Direct evidence of Fe(v) and Fe(iv) intermediates during reduction of Fe(vi) to Fe(iii): a nuclear forward scattering of synchrotron radiation approach†
Abstract
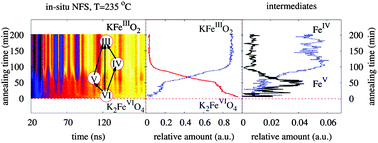
Identification of unstable high-valent iron species in electron transfer reactions of ferrate(VI) (FeVIO42−, Fe(VI)) has been an important challenge in advancing the understanding of the oxidative mechanisms of ferrates. This paper presents the first example of distinguishing various phases differing in the valence state of iron in the solid state reduction of Fe(VI) to Fe(III) oxides at 235 °C using hyperfine parameters, isomer shift and hyperfine magnetic field, obtained from nuclear forward scattering of synchrotron radiation (NFS). The NFS technique enables a fast data accumulation resulting in high time resolution of in situ experiments. The results suggest a reaction mechanism, involving Fe(V) and Fe(IV) species, in the thermal decomposition of K2FeO4 to KFeO2. The present study opens up an approach to exploring the unambiguous identification of Fe(VI), Fe(V), Fe(IV), and Fe(III) in electron-transfer reaction mechanisms of ferrates in solid and aqueous phase systems.

 Please wait while we load your content...
Please wait while we load your content...