 3.99 (t, J6″,7″ = 6.6 Hz, 2H, H-6″), 3.96–3.87 (m, 2H, H-3 and H-3′), 3.83 (dt, J4′,5′ = 3.4 Hz, J5′,6′a/b = 9.5 Hz, 1H, H-5′), 3.73–3.65 (m, 2H, H-6′), 3.63 (t, J3,4 = J4,5 = 9.1 Hz, 1H, H-4), 3.49–3.40 (m, 3H, H-2 and H-2′ and H-4′), 1.82–1.70 (m, 2H, H-7″), 1.48–1.39 (m, 2H, H-8″), 1.38–1.18 (m, 26H, H-9″–H-22″), 0.86 (t, J22″,23″ = 7.0 Hz, H-23″), 0.16, 0.15, 0.14, 0.13, 0.12, 0.11 (6s, 54H, TMS); 13C NMR (125 MHz, CDCl3) δ 166.4 (C-1″), 163.2 (C-5″), 131.8 (C-3″), 122.2 (C-2″), 114.2 (C-4″), 94.7, 94.6 (C-1 and C-1′), 73.8 (C-3), 73.4 (C-3′), 73.0 (C-5′), 72.9, 72.8 (C-2 and C-2′), 72.0 (C-4′), 71.5 (C-4), 71.0 (C-5), 68.3 (C-6″), 63.4 (C-6), 61.7 (C-6′), 32.0, 29.78, 29.75, 29.69, 29.66, 29.47, 29.45, 29.2 (C-9″–C-21″), 29.1 (C-7″), 26.1 (C-8″), 22.7 (C-22″), 14.3 (C-23′), 1.2, 1.1, 1.0, 0.9, 0.3, 0.2 (TMS); IR (film): 2955, 2924, 2854, 2091, 2052, 1988, 1719, 1607, 1581, 1510, 1458, 1387, 1313, 1251, 1167, 1109, 1076, 1009, 965, 944, 899, 972, 843, 749, 694, 646, 573, 514, 449, 421, 412 cm−1; HRMS (ESI) m/z calcd for [C55H110O13Si6 + K]+: 1185.6194; obsd.: 1185.6191.
3, v/v); [α]22.9D = +80 (c = 0.1, CH2Cl2); 1H NMR (500 MHz, CDCl3) δ 7.98 (d, J3″,4″ = 9.0 Hz, 2H, H-3″), 6.90 (d, J3″,4″ = 9.0 Hz, 2H, H-4″), 4.93 (d, J1/1′,2/2′ = 3.1 Hz, 2H, H-1 and H-1′), 4.54 (dd, J6a,b = 12.0 Hz, J5,6a 2.4 Hz, 1H, H-6a), 4.24 (dd, J6a,b = 12.0 Hz, J5,6b 2.4 Hz, 1H H-6b), 4.13–4.07 (m, 1H, H-5), 3.99 (t, J6″,7″ = 6.6 Hz, 2H, H-6″), 3.96–3.87 (m, 2H, H-3 and H-3′), 3.83 (dt, J4′,5′ = 3.4 Hz, J5′,6′a/b = 9.5 Hz, 1H, H-5′), 3.73–3.65 (m, 2H, H-6′), 3.63 (t, J3,4 = J4,5 = 9.1 Hz, 1H, H-4), 3.49–3.40 (m, 3H, H-2 and H-2′ and H-4′), 1.82–1.70 (m, 2H, H-7″), 1.48–1.39 (m, 2H, H-8″), 1.38–1.18 (m, 26H, H-9″–H-22″), 0.86 (t, J22″,23″ = 7.0 Hz, H-23″), 0.16, 0.15, 0.14, 0.13, 0.12, 0.11 (6s, 54H, TMS); 13C NMR (125 MHz, CDCl3) δ 166.4 (C-1″), 163.2 (C-5″), 131.8 (C-3″), 122.2 (C-2″), 114.2 (C-4″), 94.7, 94.6 (C-1 and C-1′), 73.8 (C-3), 73.4 (C-3′), 73.0 (C-5′), 72.9, 72.8 (C-2 and C-2′), 72.0 (C-4′), 71.5 (C-4), 71.0 (C-5), 68.3 (C-6″), 63.4 (C-6), 61.7 (C-6′), 32.0, 29.78, 29.75, 29.69, 29.66, 29.47, 29.45, 29.2 (C-9″–C-21″), 29.1 (C-7″), 26.1 (C-8″), 22.7 (C-22″), 14.3 (C-23′), 1.2, 1.1, 1.0, 0.9, 0.3, 0.2 (TMS); IR (film): 2955, 2924, 2854, 2091, 2052, 1988, 1719, 1607, 1581, 1510, 1458, 1387, 1313, 1251, 1167, 1109, 1076, 1009, 965, 944, 899, 972, 843, 749, 694, 646, 573, 514, 449, 421, 412 cm−1; HRMS (ESI) m/z calcd for [C55H110O13Si6 + K]+: 1185.6194; obsd.: 1185.6191.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) EtOAc, 1
EtOAc, 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0 → 24
0 → 24![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, v/v) to obtain the title compound 6a as a colourless waxy oil (67.4 mg, 0.056 mmol, 61%). Rf = 0.66 (pet. ether
1, v/v) to obtain the title compound 6a as a colourless waxy oil (67.4 mg, 0.056 mmol, 61%). Rf = 0.66 (pet. ether![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) EtOAc, 9
EtOAc, 9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, v/v); [α]22.7D = +100 (c = 0.1, CH2Cl2); 1H NMR (500 MHz, CDCl3) δ 5.77 (ddt, J5‴,6‴a = 17.0 Hz, J5‴,6‴b = 10.2 Hz, J4‴,5‴ = 6.7 Hz, 1H, H-5‴), 5.02 (dq, J5‴,6‴a = 17.0 Hz, J6‴a,6‴b = J6‴a,4‴ = 1.6 Hz, 1H, H-6‴a), 4.98 (ddt, J5‴,6‴b = 10.2 Hz, J6‴a,6‴b = 1.6 Hz, J6‴b,4‴ = 1.1 Hz, 2H, H-6‴b), 4.91 (d, J1/1′,2/2′ = 3.0 Hz, 2H, H-1 and H-1′), 4.29–4.26 (m, 2H, H-6a and H-6′a), 4.08–4.04 (m, 2H, H-6b and H-6′b), 3.99 (ddd, J4/4′,5/5′ = 9.1 Hz, J5/5′,6/6′b = 4.4 Hz, J5/5′,6/6′a = 2.3 Hz, 2H, H-5 and H-5′), 3.90 (t, J3/3′,4/4′ = J2/2′,3/3′ = 9.1 Hz, 2H, H-3 and H-3′), 3.48 (t, J3/3′,4/4′ = J4/4′,5/5′ = 9.1 Hz, 2H, H-4 and H-4′), 3.43 (dd, J2/2′,3/3′ = 9.1 Hz, J1/1′,2/2′ = 3.0 Hz, 2H, H-2 and H-2′), 2.41–2.28 (m, 4H, H-2″ and H-2‴), 2.12–2.07 (m, 2H, H-4‴), 1.77–1.69 (m, 2H, H-3‴), 1.65–1.58 (m, 2H, H-3″), 1.33–1.22 (m, 36H, H-4″–H-21″), 0.88 (t, J21″,22″ = 7.0 Hz, 3H, H-22″), 0.15, 0.13, 0.13 (3s, 54H, TMS); 13C NMR (125 MHz, CDCl3) δ 173.9 (C-1″/C-1‴), 173.7 (C-1″/C-1‴), 137.8 (C-5‴), 115.5 (C-6‴), 94.5 (C-1 and C-1′), 73.62 (C-3/C-3′), 73.60 (C-3/C-3′), 72.8 (C-2 and C-2′), 72.0 (C-4 and C-4′), 70.9 (C-5 and C-5′), 63.5 (C-6/C-6′), 63.4 (C-6/C-6′), 34.3 (C-2″/C-2‴), 33.5 (C-2″/C-2‴), 33.1 (C-4‴), 32.1, 29.85, 29.81, 29.78, 29.63, 29.51, 29.46, 29.31, 22.8 (C-4″–C-21″), 24.9 (C-3″), 24.0 (C-3‴), 14.3 (C-22″), 1.2, 1.0, 0.3 (TMS); IR (film): 2955, 2924, 2854, 1743, 1458, 1251, 1164, 1111, 1100, 1077, 1045, 1010, 965, 898, 873 cm−1; HRMS (ESI) m/z calcd for [C58H120O13Si6 + NH4]+: 1210.7683; obsd.: 1210.7685.
1, v/v); [α]22.7D = +100 (c = 0.1, CH2Cl2); 1H NMR (500 MHz, CDCl3) δ 5.77 (ddt, J5‴,6‴a = 17.0 Hz, J5‴,6‴b = 10.2 Hz, J4‴,5‴ = 6.7 Hz, 1H, H-5‴), 5.02 (dq, J5‴,6‴a = 17.0 Hz, J6‴a,6‴b = J6‴a,4‴ = 1.6 Hz, 1H, H-6‴a), 4.98 (ddt, J5‴,6‴b = 10.2 Hz, J6‴a,6‴b = 1.6 Hz, J6‴b,4‴ = 1.1 Hz, 2H, H-6‴b), 4.91 (d, J1/1′,2/2′ = 3.0 Hz, 2H, H-1 and H-1′), 4.29–4.26 (m, 2H, H-6a and H-6′a), 4.08–4.04 (m, 2H, H-6b and H-6′b), 3.99 (ddd, J4/4′,5/5′ = 9.1 Hz, J5/5′,6/6′b = 4.4 Hz, J5/5′,6/6′a = 2.3 Hz, 2H, H-5 and H-5′), 3.90 (t, J3/3′,4/4′ = J2/2′,3/3′ = 9.1 Hz, 2H, H-3 and H-3′), 3.48 (t, J3/3′,4/4′ = J4/4′,5/5′ = 9.1 Hz, 2H, H-4 and H-4′), 3.43 (dd, J2/2′,3/3′ = 9.1 Hz, J1/1′,2/2′ = 3.0 Hz, 2H, H-2 and H-2′), 2.41–2.28 (m, 4H, H-2″ and H-2‴), 2.12–2.07 (m, 2H, H-4‴), 1.77–1.69 (m, 2H, H-3‴), 1.65–1.58 (m, 2H, H-3″), 1.33–1.22 (m, 36H, H-4″–H-21″), 0.88 (t, J21″,22″ = 7.0 Hz, 3H, H-22″), 0.15, 0.13, 0.13 (3s, 54H, TMS); 13C NMR (125 MHz, CDCl3) δ 173.9 (C-1″/C-1‴), 173.7 (C-1″/C-1‴), 137.8 (C-5‴), 115.5 (C-6‴), 94.5 (C-1 and C-1′), 73.62 (C-3/C-3′), 73.60 (C-3/C-3′), 72.8 (C-2 and C-2′), 72.0 (C-4 and C-4′), 70.9 (C-5 and C-5′), 63.5 (C-6/C-6′), 63.4 (C-6/C-6′), 34.3 (C-2″/C-2‴), 33.5 (C-2″/C-2‴), 33.1 (C-4‴), 32.1, 29.85, 29.81, 29.78, 29.63, 29.51, 29.46, 29.31, 22.8 (C-4″–C-21″), 24.9 (C-3″), 24.0 (C-3‴), 14.3 (C-22″), 1.2, 1.0, 0.3 (TMS); IR (film): 2955, 2924, 2854, 1743, 1458, 1251, 1164, 1111, 1100, 1077, 1045, 1010, 965, 898, 873 cm−1; HRMS (ESI) m/z calcd for [C58H120O13Si6 + NH4]+: 1210.7683; obsd.: 1210.7685.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) EtOAc, 1
EtOAc, 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0 → 97
0 → 97![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3, v/v) to obtain the title compound 6b as a colourless waxy oil (64.8 mg, 0.051 mmol, 60%). Rf = 0.65 (pet. ether
3, v/v) to obtain the title compound 6b as a colourless waxy oil (64.8 mg, 0.051 mmol, 60%). Rf = 0.65 (pet. ether![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) EtOAc, 9
EtOAc, 9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, v/v); [α]22.8D = +80 (c = 0.1, CH2Cl2); 1H NMR (600 MHz, CDCl3) δ 5.80 (ddt, J10‴,11‴a = 16.9 Hz, J10‴,11‴b = 10.1 Hz, J10‴,9‴ = 6.7 Hz, 1H, H-10‴), 5.01–4.91 (m, 4H, H-1, H-1′, H-11‴a and H-11‴b), 4.27 (dd, J6/6′a,b = 11.9 Hz, J5/5′,6/6′a = 2.3 Hz, 2H, H-6a and H-6′a), 4.05 (dd, J6/6′a,b = 11.9 Hz, J5/5′,6/6′b = 4.4 Hz, 2H, H-6b and H-6′b), 4.00 (ddd, J4/4′,5/5′ = 9.2 Hz, J5/5′,6/6′b = 4.4 Hz, J5/5′,6/6′a = 2.3 Hz, 2H, H-5 and H-5′), 3.90 (t, J2/2′,3/3′ = J3/3′,4/4′ = 9.2 Hz, 2H, H-3 and H-3′), 3.48 (t, J3/3′,4/4′ = J4/4′,5/5′ = 9.2 Hz, 2H, H-4 and H-4′), 3.43 (dd, J2/2′,3/3′ = 9.2 Hz, J1/1′,2/2′ = 3.1 Hz, 2H, H-2 and H-2′), 2.38–2.29 (m, 4H, H-2″ and H-2‴), 2.05–2.01 (m, 2H, H-9‴), 1.65–1.59 (m, 4H, H-3″ and H-3‴), 1.38–1.25 (m, 46H, H-4″–H-21″ and H-4‴–H-8‴), 0.88 (t, J21″,22″ = 7.0 Hz, 3H, H-22″), 0.15, 0.13, 0.13 (3s, 54H, TMS); 13C NMR (150 MHz, CDCl3) δ 173.9 (C-1″ and C-1‴), 139.3 (C-10‴), 114.3 (C-11‴), 94.5 (C-1 and C-1′), 73.6 (C-3 and C-3′), 72.8 (C-2 and C-2′), 72.1 (C-4 and C-4′), 70.9 (C-5 and C-5′), 63.5 (C-6 and C-6′), 34.3 (C-2″ and C-2‴), 33.9, 32.1, 29.9, 29.6, 29.51, 29.45, 29.38, 29.31, 29.27, 29.21, 29.0, 22.8 (C-4″–C21″ and C-4‴–C-8‴), 24.9 (C-3″ and C-3‴), 14.3 (C-22″), 1.2, 1.0, 0.3 (TMS); IR (film): 2970, 2924, 2854, 1742, 1630, 1459, 1369, 1250, 1164, 1110, 1100, 1076, 1009, 898, 871, 840 cm−1; HRMS (ESI) m/z calcd for [C63H130O13Si6 + NH4]+: 1280.8465; obsd.: 1280.8473.
1, v/v); [α]22.8D = +80 (c = 0.1, CH2Cl2); 1H NMR (600 MHz, CDCl3) δ 5.80 (ddt, J10‴,11‴a = 16.9 Hz, J10‴,11‴b = 10.1 Hz, J10‴,9‴ = 6.7 Hz, 1H, H-10‴), 5.01–4.91 (m, 4H, H-1, H-1′, H-11‴a and H-11‴b), 4.27 (dd, J6/6′a,b = 11.9 Hz, J5/5′,6/6′a = 2.3 Hz, 2H, H-6a and H-6′a), 4.05 (dd, J6/6′a,b = 11.9 Hz, J5/5′,6/6′b = 4.4 Hz, 2H, H-6b and H-6′b), 4.00 (ddd, J4/4′,5/5′ = 9.2 Hz, J5/5′,6/6′b = 4.4 Hz, J5/5′,6/6′a = 2.3 Hz, 2H, H-5 and H-5′), 3.90 (t, J2/2′,3/3′ = J3/3′,4/4′ = 9.2 Hz, 2H, H-3 and H-3′), 3.48 (t, J3/3′,4/4′ = J4/4′,5/5′ = 9.2 Hz, 2H, H-4 and H-4′), 3.43 (dd, J2/2′,3/3′ = 9.2 Hz, J1/1′,2/2′ = 3.1 Hz, 2H, H-2 and H-2′), 2.38–2.29 (m, 4H, H-2″ and H-2‴), 2.05–2.01 (m, 2H, H-9‴), 1.65–1.59 (m, 4H, H-3″ and H-3‴), 1.38–1.25 (m, 46H, H-4″–H-21″ and H-4‴–H-8‴), 0.88 (t, J21″,22″ = 7.0 Hz, 3H, H-22″), 0.15, 0.13, 0.13 (3s, 54H, TMS); 13C NMR (150 MHz, CDCl3) δ 173.9 (C-1″ and C-1‴), 139.3 (C-10‴), 114.3 (C-11‴), 94.5 (C-1 and C-1′), 73.6 (C-3 and C-3′), 72.8 (C-2 and C-2′), 72.1 (C-4 and C-4′), 70.9 (C-5 and C-5′), 63.5 (C-6 and C-6′), 34.3 (C-2″ and C-2‴), 33.9, 32.1, 29.9, 29.6, 29.51, 29.45, 29.38, 29.31, 29.27, 29.21, 29.0, 22.8 (C-4″–C21″ and C-4‴–C-8‴), 24.9 (C-3″ and C-3‴), 14.3 (C-22″), 1.2, 1.0, 0.3 (TMS); IR (film): 2970, 2924, 2854, 1742, 1630, 1459, 1369, 1250, 1164, 1110, 1100, 1076, 1009, 898, 871, 840 cm−1; HRMS (ESI) m/z calcd for [C63H130O13Si6 + NH4]+: 1280.8465; obsd.: 1280.8473.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) EtOAc, 1
EtOAc, 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0 → 97
0 → 97![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3, v/v) to obtain the title compound 6c as a colourless waxy oil (17.5 mg, 0.013 mmol, 58%). Rf = 0.47 (pet. ether
3, v/v) to obtain the title compound 6c as a colourless waxy oil (17.5 mg, 0.013 mmol, 58%). Rf = 0.47 (pet. ether![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) EtOAc, 9
EtOAc, 9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, v/v); [α]23.4D = +60 (c = 0.1, CH2Cl2); 1H NMR (500 MHz, CDCl3) δ 5.80 (ddt, J14‴,15‴a = 16.9, J14‴,15‴b = 10.2, J13‴,14‴ = 6.7 Hz, 1H, H-14‴), 5.01–4.89 (m, 4H, H-1 and H-1′ and H-15‴a and H-15‴), 4.26 (dd, J6/6′a,b = 11.8, J5/5′,6/6′a = 2.3 Hz, 2H, H-6a and H-6′a), 4.04 (dd, J6/6′a,b = 11.9, J5/5′,6/6′b = 4.4 Hz, 2H, H-6b and H-6′b), 4.01–3.96 (m, 2H, H-5 and H-5′), 3.89 (t, J2/2′,3/3′ = J3/3′,4/4′ = 8.9 Hz, 2H, H-3 and H-3′), 3.47 (t, J3/3′,4/4′ = J4/4′,5/5′ = 9.0 Hz, 2H, H-4 and H-4′), 3.43 (dd, J2/2′,3/3′ = 9.3, J3/3′,4/4′ = 3.1 Hz, 2H, H-2 and H-2′), 2.39–2.26 (m, 4H, H-2″ and 2‴), 2.02 (q, J12‴,13‴ = J13‴,14‴ = 6.9 Hz, 2H, H-13‴), 1.68–1.57 (m, 4H, H-3″ and H-3‴), 1.43–1.33 (m, 2H, H-12‴), 1.33–1.15 (m, 52H, H-4″–H-21″ and H-4‴–H-11‴), 0.87 (t, J21″,22″ = 6.8 Hz, 3H, H-22″), 0.14, 0.12, 0.12 (3s, TMS, 54H); 13C NMR (125 MHz, CDCl3) δ 173.9 (C-1″ and C-1‴), 139.4 (C-14‴), 114.2 (C-15‴), 94.5 (C-1 and C-1′), 73.6 (C-3 and C-3′), 72.7 (C-2 and C-2′), 72.0 (C-4 and C-4′), 70.8 (H-5 and H-5′), 63.4 (C-6 and C-6′), 34.2 (C-2″ and C-2‴), 33.9 (H-13‴), 32.0 (C-20″), 29.8, 29.8, 29.7, 29.7, 29.6, 29.6, 29.5, 29.4, 29.3, 29.0, 22.8, (C-4″–C-19″ and C-4‴–C-12‴), 24.9 (C-3″ and C-3‴), 14.2 (C-22″), 1.2, 1.0, 0.3 (TMS); IR (film): 2924, 2854, 1743, 1459, 1251, 1165, 1111, 1077, 1046, 1011, 966, 931, 898, 873, 843, 749 cm−1; HRMS (ESI) m/z calcd for [C67H138O13Si6 + NH4]+: 1336.9091; obsd.: 1336.9094.
1, v/v); [α]23.4D = +60 (c = 0.1, CH2Cl2); 1H NMR (500 MHz, CDCl3) δ 5.80 (ddt, J14‴,15‴a = 16.9, J14‴,15‴b = 10.2, J13‴,14‴ = 6.7 Hz, 1H, H-14‴), 5.01–4.89 (m, 4H, H-1 and H-1′ and H-15‴a and H-15‴), 4.26 (dd, J6/6′a,b = 11.8, J5/5′,6/6′a = 2.3 Hz, 2H, H-6a and H-6′a), 4.04 (dd, J6/6′a,b = 11.9, J5/5′,6/6′b = 4.4 Hz, 2H, H-6b and H-6′b), 4.01–3.96 (m, 2H, H-5 and H-5′), 3.89 (t, J2/2′,3/3′ = J3/3′,4/4′ = 8.9 Hz, 2H, H-3 and H-3′), 3.47 (t, J3/3′,4/4′ = J4/4′,5/5′ = 9.0 Hz, 2H, H-4 and H-4′), 3.43 (dd, J2/2′,3/3′ = 9.3, J3/3′,4/4′ = 3.1 Hz, 2H, H-2 and H-2′), 2.39–2.26 (m, 4H, H-2″ and 2‴), 2.02 (q, J12‴,13‴ = J13‴,14‴ = 6.9 Hz, 2H, H-13‴), 1.68–1.57 (m, 4H, H-3″ and H-3‴), 1.43–1.33 (m, 2H, H-12‴), 1.33–1.15 (m, 52H, H-4″–H-21″ and H-4‴–H-11‴), 0.87 (t, J21″,22″ = 6.8 Hz, 3H, H-22″), 0.14, 0.12, 0.12 (3s, TMS, 54H); 13C NMR (125 MHz, CDCl3) δ 173.9 (C-1″ and C-1‴), 139.4 (C-14‴), 114.2 (C-15‴), 94.5 (C-1 and C-1′), 73.6 (C-3 and C-3′), 72.7 (C-2 and C-2′), 72.0 (C-4 and C-4′), 70.8 (H-5 and H-5′), 63.4 (C-6 and C-6′), 34.2 (C-2″ and C-2‴), 33.9 (H-13‴), 32.0 (C-20″), 29.8, 29.8, 29.7, 29.7, 29.6, 29.6, 29.5, 29.4, 29.3, 29.0, 22.8, (C-4″–C-19″ and C-4‴–C-12‴), 24.9 (C-3″ and C-3‴), 14.2 (C-22″), 1.2, 1.0, 0.3 (TMS); IR (film): 2924, 2854, 1743, 1459, 1251, 1165, 1111, 1077, 1046, 1011, 966, 931, 898, 873, 843, 749 cm−1; HRMS (ESI) m/z calcd for [C67H138O13Si6 + NH4]+: 1336.9091; obsd.: 1336.9094.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) EtOAc, 1
EtOAc, 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0 → 97
0 → 97![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3, v/v) to obtain the title compound 7a as a colourless waxy oil (113.3 mg, 0.091 mmol, 56%). Rf = 0.68 (pet. ether
3, v/v) to obtain the title compound 7a as a colourless waxy oil (113.3 mg, 0.091 mmol, 56%). Rf = 0.68 (pet. ether![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) EtOAc, 10
EtOAc, 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, v/v); [α]23.5D = +60 (c = 0.1, CH2Cl2); 1H NMR (500 MHz, CDCl3) δ 7.98 (d, J3″,4″ = 8.9 Hz, 2H, H-3″), 6.90 (d, J3″,4″ = 8.9 Hz, 2H, H-4″), 5.76 (ddt, J5‴,6‴a/b = 16.9, J5‴,6‴a/b = 10.2, J4‴,5‴ = 6.7 Hz, 1H, H-5‴), 5.04–4.91 (m, 4H, H-6‴a/b and H-1 and H-1′), 4.52 (dd, J6a,b = 12.0, J5,6a = 2.4 Hz, 1H, H-6a), 4.27 (dd, J6′a,b = 11.9, J5′,6′a = 2.3 Hz, 1H, H-6′a), 4.23 (dd, J6a,b = 12.0, J5,6b = 3.6 Hz, 1H, H-6b), 4.09 (dt, J4′,5′ = 9.6, J5′,6′a/b = 3.0 Hz, 1H), 4.06 (dd, J6′a,b = 11.9, J5′,6′b = 4.5 Hz, 1H, H-6′b), 4.02–3.97 (m, 3H, H-6′ and H-5), 3.94 (t, J3/3′,4/4′ = J2/2′,3/3′ = 8.9 Hz, 1H, H-3 or H-3′), 3.91 (t, J3/3′,4/4′ = J2/2′,3/3′ = 8.9 Hz, 1H, H-3 or H-3′), 3.63 (t, J4′,5′ = J3′,4′ = 8.9 Hz, 1H, H-4′), 3.51–3.41 (m, 3H, H-2 and H-2′ and H-4), 2.42–2.25 (m, 2H, H-2‴), 2.13–2.03 (m, 2H, H-4‴), 1.83–1.75 (m, 2H, H-7″), 1.75–1.67 (m, 2H, H-3‴), 1.49–1.40 (m, 2H, H-8″), 1.31–1.20 (m, 26H, H-9″–H-22″), 0.86 (t, J22″,23″ = 6.9 Hz, 3H, H-23″), 0.16, 0.15, 0.14, 0.12, 0.12, 0.11 (6s, 54H, CH3, TMS); 13C NMR (125 MHz, CDCl3) δ 173.6 (C-1‴), 166.4 (C-1″), 163.2 (C-5″), 137.8 (C-5‴), 131.8 (C-3″), 122.2 (C-2″), 115.5 (C-6‴), 114.2 (C-4″), 94.6 (C-1 and C-1′), 73.8, 73.6 (C-3 and C-3′), 72.79, 72.75 (C-2 and C-2′), 72.0 (C-4 and C-4′), 71.0, 70.8 (C-5 and C-5′), 68.3 (C-6″), 63.5 (C-6 and C-6′), 33.5 (C-2‴), 33.1 (C-4‴), 32.0 (C-21″ or C-22″), 29.8, 29.7, 29.7, 29.5, 29.5 (C-9″–C-20″), 29.2 (C-7″) 26.1 (C-8″), 24.0 (C-3‴), 22.8 (C-21″ or C-22″), 14.2 (C-23″), 1.2, 1.2, 1.0, 1.0, 0.3, 0.3 (TMS); IR (film): 2924, 2853, 1741, 1719, 1606, 1510, 1458, 1389, 1313, 1250, 1165, 1109, 1099, 1076, 1044, 1008, 965, 934, 898, 871, 842, 766, 748, 694, 645, 577, 536, 518, 473, 455, 423, 414 cm−1; HRMS (ESI) calcd for [C61H118O14Si6 + NH4]+: 1260.7475; obsd.: 1260.7472.
1, v/v); [α]23.5D = +60 (c = 0.1, CH2Cl2); 1H NMR (500 MHz, CDCl3) δ 7.98 (d, J3″,4″ = 8.9 Hz, 2H, H-3″), 6.90 (d, J3″,4″ = 8.9 Hz, 2H, H-4″), 5.76 (ddt, J5‴,6‴a/b = 16.9, J5‴,6‴a/b = 10.2, J4‴,5‴ = 6.7 Hz, 1H, H-5‴), 5.04–4.91 (m, 4H, H-6‴a/b and H-1 and H-1′), 4.52 (dd, J6a,b = 12.0, J5,6a = 2.4 Hz, 1H, H-6a), 4.27 (dd, J6′a,b = 11.9, J5′,6′a = 2.3 Hz, 1H, H-6′a), 4.23 (dd, J6a,b = 12.0, J5,6b = 3.6 Hz, 1H, H-6b), 4.09 (dt, J4′,5′ = 9.6, J5′,6′a/b = 3.0 Hz, 1H), 4.06 (dd, J6′a,b = 11.9, J5′,6′b = 4.5 Hz, 1H, H-6′b), 4.02–3.97 (m, 3H, H-6′ and H-5), 3.94 (t, J3/3′,4/4′ = J2/2′,3/3′ = 8.9 Hz, 1H, H-3 or H-3′), 3.91 (t, J3/3′,4/4′ = J2/2′,3/3′ = 8.9 Hz, 1H, H-3 or H-3′), 3.63 (t, J4′,5′ = J3′,4′ = 8.9 Hz, 1H, H-4′), 3.51–3.41 (m, 3H, H-2 and H-2′ and H-4), 2.42–2.25 (m, 2H, H-2‴), 2.13–2.03 (m, 2H, H-4‴), 1.83–1.75 (m, 2H, H-7″), 1.75–1.67 (m, 2H, H-3‴), 1.49–1.40 (m, 2H, H-8″), 1.31–1.20 (m, 26H, H-9″–H-22″), 0.86 (t, J22″,23″ = 6.9 Hz, 3H, H-23″), 0.16, 0.15, 0.14, 0.12, 0.12, 0.11 (6s, 54H, CH3, TMS); 13C NMR (125 MHz, CDCl3) δ 173.6 (C-1‴), 166.4 (C-1″), 163.2 (C-5″), 137.8 (C-5‴), 131.8 (C-3″), 122.2 (C-2″), 115.5 (C-6‴), 114.2 (C-4″), 94.6 (C-1 and C-1′), 73.8, 73.6 (C-3 and C-3′), 72.79, 72.75 (C-2 and C-2′), 72.0 (C-4 and C-4′), 71.0, 70.8 (C-5 and C-5′), 68.3 (C-6″), 63.5 (C-6 and C-6′), 33.5 (C-2‴), 33.1 (C-4‴), 32.0 (C-21″ or C-22″), 29.8, 29.7, 29.7, 29.5, 29.5 (C-9″–C-20″), 29.2 (C-7″) 26.1 (C-8″), 24.0 (C-3‴), 22.8 (C-21″ or C-22″), 14.2 (C-23″), 1.2, 1.2, 1.0, 1.0, 0.3, 0.3 (TMS); IR (film): 2924, 2853, 1741, 1719, 1606, 1510, 1458, 1389, 1313, 1250, 1165, 1109, 1099, 1076, 1044, 1008, 965, 934, 898, 871, 842, 766, 748, 694, 645, 577, 536, 518, 473, 455, 423, 414 cm−1; HRMS (ESI) calcd for [C61H118O14Si6 + NH4]+: 1260.7475; obsd.: 1260.7472.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) EtOAc, 1
EtOAc, 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0 → 49
0 → 49![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, v/v), which gave the title compound 7b as a colourless viscous oil (172 mg, 0.130 mmol, 80%). Rf = 0.68 (pet. ether
1, v/v), which gave the title compound 7b as a colourless viscous oil (172 mg, 0.130 mmol, 80%). Rf = 0.68 (pet. ether![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) EtOAc, 3
EtOAc, 3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 17, v/v); [α]22.0D = +60 (c = 0.1, CH2Cl2); 1H NMR (600 MHz, CDCl3) δ 7.99 (d, J3″,4″ = 8.9 Hz, 2H, H-3″), 6.91 (d, J3″,4″ = 8.9 Hz, 2H, H-4″), 5.80 (ddt, J10‴,11‴a = 16.9 Hz, J10‴,11‴b = 10.2 Hz, J10‴,9‴ = 6.7 Hz, 1H, H-10‴), 5.03–4.89 (m, 4H, H-1, H-1′, H-11‴a and b), 4.53 (dd, J6a,b = 12.0 Hz, J5,6a = 2.4 Hz, 1H, H-6a), 4.27 (dd, J6′a,b = 11.9 Hz, J5,6′a = 2.4 Hz, 1H, H-6′a), 4.24 (dd, J6a,b = 12.0 Hz, J5,6b = 3.6 Hz, 1H, H-6b), 4.11–4.09 (m, 1H, H-5), 4.06 (dd, J6′a,b = 11.9 Hz, J5,6′b = 4.5 Hz, 1H, H-6′b), 4.02–3.99 (m, 3H, H-5′ and H-6″), 3.96–3.91 (m, 2H, H-3 and 3′), 3.64 (dd, J4,5 = 9.5 Hz, J3,4 = 8.5 Hz, 1H, H-4), 3.50–3.44 (m, 3H, H-2, H-2′ and H-4′), 2.38–2.29 (m, 2H, H-2‴), 2.04–2.00 (m, 2H, H-9‴), 1.84–1.76 (m, 2H, H-7″), 1.63–1.59 (m, 2H, H-3‴), 1.48–1.43 (m, 2H, H-8″), 1.37–1.24 (m, 36H, H-9″–H-22″ and H-4‴–H-8‴), 0.88 (t, J22″,23″ = 7.0 Hz, 3H, H-23″), 0.17, 0.16, 0.15, 0.13, 0.13, 0.12 (6s, 54H, CH3-TMS); 13C NMR (150 MHz, CDCl3) δ 173.9 (C-1‴), 166.4 (C-1″), 163.2 (C-5″), 139.3 (C-10‴), 131.9 (C-3″), 122.3 (C-2″), 114.29, 114.24 (C-4″ and C-11‴), 94.7 (C-1 and C-1′), 73.8, 73.6 (C-3 and C-3′), 72.86, 72.84 (C-2 and C-2′), 72.1 (C-4 and C-4′), 71.0 (C-5), 70.9 (C-5′), 68.4 (C-6″), 63.5 (C-6 and C-6′), 34.3 (C-2‴), 33.9 (C-9‴), 32.1, 29.84, 29.75, 29.72, 29.5, 29.43, 29.38, 29.27, 29.20, 29.0, 26.2, 24.9, 22.8 (C-3‴–C-8‴ and C-7″–C-22″), 14.3 (C-23″), 1.25, 1.22, 1.04, 0.38 (TMS); IR (film): 2970, 2924, 2854, 1741, 1719, 1607, 1510, 1458, 1250, 1165, 1109, 1099, 1075, 1008, 899, 870, 838, 748 cm−1; HRMS (ESI) m/z calcd for [C66H128O14Si6 + NH4]+: 1330.8258; obsd.: 1330.8251.
17, v/v); [α]22.0D = +60 (c = 0.1, CH2Cl2); 1H NMR (600 MHz, CDCl3) δ 7.99 (d, J3″,4″ = 8.9 Hz, 2H, H-3″), 6.91 (d, J3″,4″ = 8.9 Hz, 2H, H-4″), 5.80 (ddt, J10‴,11‴a = 16.9 Hz, J10‴,11‴b = 10.2 Hz, J10‴,9‴ = 6.7 Hz, 1H, H-10‴), 5.03–4.89 (m, 4H, H-1, H-1′, H-11‴a and b), 4.53 (dd, J6a,b = 12.0 Hz, J5,6a = 2.4 Hz, 1H, H-6a), 4.27 (dd, J6′a,b = 11.9 Hz, J5,6′a = 2.4 Hz, 1H, H-6′a), 4.24 (dd, J6a,b = 12.0 Hz, J5,6b = 3.6 Hz, 1H, H-6b), 4.11–4.09 (m, 1H, H-5), 4.06 (dd, J6′a,b = 11.9 Hz, J5,6′b = 4.5 Hz, 1H, H-6′b), 4.02–3.99 (m, 3H, H-5′ and H-6″), 3.96–3.91 (m, 2H, H-3 and 3′), 3.64 (dd, J4,5 = 9.5 Hz, J3,4 = 8.5 Hz, 1H, H-4), 3.50–3.44 (m, 3H, H-2, H-2′ and H-4′), 2.38–2.29 (m, 2H, H-2‴), 2.04–2.00 (m, 2H, H-9‴), 1.84–1.76 (m, 2H, H-7″), 1.63–1.59 (m, 2H, H-3‴), 1.48–1.43 (m, 2H, H-8″), 1.37–1.24 (m, 36H, H-9″–H-22″ and H-4‴–H-8‴), 0.88 (t, J22″,23″ = 7.0 Hz, 3H, H-23″), 0.17, 0.16, 0.15, 0.13, 0.13, 0.12 (6s, 54H, CH3-TMS); 13C NMR (150 MHz, CDCl3) δ 173.9 (C-1‴), 166.4 (C-1″), 163.2 (C-5″), 139.3 (C-10‴), 131.9 (C-3″), 122.3 (C-2″), 114.29, 114.24 (C-4″ and C-11‴), 94.7 (C-1 and C-1′), 73.8, 73.6 (C-3 and C-3′), 72.86, 72.84 (C-2 and C-2′), 72.1 (C-4 and C-4′), 71.0 (C-5), 70.9 (C-5′), 68.4 (C-6″), 63.5 (C-6 and C-6′), 34.3 (C-2‴), 33.9 (C-9‴), 32.1, 29.84, 29.75, 29.72, 29.5, 29.43, 29.38, 29.27, 29.20, 29.0, 26.2, 24.9, 22.8 (C-3‴–C-8‴ and C-7″–C-22″), 14.3 (C-23″), 1.25, 1.22, 1.04, 0.38 (TMS); IR (film): 2970, 2924, 2854, 1741, 1719, 1607, 1510, 1458, 1250, 1165, 1109, 1099, 1075, 1008, 899, 870, 838, 748 cm−1; HRMS (ESI) m/z calcd for [C66H128O14Si6 + NH4]+: 1330.8258; obsd.: 1330.8251.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) EtOAc, 1
EtOAc, 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0 → 97
0 → 97![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3, v/v). The title compound 7c was obtained as a colourless viscous oil (78.7 mg, 0.057 mmol, 70%). Rf = 0.48 (pet. ether
3, v/v). The title compound 7c was obtained as a colourless viscous oil (78.7 mg, 0.057 mmol, 70%). Rf = 0.48 (pet. ether![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) EtOAc, 9
EtOAc, 9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, v/v); [α]27.4D = +51.7 (c = 1.0, CH2Cl2); 1H NMR (500 MHz, CDCl3) δ 7.97 (d, J3″,4″ = 8.8 Hz, 2H, H-3″), 6.90 (d, J3″,4″ = 8.8 Hz, 2H, H-4″), 5.84–5.80 (m, 1H, H-14‴), 5.00–4.89 (m, 4H, H-1 and H-1′ and H-15‴), 4.52 (dd, J6a,b = 12.1 Hz, J5,6a = 2.1 Hz, 1H, H-6a), 4.30–4.20 (m, 2H, H-6′a and H-6b), 4.1 (dt, J4,5 = 9.5 Hz, J5,6a/b = 2.7 Hz, 1H, H-5), 4.10 (dd, J6′a,b = 11.9 Hz, J5′,6′b = 4.4 Hz, 1H, H-6′b), 4.02–4.00 (m, 3H, H-5′ and H-6″), 3.94 (t, J2/2′,3/3′ = J3/3′,4/4′ = 8.9 Hz, 1H, H-3 or H-3′), 3.91 (t, J2/2′,3/3′ = J3/3′,4/4′ = 8.9 Hz, 1H, H-3 or H-3′), 3.63 (t, J3,4 = J4,5 = 9.0 Hz, 1H, H-4), 3.50–3.41 (m, 3H, H-2 and H-2′ and H-4′), 2.38–2.26 (m, 2H, H-2‴), 2.06–1.99 (m, 2H, H-13‴), 1.83–1.75 (m, 2H, H-7″), 1.65–1.57 (m, 2H, H-3‴), 1.49–1.40 (m, 2H, H-8″), 1.39–1.32 (m, 2H, H-9″), 1.32–1.17 (m, 44H, H-4‴-H–12‴ and H-10″–H-22″), 0.87 (t, J22″,23″ = 6.9 Hz), 0.16, 0.15, 0.14, 0.12, 0.11 (6s, 54H, CH3, TMS); 13C NMR (125 MHz, CDCl3) δ 173.9 (C-1‴), 166.4 (C-1″), 163.2 (C-5″), 139.4 (C-14‴), 131.8 (C-3″), 122.2 (C-2″), 114.2 (C-4″ and C-15‴), 94.6 (C-1 and C-1′), 73.8 (C-3 or C-3′), 73.6 (C-3 or C-3′), 72.8, 72.8 (C-2 and C-2′ and C-4′), 72.0 (C-4), 71.0 (C-5), 70.8 (C-5′), 68.3 (C-6″), 63.5, 63.4 (C-6 and C-6′), 34.2 (C-2‴), 33.9 (C-13‴), 32.0, 29.8, 29.8, 29.7, 29.7, 29.6, 29.6, 29.5, 29.5, 29.4, 29.2, 29.2, 29.0, 22.8 (C-7″ and C-9″–C-22″ and C-5‴–C-12‴), 26.1 (C-8″), 24.9 (C-3‴), 14.2 (C-23″), 1.2, 1.2, 1.0, 0.3, 0.3 (TMS); IR (film): 2924, 2854, 1742, 1720, 1607, 1511, 1458, 1250, 1165, 1109, 1099, 1075, 1044, 1008, 965, 934, 898, 870, 838, 748, 695, 536, 519 cm−1; HRMS (ESI) m/z calcd for [C70H136O14Si6 + NH4]+: 1386.8884; obsd.: 1386.8844.
1, v/v); [α]27.4D = +51.7 (c = 1.0, CH2Cl2); 1H NMR (500 MHz, CDCl3) δ 7.97 (d, J3″,4″ = 8.8 Hz, 2H, H-3″), 6.90 (d, J3″,4″ = 8.8 Hz, 2H, H-4″), 5.84–5.80 (m, 1H, H-14‴), 5.00–4.89 (m, 4H, H-1 and H-1′ and H-15‴), 4.52 (dd, J6a,b = 12.1 Hz, J5,6a = 2.1 Hz, 1H, H-6a), 4.30–4.20 (m, 2H, H-6′a and H-6b), 4.1 (dt, J4,5 = 9.5 Hz, J5,6a/b = 2.7 Hz, 1H, H-5), 4.10 (dd, J6′a,b = 11.9 Hz, J5′,6′b = 4.4 Hz, 1H, H-6′b), 4.02–4.00 (m, 3H, H-5′ and H-6″), 3.94 (t, J2/2′,3/3′ = J3/3′,4/4′ = 8.9 Hz, 1H, H-3 or H-3′), 3.91 (t, J2/2′,3/3′ = J3/3′,4/4′ = 8.9 Hz, 1H, H-3 or H-3′), 3.63 (t, J3,4 = J4,5 = 9.0 Hz, 1H, H-4), 3.50–3.41 (m, 3H, H-2 and H-2′ and H-4′), 2.38–2.26 (m, 2H, H-2‴), 2.06–1.99 (m, 2H, H-13‴), 1.83–1.75 (m, 2H, H-7″), 1.65–1.57 (m, 2H, H-3‴), 1.49–1.40 (m, 2H, H-8″), 1.39–1.32 (m, 2H, H-9″), 1.32–1.17 (m, 44H, H-4‴-H–12‴ and H-10″–H-22″), 0.87 (t, J22″,23″ = 6.9 Hz), 0.16, 0.15, 0.14, 0.12, 0.11 (6s, 54H, CH3, TMS); 13C NMR (125 MHz, CDCl3) δ 173.9 (C-1‴), 166.4 (C-1″), 163.2 (C-5″), 139.4 (C-14‴), 131.8 (C-3″), 122.2 (C-2″), 114.2 (C-4″ and C-15‴), 94.6 (C-1 and C-1′), 73.8 (C-3 or C-3′), 73.6 (C-3 or C-3′), 72.8, 72.8 (C-2 and C-2′ and C-4′), 72.0 (C-4), 71.0 (C-5), 70.8 (C-5′), 68.3 (C-6″), 63.5, 63.4 (C-6 and C-6′), 34.2 (C-2‴), 33.9 (C-13‴), 32.0, 29.8, 29.8, 29.7, 29.7, 29.6, 29.6, 29.5, 29.5, 29.4, 29.2, 29.2, 29.0, 22.8 (C-7″ and C-9″–C-22″ and C-5‴–C-12‴), 26.1 (C-8″), 24.9 (C-3‴), 14.2 (C-23″), 1.2, 1.2, 1.0, 0.3, 0.3 (TMS); IR (film): 2924, 2854, 1742, 1720, 1607, 1511, 1458, 1250, 1165, 1109, 1099, 1075, 1044, 1008, 965, 934, 898, 870, 838, 748, 695, 536, 519 cm−1; HRMS (ESI) m/z calcd for [C70H136O14Si6 + NH4]+: 1386.8884; obsd.: 1386.8844.
General procedure for olefin metathesis
The olefin starting material was co-evaporated with dry toluene (2 × 40 mL mmol−1) and re-dissolved in dry CH2Cl2 (20 mL mmol−1). To this was added Grubbs’ catalyst (2nd generation, 1 mol%) and the mixture heated under reflux for 24 hours. At this point, the reaction was deemed complete, as confirmed by TLC and the reaction mixture was concentrated under reduced pressure.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) EtOAc, 1
EtOAc, 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0 → 93
0 → 93![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 7, v/v) to obtain the title compound 16a as a colourless viscous oil (32.4 mg, 0.014 mmol, 64%). Rf = 0.68 (pet. ether
7, v/v) to obtain the title compound 16a as a colourless viscous oil (32.4 mg, 0.014 mmol, 64%). Rf = 0.68 (pet. ether![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) EtOAc, 9
EtOAc, 9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, v/v); [α]29.5D = +80 (c = 0.1, CH2Cl2); 1H NMR (500 MHz, CDCl3) δ 5.45–5.34 (m, 2H, H-5‴), 4.90 (d, J1/1′,2/2′ = 3.0 Hz, 4H, H-1 and H-1′), 4.26 (dd, J6/6′a,b = 11.9 Hz, J5/5′,6/6′a = 2.3 Hz, 4H, H-6a and H-6′a), 4.04 (dd, J6/6′a,b = 11.9 Hz, J5/5′,6/6′b = 4.4 Hz, 4H, H-6b and H-6′b), 3.98 (ddd, J4/4′,5/5′ = 9.2 Hz, J5/5′,6/6′b = 4.4 Hz, J5/5′,6/6′a = 2.3 Hz, 4H, H-5 and H-5′), 3.88 (t, J2/2′,3/3′ = J3/3′,4/4′ = 9.2 Hz, 4H, H-3 and H-3′), 3.47 (t, J3/3′,4/4′ = J4/4′,5/5′ = 9.2 Hz, 4H, H-4 and H-4′), 3.42 (dd, J2/2′,3/3′ = 9.2 Hz, J1/1′,2/2′ = 3.1 Hz, 4H, H-2 and H-2′), 2.41–2.27 (m, 8H, H-2″ and H-2‴), 2.07–2.00 (m, 4H, H-4‴), 1.70–1.57 (m, 8H, H-3″ and H-3‴), 1.31–1.23 (m, 72H, H-4″–H-21″), 0.86 (t, J21″,22″ = 6.9 Hz, 6H, H-22), 0.13, 0.12, 0.11, 0.11 (4s, 108H, TMS); 13C NMR (125 MHz, CDCl3) δ 173.9 (C-1″/C-1‴), 173.6 (C-1″/C-1‴), 130.2 (C-5‴), 94.6 (C-1 and C-1′), 73.6 (C-3 and C-3′), 72.7 (C-2 and C-2′), 72.0 (C-4 and C-4′), 70.8 (C-5 and C-5′), 63.42 (C-6/C-6′), 63.38 (C-6/C-6′), 34.2 (C-2″/C-2‴), 33.6 (C-2″/C-2‴), 32.01(C-4‴, Z/E), 31.97 (C-4‴, Z/E), 29.79, 29.74, 29.58, 29.45, 29.41, 29.26, 22.8 (C-4″–C-21″), 24.9 (C-3″), 24.6 (C-3‴), 14.2 (C-22″), 1.1, 1.0, 0.3 (TMS); IR (film): 2956, 2923, 2853, 1741, 1720, 1659, 1633, 1607, 1511, 1467, 1420, 1314, 1251, 1166, 1110, 1099, 1076, 1044, 1009, 966, 935, 899, 871, 841, 767, 748 cm−1; HRMS (ESI) m/z calcd for [C114H236O26Si12 + Na]+: 2381.4302; obsd.: 2381.4366.
1, v/v); [α]29.5D = +80 (c = 0.1, CH2Cl2); 1H NMR (500 MHz, CDCl3) δ 5.45–5.34 (m, 2H, H-5‴), 4.90 (d, J1/1′,2/2′ = 3.0 Hz, 4H, H-1 and H-1′), 4.26 (dd, J6/6′a,b = 11.9 Hz, J5/5′,6/6′a = 2.3 Hz, 4H, H-6a and H-6′a), 4.04 (dd, J6/6′a,b = 11.9 Hz, J5/5′,6/6′b = 4.4 Hz, 4H, H-6b and H-6′b), 3.98 (ddd, J4/4′,5/5′ = 9.2 Hz, J5/5′,6/6′b = 4.4 Hz, J5/5′,6/6′a = 2.3 Hz, 4H, H-5 and H-5′), 3.88 (t, J2/2′,3/3′ = J3/3′,4/4′ = 9.2 Hz, 4H, H-3 and H-3′), 3.47 (t, J3/3′,4/4′ = J4/4′,5/5′ = 9.2 Hz, 4H, H-4 and H-4′), 3.42 (dd, J2/2′,3/3′ = 9.2 Hz, J1/1′,2/2′ = 3.1 Hz, 4H, H-2 and H-2′), 2.41–2.27 (m, 8H, H-2″ and H-2‴), 2.07–2.00 (m, 4H, H-4‴), 1.70–1.57 (m, 8H, H-3″ and H-3‴), 1.31–1.23 (m, 72H, H-4″–H-21″), 0.86 (t, J21″,22″ = 6.9 Hz, 6H, H-22), 0.13, 0.12, 0.11, 0.11 (4s, 108H, TMS); 13C NMR (125 MHz, CDCl3) δ 173.9 (C-1″/C-1‴), 173.6 (C-1″/C-1‴), 130.2 (C-5‴), 94.6 (C-1 and C-1′), 73.6 (C-3 and C-3′), 72.7 (C-2 and C-2′), 72.0 (C-4 and C-4′), 70.8 (C-5 and C-5′), 63.42 (C-6/C-6′), 63.38 (C-6/C-6′), 34.2 (C-2″/C-2‴), 33.6 (C-2″/C-2‴), 32.01(C-4‴, Z/E), 31.97 (C-4‴, Z/E), 29.79, 29.74, 29.58, 29.45, 29.41, 29.26, 22.8 (C-4″–C-21″), 24.9 (C-3″), 24.6 (C-3‴), 14.2 (C-22″), 1.1, 1.0, 0.3 (TMS); IR (film): 2956, 2923, 2853, 1741, 1720, 1659, 1633, 1607, 1511, 1467, 1420, 1314, 1251, 1166, 1110, 1099, 1076, 1044, 1009, 966, 935, 899, 871, 841, 767, 748 cm−1; HRMS (ESI) m/z calcd for [C114H236O26Si12 + Na]+: 2381.4302; obsd.: 2381.4366.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) EtOAc, 1
EtOAc, 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0 → 24
0 → 24![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, v/v) to obtain the title compound 16b as a colourless viscous oil (25.7 mg, 0.010 mmol, 51%). Rf = 0.55 (pet. ether
1, v/v) to obtain the title compound 16b as a colourless viscous oil (25.7 mg, 0.010 mmol, 51%). Rf = 0.55 (pet. ether![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) EtOAc, 9
EtOAc, 9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, v/v); [α]23.3D = +60 (c = 0.1, CH2Cl2); 1H NMR (600 MHz, CDCl3) δ 5.39–5.32 (m, 2H, H-10‴), 4.92 (d, J1/1′,2/2′ = 3.1 Hz, 4H, H-1 and H-1′), 4.27 (dd, J6/6′a,b = 11.9 Hz, J5/5′,6/6′a = 2.3 Hz, 4H, H-6a and H-6′a), 4.05 (dd, J6/6′a,b = 11.9 Hz, J5/5′,6/6′b = 4.4 Hz, 4H, H-6b and H-6′b), 4.00 (ddd, J4/4′,5/5′ = 9.2 Hz, J5/5′,6/6′b = 4.4 Hz, J5/5′,6/6′a = 2.3 Hz, 4H, H-5 and H-5′), 3.90 (t, J2/2′,3/3′ = J3/3′,4/4′ = 9.2 Hz, 4H, H-3 and H-3′), 3.48 (t, J3/3′,4/4′ = J4/4′,5/5′ = 9.2 Hz, 4H, H-4 and H-4′), 3.44 (dd, J2/2′,3/3′ = 9.2 Hz, J1/1′,2/2′ = 3.1 Hz, 4H, H-2 and H-2′), 2.38–2.29 (m, 8H, H-2″ and H-2‴), 2.02–1.93 (m, 4H, H-9‴), 1.64–1.60 (m, 8H, H-3″ and H-3‴), 1.33–1.22 (m, 92H, H-4″–H-21″ and H-4‴–H-8‴), 0.88 (t, J21″,22″ = 7.0 Hz, 6H, H-22″), 0.15, 0.13, 0.13 (3s, 108H, TMS); 13C NMR (150 MHz, CDCl3) δ 173.94, 173.92 (C-1″ and C-1‴), 130.5 (C-10‴), 94.6 (C-1 and C-1′), 73.6 (C-3 and C-3′), 72.8 (C-2 and C-2′), 72.1 (C-4 and C-4′), 70.9 (C-5 and C-5′), 63.4 (C-6 and C-6′), 34.3 (C-2″ and C-2‴), 32.8 (C-9‴), 32.1, 29.85, 29.81, 29.79, 29.64, 29.51, 29.46, 29.3, 22.8 (C-4″–C21″ and C-4‴–C-8‴), 24.9 (C-3″ and C-3‴), 14.3 (C-22″), 1.2, 1.0, 0.3 (TMS); IR (film): 2970, 2923, 2853, 1742, 1459, 1368, 1250, 1163, 1110, 1099, 1076, 1045, 1009, 871, 837, 747 cm−1; HRMS (ESI) m/z calcd for [C124H256O26Si12 + 2NH4]2+: 1267.3326; obsd.: 1267.3320.
1, v/v); [α]23.3D = +60 (c = 0.1, CH2Cl2); 1H NMR (600 MHz, CDCl3) δ 5.39–5.32 (m, 2H, H-10‴), 4.92 (d, J1/1′,2/2′ = 3.1 Hz, 4H, H-1 and H-1′), 4.27 (dd, J6/6′a,b = 11.9 Hz, J5/5′,6/6′a = 2.3 Hz, 4H, H-6a and H-6′a), 4.05 (dd, J6/6′a,b = 11.9 Hz, J5/5′,6/6′b = 4.4 Hz, 4H, H-6b and H-6′b), 4.00 (ddd, J4/4′,5/5′ = 9.2 Hz, J5/5′,6/6′b = 4.4 Hz, J5/5′,6/6′a = 2.3 Hz, 4H, H-5 and H-5′), 3.90 (t, J2/2′,3/3′ = J3/3′,4/4′ = 9.2 Hz, 4H, H-3 and H-3′), 3.48 (t, J3/3′,4/4′ = J4/4′,5/5′ = 9.2 Hz, 4H, H-4 and H-4′), 3.44 (dd, J2/2′,3/3′ = 9.2 Hz, J1/1′,2/2′ = 3.1 Hz, 4H, H-2 and H-2′), 2.38–2.29 (m, 8H, H-2″ and H-2‴), 2.02–1.93 (m, 4H, H-9‴), 1.64–1.60 (m, 8H, H-3″ and H-3‴), 1.33–1.22 (m, 92H, H-4″–H-21″ and H-4‴–H-8‴), 0.88 (t, J21″,22″ = 7.0 Hz, 6H, H-22″), 0.15, 0.13, 0.13 (3s, 108H, TMS); 13C NMR (150 MHz, CDCl3) δ 173.94, 173.92 (C-1″ and C-1‴), 130.5 (C-10‴), 94.6 (C-1 and C-1′), 73.6 (C-3 and C-3′), 72.8 (C-2 and C-2′), 72.1 (C-4 and C-4′), 70.9 (C-5 and C-5′), 63.4 (C-6 and C-6′), 34.3 (C-2″ and C-2‴), 32.8 (C-9‴), 32.1, 29.85, 29.81, 29.79, 29.64, 29.51, 29.46, 29.3, 22.8 (C-4″–C21″ and C-4‴–C-8‴), 24.9 (C-3″ and C-3‴), 14.3 (C-22″), 1.2, 1.0, 0.3 (TMS); IR (film): 2970, 2923, 2853, 1742, 1459, 1368, 1250, 1163, 1110, 1099, 1076, 1045, 1009, 871, 837, 747 cm−1; HRMS (ESI) m/z calcd for [C124H256O26Si12 + 2NH4]2+: 1267.3326; obsd.: 1267.3320.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) EtOAc, 1
EtOAc, 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0 → 24
0 → 24![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, v/v) to obtain the title compound 16a as a colourless viscous oil (18.4 mg, 0.007 mmol, 57%). Rf = 0.60 (pet. ether
1, v/v) to obtain the title compound 16a as a colourless viscous oil (18.4 mg, 0.007 mmol, 57%). Rf = 0.60 (pet. ether![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) EtOAc, 9
EtOAc, 9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, v/v); [α]24.3D = +60 (c = 0.1, CH2Cl2); 1H NMR (500 MHz, CDCl3) δ 5.41–5.31 (m, 2H, H-14‴), 4.91 (d, J1/1′,2/2′ = 2.8 Hz, 4H, H-1 and H-1′), 4.26 (dd, J6/6′a,b = 11.8, J5/5′,6/6′a = 2.1 Hz, 4H, H-6a and H-6′a), 4.04 (dd, J6/6′a,b = 12.0, J5/5′,6/6′b = 4.3 Hz, 4H, H-6b and H-6′b), 3.98 (ddd, J4/4′,5/5′ = 9.6, J5/5′,6/6′b = 4.2, J5/5′,6/6′a = 2.1 Hz, 4H, H-5 and H-5′), 3.89 (t, J2/2′,3/3′ = J3/3′,4/4′ = 9.0 Hz, 4H, H-3 and H-3′), 3.47 (t, J3/3′,4/4′ = J4/4′,5/5′ = 9.0 Hz, 4H, H-4 and H-4′), 3.43 (dd, J2/2′,3/3′ = 9.1, J1/1′,2/2′ = 2.9 Hz, 4H, H-2 and H-2′), 2.38–2.26 (m, 8H, H-2″ and H-2‴), 2.02–1.91 (m, 4H, H-13‴), 1.67–1.56 (m, 8H, H-3″ and H-3‴), 1.34–1.20 (m, 108H, H-4″–H-21″ and H-4‴–H-12‴), 0.87 (t, J21″,22″ = 7.0 Hz, H-22″), 0.14, 0.12, 0.12 (3s, 108H, TMS); 13C NMR (125 MHz, CDCl3) δ 173.9 (C-1″ and C-1‴), 130.4 (C-14‴), 94.5 (C-1 and C-1′), 73.6 (C-3 and C-3′), 72.7 (C-2 and C-2′), 72.0 (C-4 and C-4′), 70.8 (C-5 and C-5′), 63.4 (C-6 and C-6′), 34.2 (C-2″ and C-2‴), 32.7 (C-13″), 32.0, 29.8, 29.8, 29.6, 29.5, 29.4, 29.3, 29.3, 22.8 (C-4″–C-21″ and C-4‴–C-12‴), 24.9 (C-3″ and C-3‴), 14.2 (C-22″), 1.2, 1.0, 0.3 (TMS); IR (film): 2923, 2853, 1742, 1632, 1459, 1411, 1250, 1164, 1110, 1099, 1076, 1045, 1009, 965, 897, 871, 839, 747, 684, 624, 579, 536, 518 cm−1; HRMS (ESI) m/z calcd for [C132H272O26Si12 + 2Na]2+: 1327.8489; obsd.: 1327.8482.
1, v/v); [α]24.3D = +60 (c = 0.1, CH2Cl2); 1H NMR (500 MHz, CDCl3) δ 5.41–5.31 (m, 2H, H-14‴), 4.91 (d, J1/1′,2/2′ = 2.8 Hz, 4H, H-1 and H-1′), 4.26 (dd, J6/6′a,b = 11.8, J5/5′,6/6′a = 2.1 Hz, 4H, H-6a and H-6′a), 4.04 (dd, J6/6′a,b = 12.0, J5/5′,6/6′b = 4.3 Hz, 4H, H-6b and H-6′b), 3.98 (ddd, J4/4′,5/5′ = 9.6, J5/5′,6/6′b = 4.2, J5/5′,6/6′a = 2.1 Hz, 4H, H-5 and H-5′), 3.89 (t, J2/2′,3/3′ = J3/3′,4/4′ = 9.0 Hz, 4H, H-3 and H-3′), 3.47 (t, J3/3′,4/4′ = J4/4′,5/5′ = 9.0 Hz, 4H, H-4 and H-4′), 3.43 (dd, J2/2′,3/3′ = 9.1, J1/1′,2/2′ = 2.9 Hz, 4H, H-2 and H-2′), 2.38–2.26 (m, 8H, H-2″ and H-2‴), 2.02–1.91 (m, 4H, H-13‴), 1.67–1.56 (m, 8H, H-3″ and H-3‴), 1.34–1.20 (m, 108H, H-4″–H-21″ and H-4‴–H-12‴), 0.87 (t, J21″,22″ = 7.0 Hz, H-22″), 0.14, 0.12, 0.12 (3s, 108H, TMS); 13C NMR (125 MHz, CDCl3) δ 173.9 (C-1″ and C-1‴), 130.4 (C-14‴), 94.5 (C-1 and C-1′), 73.6 (C-3 and C-3′), 72.7 (C-2 and C-2′), 72.0 (C-4 and C-4′), 70.8 (C-5 and C-5′), 63.4 (C-6 and C-6′), 34.2 (C-2″ and C-2‴), 32.7 (C-13″), 32.0, 29.8, 29.8, 29.6, 29.5, 29.4, 29.3, 29.3, 22.8 (C-4″–C-21″ and C-4‴–C-12‴), 24.9 (C-3″ and C-3‴), 14.2 (C-22″), 1.2, 1.0, 0.3 (TMS); IR (film): 2923, 2853, 1742, 1632, 1459, 1411, 1250, 1164, 1110, 1099, 1076, 1045, 1009, 965, 897, 871, 839, 747, 684, 624, 579, 536, 518 cm−1; HRMS (ESI) m/z calcd for [C132H272O26Si12 + 2Na]2+: 1327.8489; obsd.: 1327.8482.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) EtOAc, 1
EtOAc, 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0 → 93
0 → 93![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 7, v/v) to obtain the title compound 17a as a colourless viscous oil (40.5 mg, 0.016 mmol, 75%). Rf = 0.53 (pet. ether
7, v/v) to obtain the title compound 17a as a colourless viscous oil (40.5 mg, 0.016 mmol, 75%). Rf = 0.53 (pet. ether![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) EtOAc, 9
EtOAc, 9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, v/v); [α]27.3D = +40 (c = 0.1, CH2Cl2); 1H NMR (500 MHz, CDCl3) δ 7.97 (d, J3′,4′ = 9.1 Hz, 4H, H-3″), 6.90 (d, J3′,4′ = 9.0 Hz, 4H, H-4″), 5.42–5.34 (m, 2H, H-5‴), 4.93 (t, J1/1′,2/2′ = 3.5 Hz, 4H, H-1 and H-1′), 4.52 (dd, J6a,b = 12.1, J5,6a = 2.4 Hz, 2H, H-6a), 4.28–4.20 (m, 4H, H-6b and H-6a′), 4.12–4.07 (m, 2H, H-5′), 4.05 (dd, J6′a,b = 11.8, J5′,6′b = 4.4 Hz, 2H, H-6′b), 4.02–3.97 (m, 6H, H-5 and H-6″), 3.93 (t, J2/2′,3/3′ = J3/3′,4/4′ = 8.9 Hz, 2H, H-3 or H-3′), 3.90 (t, J2/2′,3/3′ = J3/3′,4/4′ = 9.0 Hz, 2H, H-3 or H-3′), 3.64 (t, J3′,4′ = J4′,5′ = 9.0 Hz, 2H, H-4′), 3.50–3.41 (m, 6H, H-2 and H-2′ and H-4), 2.47–2.22 (m, 5H, H-2‴), 2.12–1.93 (m, 4H, H-4‴), 1.84–1.74 (m, 5H, H-7″), 1.70–1.62 (m, 4H, H-3‴), 1.48–1.40 (m, 4H, H-8″), 1.37–1.17 (m, 52H, H-9″–H-22″), 0.86 (t, J22″,23″ = 6.9 Hz, 6H, H-23″), 0.15, 0.14, 0.13, 0.11, 0.10 (6s, 108H, CH3, TMS); 13C NMR (125 MHz, CDCl3) δ 173.7 (C-1‴), 166.3 (C-1″), 163.2 (C-5″), 131.8 (C-3″), 130.2 (C-5‴), 122.2 (C-2″), 114.2 (C-4″), 94.7 (C-1 and C-1′), 73.8, 73.6 (C-3 and C-3′), 72.8, 72.7 (C-2 and C-2′), 72.0 (C-4 and C-4″), 70.9, 70.8 (C-5 and C-5′), 68.3 (C-6″), 63.5 (C-6 and C-6′), 33.6 (C-2‴), 32.0 (C-4‴), 29.8, 29.8, 29.7, 29.7, 29.5, 29.5, 22.8 (C-9″–C-22″), 29.2 (C-7″), 26.1 (C-8″), 24.6 (C-3‴), 14.2 (C-23″), 1.2, 1.2, 1.0, 0.3, 0.3 (TMS); IR (film): 2924, 2854, 1740, 1719, 1606, 1511, 1457, 1249, 1165, 1099, 1075, 1044, 1008, 966, 934, 898, 870, 835, 748, 695, 537, 518 cm−1; HRMS (ESI) m/z calcd for [C120H232O28Si12 + 2NH4]2+: 1247.2336; obsd.: 1247.2339.
1, v/v); [α]27.3D = +40 (c = 0.1, CH2Cl2); 1H NMR (500 MHz, CDCl3) δ 7.97 (d, J3′,4′ = 9.1 Hz, 4H, H-3″), 6.90 (d, J3′,4′ = 9.0 Hz, 4H, H-4″), 5.42–5.34 (m, 2H, H-5‴), 4.93 (t, J1/1′,2/2′ = 3.5 Hz, 4H, H-1 and H-1′), 4.52 (dd, J6a,b = 12.1, J5,6a = 2.4 Hz, 2H, H-6a), 4.28–4.20 (m, 4H, H-6b and H-6a′), 4.12–4.07 (m, 2H, H-5′), 4.05 (dd, J6′a,b = 11.8, J5′,6′b = 4.4 Hz, 2H, H-6′b), 4.02–3.97 (m, 6H, H-5 and H-6″), 3.93 (t, J2/2′,3/3′ = J3/3′,4/4′ = 8.9 Hz, 2H, H-3 or H-3′), 3.90 (t, J2/2′,3/3′ = J3/3′,4/4′ = 9.0 Hz, 2H, H-3 or H-3′), 3.64 (t, J3′,4′ = J4′,5′ = 9.0 Hz, 2H, H-4′), 3.50–3.41 (m, 6H, H-2 and H-2′ and H-4), 2.47–2.22 (m, 5H, H-2‴), 2.12–1.93 (m, 4H, H-4‴), 1.84–1.74 (m, 5H, H-7″), 1.70–1.62 (m, 4H, H-3‴), 1.48–1.40 (m, 4H, H-8″), 1.37–1.17 (m, 52H, H-9″–H-22″), 0.86 (t, J22″,23″ = 6.9 Hz, 6H, H-23″), 0.15, 0.14, 0.13, 0.11, 0.10 (6s, 108H, CH3, TMS); 13C NMR (125 MHz, CDCl3) δ 173.7 (C-1‴), 166.3 (C-1″), 163.2 (C-5″), 131.8 (C-3″), 130.2 (C-5‴), 122.2 (C-2″), 114.2 (C-4″), 94.7 (C-1 and C-1′), 73.8, 73.6 (C-3 and C-3′), 72.8, 72.7 (C-2 and C-2′), 72.0 (C-4 and C-4″), 70.9, 70.8 (C-5 and C-5′), 68.3 (C-6″), 63.5 (C-6 and C-6′), 33.6 (C-2‴), 32.0 (C-4‴), 29.8, 29.8, 29.7, 29.7, 29.5, 29.5, 22.8 (C-9″–C-22″), 29.2 (C-7″), 26.1 (C-8″), 24.6 (C-3‴), 14.2 (C-23″), 1.2, 1.2, 1.0, 0.3, 0.3 (TMS); IR (film): 2924, 2854, 1740, 1719, 1606, 1511, 1457, 1249, 1165, 1099, 1075, 1044, 1008, 966, 934, 898, 870, 835, 748, 695, 537, 518 cm−1; HRMS (ESI) m/z calcd for [C120H232O28Si12 + 2NH4]2+: 1247.2336; obsd.: 1247.2339.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) EtOAc, 1
EtOAc, 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0 → 19
0 → 19![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, v/v) to obtain the title compound 17b as a colourless viscous oil (41.8 mg, 0.016 mmol, 60%) Rf = 0.75 (pet. ether
1, v/v) to obtain the title compound 17b as a colourless viscous oil (41.8 mg, 0.016 mmol, 60%) Rf = 0.75 (pet. ether![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) EtOAc, 3
EtOAc, 3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 22, v/v); [α]21.0D = +40 (c = 0.1, CH2Cl2); 1H NMR (500 MHz, CDCl3) δ 7.99 (d, J3″,4″ = 8.9 Hz, 4H, H-3″), 6.91 (d, J3″,4″ = 8.9 Hz, 4H, H-4″), 5.37–5.32 (m, 2H, Z- and E-H-10‴), 4.95 (d, J1/1′,2/2′ = 3.5 Hz, 2H, H-1/1′), 4.94 (d, J1/1′,2/2′ = 3.5 Hz, 2H, H-1/1′), 4.53 (dd, J6a,b = 12.0 Hz, J5,6a = 2.4 Hz, 2H, H-6a), 4.28–4.23 (m, 4H, H-6b and H-6′a), 4.11–4.05 (m, 4H, H-5 and H-6′b), 4.03–3.99 (m, 6H, H-5′ and H-6″), 3.97–3.90 (m, 4H, H-3 and 3′), 3.65 (t, J3,4 = J4,5 = 9.1 Hz, 2H, H-4), 3.50–3.43 (m, 6H, H-2, H-2′ and H-4′), 2.37–2.30 (m, 4H, H-2‴), 2.00–1.90 (m, 4H, H-9‴), 1.83–1.77 (m, 4H, H-7″), 1.63–1.59 (m, 4H, H-3‴), 1.46 (p, J7″,8″ = J8″,9″ = 7.2 Hz, 4H, H-8″), 1.36–1.21 (m, 76H, H-9″–H-22″ and H-4‴–H-8‴), 0.88 (t, J22″,23″ = 7.1 Hz, 6H, H-23″), 0.17, 0.16, 0.15, 0.13, 0.12 (5s, 54H, TMS); 13C NMR (150 MHz, CDCl3) δ 174.0 (C-1‴), 166.4 (C-1″), 163.2 (C-5″), 131.8 (C-3″), 130.5 (C-10‴), 122.3 (C-2″), 114.2 (C-4″), 94.7 (C-1 and C-1′), 73.8, 73.6 (C-3 and C-3′), 72.84, 72.79 (C-2 and C-2′), 72.0 (C-4 and C-4′), 70.98 (C-5), 70.86 (C-5′), 68.4 (C-6″), 63.51 (C-6′), 63.45 (C-6), 34.3 (C-2‴), 32.8(C-9‴), 32.1, 29.84, 29.81, 29.75, 29.72, 29.53, 29.51, 29.49, 29.45, 29.32, 29.26, 26.1, 24.9, 22.8 (C-3‴–C-8‴ and C-7″–C-22″), 14.3 (C-23″), 1.24, 1.21, 1.04, 1.03, 0.38, 0.36 (TMS); IR (film): 2970, 2925, 2854, 1741, 1720, 1607, 1511, 1459, 1251, 1166, 1110, 1100, 1076, 1045, 1009, 899, 872, 843 cm−1; HRMS (ESI) m/z calcd for [C130H252O28Si12 + HCOO]−: 2642.5509; obsd.: 2642.5522.
22, v/v); [α]21.0D = +40 (c = 0.1, CH2Cl2); 1H NMR (500 MHz, CDCl3) δ 7.99 (d, J3″,4″ = 8.9 Hz, 4H, H-3″), 6.91 (d, J3″,4″ = 8.9 Hz, 4H, H-4″), 5.37–5.32 (m, 2H, Z- and E-H-10‴), 4.95 (d, J1/1′,2/2′ = 3.5 Hz, 2H, H-1/1′), 4.94 (d, J1/1′,2/2′ = 3.5 Hz, 2H, H-1/1′), 4.53 (dd, J6a,b = 12.0 Hz, J5,6a = 2.4 Hz, 2H, H-6a), 4.28–4.23 (m, 4H, H-6b and H-6′a), 4.11–4.05 (m, 4H, H-5 and H-6′b), 4.03–3.99 (m, 6H, H-5′ and H-6″), 3.97–3.90 (m, 4H, H-3 and 3′), 3.65 (t, J3,4 = J4,5 = 9.1 Hz, 2H, H-4), 3.50–3.43 (m, 6H, H-2, H-2′ and H-4′), 2.37–2.30 (m, 4H, H-2‴), 2.00–1.90 (m, 4H, H-9‴), 1.83–1.77 (m, 4H, H-7″), 1.63–1.59 (m, 4H, H-3‴), 1.46 (p, J7″,8″ = J8″,9″ = 7.2 Hz, 4H, H-8″), 1.36–1.21 (m, 76H, H-9″–H-22″ and H-4‴–H-8‴), 0.88 (t, J22″,23″ = 7.1 Hz, 6H, H-23″), 0.17, 0.16, 0.15, 0.13, 0.12 (5s, 54H, TMS); 13C NMR (150 MHz, CDCl3) δ 174.0 (C-1‴), 166.4 (C-1″), 163.2 (C-5″), 131.8 (C-3″), 130.5 (C-10‴), 122.3 (C-2″), 114.2 (C-4″), 94.7 (C-1 and C-1′), 73.8, 73.6 (C-3 and C-3′), 72.84, 72.79 (C-2 and C-2′), 72.0 (C-4 and C-4′), 70.98 (C-5), 70.86 (C-5′), 68.4 (C-6″), 63.51 (C-6′), 63.45 (C-6), 34.3 (C-2‴), 32.8(C-9‴), 32.1, 29.84, 29.81, 29.75, 29.72, 29.53, 29.51, 29.49, 29.45, 29.32, 29.26, 26.1, 24.9, 22.8 (C-3‴–C-8‴ and C-7″–C-22″), 14.3 (C-23″), 1.24, 1.21, 1.04, 1.03, 0.38, 0.36 (TMS); IR (film): 2970, 2925, 2854, 1741, 1720, 1607, 1511, 1459, 1251, 1166, 1110, 1100, 1076, 1045, 1009, 899, 872, 843 cm−1; HRMS (ESI) m/z calcd for [C130H252O28Si12 + HCOO]−: 2642.5509; obsd.: 2642.5522.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) EtOAc, 1
EtOAc, 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0 → 19
0 → 19![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, v/v) to obtain the title compound 17c as a colourless viscous oil (21.8 mg, 0.008 mmol, 46%). Rf = 0.67 (pet. ether
1, v/v) to obtain the title compound 17c as a colourless viscous oil (21.8 mg, 0.008 mmol, 46%). Rf = 0.67 (pet. ether![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) EtOAc, 9
EtOAc, 9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, v/v); [α]23.2D = +49.4 (c = 0.5, CH2Cl2); 1H NMR (500 MHz, CDCl3) δ 7.98 (d, J3″,4″ = 8.7 Hz, 4H, H-3″), 6.90 (d, J3″,4″ = 8.7 Hz, 4H, H-4″), 5.38–5.31 (m, 2H, H-14‴), 4.94 (d, J1/1′,2/2′ = 3.5 Hz, 2H, H-1/1′), 4.93 (d, J1/1′,2/2′ = 3.5 Hz, H-1/1′), 4.52 (dd, J6a,b = 12.0, J5,6a = 2.4 Hz, 2H, H-6a), 4.29–4.20 (m, 4H, H-6b and H-6′a), 4.09 (dt, J4,5 = 9.5, J5,6a/b = 2.8 Hz, 2H, H-5), 4.05 (dd, J6′a,b = 11.9 Hz, J5′,6′b = 4.4 Hz, 2H, H-6′b), 4.02–3.97 (m, 6H, H-5′ and H-6″), 3.94 (t, J2/2′,3/3′ = J3/3′,4/4′ = 8.9 Hz, 2H, H-3/3′), 3.91 (t, J2/2′,3/3′ = J3/3′,4/4′ = 8.9 Hz, 2H, H-3/3′), 3.63 (t, J3,4 = J4,5 = 9.0 Hz, 2H, H-4), 3.50–3.41 (m, 6H, H-2 and H-2′ and H-4′), 2.38–2.26 (m, 4H, H-2‴), 2.02–1.90 (m, 4H, H-13‴), 1.82–1.75 (m, 4H, H-7″), 1.65–1.56 (m, 4H, H-3‴), 1.48–1.40 (m, 4H, H-8″), 1.38–1.17 (m, 92H, H-9″–H-22″ and H-4‴–H-12‴), 0.87 (t, J22″,23″ = 6.8H, 6H, H-23″), 0.16, 0.15, 0.14, 0.12, 0.11 (6s, 54H, TMS); 13C NMR (125 MHz, CDCl3) δ 173.9 (C-1‴), 166.3 (C-1″), 163.2 (C-5″), 131.8 (C-3″), 130.4 (C-14‴), 122.2 (C-2″), 114.2 (C-4″), 94.6 (C-1 and C-1′), 73.8, 73.6 (C-3 and C-3′), 72.8 (C-2 and C-2′), 72.0 (C-4 and C-4′), 71.0, 70.8 (C-5 and C-5′), 68.3 (C-6″), 63.5, 63.4 (C-6 and C-6′), 34.2 (C-2‴), 32.7 (C-13‴), 32.0, 29.8, 29.8, 29.7, 29.7, 29.7, 29.6, 29.5, 29.5, 29.4, 29.3, 29.3, 29.2, 22.8 (C-7″ and C-9″-C-22″ and C-4‴-C-12‴), 26.1 (C-8″), 24.9 (C-3‴), 14.2 (C-23″), 1.2, 1.2, 1.0, 1.0, 0.3, 0.3 (TMS); IR (film): 2924, 2854, 1742, 1720, 1607, 1511, 1458, 1313, 1251, 1166, 1110, 1100, 1077, 1045, 1010, 966, 934, 899, 872, 843, 767, 748 cm−1.
1, v/v); [α]23.2D = +49.4 (c = 0.5, CH2Cl2); 1H NMR (500 MHz, CDCl3) δ 7.98 (d, J3″,4″ = 8.7 Hz, 4H, H-3″), 6.90 (d, J3″,4″ = 8.7 Hz, 4H, H-4″), 5.38–5.31 (m, 2H, H-14‴), 4.94 (d, J1/1′,2/2′ = 3.5 Hz, 2H, H-1/1′), 4.93 (d, J1/1′,2/2′ = 3.5 Hz, H-1/1′), 4.52 (dd, J6a,b = 12.0, J5,6a = 2.4 Hz, 2H, H-6a), 4.29–4.20 (m, 4H, H-6b and H-6′a), 4.09 (dt, J4,5 = 9.5, J5,6a/b = 2.8 Hz, 2H, H-5), 4.05 (dd, J6′a,b = 11.9 Hz, J5′,6′b = 4.4 Hz, 2H, H-6′b), 4.02–3.97 (m, 6H, H-5′ and H-6″), 3.94 (t, J2/2′,3/3′ = J3/3′,4/4′ = 8.9 Hz, 2H, H-3/3′), 3.91 (t, J2/2′,3/3′ = J3/3′,4/4′ = 8.9 Hz, 2H, H-3/3′), 3.63 (t, J3,4 = J4,5 = 9.0 Hz, 2H, H-4), 3.50–3.41 (m, 6H, H-2 and H-2′ and H-4′), 2.38–2.26 (m, 4H, H-2‴), 2.02–1.90 (m, 4H, H-13‴), 1.82–1.75 (m, 4H, H-7″), 1.65–1.56 (m, 4H, H-3‴), 1.48–1.40 (m, 4H, H-8″), 1.38–1.17 (m, 92H, H-9″–H-22″ and H-4‴–H-12‴), 0.87 (t, J22″,23″ = 6.8H, 6H, H-23″), 0.16, 0.15, 0.14, 0.12, 0.11 (6s, 54H, TMS); 13C NMR (125 MHz, CDCl3) δ 173.9 (C-1‴), 166.3 (C-1″), 163.2 (C-5″), 131.8 (C-3″), 130.4 (C-14‴), 122.2 (C-2″), 114.2 (C-4″), 94.6 (C-1 and C-1′), 73.8, 73.6 (C-3 and C-3′), 72.8 (C-2 and C-2′), 72.0 (C-4 and C-4′), 71.0, 70.8 (C-5 and C-5′), 68.3 (C-6″), 63.5, 63.4 (C-6 and C-6′), 34.2 (C-2‴), 32.7 (C-13‴), 32.0, 29.8, 29.8, 29.7, 29.7, 29.7, 29.6, 29.5, 29.5, 29.4, 29.3, 29.3, 29.2, 22.8 (C-7″ and C-9″-C-22″ and C-4‴-C-12‴), 26.1 (C-8″), 24.9 (C-3‴), 14.2 (C-23″), 1.2, 1.2, 1.0, 1.0, 0.3, 0.3 (TMS); IR (film): 2924, 2854, 1742, 1720, 1607, 1511, 1458, 1313, 1251, 1166, 1110, 1100, 1077, 1045, 1010, 966, 934, 899, 872, 843, 767, 748 cm−1.
General procedure for the desilylation
To a solution of the TMS protected trehalose ester in CH2Cl2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) MeOH (20 mL mmol−1, 1
MeOH (20 mL mmol−1, 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, v/v) was added Dowex-H+ until the pH of the reaction mixture reached 2–3. The mixture was then stirred at room temperature for 1.5–2.5 hours until the reaction was deemed complete, as confirmed by TLC. The reaction mixture was then diluted with CH2Cl2
1, v/v) was added Dowex-H+ until the pH of the reaction mixture reached 2–3. The mixture was then stirred at room temperature for 1.5–2.5 hours until the reaction was deemed complete, as confirmed by TLC. The reaction mixture was then diluted with CH2Cl2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) MeOH (1
MeOH (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, v/v), filtered, washed with CH2Cl2
1, v/v), filtered, washed with CH2Cl2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) MeOH (1
MeOH (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, v/v), and the filtrate was concentrated under vacuum.
1, v/v), and the filtrate was concentrated under vacuum.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) MeOH, 1
MeOH, 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0 → 41
0 → 41![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 9, v/v) to obtain the final glycolipid 3a as a thin white film (10.6 mg, 0.0071 mmol, 91%). Rf = 0.52 (CH2Cl2
9, v/v) to obtain the final glycolipid 3a as a thin white film (10.6 mg, 0.0071 mmol, 91%). Rf = 0.52 (CH2Cl2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) MeOH, 4
MeOH, 4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, v/v); [α]25.2D= + 40 (c = 0.1, pyridine); 1H NMR (500 MHz, C5D5N) δ 5.89 (2d, J1/1′,2/2′ = 3.8 Hz, 4H, H-1 and H-1′), 5.30–5.28 (m, 2H, H-5‴), 5.12–5.06 (m, 4H, H-5 and H-5′), 5.00–4.98 (m, 4H, H-6a and H-6′a), 4.86–4.81 (m, 4H, H-6b and H-6′b), 4.75 (t, J2/2′,3/3′ = J3/3′,4/4′ = 9.3 Hz, 4H, H-3 and H-3′), 4.32 (dd, J2/2′,3/3′ = 9.3 Hz, J1/1′,2/2′ = 3.8 Hz, 4H, H-2 and H-2′), 4.21–4.16 (m, 4H, H-4 and H-4′), 2.35–2.29 (m, 8H, H-2″ and H-2‴), 1.94–1.90 (m, 4H, H-4‴), 1.69–1.59 (m, 8H, H-3″ and H-3‴), 1.32–1.19 (m, 72H, H-4″–H-21″), 0.87 (t, 6H, H-22″); 13C NMR (125 MHz, C5D5N) δ 174.1 (C-1″/C-1‴), 173.9 (C-1″/C-1‴), 130.8 (C-5‴), 96.3 (C-1 and C-1′), 75.3 (C-3 and C-3′), 73.7 (C-2 and C-2′), 72.4 (C-4 and C-4′), 72.0 (C-5 and C-5′), 64.8 (C-6 and C-6′), 34.8 (C-2‴), 34.2 (C-2″), 32.5 (C-4‴), 30.4, 30.3, 30.2, 29.98, 29.76, 23.3 (C-4″–C21″), 25.7 (C-3″), 25.4 (C-3‴), 14.7 (C-22″). IR (film): 3358 (broad), 2917, 2850, 1716, 1717, 1467, 1240, 1174, 1148, 1103, 1076, 1050, 1020, 987 cm−1; HRMS (ESI) m/z calcd for [C78H140O26 + NH4]+: 1510.9971; obsd.: 1510.9970.
1, v/v); [α]25.2D= + 40 (c = 0.1, pyridine); 1H NMR (500 MHz, C5D5N) δ 5.89 (2d, J1/1′,2/2′ = 3.8 Hz, 4H, H-1 and H-1′), 5.30–5.28 (m, 2H, H-5‴), 5.12–5.06 (m, 4H, H-5 and H-5′), 5.00–4.98 (m, 4H, H-6a and H-6′a), 4.86–4.81 (m, 4H, H-6b and H-6′b), 4.75 (t, J2/2′,3/3′ = J3/3′,4/4′ = 9.3 Hz, 4H, H-3 and H-3′), 4.32 (dd, J2/2′,3/3′ = 9.3 Hz, J1/1′,2/2′ = 3.8 Hz, 4H, H-2 and H-2′), 4.21–4.16 (m, 4H, H-4 and H-4′), 2.35–2.29 (m, 8H, H-2″ and H-2‴), 1.94–1.90 (m, 4H, H-4‴), 1.69–1.59 (m, 8H, H-3″ and H-3‴), 1.32–1.19 (m, 72H, H-4″–H-21″), 0.87 (t, 6H, H-22″); 13C NMR (125 MHz, C5D5N) δ 174.1 (C-1″/C-1‴), 173.9 (C-1″/C-1‴), 130.8 (C-5‴), 96.3 (C-1 and C-1′), 75.3 (C-3 and C-3′), 73.7 (C-2 and C-2′), 72.4 (C-4 and C-4′), 72.0 (C-5 and C-5′), 64.8 (C-6 and C-6′), 34.8 (C-2‴), 34.2 (C-2″), 32.5 (C-4‴), 30.4, 30.3, 30.2, 29.98, 29.76, 23.3 (C-4″–C21″), 25.7 (C-3″), 25.4 (C-3‴), 14.7 (C-22″). IR (film): 3358 (broad), 2917, 2850, 1716, 1717, 1467, 1240, 1174, 1148, 1103, 1076, 1050, 1020, 987 cm−1; HRMS (ESI) m/z calcd for [C78H140O26 + NH4]+: 1510.9971; obsd.: 1510.9970.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) MeOH, 1
MeOH, 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0 → 41
0 → 41![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 9, v/v) to obtain glycolipid 3b as a thin white film (10.1 mg, 0.0062 mmol, 83%). Rf = 0.63 (CH2Cl2
9, v/v) to obtain glycolipid 3b as a thin white film (10.1 mg, 0.0062 mmol, 83%). Rf = 0.63 (CH2Cl2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) MeOH, 4
MeOH, 4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, v/v); [α]23.5D = +60 (c = 0.1, pyridine); 1H NMR (600 MHz, C5D5N) δ 5.90 (d, J1/1′,2/2′ = 3.7 Hz, 4H, H-1 and H-1′), 5.53–5.48 (m, 2H, H-10‴), 5.11–5.08 (m, 4H, H-5 and H-5′), 5.02–4.99 (m, 4H, H-6a and H-6′a), 4.84 (dd, J6/6′a,b = 11.9 Hz, J5/5′,6/6′b = 5.3 Hz, 4H, H-6b and H-6′b), 4.76 (t, J2/2′,3/3′ = J3/3′,4/4′ = 9.3 Hz, 4H, H-3 and H-3′), 4.32 (dd, J2/2′,3/3′ = 9.3 Hz, J1/1′,2/2′ = 3.7 Hz, 4H, H-2 and H-2′), 4.18 (t, J3/3′,4/4′ = J4/4′,5/5′ = 9.3 Hz, 4H, H-4 and H-4′), 2.36–2.29 (m, 8H, H-2″ and H-2‴), 2.12–2.03 (m, 4H, H-9‴), 1.65–1.59 (m, 8H, H-3″ and H-3‴), 1.38–1.15 (m, 92H, H-4″–H-21″ and H-4‴–H-8‴), 0.88 (t, J21″,22″ = 6.9 Hz, 6H, H-22″); 13C NMR (150 MHz, C5D5N) δ 174.1 (C-1″ and C-1‴), 131.1 (C-10‴), 96.2 (C-1 and C-1′), 75.3 (C-3 and C-3′), 73.8 (C-2 and C-2′), 72.4 (C-4 and C-4′), 71.9 (C-5 and C-5′), 64.7 (C-6 and C-6′), 34.8 (C-2″ and C-2‴), 33.4 (C-9‴), 32.5, 30.40, 30.36, 30.30, 30.15, 29.98, 29.94, 29.84, 29.75, 23.3 (C-4″–C21″ and C-4‴–C-8‴), 25.6 (C-3″ and C-3‴), 14.7 (C-22″); IR (film): 3343 (broad), 2918, 2850, 1736, 1735, 1466, 1354, 1241, 1175, 1150, 1103, 1077, 1053, 1019, 989, 941 cm−1; HRMS (ESI) m/z calcd for [C88H160O26 + HCOO]−: 1679.1213; obsd.: 1679.1188.
1, v/v); [α]23.5D = +60 (c = 0.1, pyridine); 1H NMR (600 MHz, C5D5N) δ 5.90 (d, J1/1′,2/2′ = 3.7 Hz, 4H, H-1 and H-1′), 5.53–5.48 (m, 2H, H-10‴), 5.11–5.08 (m, 4H, H-5 and H-5′), 5.02–4.99 (m, 4H, H-6a and H-6′a), 4.84 (dd, J6/6′a,b = 11.9 Hz, J5/5′,6/6′b = 5.3 Hz, 4H, H-6b and H-6′b), 4.76 (t, J2/2′,3/3′ = J3/3′,4/4′ = 9.3 Hz, 4H, H-3 and H-3′), 4.32 (dd, J2/2′,3/3′ = 9.3 Hz, J1/1′,2/2′ = 3.7 Hz, 4H, H-2 and H-2′), 4.18 (t, J3/3′,4/4′ = J4/4′,5/5′ = 9.3 Hz, 4H, H-4 and H-4′), 2.36–2.29 (m, 8H, H-2″ and H-2‴), 2.12–2.03 (m, 4H, H-9‴), 1.65–1.59 (m, 8H, H-3″ and H-3‴), 1.38–1.15 (m, 92H, H-4″–H-21″ and H-4‴–H-8‴), 0.88 (t, J21″,22″ = 6.9 Hz, 6H, H-22″); 13C NMR (150 MHz, C5D5N) δ 174.1 (C-1″ and C-1‴), 131.1 (C-10‴), 96.2 (C-1 and C-1′), 75.3 (C-3 and C-3′), 73.8 (C-2 and C-2′), 72.4 (C-4 and C-4′), 71.9 (C-5 and C-5′), 64.7 (C-6 and C-6′), 34.8 (C-2″ and C-2‴), 33.4 (C-9‴), 32.5, 30.40, 30.36, 30.30, 30.15, 29.98, 29.94, 29.84, 29.75, 23.3 (C-4″–C21″ and C-4‴–C-8‴), 25.6 (C-3″ and C-3‴), 14.7 (C-22″); IR (film): 3343 (broad), 2918, 2850, 1736, 1735, 1466, 1354, 1241, 1175, 1150, 1103, 1077, 1053, 1019, 989, 941 cm−1; HRMS (ESI) m/z calcd for [C88H160O26 + HCOO]−: 1679.1213; obsd.: 1679.1188.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) MeOH, 1
MeOH, 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0 → 41
0 → 41![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 9, v/v) to obtain the final glycolipid 3c as a thin white film (9.6 mg, 0.0055 mmol, quant.). Rf = 0.26 (CH2Cl2
9, v/v) to obtain the final glycolipid 3c as a thin white film (9.6 mg, 0.0055 mmol, quant.). Rf = 0.26 (CH2Cl2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) MeOH, 4
MeOH, 4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, v/v); [α]24.9D = +65.8 (c = 0.1, pyridine); 1H NMR (500 MHz, C5D5N) δ 5.83 (d, J1/1′,2/2′ = 3.4 Hz, 4H, H-1 and H-1′), 5.50–5.43 (m, 2H, H-14‴), 5.06–5.00 (m, 4H, H-5 and H-5′), 4.97–4.91 (m, 4H, H-6a and H-6′a), 4.77 (dd, J6/6′a,b = 11.7, J5/5′,6/6′b = 5.3 Hz, 4H, H-6b and H-6′b), 4.69 (t, J2/2′,3/3′ = J3/3′,4/4′ = 9.1 Hz, 4H, H-3 and H-3′), 4.25 (dd, J2/2′,3/3′ = 9.7 Hz, J1/1′,2/2′ = 3.3 Hz, 4H, H-2 and H-2′), 4.11 (t, J3/3′,4/4′ = J4/4′,5/5′ = 9.4 Hz, 4H, H-4 and H-4′), 2.30–2.22 (m, 8H, H-2″ and H-2‴), 2.10–1.97 (m, 4H, H-13‴), 1.60–1.52 (m, 8H, H-3″ and H-3‴), 1.39–1.03 (m, 108H, H-4″–H-21″ and H-4‴–H-12‴), 0.81 (t, J21″,22″ = 6.9 Hz, 6H, H-22″); 13C NMR (125 MHz, C5D5N) δ 173.5 (C-1″ and C-1‴), 130.5 (C-14‴), 95.7 (C-1 and C-1′), 74.7 (C-3 and C-3′), 73.2 (C-2 and C-2′), 71.9 (C-4 and C-4′), 71.4 (C-5 and C-5′), 64.2 (C-6 and C-6′), 34.3 (C-2″ and C-2‴), 32.9 (C-13‴), 32.0, 29.9, 29.8, 29.8, 29.7, 29.6, 29.5, 29.4, 29.4, 29.2, 22.8 (C-4″–C-21″ and C-4‴–C12‴), 25.1 (C-3″ and C-3‴), 14.1 (C-22″); IR (film): 3353 (broad), 2917, 2849, 2337, 2258, 2224, 2197, 2136, 2021, 1736, 1467, 1376, 1260, 12223, 1175, 1149, 1102, 1076, 1052, 1018, 988, 941, 805, 720, 661, 645, 578, 521, 457, 436 cm−1; HRMS (ESI) m/z calcd for [C98H176O26 + NH4]+: 1764.2822; obsd.: 1764.2879.
1, v/v); [α]24.9D = +65.8 (c = 0.1, pyridine); 1H NMR (500 MHz, C5D5N) δ 5.83 (d, J1/1′,2/2′ = 3.4 Hz, 4H, H-1 and H-1′), 5.50–5.43 (m, 2H, H-14‴), 5.06–5.00 (m, 4H, H-5 and H-5′), 4.97–4.91 (m, 4H, H-6a and H-6′a), 4.77 (dd, J6/6′a,b = 11.7, J5/5′,6/6′b = 5.3 Hz, 4H, H-6b and H-6′b), 4.69 (t, J2/2′,3/3′ = J3/3′,4/4′ = 9.1 Hz, 4H, H-3 and H-3′), 4.25 (dd, J2/2′,3/3′ = 9.7 Hz, J1/1′,2/2′ = 3.3 Hz, 4H, H-2 and H-2′), 4.11 (t, J3/3′,4/4′ = J4/4′,5/5′ = 9.4 Hz, 4H, H-4 and H-4′), 2.30–2.22 (m, 8H, H-2″ and H-2‴), 2.10–1.97 (m, 4H, H-13‴), 1.60–1.52 (m, 8H, H-3″ and H-3‴), 1.39–1.03 (m, 108H, H-4″–H-21″ and H-4‴–H-12‴), 0.81 (t, J21″,22″ = 6.9 Hz, 6H, H-22″); 13C NMR (125 MHz, C5D5N) δ 173.5 (C-1″ and C-1‴), 130.5 (C-14‴), 95.7 (C-1 and C-1′), 74.7 (C-3 and C-3′), 73.2 (C-2 and C-2′), 71.9 (C-4 and C-4′), 71.4 (C-5 and C-5′), 64.2 (C-6 and C-6′), 34.3 (C-2″ and C-2‴), 32.9 (C-13‴), 32.0, 29.9, 29.8, 29.8, 29.7, 29.6, 29.5, 29.4, 29.4, 29.2, 22.8 (C-4″–C-21″ and C-4‴–C12‴), 25.1 (C-3″ and C-3‴), 14.1 (C-22″); IR (film): 3353 (broad), 2917, 2849, 2337, 2258, 2224, 2197, 2136, 2021, 1736, 1467, 1376, 1260, 12223, 1175, 1149, 1102, 1076, 1052, 1018, 988, 941, 805, 720, 661, 645, 578, 521, 457, 436 cm−1; HRMS (ESI) m/z calcd for [C98H176O26 + NH4]+: 1764.2822; obsd.: 1764.2879.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) MeOH, 1
MeOH, 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0 → 41
0 → 41![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 9, v/v) to obtain the title compound 4a as a thin white film (10.1 mg, 0.00063 mmol, 82%). Rf = 0.59 (CH2Cl2
9, v/v) to obtain the title compound 4a as a thin white film (10.1 mg, 0.00063 mmol, 82%). Rf = 0.59 (CH2Cl2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) MeOH, 4
MeOH, 4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, v/v); [α]26.2D = +45.8 (c = 0.1, pyridine); 1H NMR (500 MHz, C5D5N) δ 8.21 (d, J3″,4″ = 8.9 Hz, 4H, H-3″), 6.90 (d, J3″,4″ = 8.9 Hz, 4H, H-4″), 5.87 (d, J1/1′,2/2′ = 3.6 Hz, 2H, H-1/1′), 5.83 (d, J1/1′,2/2′ = 3.6 Hz, 2H, H-1/1′), 5.21–5.17 (m, 2H, H-5), 5.12 (dd, J6a,b = 11.7, J5,6a = 2.1 Hz, 2H, H-6a), 5.05–4.97 (m, 4H, H-5′ and H-6b), 4.90 (dd, J6′a,b = 11.7, J5′,6′a = 3.3 Hz, 2H, H-6′a), 4.77–4.66 (m, 6H, H-3 and H-3′ and H-6′b), 4.31–4.24 (m, 4H, H-2 and H-2′), 4.21 (t, J3,4 = J4,5 = 9.4 Hz, 2H, H-4), 4.10 (t, J3′,4′ = J4′,5′ = 9.4 Hz, 2H, H-4′), 3.84 (t, J6″,7″ = 6.4 Hz, 4H, H-6″), 2.29–2.15 (m, 4H, H-2‴), 1.89–1.77 (m, 4H, H-4‴), 1.70–1.62 (m, 4H, H-7″), 1.60–1.51 (m, 4H, H-3‴), 1.38–1.30 (m, 4H, H-8″), 1.27–1.11 (m, 60H, H-9″–H-22″), 0.80 (t, J22″,23″ = 7.0 Hz, 6H, H-23″); 13C NMR (125 MHz, C5D5N) δ 173.4 (C-1‴), 166.4 (C-1″), 163.2 (C-5″), 131.9 (C-3″), 130.2 (C-5‴), 114.4 (C-4″), 95.8 (C-1/1′), 74.9, 74.7 (C-3/3′), 73.2 (C-2/2′), 71.9, 71.8 (C-4/4′), 71.5, 71.4 (C-5/5′), 68.3 (C-6″), 64.8 (C-6), 64.2 (C-6′), 33.6 (C-2‴), 32.0 (C-4‴), 29.8, 29.8, 29.7, 29.5, 29.5, 22.8 (C-9″–C-22″), 29.2 (C-7″), 26.1 (C-8″), 24.8 (C-3‴), 14.1 (C-23″); IR (film): 3340 (broad), 2921, 2852, 1711, 1605, 1580, 1510, 1453, 1422, 1378, 1276, 1253, 1168, 1148, 1100, 1075, 1049, 1019, 983, 911, 846, 805, 769, 720, 696, 647, 578, 509, 444 cm−1; HRMS (ESI) m/z calcd for [C84H136O28 + NH4]+: 1610.9556; obsd.: 1610.9544.
1, v/v); [α]26.2D = +45.8 (c = 0.1, pyridine); 1H NMR (500 MHz, C5D5N) δ 8.21 (d, J3″,4″ = 8.9 Hz, 4H, H-3″), 6.90 (d, J3″,4″ = 8.9 Hz, 4H, H-4″), 5.87 (d, J1/1′,2/2′ = 3.6 Hz, 2H, H-1/1′), 5.83 (d, J1/1′,2/2′ = 3.6 Hz, 2H, H-1/1′), 5.21–5.17 (m, 2H, H-5), 5.12 (dd, J6a,b = 11.7, J5,6a = 2.1 Hz, 2H, H-6a), 5.05–4.97 (m, 4H, H-5′ and H-6b), 4.90 (dd, J6′a,b = 11.7, J5′,6′a = 3.3 Hz, 2H, H-6′a), 4.77–4.66 (m, 6H, H-3 and H-3′ and H-6′b), 4.31–4.24 (m, 4H, H-2 and H-2′), 4.21 (t, J3,4 = J4,5 = 9.4 Hz, 2H, H-4), 4.10 (t, J3′,4′ = J4′,5′ = 9.4 Hz, 2H, H-4′), 3.84 (t, J6″,7″ = 6.4 Hz, 4H, H-6″), 2.29–2.15 (m, 4H, H-2‴), 1.89–1.77 (m, 4H, H-4‴), 1.70–1.62 (m, 4H, H-7″), 1.60–1.51 (m, 4H, H-3‴), 1.38–1.30 (m, 4H, H-8″), 1.27–1.11 (m, 60H, H-9″–H-22″), 0.80 (t, J22″,23″ = 7.0 Hz, 6H, H-23″); 13C NMR (125 MHz, C5D5N) δ 173.4 (C-1‴), 166.4 (C-1″), 163.2 (C-5″), 131.9 (C-3″), 130.2 (C-5‴), 114.4 (C-4″), 95.8 (C-1/1′), 74.9, 74.7 (C-3/3′), 73.2 (C-2/2′), 71.9, 71.8 (C-4/4′), 71.5, 71.4 (C-5/5′), 68.3 (C-6″), 64.8 (C-6), 64.2 (C-6′), 33.6 (C-2‴), 32.0 (C-4‴), 29.8, 29.8, 29.7, 29.5, 29.5, 22.8 (C-9″–C-22″), 29.2 (C-7″), 26.1 (C-8″), 24.8 (C-3‴), 14.1 (C-23″); IR (film): 3340 (broad), 2921, 2852, 1711, 1605, 1580, 1510, 1453, 1422, 1378, 1276, 1253, 1168, 1148, 1100, 1075, 1049, 1019, 983, 911, 846, 805, 769, 720, 696, 647, 578, 509, 444 cm−1; HRMS (ESI) m/z calcd for [C84H136O28 + NH4]+: 1610.9556; obsd.: 1610.9544.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) MeOH, 1
MeOH, 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0 → 41
0 → 41![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 9, v/v) to obtain the title compound 4b as a thin white film (13.9 mg, 0.0080 mmol, 89%). Rf = 0.42 (CH2Cl2
9, v/v) to obtain the title compound 4b as a thin white film (13.9 mg, 0.0080 mmol, 89%). Rf = 0.42 (CH2Cl2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) MeOH, 4
MeOH, 4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, v/v); [α]20.8D = +60 (c = 0.1, pyridine); 1H NMR (600 MHz, C5D5N) δ 8.30 (d, J3″,4″ = 8.8 Hz, 4H, H-3″), 6.98 (d, J3″,4″ = 8.8 Hz, 4H, H-4″), 5.96 (d, J1/1′,2/2′ = 3.6 Hz, 2H, H-1/1′), 5.93 (d, J1/1′,2/2′ = 3.6 Hz, 2H, H-1/1′), 5.57–5.46 (m, 2H, Z- and E-H-10‴), 5.27 (dd, J4/4′,5/5′ = 10.2 Hz, J5/5′,6/6′-a/b = 5.3 Hz, 2H, H-5/5′), 5.21–5.00 (m, 6H, H-5/5′, H-6a, H-6b, and H-6′a), 4.88–4.76 (m, 6H, H-3, H-3′, and H-6′b), 4.39–4.35 (m, 4H, H-2 and H-2′), 4.29 (dd, J4/4′,5/5′ = 10.2 Hz, J3/3′,4/4′ = 9.3 Hz, 2H, H-4/4′), 4.20 (dd, J4/4′,5/5′ = 10.2 Hz, J3/3′,4/4′ = 9.3 Hz, 2H, H-4/4′), 3.93 (t, J6″,7″ = 6.7 Hz, 4H, H-6″), 2.34–2.30 (m, 4H, H-2‴), 2.11–2.02 (m, 4H, H-9‴), 1.74 (p, J6″,7″ = J7″,8″ = 6.7 Hz, 4H, H-7″), 1.61 (p, J2‴,3‴ = J3‴,4‴ = 7.5 Hz, 4H, H-3‴), 1.44–1.17 (m, 80H, H-8″–H-22″ and H-4‴–H-8‴), 0.88 (t, J22″,23″ = 7.0 Hz, 6H, H-23″); 13C NMR (150 MHz, C5D5N) δ 174.1 (C-1‴), 166.9 (C-1″), 163.7 (C-5″), 132.4 (C-3″), 131.1 (C-10‴), 123.6 (C-2″), 114.9 (C-4″), 96.3 (C-1 and C-1′), 75.4 (C-3/3′), 75.3 (C-3/3′), 73.8 (C-2 and C-2′), 72.5 (C-4/4′), 72.4 (C-4/4′), 72.1 (C-5/5′), 71.9 (C-5/5′), 68.8 (C-6″), 65.3 (C-6/6′), 64.7 (C-6/6′), 34.7 (C-2‴), 33.3 (C-9‴), 32.5, 30.4, 30.27, 30.26, 30.23, 30.0, 29.97, 29.89, 29.80, 29.74, 29.70, 26.6, 25.6, 23.3 (C-3‴–C-8‴ and C-7″–C-22″), 14.6 (C-23″); IR (film): 3343 (broad), 2919, 2851, 1712, 1606, 1279, 1254, 1170, 1149, 1101, 1076, 1050, 1019, 987, 769 cm−1; HRMS (ESI) m/z calcd for [C94H156O28 + NH4]+: 1751.1121; obsd.: 1751.1198.
1, v/v); [α]20.8D = +60 (c = 0.1, pyridine); 1H NMR (600 MHz, C5D5N) δ 8.30 (d, J3″,4″ = 8.8 Hz, 4H, H-3″), 6.98 (d, J3″,4″ = 8.8 Hz, 4H, H-4″), 5.96 (d, J1/1′,2/2′ = 3.6 Hz, 2H, H-1/1′), 5.93 (d, J1/1′,2/2′ = 3.6 Hz, 2H, H-1/1′), 5.57–5.46 (m, 2H, Z- and E-H-10‴), 5.27 (dd, J4/4′,5/5′ = 10.2 Hz, J5/5′,6/6′-a/b = 5.3 Hz, 2H, H-5/5′), 5.21–5.00 (m, 6H, H-5/5′, H-6a, H-6b, and H-6′a), 4.88–4.76 (m, 6H, H-3, H-3′, and H-6′b), 4.39–4.35 (m, 4H, H-2 and H-2′), 4.29 (dd, J4/4′,5/5′ = 10.2 Hz, J3/3′,4/4′ = 9.3 Hz, 2H, H-4/4′), 4.20 (dd, J4/4′,5/5′ = 10.2 Hz, J3/3′,4/4′ = 9.3 Hz, 2H, H-4/4′), 3.93 (t, J6″,7″ = 6.7 Hz, 4H, H-6″), 2.34–2.30 (m, 4H, H-2‴), 2.11–2.02 (m, 4H, H-9‴), 1.74 (p, J6″,7″ = J7″,8″ = 6.7 Hz, 4H, H-7″), 1.61 (p, J2‴,3‴ = J3‴,4‴ = 7.5 Hz, 4H, H-3‴), 1.44–1.17 (m, 80H, H-8″–H-22″ and H-4‴–H-8‴), 0.88 (t, J22″,23″ = 7.0 Hz, 6H, H-23″); 13C NMR (150 MHz, C5D5N) δ 174.1 (C-1‴), 166.9 (C-1″), 163.7 (C-5″), 132.4 (C-3″), 131.1 (C-10‴), 123.6 (C-2″), 114.9 (C-4″), 96.3 (C-1 and C-1′), 75.4 (C-3/3′), 75.3 (C-3/3′), 73.8 (C-2 and C-2′), 72.5 (C-4/4′), 72.4 (C-4/4′), 72.1 (C-5/5′), 71.9 (C-5/5′), 68.8 (C-6″), 65.3 (C-6/6′), 64.7 (C-6/6′), 34.7 (C-2‴), 33.3 (C-9‴), 32.5, 30.4, 30.27, 30.26, 30.23, 30.0, 29.97, 29.89, 29.80, 29.74, 29.70, 26.6, 25.6, 23.3 (C-3‴–C-8‴ and C-7″–C-22″), 14.6 (C-23″); IR (film): 3343 (broad), 2919, 2851, 1712, 1606, 1279, 1254, 1170, 1149, 1101, 1076, 1050, 1019, 987, 769 cm−1; HRMS (ESI) m/z calcd for [C94H156O28 + NH4]+: 1751.1121; obsd.: 1751.1198.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) MeOH, 1
MeOH, 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0 → 41
0 → 41![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 9, v/v) to obtain the title compound 4c as a thin white film (6.2 mg, 0.0033 mmol, 71%). Rf = 0.23 (CH2Cl2
9, v/v) to obtain the title compound 4c as a thin white film (6.2 mg, 0.0033 mmol, 71%). Rf = 0.23 (CH2Cl2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) MeOH, 4
MeOH, 4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, v/v); [α]25.2D = +46.6 (c = 0.1, pyridine); 1H NMR (500 MHz, CDCl3) δ 8.22 (d, J3″,4″ = 8.9 Hz, 4H, H-3″), 6.90 (d, J3″,4″ = 8.9 Hz, 4H, H-4″), 5.89 (d, J1/1′,2/2′ = 3.7 Hz, 2H, H-1/H-1′), 5.85 (d, J1/1′,2/2′ = 3.7 Hz, 2H, H-1/1′), 5.50–5.43 (m, 2H, H-14″), 5.22–5.17 (m, 2H, H-5/5′), 5.13 (dd, J6a,b = 11.7, J5,6a = 2.0 Hz, 2H, H-6a), 5.07–4.81 (m, 6H, H-5/5′ and H-6b and H-6′a), 4.79–4.68 (m, 6H, H-6′, H-3 and H-3′), 4.32–4.25 (m, 4H, H-2 and H-2′), 4.21 (t, J3/3′,4/4′ = J4/4′,5/5′ = 9.1 Hz, 2H, H-4/4′), 4.12 (t, J3/3′,4/4′ = J4/4′,5/5′ = 9.3 Hz, 2H, H-4/4′), 3.85 (t, J6″,7″ = 6.6 Hz, 4H, H-6″), 2.30–2.20 (m, 4H, H-2‴), 2.10–1.97 (m, 4H, H-13‴), 1.67 (p, J6″,7″ = J7″,8″ = 6.7 Hz, 4H, H-7″), 1.55 (p, J2‴,3‴ = J3‴,4‴ = 7.4 Hz, 4H, H-3‴), 1.40–1.05 (m, 96H, H-8″–H-22″ and H-4‴–H-12‴), 0.81 (t, J22″,23″ = 7.0 Hz, 6H, H-23″); 13C NMR (125 MHz, CDCl3) δ 173.5 (C-1‴), 166.4 (C-1″), 163.2 (C-5″), 131.9 (C-3″), 130.6 (C-14‴), 114.4 (C-4″), 95.7 (C-1/1′), 75.6, 75.4 (C-3/3′), 73.3 (C-2 and C-2′), 72.8, 72.6 (C-4/4′), 72.0, 71.4 (C-5/5′), 68.3 (C-6″), 64.2 (C-6 and C-6′), 34.2 (C-2‴), 32.9 (C-13‴), 32.0 (C-7″), 29.8, 29.8, 29.7, 29.5, 29.2, 26.1, 25.1, 22.8 (C-3‴–C-12‴ and C-7″–C-22″), 14.1 (C-23″); IR (film): 3354 (broad), 2948, 2919, 2838, 1647, 1450, 1407, 1253, 1168, 1109, 1078, 1016, 568, 475, 441, 421 cm−1; HRMS: (ESI) m/z calcd for [C102H172O28 + NH4]+: 1864.2407, obsd. = 1864.23732.
1, v/v); [α]25.2D = +46.6 (c = 0.1, pyridine); 1H NMR (500 MHz, CDCl3) δ 8.22 (d, J3″,4″ = 8.9 Hz, 4H, H-3″), 6.90 (d, J3″,4″ = 8.9 Hz, 4H, H-4″), 5.89 (d, J1/1′,2/2′ = 3.7 Hz, 2H, H-1/H-1′), 5.85 (d, J1/1′,2/2′ = 3.7 Hz, 2H, H-1/1′), 5.50–5.43 (m, 2H, H-14″), 5.22–5.17 (m, 2H, H-5/5′), 5.13 (dd, J6a,b = 11.7, J5,6a = 2.0 Hz, 2H, H-6a), 5.07–4.81 (m, 6H, H-5/5′ and H-6b and H-6′a), 4.79–4.68 (m, 6H, H-6′, H-3 and H-3′), 4.32–4.25 (m, 4H, H-2 and H-2′), 4.21 (t, J3/3′,4/4′ = J4/4′,5/5′ = 9.1 Hz, 2H, H-4/4′), 4.12 (t, J3/3′,4/4′ = J4/4′,5/5′ = 9.3 Hz, 2H, H-4/4′), 3.85 (t, J6″,7″ = 6.6 Hz, 4H, H-6″), 2.30–2.20 (m, 4H, H-2‴), 2.10–1.97 (m, 4H, H-13‴), 1.67 (p, J6″,7″ = J7″,8″ = 6.7 Hz, 4H, H-7″), 1.55 (p, J2‴,3‴ = J3‴,4‴ = 7.4 Hz, 4H, H-3‴), 1.40–1.05 (m, 96H, H-8″–H-22″ and H-4‴–H-12‴), 0.81 (t, J22″,23″ = 7.0 Hz, 6H, H-23″); 13C NMR (125 MHz, CDCl3) δ 173.5 (C-1‴), 166.4 (C-1″), 163.2 (C-5″), 131.9 (C-3″), 130.6 (C-14‴), 114.4 (C-4″), 95.7 (C-1/1′), 75.6, 75.4 (C-3/3′), 73.3 (C-2 and C-2′), 72.8, 72.6 (C-4/4′), 72.0, 71.4 (C-5/5′), 68.3 (C-6″), 64.2 (C-6 and C-6′), 34.2 (C-2‴), 32.9 (C-13‴), 32.0 (C-7″), 29.8, 29.8, 29.7, 29.5, 29.2, 26.1, 25.1, 22.8 (C-3‴–C-12‴ and C-7″–C-22″), 14.1 (C-23″); IR (film): 3354 (broad), 2948, 2919, 2838, 1647, 1450, 1407, 1253, 1168, 1109, 1078, 1016, 568, 475, 441, 421 cm−1; HRMS: (ESI) m/z calcd for [C102H172O28 + NH4]+: 1864.2407, obsd. = 1864.23732.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) EtOAc, 1
EtOAc, 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0 → 19
0 → 19![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, v/v) to obtain the title compound 18 as a colourless viscous oil (217 mg, 0.196 mmol, 86%). Rf = 0.69 (pet. ether
1, v/v) to obtain the title compound 18 as a colourless viscous oil (217 mg, 0.196 mmol, 86%). Rf = 0.69 (pet. ether![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) EtOAc, 9
EtOAc, 9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, v/v); [α]21.7D = +76 (c = 1, CH2Cl2); 1H NMR (500 MHz, CDCl3) δ 5.80 (ddt, J16,17a = 16.9 Hz, J16,17b = 10.2 Hz, J15,16 = 6.7 Hz, 2H, H-16), 5.01–4.91 (m, 6H, H-17a, H-17b and H-1), 4.27 (dd, J6a,b = 11.9 Hz, J5,6a = 2.3 Hz, 2H, H-6a), 4.05 (dd, J6a,b = 11.9 Hz, J5,6b = 4.4 Hz, 2H, H-6b), 3.99 (ddd, J4,5 = 9.4 Hz, J5,6b = 4.4, J5,6a = 2.3 Hz, 2H, H-5), 3.90 (t, J2,3 = J3,4 = 8.9 Hz, 2H, H-3), 3.49–3.42 (m, 4H, H-2 and H-4), 2.39–2.28 (m, 4H, H-8), 2.05–2.01 (m, 4H, H-15), 1.64–1.59 (m, 4H, H-9), 1.38–1.27 (m, 20H, H-10–H-14), 0.15, 0.13, 0.13 (3s, 54H, TMS); 13C NMR (125 MHz, CDCl3) δ 173.9 (C-7), 139.3 (C-16), 114.3 (C-17), 94.5 (C-1), 73.6 (C-3), 72.8 (C-2), 72.0 (C-4), 70.9 (C-5), 63.4 (C-6), 34.3 (C-8), 33.9 (C-15), 29.43, 29.38, 29.26, 29.21, 29.0 (C-10–C-14), 24.9 (C-9), 1.2, 1.0, 0.3 (TMS); IR (film): 2971, 2926, 2855, 1741, 1640, 1457, 1415, 1250, 1075, 870, 836 cm−1; HRMS (ESI) m/z calcd for [C52H106O13Si6 + NH4]+: 1124.6587; obsd.: 1124.6597.
1, v/v); [α]21.7D = +76 (c = 1, CH2Cl2); 1H NMR (500 MHz, CDCl3) δ 5.80 (ddt, J16,17a = 16.9 Hz, J16,17b = 10.2 Hz, J15,16 = 6.7 Hz, 2H, H-16), 5.01–4.91 (m, 6H, H-17a, H-17b and H-1), 4.27 (dd, J6a,b = 11.9 Hz, J5,6a = 2.3 Hz, 2H, H-6a), 4.05 (dd, J6a,b = 11.9 Hz, J5,6b = 4.4 Hz, 2H, H-6b), 3.99 (ddd, J4,5 = 9.4 Hz, J5,6b = 4.4, J5,6a = 2.3 Hz, 2H, H-5), 3.90 (t, J2,3 = J3,4 = 8.9 Hz, 2H, H-3), 3.49–3.42 (m, 4H, H-2 and H-4), 2.39–2.28 (m, 4H, H-8), 2.05–2.01 (m, 4H, H-15), 1.64–1.59 (m, 4H, H-9), 1.38–1.27 (m, 20H, H-10–H-14), 0.15, 0.13, 0.13 (3s, 54H, TMS); 13C NMR (125 MHz, CDCl3) δ 173.9 (C-7), 139.3 (C-16), 114.3 (C-17), 94.5 (C-1), 73.6 (C-3), 72.8 (C-2), 72.0 (C-4), 70.9 (C-5), 63.4 (C-6), 34.3 (C-8), 33.9 (C-15), 29.43, 29.38, 29.26, 29.21, 29.0 (C-10–C-14), 24.9 (C-9), 1.2, 1.0, 0.3 (TMS); IR (film): 2971, 2926, 2855, 1741, 1640, 1457, 1415, 1250, 1075, 870, 836 cm−1; HRMS (ESI) m/z calcd for [C52H106O13Si6 + NH4]+: 1124.6587; obsd.: 1124.6597.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) EtOAc, 1
EtOAc, 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0 → 97
0 → 97![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3, v/v) to obtain the title compound 19 as a colourless viscous oil (23.7 mg, 0.022 mmol, 28%). Rf = 0.56 (pet. ether
3, v/v) to obtain the title compound 19 as a colourless viscous oil (23.7 mg, 0.022 mmol, 28%). Rf = 0.56 (pet. ether![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) EtOAc, 9
EtOAc, 9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, v/v); [α]21.9D = +80 (c = 0.1, CH2Cl2); 1H NMR (600 MHz, CDCl3) δ 5.37–5.33 (m, 2H, Z/E-H-16), 4.95 and 4.91 (2d, J1,2 = 3.1 Hz, 2H, H-1), 4.33–4.23 (m, 2H, H-6a), 4.07–4.03 (m, 2H, H-6b), 3.98–3.91 (m, 4H, H-3 and H-5), 3.48–3.38 (m, 4H, H-2 and H-4), 2.33–2.30 (m, 4H, H-8), 2.04–1.95 (m, 4H, H-15), 1.65–1.60 (m, 4H, H-9), 1.33–1.25 (m, 20H, H-10–H-14), 0.15 and 0.14 (2s, 54H, TMS); 13C NMR (150 MHz, CDCl3) δ 174.0 (C-7), 130.5 (C-16), 93.5 (C-1), 73.4 (C-3/5), 72.8 (C-2), 72.5 (C-4), 70.9 (C-3/5), 63.8 (C-6), 34.2 (C-8), 32.5 (C-15), 29.36, 29.26, 28.97, 28.73 (C-10–C-14), 24.9 (C-9), 1.17, 1.03, 0.26 (TMS); IR (film): 2971, 2927, 2855, 1742, 1457, 1406, 1251, 1112, 1077, 873, 842 cm−1; HRMS (ESI) m/z calcd for [C50H102O13Si6 + NH4]+: 1096.6274; obsd.: 1096.6283.
1, v/v); [α]21.9D = +80 (c = 0.1, CH2Cl2); 1H NMR (600 MHz, CDCl3) δ 5.37–5.33 (m, 2H, Z/E-H-16), 4.95 and 4.91 (2d, J1,2 = 3.1 Hz, 2H, H-1), 4.33–4.23 (m, 2H, H-6a), 4.07–4.03 (m, 2H, H-6b), 3.98–3.91 (m, 4H, H-3 and H-5), 3.48–3.38 (m, 4H, H-2 and H-4), 2.33–2.30 (m, 4H, H-8), 2.04–1.95 (m, 4H, H-15), 1.65–1.60 (m, 4H, H-9), 1.33–1.25 (m, 20H, H-10–H-14), 0.15 and 0.14 (2s, 54H, TMS); 13C NMR (150 MHz, CDCl3) δ 174.0 (C-7), 130.5 (C-16), 93.5 (C-1), 73.4 (C-3/5), 72.8 (C-2), 72.5 (C-4), 70.9 (C-3/5), 63.8 (C-6), 34.2 (C-8), 32.5 (C-15), 29.36, 29.26, 28.97, 28.73 (C-10–C-14), 24.9 (C-9), 1.17, 1.03, 0.26 (TMS); IR (film): 2971, 2927, 2855, 1742, 1457, 1406, 1251, 1112, 1077, 873, 842 cm−1; HRMS (ESI) m/z calcd for [C50H102O13Si6 + NH4]+: 1096.6274; obsd.: 1096.6283.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) MeOH, 1
MeOH, 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0 → 17
0 → 17![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3, v/v) to obtain the final glycolipid 5 as a thin white film (9.40 mg, 0.015 mmol, quantitative). Rf = 0.38 (CH2Cl2
3, v/v) to obtain the final glycolipid 5 as a thin white film (9.40 mg, 0.015 mmol, quantitative). Rf = 0.38 (CH2Cl2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) MeOH, 17
MeOH, 17![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3, v/v); [α]22.3D = +80 (c = 0.1, pyridine); 1H NMR (600 MHz, C5D5N) δ 5.90 and 5.91 (2d, 2H, Z/E-H-1), 5.51–5.46 (m, 2H, Z/E-H-16), 5.06–4.98 (m, 4H, H-5 and H-6a), 4.80–4.75 (m, 4H, H-3 and H-6b), 4.32–4.29 (m, 2H, H-2), 4.21–4.11 (m, 2H, H-4), 2.32–2.29 (m, 4H, H-8), 2.12–2.00 (m, 4H, H-15), 1.64 (p, J8,9 = J9,10 = 7.5 Hz, 4H, H-9), 1.38–1.16 (m, 20H, H-10–H-14); 13C NMR (150 MHz, C5D5N) δ 174.0 (C-7), 131.2 (Z/E-C-16), 130.7 (Z/E-C-16), 95.4 (Z/E-C-1), 95.3 (Z/E-C-1), 75.2(C-3), 73.8 (C-2), 72.5 (C-4), 71.9 (C-5), 64.7 (Z/E-C-6), 64.4 (Z/E-C-6), 34.84 (C-8), 33.0 (C-15), 29.8, 29.7, 29.5, 29.4, 29.1 (C-10–C-14), 25.6 (C-9). IR (film): 3346 (broad), 2924, 2853, 1736, 1552, 1442, 1349, 1262, 1183, 1150, 1105, 1077, 1052, 1020, 991, 807 cm−1; HRMS (ESI) m/z calcd for [C32H54O13 + NH4]+: 664.3903; obsd.: 664.3907.
3, v/v); [α]22.3D = +80 (c = 0.1, pyridine); 1H NMR (600 MHz, C5D5N) δ 5.90 and 5.91 (2d, 2H, Z/E-H-1), 5.51–5.46 (m, 2H, Z/E-H-16), 5.06–4.98 (m, 4H, H-5 and H-6a), 4.80–4.75 (m, 4H, H-3 and H-6b), 4.32–4.29 (m, 2H, H-2), 4.21–4.11 (m, 2H, H-4), 2.32–2.29 (m, 4H, H-8), 2.12–2.00 (m, 4H, H-15), 1.64 (p, J8,9 = J9,10 = 7.5 Hz, 4H, H-9), 1.38–1.16 (m, 20H, H-10–H-14); 13C NMR (150 MHz, C5D5N) δ 174.0 (C-7), 131.2 (Z/E-C-16), 130.7 (Z/E-C-16), 95.4 (Z/E-C-1), 95.3 (Z/E-C-1), 75.2(C-3), 73.8 (C-2), 72.5 (C-4), 71.9 (C-5), 64.7 (Z/E-C-6), 64.4 (Z/E-C-6), 34.84 (C-8), 33.0 (C-15), 29.8, 29.7, 29.5, 29.4, 29.1 (C-10–C-14), 25.6 (C-9). IR (film): 3346 (broad), 2924, 2853, 1736, 1552, 1442, 1349, 1262, 1183, 1150, 1105, 1077, 1052, 1020, 991, 807 cm−1; HRMS (ESI) m/z calcd for [C32H54O13 + NH4]+: 664.3903; obsd.: 664.3907.
Biological methods
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1
1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 40 mineral oil
40 mineral oil![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) TWEEN 80
TWEEN 80![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) PBS emulsion and then subsequently diluted with 1
PBS emulsion and then subsequently diluted with 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 PBS containing 50 µg EndoGrade OVA purchased from Lionex Diagnostics and Therapeutics.
1 PBS containing 50 µg EndoGrade OVA purchased from Lionex Diagnostics and Therapeutics.
Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article have been included in the article and in the supplementary information (SI). Supplementary information: flow cytometry data and NMR data for all new compounds. See DOI: https://doi.org/10.1039/d5ob01835h.Acknowledgements
We would like to thank the Health Research Council of New Zealand (HRC, Consolidator Grant, VUW20/928) for funding. We would also like to thank Professor Sho Yamasaki (Osaka University) for kindly providing the Mincle reporter cell lines and Mincle−/− mice.References
- A. F. Weth, E. M. Dangerfield, M. S. M. Timmer and B. L. Stocker, Vaccines, 2024, 12, 1320, DOI:10.3390/vaccines12121320.
- G. K. Pedersen, P. Andersen and D. Christensen, Semin. Immunol., 2018, 39, 4–13, DOI:10.1016/j.smim.2018.10.003.
- R. R. Rapaka, A. S. Cross and M. A. McArthur, Vaccines, 2021, 9, 820, DOI:10.3390/vaccines9080820.
- C. D. Braganza, T. Teunissen, M. S. M. Timmer and B. L. Stocker, Front. Immunol., 2018, 8, 1940, DOI:10.3389/fimmu.2017.01940.
- J. H. Bird, A. A. Khan, N. Nishimura, S. Yamasaki, M. S. M. Timmer and B. L. Stocker, J. Org. Chem., 2018, 83, 7593–7605, DOI:10.1021/acs.joc.7b03269.
- A. J. Foster, M. Nagata, X. Lu, A. T. Lynch, Z. Omahdi, E. Ishikawa, S. Yamasaki, M. S. M. Timmer and B. L. Stocker, J. Med. Chem., 2018, 61, 1045–1060, DOI:10.1021/acs.jmedchem.7b01468.
- A. J. Foster, K. Kodar, M. S. M. Timmer and B. L. Stocker, Org. Biomol. Chem., 2020, 18, 1095–1103, 10.1039/c9ob02397f.
- E. M. Dangerfield, A. T. Lynch, K. Kodar, B. L. Stocker and M. S. M. Timmer, Carbohydr. Res., 2022, 511, 108461, DOI:10.1016/j.carres.2021.108461.
- O. K. Rasheed, G. Ettenger, C. Buhl, R. Child, S. M. Miller, J. T. Evans and K. T. Ryter, Bioorg. Med. Chem., 2020, 28, 115564, DOI:10.1016/j.bmc.2020.115564.
- A. M. S. Riel, V. Rungelrath, T. A. Elwaie, O. K. Rasheed, L. Hicks, G. Ettenger, D. C. You, M. Smith, C. Buhl, W. Abdelwahab, S. M. Miller, A. J. Smith, D. Burkhart, J. T. Evans and K. T. Ryter, Int. J. Mol. Sci., 2024, 25, 10031, DOI:10.3390/ijms251810031.
- K. T. Ryter, G. Ettenger, O. K. Rasheed, C. Buhl, R. Child, S. M. Miller, D. Holley, A. J. Smith and J. T. Evans, J. Med. Chem., 2020, 63, 309–320, DOI:10.1021/acs.jmedchem.9b01598.
- W. M. Abdelwahab, B. Le-Vinh, A. Riffey, L. Hicks, C. Buhl, G. Ettenger, K. J. Jackson, A. M. Weiss, S. Miller, K. Ryter, J. T. Evans and D. J. Burkhart, ACS Appl. Bio Mater., 2024, 7, 3877–3889, DOI:10.1021/acsabm.4c00245.
- S. K. Gupta, N. Parlane, B. Bridgeman, A. T. Lynch, E. M. Dangerfield, M. S. M. Timmer, B. L. Stocker and D. N. Wedlock, PLoS One, 2023, 18, e0278853, DOI:10.1371/journal.pone.0278853.
- B. L. Stocker, E. M. Dangerfield, S. K. Gupta, N. A. Parlane, A. J. Foster, D. N. Wedlock and M. S. M. Timmer, Pure Appl. Chem., 2023, 95, 979–990, DOI:10.1515/pac-2023-0116.
- K. Werninghaus, A. Babiak, O. Groß, C. Hölscher, H. Dietrich, E. M. Agger, J. Mages, A. Mocsai, H. Schoenen, K. Finger, F. Nimmerjahn, G. D. Brown, C. Kirschning, A. Heit, P. Andersen, H. Wagner, J. Ruland and R. Lang, J. Exp. Med., 2009, 206, 89–97, DOI:10.1084/jem.20081445.
- H. Schoenen, B. Bodendorfer, K. Hitchens, S. Manzanero, K. Werninghaus, F. Nimmerjahn, E. M. Agger, S. Stenger, P. Andersen, J. Ruland, G. D. Brown, C. Wells and R. Lang, J. Immunol., 2010, 184, 2756–2760, DOI:10.4049/jimmunol.0904013.
- E. Ishikawa, T. Ishikawa, Y. S. Morita, K. Toyonaga, H. Yamada, O. Takeuchi, T. Kinoshita, S. Akira, Y. Yoshikai and S. Yamasaki, J. Exp. Med., 2009, 206, 2879–2888, DOI:10.1084/jem.20091750.
- K. Kodar, J. L. Harper, M. J. McConnell, M. S. M. Timmer and B. L. Stocker, Immun., Inflammation Dis., 2017, 5, 503–514, DOI:10.1002/iid3.186.
- E. M. Dangerfield, S. Ishizuka, K. Kodar, S. Yamasaki, M. S. M. Timmer and B. L. Stocker, J. Med. Chem., 2024, 67, 5373–5390, DOI:10.1021/acs.jmedchem.3c01840.
- A. Lobato-Pascual, P. C. Saether, S. Fossum, E. Dissen and M. R. Daws, Eur. J. Immunol., 2013, 43, 3167–3317, DOI:10.1002/eji.201343752.
- S. Yamasaki, Eur. J. Immunol., 2013, 43, 3156–3158, DOI:10.1002/eji.201344131.
- Y. Miyake, K. Toyonaga, D. Mori, S. Kakuta, Y. Hoshino, A. Oyamada, H. Yamada, K. Ono, M. Suyama, Y. Iwakura, Y. Yoshikai and S. Yamasaki, Immunity, 2013, 38, 1050–1062, DOI:10.1016/j.immuni.2013.03.010Y.
- Y. Liu, K. Drickamer and M. E. Taylor, Glycobiol, 2024, 34, cwae083, DOI:10.1093/glycob/cwae083.
- R. Kiyotake, M. Oh-hora, E. Ishikawa, T. Miyamoto, T. Ishibashi and S. Yamasaki, J. Biol. Chem., 2015, 290, 25322–25332, DOI:10.1074/jbc.M115.645234.
- M. A. T. P. Manthrirathna, E. M. Dangerfield, S. Ishizuka, A. Woods, B. S. Luong, S. Yamasaki, M. S. M. Timmer and B. L. Stocker, Front. Mol. Biosci., 2022, 9, 1015210, DOI:10.3389/fmolb.2022.1015210.
- B. L. Stocker, K. Kodar, K. Wahi, A. J. Foster, J. L. Harper, D. Mori, S. Yamaski and M. S. M. Timmer, Glycoconjugate J., 2019, 36, 36–78, DOI:10.1007/s10719-018-09857-9.
- S. Ishizuka, J. H. M. van Dijk, T. Kawakita, Y. Miyamoto, Y. Maeda, M. Goto, G. Le Calvez, L. M. Groot, M. D. Witte, A. J. Minnaard, G. A. van der Marel, M. Ato, M. Nagae, J. D. C. Codée and S. Yamasaki, ACS Cent. Sci., 2023, 9, 1388–1399, DOI:10.1021/acscentsci.3c00040.
- A. Furukawa, J. Kamishikiryo, D. Mori, K. Toyonaga, Y. Okabe, A. Toji, R. Kanda, Y. Miyake, T. Ose, S. Yamasaki and K. Maenaka, Proc. Natl. Acad. Sci., 2013, 110, 17438–17443, DOI:10.1073/pnas.1312649110.
- H. Feinberg, S. A. Jégouzo, T. J. Rowntree, Y. Guan, M. A. Brash, M. E. Taylor, W. I. Weis and K. Drickamer, J. Biol. Chem., 2013, 288, 28457–28465, DOI:10.1074/jbc.M113.497149.
- H. Feinberg, N. D. Rambaruth, S. A. Jégouzo, K. M. Jacobsen, R. Djurhuus, T. B. Poulsen, W. I. Weis, M. E. Taylor and K. Drickamer, J. Biol. Chem., 2016, 291, 21222–21233, DOI:10.1074/jbc.M116.749515.
- M. Stephens, K. Keane, S. Roizes, S. Liao and P. V. Weid, Mol. Ther. – Nucleic Acids, 2022, 28, 935–947, DOI:10.1016/j.omtn.2022.05.026.
- B. L. Stocker, A. A. Khan, S. H. Chee, F. Kamena and M. S. M. Timmer, ChemBioChem, 2014, 15, 382–388, DOI:10.1002/cbic.201300674.
- K. Kodar, E. M. Dangerfield, A. J. Foster, D. Forsythe, S. Ishizuka, M. J. McConnell, S. Yamasaki, M. S. M. Timmer and B. L. Stocker, Inflammation, 2023, 46, 1365–1380, DOI:10.1007/s10753-023-01814-5.
- A. T. Lynch, C. Motozono, A. J. Foster, K. Kodar, E. M. Dangerfield, S. Yamasaki, D. N. Wedlock, M. S. M. Timmer and B. L. Stocker, Bioorg. Chem., 2021, 110, 104747, DOI:10.1016/j.bioorg.2021.104747.
- A. Khan, K. Kodar, M. S. M. Timmer and B. L. Stocker, Tetrahedron, 2018, 74, 1269–1277, DOI:10.1016/j.tet.2017.11.076.
- M. Luchner, S. Reinke and A. Milicic, Pharmaceutics, 2021, 13, 142, DOI:10.3390/pharmaceutics13020142.
- B. Su, J. Wang, X. Wang, H. Jin, G. Zhao, Z. Ding, Y. Kang and B. Wang, Vaccine, 2008, 26, 5111–5122, DOI:10.1016/j.vaccine.2008.03.089.
- B. van Den Eeckhout, J. Tavernier and S. Gerlo, Front. Immunol., 2021, 11, 621931, DOI:10.3389/fimmu.2020.621931.
- J. M. Trevejo, M. W. Marino, N. Philpott, R. Josien, E. C. Richards, K. B. Elkon and E. Falck-Pedersen, Proc. Natl. Acad. Sci. U. S. A., 2001, 98, 12162–12167, DOI:10.1073/pnas.211423598.
- H. Clay, H. E. Volkman and L. Ramakrishnan, Immunity, 2008, 29, 283–294, DOI:10.1016/j.immuni.2008.06.011.
- L. L. Laichalk, S. L. Kunkel, R. M. Strieter, J. M. Danforth, M. B. Bailie and T. J. Standiford, Infect. Immun., 1996, 64, 5211–5218, DOI:10.1128/iai.64.12.5211-5218.1996.
- N. Parameswaran and S. Patial, Crit. Rev. Eukaryotic Gene Expression, 2010, 20, 87–103, DOI:10.1615/critreveukargeneexpr.v20.i2.10.
- I. Montgomerie, T. W. Bird, O. R. Palmer, N. C. Mason, T. E. Pankhurst, B. Lawley, L. C. Hernández, R. Harfoot, A. Authier-Hall, D. E. Anderson, K. L. Hilligan, K. H. Buick, N. M. Mbenza, G. Mittelstädt, S. Maxwell, S. Sinha, J. Kuang, K. Subbarao, E. J. Parker, A. Sher, I. F. Hermans, J. E. Ussher, M. E. Quiñones-Mateu, D. Comoletti and L. M. Connor, iScience, 2023, 26, 106256, DOI:10.1016/j.isci.2023.106256.
- M. A. Podestà, C. B. Cavazzoni, B. L. Hanson, E. D. Bechu, G. Ralli, R. L. Clement, H. Zhang, P. Chandrakar, J. M. Lee, T. Reyes-Robles, R. Abdi, A. Diallo, D. R. Sen and P. T. Sage, Nat. Commun., 2023, 14, 7712, DOI:10.1038/s41467-023-43427-4.
- A. A. Khan, S. H. Chee, R. J. McLaughlin, J. L. Harper, F. Kamena, M. S. M. Timmer and B. L. Stocker, ChemBioChem, 2011, 12, 2572–2576, DOI:10.1002/cbic.201100451.
- B. L. Stocker, E. M. Dangerfield, S. K. Gupta, N. A. Parlane, A. J. Foster, D. N. Wedlock and M. S. M. Timmer, Pure Appl. Chem., 2023, 95, 979–990, DOI:10.1371/journal.pone.0278853.
- B. L. Stocker, A. A. Khan, S. H. Chee, F. Kamena and M. S. M. Timmer, ChemBioChem, 2014, 15, 382–388, DOI:10.1002/cbic.201300674.
- S. Yamasaki, E. Ishikawa, M. Sakuma, H. Hara, K. Ogata and T. Saito, Nat. Immunol., 2008, 9, 1179–1188, DOI:10.1038/ni.1651.
- B. L. Stocker, K. Kodar, K. Wahi, A. J. Foster, J. L. Harper, D. Mori, S. Yamaski and M. S. M. Timmer, Glycoconjugate J., 2019, 36, 36–78, DOI:10.1007/s10719-018-09857-9.
Footnote |
| † These authors contributed equally. |
| This journal is © The Royal Society of Chemistry 2026 |