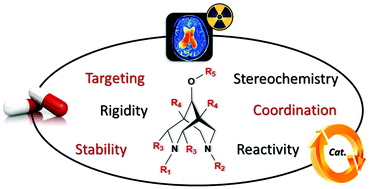
Bispidones (3,7-diazabicyclo[3.3.1]nonan-9-one) are bicyclic analogues of the natural antiarrhythmic agent, spartein. They can straightforwardly be obtained from two successive Mannich reactions. Reduction of the ketone gives the corresponding bispidol. Substituted bispidones and bispidols offer a large playground by varying the substituents, the configuration of the carbon atoms in position 2 and 4 as well as the conformation of the bicycle. While chair–boat conformers display a strong affinity for κ-opioid receptors, chair–chair bispidines provide adaptable coordination spheres for transition metal and rare-earth ions. Because of their very rich coordination chemistry, substituted bispidines have emerged in various applications of coordination chemistry, such as catalysis, magnetism and medical imaging.