Self-assembly beyond semifluorinated alkanes in a semifluorinated benzene derivative†
Abstract
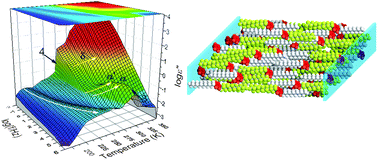
Tethering amphiphiles symmetrically to a small benzene core – in analogy to semifluorinated alkanes (SFAs) – gives rise to self-assembly far more rich than any previously investigated SFA. The modulation of intermolecular interactions between hydrocarbon and fluorocarbon chains as well as additional π–π interactions of the cores are thought to be responsible for the rich self-assembly. The system is investigated with respect to thermodynamics, structure and dynamics, respectively, with differential scanning calorimetry, X-ray scattering and dielectric spectroscopy. The mechanical and nano-mechanical properties are studied with rheology and Brillouin light scattering covering a broad frequency range and spatial resolution. Starting from lower temperatures distinct phases are identified consisting of nanophase separated domains comprising crystalline or melted fluorocarbon/hydrocarbon domains. At intermediate temperatures lamellar phases are identified with molecular and supramolecular order. At higher temperatures domains composed of thermodynamically unfavorable yet kinetically mixed configurations exist. The latter phase is prone to annealing as these trapped configurations convert from locally segregated to more homogeneously mixed molecules. At higher temperatures a quasi-isotropic phase is formed composed of orientationally correlated dipoles.

 Please wait while we load your content...
Please wait while we load your content...