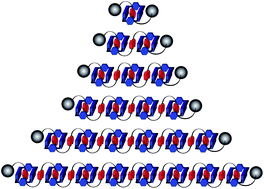
We describe in detail a strategy for creating foldamers in which interactions between mechanically interlocked components dictate the single-molecule assembly of a folded secondary structure. This unique folding motif is based on a flexible polyether dumbbell bearing 1,5-dioxynaphthalene (DNP) donors, which folds its way through a series of cyclobis(paraquat-p-phenylene) (CBPQT4+) acceptor rings in a serpentine fashion to enable extended donor–acceptor (D–A) stacking between DNP and the electron-poor 4,4′-bipyridinium (BIPY2+) units in CBPQT4+. These oligorotaxanes can be prepared in a wide range of sizes, with molecular weights up to >15 000 Da, on account of novel one-pot reactions we developed to generate the necessary oligo-DNP precursors. The product distributions from the final kinetically controlled stoppering reactions are highly biased towards oligorotaxanes in which approximately half of the DNP units are encircled by rings, a fact which can be rationalized if the dominant solution-state structures of the pseudorotaxane precursors reflect the solid-state superstructures of analogous compounds, which express 50% recognition site occupancy because of their proclivity to pack into continuous D–A–D–A stacks. The presence of well-defined folded structures in solution have been confirmed by 1H NMR spectroscopy in CD3CN. Moreover, we discovered an empirical selection rule forbidding CBPQT4+ rings to occupy adjacent DNP sites, which elegantly explains both the product distributions and the 1H NMR spectra. Depending on their adherence to this selection rule, all of the oligorotaxanes belong to one of three families: whereas ‘Confused’ oligorotaxanes adopt multiple translational isomers that satisfy the rule and ‘Frustrated’ species cannot obey it at all, members of the ‘Happy’ family each express only one rule-compliant ‘Goldilocks’ isomer. The NMR spectra of these oligorotaxanes also shed light on their dynamics; rapid 180° rotations of DNP units cause pairs of heterotopic BIPY2+ protons in the accompanying CBPQT4+ rings to exchange sites, giving rise to time-averaged signals. This process, which we term ‘superrotation’, will apply much more generally to other mechanically interlocked systems.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...