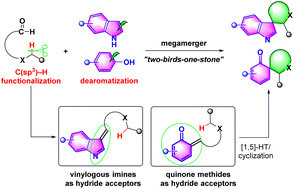
Dearomatization and direct C(sp3)–H functionalization represent two hot research fields in organic synthesis, the merging of which is among the most challenging but appealing topics. The emerging hydride transfer-involved dearomatization reaction can achieve this goal in an efficient redox-neutral manner, and encouraging progress has been made in this area during the past decade. Herein, we would like to give a timely account of the topic, and this review is categorized according to the hydride acceptors, namely vinylogous imines and quinone methides, which initiate the cascade hydride transfer/dearomative cyclization process to dearomatize indoles and phenols, respectively. Moreover, the limitations, challenges and future directions are also pointed out.