Solid-state excimer emission of o-carborane derivatives and applications for stimuli-responsive luminescent materials
Kazuo
Tanaka

Department of Polymer Chemistry, Graduate School of Engineering, Kyoto University, Katsura, Nishikyo-ku, Kyoto 615-8510, Japan. E-mail: tanaka@poly.synchem.kyoto-u.ac.jp; Tel: +81-75-383-2604
First published on 15th May 2024
Abstract
o-Carborane is an icosahedral cluster containing boron and has been paid attention as a versatile platform for constructing stimuli-responsive materials with solid-state luminescence properties by suppressing aggregation-caused quenching (ACQ), which is the emission annihilation process observed in condensed states, such as concentrated solutions, solids and films. Non-specific intermolecular interactions in the condensed states resulting in ACQ are disturbed by the spherical boron cluster. More recently, it has been demonstrated from several reports that o-carborane is capable of assisting solid-state excimer emission in aryl-modified o-carboranes. Moreover, some of these materials provide unique stimuli-responsiveness. In the commodity design for solid-state luminescent materials, o-carboranes are responsible for disturbing intermolecular interactions and subsequently presenting solid-state luminescence. On the other hand, excimer emission, which is inducible from the intermolecular photophysical process, has been discovered in the solid state based on o-carborane materials. In this manuscript, recent progress in stimuli-responsive luminescent materials via excimer formation will be described. Moreover, as the further potential of o-carborane materials, recent findings on the photo-salient effect which can be induced through excimer formation followed by photoreaction will also be explained.
1. Introduction
Demands for luminescent organic materials are still high for the development of next-generation technologies for optoelectronic organic devices. As examples, luminescent sensors are proposed. Stimuli-responsive materials that change their optical properties, such as emission color, in response to environmental changes or external stimuli can be expected to be directly applied as a sensing material. Therefore, many researchers have devoted their efforts to explore new molecular skeletons. However, there is a problem to be solved in the development of luminescent devices and materials based on organic luminescent dyes. In general, the luminescence properties of organic dyes are critically lowered in the solid state due to ACQ caused by non-specific intermolecular interactions. In particular, in using a chemical sensor, it is assumed that luminescent dyes are generally used as a film form. Therefore, to obtain advanced luminescent sensors based on organic materials, it is essential to overcome ACQ without loss of stimulus responsiveness.According to recent studies, aryl-modified o-carboranes are recognized as promising scaffolds for constructing stimuli-responsive materials with solid-state luminescence properties.1–5o-Carborane is an icosahedral cluster composed of two carbon and ten boron atoms.6–9 Owing to the steric hindrance, ACQ can be often avoided. Consequently, various solid-state luminescent materials with unique stimuli-responsiveness have been developed by utilizing o-carborane as “an element-block”, which is the functional building block containing inorganic elements for constructing advanced luminescent materials, such as chemical and environmental sensors.10–14 When linked with an aromatic ring at the carbon atom in the cluster, the o-carborane unit can work as an electron-acceptor group and form robust electron-donor–acceptor interactions.15 As a result, the intramolecular charge transfer (CT) process followed by intense emission can occur. It should be noted that since intermolecular interactions are disturbed even in the condensed state because of the steric hindrance of the spherical structure of the o-carborane unit, ACQ can be suppressed, and intense emission with the CT character can be observed in the solid and aggregation states.
Furthermore, various environment-sensitive and/or stimuli-responsive luminescent materials have been developed based on the molecular motions in the excited state concerning o-carborane units, such as the C–C bond expansion in the cluster and the intramolecular rotation of the cluster relative to the substituent group. For example, the former is known to be involved in the expression of aggregation-induced emission (AIE) behaviors.16 In the solution state, non-radiative decay proceeds via the extension of the C–C bond, resulting in emission annihilation.17 It should be noted that when the AIE properties were firstly discovered from aryl-modified o-carboranes, it was assumed that emission annihilation might be induced by C–C bond vibration.18 Recently, according to quantum calculation data, we have suggested that C–C bond extension rather than vibration is responsible for emission annihilation.17 On the other hand, in the aggregation state, because of structural restriction caused by molecular packing, luminescence is observed. It is known that AIE is expressed by combining these behaviors in solution and aggregation.16 That is, most organic luminescent dyes emit light only in dilute solutions. Conversely, to achieve AIE behavior, it is necessary to exhibit luminescence enhancement in the solid state simultaneously with luminescence quenching in the solution state. In each process, different factors are important and sometimes are different. For example, molecular motion can cause luminescence quenching. To induce solid-state luminescence, it is necessary not only to suppress molecular motion but also to inhibit intermolecular interactions. If a single molecule can exhibit these two behaviors, that molecule can be called an AIE-active molecule. The C–C bond extension in o-carborane causes solution quenching and is therefore directly involved in AIE behavior.
The latter issue concerning intramolecular rotation is a crucial factor related to the luminescence properties such as color in the solid state. We have reported that aryl-modified o-carboranes undergo twisted intramolecular charge transfer (TICT) even in crystals.19 When the C–C bond and the π-conjugated system are present in the same plane, emission from the locally excited (LE) state of the aromatic ring is observed, while in the excited state, the o-carborane unit rotates 90° to form the TICT state and subsequently emission is observed. Based on this mechanism, various luminochromic solid materials have been developed. For instance, we succeeded in tuning the thermochromic luminescence properties by introducing substituents around the connection bond between aromatics and the o-carborane unit to modulate the height of the rotation barrier and permitting rotational motion at a certain temperature.20–22 These solid-state luminescence properties can basically be explained by electronic properties in the single molecular level such as structural relaxation and rotational motion of the cluster units.
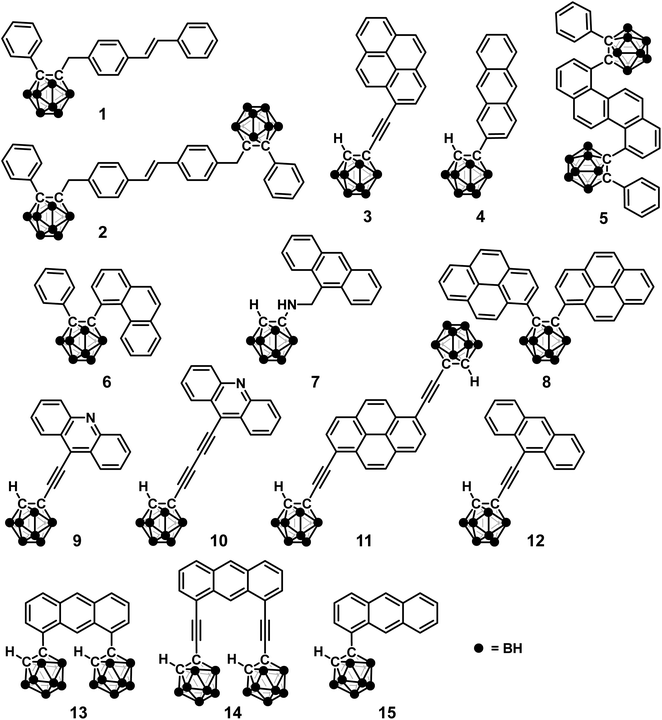
As described above, the o-carborane unit contributes to the isolation of the molecules even in the condensed state followed by solid-state luminescence properties. In contrast, recently, it has become possible to control the electronic properties in the solid state and to induce luminochromic behaviors triggered by through-space intermolecular interactions. Significantly, excimer emission has been revealed, and diverse unique behaviors have been discovered (Fig. 1). Excimers are short-lived dimeric molecules that form only when one of the dimer components is in an excited state. Each molecule needs to be close to each other; however ACQ often occurs in the solid state, competing with conventional emission processes in a single molecule. Interestingly, some materials showed solid-state excimer emission and exhibited more sensitive responsiveness than conventional stimulus-responsive materials. From these results, applications in next-generation sensing technology are envisioned. In this manuscript, recent examples on aryl-modified o-carboranes having solid-state excimer emission are surveyed. Mechanisms in these materials and attempts to control the emission properties will be illustrated. Moreover, the photoreaction via the excimer state and subsequently the photo-salient effect, where crystal freaks show physical movements such as jumping, are explained. In the conventional design of solid-state luminescent materials, intermolecular interactions have been recognized as a cause of ACQ that should be avoided; meanwhile solid-state excimer emission properties assisted by o-carborane could lead to the establishment of new molecular design strategies for controlling properties and creating new materials.
 | ||
| Fig. 1 Schematic illustration of stimuli-responsive luminochromic behaviors via intramolecular charge transfer (ICT) and excimer states. | ||
2. Stimuli-responsive luminochromic materials
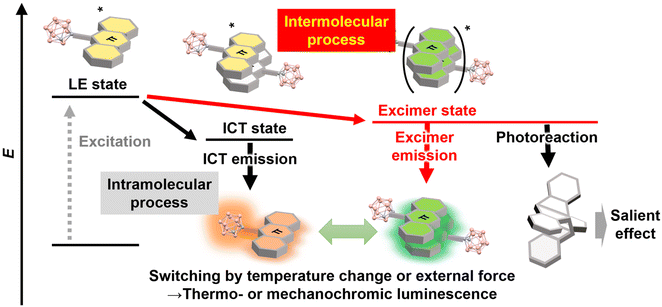
In Fig. 2, the series of aryl-modified o-carboranes presenting solid-state excimer emission are listed. Solid-state excimer emission was firstly reported by Núñez et al. in 2017 from the stilbene-modified o-carboranes 1 and 2.23 In the solution state, both compounds show LE emissions; meanwhile enhanced emissions were observed in the condensed states, such as film, aggregate and solid states. Finally, aggregation-induced excimer emission was obtained from aryl-modified o-carboranes.In 2017, it was found that pyrene-modified o-carborane linked by the ethynyl linker 3 exhibits excimer emission in the crystalline state (Fig. 3).24 At room temperature, orange emission is attributable to CT emission, similar to the conventional aryl-modified o-carboranes; meanwhile at 77 K, LE and excimer emission were observed. Corresponding to the mechanism change, the emission color turned to green. This is the first example in which the intramolecular emission process through the CT state is switched to the intermolecular excimer process in the solid state by temperature (Fig. 1). After this report, it was also found that 3 can exhibit excimer emission accompanying aggregation in the presence of a small amount of water in organic solvents.25 Conventional molecular sensors for measuring water content in solvents are broadly classified into those that measure extremely small amounts of water (less than 1%) and those that measure relatively high amounts of water (more than 1%). It was shown that all these water contents can be measured by the color change when using the pyrene-ethynyl-o-carborane molecule.
 | ||
| Fig. 3 Thermochromic luminescence caused by switching from CT to excimer emission by cooling to 77 K. Reprinted with permission from ref. 24. Copyright 2017 Wiley-VCH Verlag GmbH & Co. KGaA, Weinheim. | ||
In 2018, Guo and Jia et al. reported mechanochromic luminescence by eliciting solid-state excimer emission triggered by mechanical forces from the modified o-carborane derivative 4 attached to the 2-position of anthracene.26 When the LE-derived blue luminescent crystal of 4 was ground with a weak force, the emission band from the excimer was observed around 530 nm, and the emission color of the solid sample changed to yellow. When the crystalline sample was ground with a stronger force, the emission band from the TICT state was enhanced around 625 nm, resulting in pink emission. Interestingly, the combination of three different emission bands resulted in complex emission colors and sensitive responses.
In 2018, Stavrinou and Heeney et al. also reported that the distorted chrysene derivative 5 modified with two phenyl-o-carboranes exhibits solid-state excimer emission.27 Since it is the first excimer-emitting molecule based on chrysene, it can be said that they successfully demonstrated that o-carborane is a building block for deriving not only CT emission but also excimer emission. After that, they reported that three emission bands, LE, CT and excimer, were observed in the phenanthrene-substituted o-carborane derivative 6.28
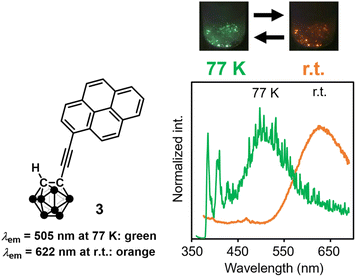
Starting from these studies, solid excimers were found in various aryl-modified o-carboranes. Mechanochromic luminescence was detected from the amine-linked o-carborane derivative 7,29 and thermochromic luminescence was observed from the pyrene-modified molecule 8.30 As mentioned above, pyrene-ethynyl-o-carborane provides excimer emission at low temperature,24 whereas in acridine-ethynyl-o-carborane 9, dimer formation was reinforced by hydrogen bonds based on the polarization of the o-carborane, and excimer luminescence in the yellow region was observed at room temperature (Fig. 4a and b).31 Based on thermochromic luminescence properties, thermometers were demonstrated by using 9 as a paint-type sensor (Fig. 4c). As shown in 10, when the linker was additionally extended, excimer luminescence was obtained in a relatively longer wavelength region (orange), and gradual changes in the emission color were observed with temperature changes.32 It is suggested that the extension of π-conjugated systems through ethynyl linkers is responsible for the longer wavelength emission band of 10. Moreover, it is proposed that the stacking area could be expanded. As a result, a narrower energy gap, followed by longer-wavelength emission can be induced.
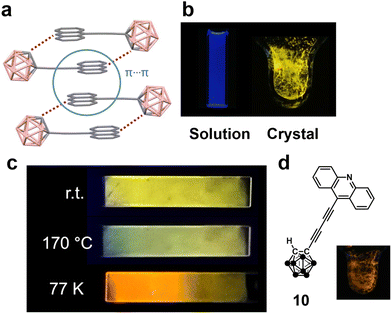 | ||
| Fig. 4 (a) The origin of excimer emission in the crystal packing of 9. (b) Appearances of 9 in solution and crystals. (c) Thermochromic luminescence properties of 9 on the glass substrate. Reprinted with permission from ref. 30. Copyright 2019 Wiley-VCH Verlag GmbH & Co. KGaA. (d) Solid-state excimer emission of 10. Adapted with permission from ref. 31. Copyright 2021 American Chemical Society. | ||
Bis-o-carborane-substituted compounds showed unique stimuli-responsiveness toward environmental changes. Sensitivity toward external forces was enhanced as shown in the pyrene derivative 11.33 Although the mono-substituted anthracene-ethynyl-o-carborane 12 showed mechanochromic luminescence only by a strong force and 3 was insensitive, the emission color of 11 was changed just by touching the crystalline sample of the bis-substituted one with a spatula (Fig. 5). It is proposed that the crystal packing could be sparse because of the two o-carboranes and the sensitivity might be improved. In addition to thermochromic luminescence observed in the modified anthracene 13,34 it was discovered that the direction of wavelength changes in mechanochromic luminescence was converted in 14 having the ethynyl linkers (Fig. 6).35 In the case of 13, the bathochromic emission band was observed after grinding; meanwhile the hypsochromic emission band appeared from 14. Moreover, self-recovery of the luminescent color proceeded in 13 at room temperature but in 14 it proceeded at 323 K. In the bis-o-carborane systems, similar to a previous report in which an excimer was not mentioned,36 the self-recovering behaviors from the ground color to the pristine color were observed after the mechanical treatments.34 This fact supports the speculation that bis-o-carborane substitutions could form sparse crystalline packings followed by sensitive stimuli-responsiveness.
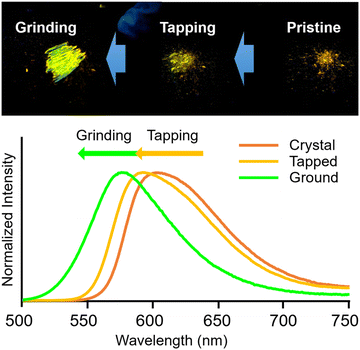 | ||
| Fig. 5 Multi-step mechanochromic luminescence of 11 by tapping and subsequently grinding with the pristine crystalline sample. Adapted with permission from ref. 32. Copyright 2022 Elsevier. | ||
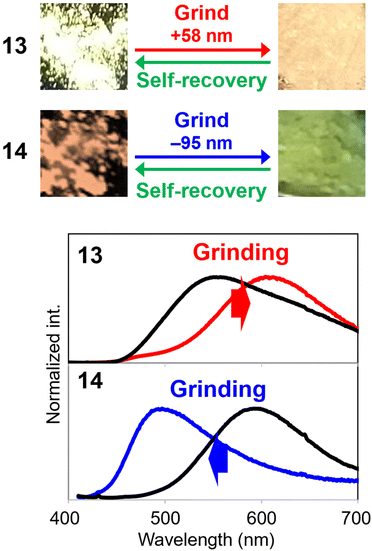 | ||
| Fig. 6 Mechanochromic luminescence and self-recovery of 13 and 14. Reprinted with permission from ref. 34. Copyright 2023 Wiley-VCH Verlag GmbH & Co. KGaA, Weinheim. | ||
From carboranes other than o-carborane, a solid-state excimer and its related luminochromic behaviors have been observed. AIE behaviors were observed from the anthracene-modified m-carborane derivative with the methylene linker, and these emission bands were similar to those of the excimer of anthracene.37 On the other hand, excimer formation worked as a quenching process in the directly connected anthracenyl m- and p-carboranes.38 Solid-state excimer emission was also found in nido-carborane derivatives, and it has also been reported that the optical properties were controllable with counter cations.39
3. Tuning of emission color
Next, modulation of emission color is described. From acridine-ethynyl-o-carborane, excimer emission was able to be induced at room temperature by assisting hydrogen bond formation between the nitrogen atom and electron deficient C–H bond in o-carborane40 in the dimer unit.31 Based on this chemistry, tuning of luminescence properties concerning excimer emission was achieved. Three kinds of derivatives, Me0, Me2, and Me8, with variable numbers of methyl groups in the o-carborane unit were synthesized (Fig. 7).41 All derivatives exhibited a columnar crystal structure including a dimer as a basic unit and showed significant solid-state emission. In addition, although the number of methyl groups hardly affect the electronic properties of each derivative according to the series of measurements in solution, it exhibited various emission colors from light blue to green in the solid state. According to the optical parameters obtained from measurements and lifetime analyses, it was concluded that excimer emission can be obtained. From the X-ray structural analyses, it was clarified that the distance between the π-planes and the overlapping area is slightly different because of the steric hindrance of the methyl group. In Me8, there is no overlap between dimers, and intermolecular interactions in the crystal packing have different strengths and ratios among Me0, Me2, and Me8. There are two types of π–π interactions in the crystal structure, “intra-dimer” and “inter-dimer”, and the emission color is determined by the degree of combination of these interactions.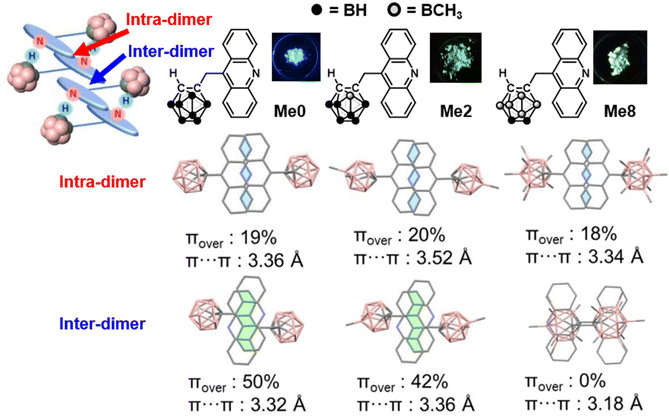 | ||
| Fig. 7 Structure–property relationship between the stacking areas and color of excimer emission in the o-carborane derivatives with different numbers of methyl groups in the cluster unit. Reprinted with permission from ref. 40. Copyright 2023 Wiley-VCH Verlag GmbH & Co. KGaA, Weinheim. | ||
4. Photo-triggered dimerization and the photo-salient effect
In the above sections, excimer emission properties and modulation of these behaviors have been explained. Meanwhile, there also exists the photochemical process in which a photoreaction occurs from the excimer state through non-radiative decay (Fig. 8a). As a representative example, the dimerization reaction of anthracene in the crystal can occur through this process. In particular, it has recently been discovered that an o-carborane-modified anthracene derivative exhibits photo-triggered dimerization and a subsequent photosalient effect.42 In this section, the mechanism is described.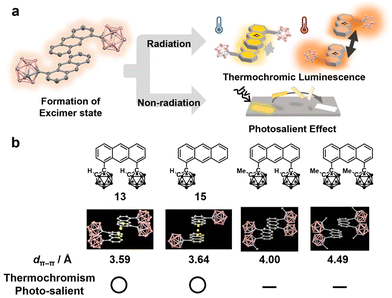 | ||
| Fig. 8 (a) Schematic illustration of photochemical processes from the excimer state. (b) Structure and property relationship on the photo-salient effect based on the o-carborane-modified anthracene derivatives. Reprinted with permission from ref. 41. Copyright 2024 Wiley-VCH Verlag GmbH & Co. KGaA, Weinheim. | ||
Four kinds of o-carborane-modified anthracene derivatives involving 13 and 15 were synthesized with different substitution positions and numbers of the o-carborane unit (Fig. 8b).42 It was found that 13 and 15 form the characteristic crystal structure involving the columnar π-stacking structures of the anthracene units. In particular, it was shown that these crystals can exhibit the photosalient effect accompanied by photochemical [4 + 4] cycloaddition and temperature-dependent photophysical dual-emission properties including excimer emission of anthracene. These behaviors were classified as non-radiative and radiative deactivation pathways via the excimer formation in the excited state. It was proposed that distortion energy is accumulated in each crystal freak by the photo-triggered dimerization inside crystal packings. As a result, these distortions could be released by jumping and physical movements. The photo-salient effect might be paid attention as a key material for a light-driven actuator.
Based on anthracene analogs, we aimed to clarify the structural factors which are required for the excimer formation.42 As a result, we found that two structural features are essential. The first is that aromatic ring moieties need to stack and form a columnar structure. The second is that the distance between stacking is less than a threshold value. It is assumed that the latter threshold should be changed by replacing the structure of the aromatic ring, but these two structural features are considered to be general factors.
5. Conclusion and perspective
In this manuscript, recent progress in solid-state excimer luminescence from a series of aryl-modified o-carboranes is described. So far, there have been many reports on CT emission-based stimuli-responsive materials, and their luminescence mechanism including luminochromic behaviors is completed in the single molecule. Furthermore, the photochemical reaction via the excimer state followed by the photo-salient effect was also introduced. In contrast, the use of o-carborane has opened up the control of luminescence properties through the intermolecular processes. Although it is difficult to precisely control the crystal structure of organic compounds, it was possible to modulate the emission color by adjusting the molecular distribution in the crystal packing to some extent by changing the substituents and replacing carbons in the molecular skeleton with other elements. Hence, the use of o-carborane platforms for expressing intermolecular processes is expected to lead to the creation of multichromic optical materials with more sensitive and diverse responsiveness.Conflicts of interest
There are no conflicts to declare.Acknowledgements
This work was partially supported by The Asahi Glass Foundation and JSPS KAKENHI Grant Number 24K01570.References
- R. Núñez, Mà. Tarrés, A. Ferrer-Ugalde, F. F. de Biani and F. Teixidor, Chem. Rev., 2016, 116, 14307–14378 CrossRef PubMed.
- S. Mukherjee and P. Thilagar, Chem. Commun., 2016, 52, 1070–1093 RSC.
- D. Shimoyama and F. Jäkle, Aggregate, 2022, 3, e149 CrossRef CAS.
- X. Li, H. Yan and Q. Zhao, Chem. – Eur. J., 2016, 22, 1888–1898 CrossRef CAS PubMed.
- B. Dong, A. Oyelade and J. A. Kelber, Phys. Chem. Chem. Phys., 2017, 19, 10986–10997 RSC.
- J. M. Oliva, L. Serrano-Andrés, D. J. Klein, P. v. R. Schleyer and J. Michl, Int. J. Photoenergy, 2009, 292393 Search PubMed.
- R. Núñez, I. Romero, F. Teixidor and C. Viñas, Chem. Soc. Rev., 2016, 45, 5147–5173 RSC.
- R. Núñez, M. Tarrés, A. Ferrer-Ugalde, F. F. d. Biani and F. Teixidor, Chem. Rev., 2016, 116, 14307–14378 CrossRef PubMed.
- D. Zhao and Z. Xie, Coord. Chem. Rev., 2016, 314, 14–33 CrossRef CAS.
- K. Tanaka, M. Gon, S. Ito, J. Ochi and Y. Chujo, Coord. Chem. Rev., 2022, 472, 214779 CrossRef CAS.
- K. Tanaka and Y. Chujo, Polym. J., 2023, 55, 353–364 CrossRef CAS.
- M. Gon, K. Tanaka and Y. Chujo, Bull. Chem. Soc. Jpn., 2019, 92, 7–18 CrossRef CAS.
- M. Gon, K. Tanaka and Y. Chujo, Polym. J., 2018, 50, 109–126 CrossRef CAS.
- Y. Chujo and K. Tanaka, Bull. Chem. Soc. Jpn., 2015, 88, 633–643 CrossRef CAS.
- K. Nishino, H. Yamamoto, K. Tanaka and Y. Chujo, Org. Lett., 2016, 18, 4064–4067 CrossRef CAS PubMed.
- J. Ochi, K. Tanaka and Y. Chujo, Angew. Chem., 2020, 132, 9841–9845 CrossRef PubMed.
- J. Ochi, K. Tanaka and Y. Chujo, Dalton Trans., 2021, 50, 1025–1033 RSC.
- K. Kokado and Y. Chujo, Macromolecules, 2009, 42, 1418–1420 CrossRef CAS.
- H. Naito, K. Nishino, Y. Morisaki, K. Tanaka and Y. Chujo, Angew. Chem., Int. Ed., 2017, 56, 254–259 CrossRef CAS PubMed.
- J. Ochi, K. Yuhara, K. Tanaka and Y. Chujo, Chem. – Eur. J., 2022, 28, e202200155 CrossRef CAS PubMed.
- J. Ochi, T. Yanagihara, K. Tanaka and Y. Chujo, Phys. Chem. Chem. Phys., 2023, 25, 11839–11844 RSC.
- K. Nishino, K. Tanaka and Y. Chujo, Asian J. Org. Chem., 2019, 8, 2228–2232 CrossRef CAS.
- A. Ferrer-Ugalde, J. Cabrera-González, E. J. Juárez-Pérez, F. Teixidor, E. Pérez-Inestrosa, J. M. Montenegro, R. Sillanpää, M. Haukka and R. Núñez, Dalton Trans., 2017, 46, 2091–2104 RSC.
- K. Nishino, H. Yamamoto, K. Tanaka and Y. Chujo, Asian J. Org. Chem., 2017, 6, 1818–1822 CrossRef CAS.
- K. Nishino, H. Yamamoto, J. Ochi, K. Tanaka and Y. Chujo, Chem. – Asian J., 2019, 14, 1577–1581 CrossRef CAS PubMed.
- X. Wu, J. Guo, Y. Cao, J. Zhao, W. Jia, Y. Chen and D. Jia, Chem. Sci., 2018, 9, 5270–5277 RSC.
- A. V. Marsh, N. J. Cheetham, M. Little, M. Dyson, A. J. P. White, P. Beavis, C. N. Warriner, A. C. Swain, P. N. Stavrinou and M. Heeney, Angew. Chem., Int. Ed., 2018, 57, 10640–10645 CrossRef CAS PubMed.
- A. V. Marsh, M. Little, N. J. Cheetham, M. J. Dyson, M. Bidwell, A. J. P. White, C. N. Warriner, A. C. Swain, I. McCulloch, P. N. Stavrinou and M. Heeney, Chem. – Eur. J., 2021, 27, 1970–1975 CrossRef CAS PubMed.
- C. Xu, T. Li, J. Miao, K. Liu, Y. Nie, G. Liu and X. Jiang, New J. Chem., 2023, 47, 4448–4456 RSC.
- W. Wu, J. Guo, J. Zhao, Y. Che, D. Jia and Y. Chen, Dyes Pigm., 2018, 154, 44–51 CrossRef.
- J. Ochi, K. Tanaka and Y. Chujo, Eur. J. Org. Chem., 2019, 2984–2988 CrossRef CAS.
- J. Ochi, K. Tanaka and Y. Chujo, Inorg. Chem., 2021, 60, 8990–8997 CrossRef CAS PubMed.
- H. Yamamoto, J. Ochi, K. Yuhara, K. Tanaka and Y. Chujo, Cell Rep. Phys. Sci., 2022, 3, 100758 CrossRef CAS.
- K. Yuhara, K. Tanaka and Y. Chujo, Mater. Chem. Front., 2022, 6, 1414–1420 RSC.
- K. Yuhara and K. Tanaka, Chem. – Eur. J., 2023, 29, e202301189 CrossRef CAS PubMed.
- D. Tu, P. Leong, Z. Li, R. Hu, C. Shi, K. Y. Zhang, H. Yan and Q. Zhao, Chem. Commun., 2016, 52, 12494–12497 RSC.
- M. Chaari, Z. Kelemen, D. Choquesillo-Lazarte, N. Gaztelumendi, F. Teixidor, C. Viñas, C. Nogués and R. Núñez, Biomater. Sci., 2019, 7, 5324–5337 RSC.
- A. V. Marsh, M. J. Dyson, N. J. Cheetham, M. Bidwell, M. Little, A. J. P. White, C. N. Warriner, A. C. Swain, I. McCulloch, P. N. Stavrinou, S. C. J. Meskers and M. Heeney, Adv. Electron. Mater., 2020, 6, 2000312 CrossRef CAS.
- T. Yanagihara and K. Tanaka, Adv. Opt. Mater., 2023, 11, 2300492 CrossRef CAS.
- A. M. Spokoyny, C. W. Machan, D. J. Clingerman, M. S. Rosen, M. J. Wiester, R. D. Kennedy, C. L. Stern, A. A. Sarjeant and C. A. Mirkin, Nat. Chem., 2011, 3, 590–596 CrossRef CAS PubMed.
- J. Ochi, K. Tanaka and Y. Chujo, Angew. Chem., Int. Ed., 2023, 62, e202214397 CrossRef CAS PubMed.
- K. Yuhara and K. Tanaka, Angew. Chem., Int. Ed., 2024, 63, e202319712 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2024 |