Open Access Article
Open Access ArticleNanowaste management in laboratory practice – a technical guideline
Fabienne
Schwab
 *ab,
Barbara
Rothen-Rutishauser
*ab,
Barbara
Rothen-Rutishauser
 a,
Aline
Scherz
c,
Thierry
Meyer
d,
Bedia Begüm
Karakoçak
a,
Aline
Scherz
c,
Thierry
Meyer
d,
Bedia Begüm
Karakoçak
 a and
Alke
Petri-Fink
a and
Alke
Petri-Fink
 *a
*a
aAdolphe Merkle Institute, University of Fribourg, Fribourg, Switzerland. E-mail: alke.fink@unifr.ch; Tel: +41 26 300 9501
bLucerne University of Applied Sciences and Arts, Lucerne, Switzerland. E-mail: fabienne.schwab@alumni.ethz.ch; Tel: +41 78 736 0019
cAdministrative Direction, University of Fribourg, Fribourg, Switzerland
dGroup of Chemical and Physical Safety, Ecole Polytechnique Fédérale de Lausanne, Lausanne, Switzerland
First published on 19th February 2026
Abstract
The management of waste containing nanomaterials, here termed “nanowaste”, is not yet sufficiently regulated at both national and international levels. Here, we provide a comprehensive review of nanowaste management, situating laboratory practices within the broader regulatory context, with special attention to the Basel Convention. We then discuss potential measures to avoid or minimize nanowaste, options for nanowaste recovery and recycling, nanowaste risk assessment, protective equipment, categorization, collection, storage, labeling, and ultimately, disposal. Building on occupational health legislation and practical laboratory experience, we propose initial technical guidelines tailored to research environments and small and medium-sized enterprises (SMEs), where relatively small but highly diverse volumes of nanowaste are generated. To illustrate their application, we supplement four case studies, including the disposal of orphaned samples, small-scale and large-scale disposal, and nanomaterial spills. To strengthen trust in nanotechnology and support responsible innovation, we emphasize the importance of applying the precautionary principle and treating nanowaste with unknown properties as potentially hazardous to both human health and the environment. By explicitly linking laboratory-level practices with national and international frameworks, this guideline serves both as an immediately applicable tool for researchers and SMEs and as a technical foundation to inform future Basel Convention Technical Guidelines on nanowaste.
Environmental significanceEngineered nanomaterials are increasingly embedded in industrial products and research applications, yet their end-of-life management remains insufficiently defined. While research laboratories generate comparatively small volumes of nanowaste, these streams are highly diverse, frequently novel, and often lack comprehensive hazard data, creating uncertainty for human and environmental protection. Early-stage handling practices are therefore critical to preventing unintentional environmental release and occupational exposure. This tutorial provides structured, precautionary guidance for nanowaste management in laboratories and SMEs, linking bench-level decision-making with international waste governance frameworks. By strengthening waste practices at the point of material innovation, the work supports responsible nanotechnology development and contributes to long-term environmental stewardship. |
Introduction and objectives
Waste containing nanomaterials, here termed nanowaste, can no longer be considered an exception. Engineered nanomaterials already represent a significant and rapidly growing1–3 fraction of global material flows, with a market growth rate of 18.2% between 2016 and 2021.4 In 2010, it was estimated that ∼63–91% of globally produced nanomaterials, i.e., up to 281![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 metric tons, ended up in landfills,2 raising concerns about long-term environmental pollution, while highlighting the need for proportionate and precautionary management of nanowaste generated in research environments. These estimates do not even consider the emerging category of nanomaterials such as nanoplastics, which is now recognized as a major environmental issue.2,5 It should be noted that the environment itself contains enormous quantities of naturally occurring nanoparticles that play key roles in geochemical and biological processes; however, engineered nanomaterials differ in composition, functionality, and exposure pathways, necessitating specific consideration of their waste management.6,7
000 metric tons, ended up in landfills,2 raising concerns about long-term environmental pollution, while highlighting the need for proportionate and precautionary management of nanowaste generated in research environments. These estimates do not even consider the emerging category of nanomaterials such as nanoplastics, which is now recognized as a major environmental issue.2,5 It should be noted that the environment itself contains enormous quantities of naturally occurring nanoparticles that play key roles in geochemical and biological processes; however, engineered nanomaterials differ in composition, functionality, and exposure pathways, necessitating specific consideration of their waste management.6,7
While large-scale industrial sources contribute to these numbers, a notable share of engineered nanomaterials is produced in research laboratories and SMEs, where novel materials are designed, synthesized, and tested. These environments are therefore not only a starting point of innovation but also at the frontline of nanowaste generation and its safe management during the lifecycle in a laboratory.
An estimated 1–20% of the engineered nanomaterials are produced by academia and other research institutions worldwide, primarily at laboratory scale and for research purposes.2 Research laboratories not only generate nanowaste but also train the next generation of scientists in how to handle it. As such, they play a dual role: developing disposal practices for novel materials and transmitting these practices to a broader community. To date, however, beyond the commercially available CEN/TS 17275:2018 standard (which mainly addresses terminology and definitions)8 and a number of gap analyses and classification proposals,3,9–14 there are no practical guidelines for nanowaste management tailored to laboratory practice.3,15 This lack of guidance is striking because laboratories often deal with small but diverse waste streams that require rapid, pragmatic solutions.
Unlike conventional laboratory waste streams, nanowaste can consist of complex mixtures, including (but not limited to) carbon, polymers (nanoplastics), and heavy metals, and can have unique shapes and structures as in the cases of nanotubes and -wires, fullerenes (buckyballs), graphene (sheets), and quantum dots (quasi-spherical) with partially still unknown hazard properties (Fig. S1).1,16 Due to the different properties such as the nanomaterials' size, composition, surface charge, shape, and dissolution rate in aqueous and lipid-rich environments, nanowaste can exhibit vastly different physicochemical behaviors and hazards compared to the bulk material waste, and therefore requires case-by-case assessment and, where appropriate, different treatment compared to regular chemical or biological waste. Importantly, not all nanomaterials are intrinsically hazardous; however, in research settings, hazard data are often incomplete, justifying a precautionary approach.
To ensure the sustainable and safe utilization of nanomaterials in research environments, a thorough evaluation of potential hazards and risks is required throughout their entire lifecycle, from resource extraction and production to use and disposal. Anticipating risks early allows researchers to make informed design and handling choices and to embed mitigation strategies directly into material development, thereby protecting people and the environment.17 In recent years, user-friendly methods for assessing precautionary risk levels in laboratory settings have been developed and should be applied at the earliest stages of research.4,18 Building on this foundation, the present tutorial review examines existing and missing regulations and provides practical guidance for nanowaste disposal, including risk assessment, classification, labeling, collection, storage, transport, recycling, deactivation, and elimination.
Our focus is on practices grounded in the precautionary principle and applicable to a broad range of nanomaterials: nanoparticles with external dimensions between 1–100 nm, other nano-objects such as graphene and most multi-walled carbon nanotubes ≤500 nm in diameter, as well as larger hazardous respirable fibers (2–50 μm long, ≤3 μm in diameter, aspect ratio L/D > 3).19,20 The disposal practices discussed here are intended for intentionally engineered nanoparticles and nanomaterials.18 For brevity, the term “nanomaterials” will be used throughout, while “nanowaste” will refer to waste generated from nanomaterial production, nano-enabled products at the end of life, and materials contaminated by nanomaterials (see SI section “What is Nanowaste?”).9,15
This tutorial review serves as a technical guideline for researchers, laboratory managers, technical staff, safety officers, regulatory specialists, and other stakeholders seeking in-depth information on nanowaste management in laboratories. At the same time, it is written with a second audience in mind: policymakers and regulators, especially those engaged with the Basel Convention. Laboratory practices cannot be considered in isolation, since effective nanowaste management must ultimately align with international governance frameworks. Although the Convention regulates the movement and disposal of hazardous wastes, it does not yet explicitly address nanomaterials, leaving considerable ambiguity. By situating laboratory-level practices in this wider context, the tutorial serves a dual purpose: providing actionable guidance for researchers while contributing to the policy discussions that may lead to future Technical Guidelines on nanowaste under the Basel Convention. It should be noted that waste streams from consumer products are not addressed here, as the scope of this guideline is explicitly limited to laboratory and SME environments.
History and regulatory context – the Basel Convention
Early awareness of nanoscale hazards
Before nanotechnology was defined as such, particulate materials with nanoscale fractions were already recognized as hazardous in the workplace. A prominent example is cellulose fibers from sugarcane or cotton dust in textile factories, which caused respiratory disease and prompted some of the earliest workplace hygiene measures more than a century ago.21,22 This historical case illustrates that nanoscale exposures are not new phenomena, even if their classification as “nanomaterials” is recent. Today, research laboratories and SMEs occupy a similar frontier: they handle novel engineered nanomaterials with unknown hazards and risks and must therefore implement protective practices in the absence of explicit regulatory guidance. This also illustrates that prior occupational hygiene knowledge (‘prior art’) on dusts and fibers remains directly relevant and can inform today's nanowaste management practices.The Basel Convention as global framework
Internationally, the Basel Convention provides the principal framework for controlling hazardous waste management and transboundary movement, as illustrated in Fig. 1.23–25 Entering into force in 1992, it regulates most categories of hazardous chemical waste, with exceptions such as radioactive materials and ship discharges covered elsewhere.25 The Convention has been continuously amended through annual meetings of its parties,23 and most recently strengthened by the 2019 Ban Amendment, which prohibits the export of hazardous waste from OECD and EU countries to less wealthy nations.23,24,26 (As of 2024, the Convention has 191 Parties, the majority of which are low- and middle-income countries, underscoring the importance of guidance that is applicable across diverse resource settings.) Although primarily focused on large-scale industrial waste flows, the Convention indirectly shapes how all countries, including those hosting research institutions and SMEs, classify and dispose of waste, even if nanowaste is not yet explicitly mentioned. | ||
| Fig. 1 Graphical abstract of the Basel Convention. Adapted from the 2019 edition of the Basel Convention.25 White boxes show the decision scheme in article 1 of the convention. Colored boxes represent the three main waste groups (hazardous, non-hazardous, other waste), all of which can implicitly cover nanomaterials. Page numbers refer to the 2019 edition. Abbreviations: UN = United Nations. | ||
How nanowaste fits — implicitly25
To analyze how nanowaste might be covered, it is necessary to examine the Basel annexes. Annex I lists 45 waste streams and constituents requiring control, including laboratory waste from R&D and teaching (Y14) and dusts and fibers (Y36). Annex II covers “other waste”, such as household and plastic waste; annex III defines hazard characteristics (e.g. explosive H1, toxic chronic H11, ecotoxic H12); and annexes VIII and IX list specific hazardous and non-hazardous wastes. (For completeness, though not directly relevant for nanowaste, annex IV lists disposal operations, annex V specifies the information required for notification and movement documents, annex VI sets arbitration procedures, and annex VII lists parties subject to additional obligations.) Article 1 of the Convention specifies that wastes subject to transboundary movement are deemed hazardous if they fall within annex I and display annex III hazard properties, or if a country of import/export deems them hazardous.26 In this sense, nanowaste can already fall into any of these categories, i.e., hazardous, non-hazardous, or “other waste”, depending on its composition and behavior.Case example: nano-TiO2
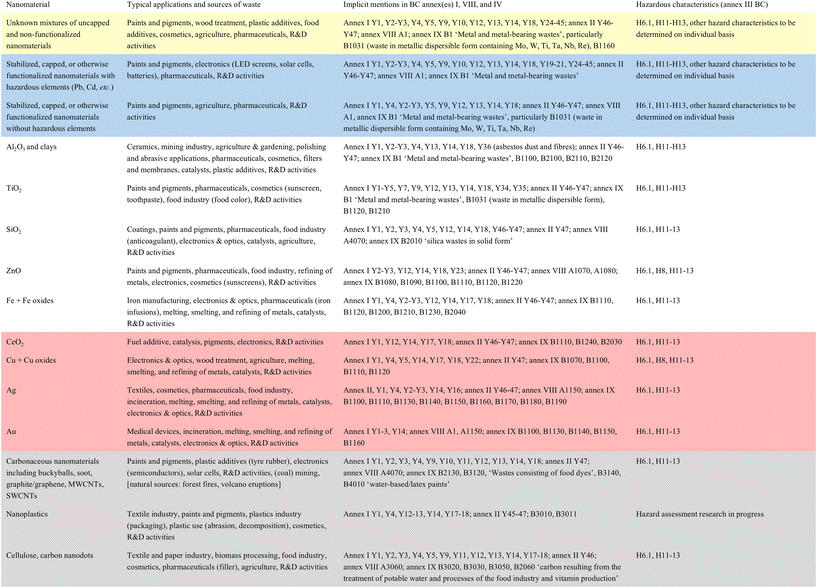
The ambiguity can be illustrated by the widely used nanomaterial titanium dioxide (nano-TiO2), found in paints, cosmetics, pharmaceuticals, and sunscreens (Table 1). While not explicitly mentioned in the Convention,25 nano-TiO2 can occur in multiple annex I streams (e.g., Y1–Y5, Y7, Y9, Y12–Y14, Y18, Y34–Y35), as well as annex II household/plastics categories (Y46–Y47). Many of these streams can also display hazard properties under annex III (e.g., H6.1, H11–H13). Therefore, major waste streams and constituents discussed in the Basel Convention implicitly cover nano-TiO2 waste. This regulatory gray area raises a practical question: can nanowaste in annex II (e.g., household waste) or annex IX (e.g., B1120 spent catalysts) be genuinely treated as non-hazardous?For laboratory and SME environments, this means that a common material such as nano-TiO2 can shift between hazardous and non-hazardous status depending on interpretation, leaving researchers and safety officers with considerable uncertainty. This ambiguity complicates compliance and may result in inconsistent practices across institutions and countries. Similar ambiguities exist for other nanomaterials. As with nano-TiO2, they are implicitly covered by the Basel Convention due to their presence in annex I waste streams and their annex III hazard characteristics. As a result, many nanomaterials are de facto included in both annex VIII (hazardous wastes) and annex IX (non-hazardous wastes). Table 1 summarizes these nanomaterials, their implicit coverage under the Basel Convention, and their hazard characteristics.
Regulatory gaps and calls for action
Awareness of these gaps is not new. Since 2015,27 reports have highlighted the uncertainty of nanowaste coverage under the Basel Convention,15,28 and in 2019 the parties issued Decision BC-14/14, inviting submissions on national activities and strategies for nanowaste management.26 These inputs, alongside work by the OECD (Organisation for Economic Co-operation and Development), UNITAR (United Nations Institute for Training and Research), WHO (World Health Organization), and ILO (International Labour Organization), are now being discussed at the level of Basel's Open-ended Working Group. The objective is to reduce ambiguity and provide clearer international guidance. Two main pathways exist:1. Explicit inclusion of nanowaste in one or more Basel annexes. This would carry legal weight but would require consensus among all parties, a process that could take many years.
2. Development of Technical Guidelines on nanowaste, modeled on earlier guidance for used oil (1995), tires (2011), and persistent organic pollutants (2019).23,29 These guidelines, while not legally binding, provide practice-oriented standards that accelerate harmonized implementation across countries.
Implications for laboratories and SMEs
For research and SME environments, the current situation means that nanowaste is implicitly regulated but poorly defined, leaving safety officers and regulators with significant interpretive leeway. This makes pragmatic laboratory guidance essential, as provided in this tutorial. At the same time, laboratory-level practices, once standardized, could directly inform the drafting of future Basel Technical Guidelines on nanowaste. In this way, the research and SME community can play a bottom-up role in shaping national and international policy, while ensuring immediate protection of workers and the environment.Pragmatic approaches to determine nanowaste risks
When regulatory data about a nanomaterial are missing, as is often the case in research laboratories, the risk of a material for people and the environment may need to be assessed under extreme time pressure, sometimes for many nanomaterials without available safety data sheets (SDS).14 In such cases, performing full nanomaterial hazard and risk assessments4,18 can be challenging in practice, particularly due to data gaps and time constraints. Instead, it is often necessary to understand the potential hazards while handling nanowaste, based on the waste type (Table S1).16 In general, the risk posed by nanowaste decreases as the proportion of “free” nanomaterials diminishes: from dry powders (high risk), to aerosols, suspensions, and finally composites (lower risk).14,16 If more time is available, and to establish more general rules for the laboratory, calculating a risk quotient can help to judge the safety of a nanomaterial1 or a nanowaste disposal practice.14 The risk quotient (sometimes called risk characterization ratio) is the ratio of the predicted environmental concentration (PEC) of the nanomaterial to its “predicted no-effect concentration (PNEC)”.1 A lower risk quotient (<1) indicates a lower level of concern.1 Where human toxicity data are unavailable, a safety factor (typically 10–1000) must be applied. A worked case study demonstrating how to calculate the risk quotient for transmission electron microscopy (TEM) grid disposal with nanomaterials is provided in the SI.Nanomaterials suspected of having a high risk of causing harm to human health or the environment, for example, respirable fibers 2–50 μm long with diameters ≤3 μm, or with aspect ratios L/D > 3,19,20 as well as nanomaterials containing highly hazardous substances, should be treated as hazardous nanowaste according to the precautionary principle.30,31 For nanowaste with concerning or entirely unknown toxicological properties, a more detailed risk assessment can be conducted based on the bulk material's SDS, acknowledging the assumptions involved, as described by Groso and co-workers18 and Buitrago and co-workers.4 For larger-scale applications, manufacturers and traders can also use governmental tools such as Switzerland's Precautionary Matrix for synthetic nanomaterials32 to evaluate nanomaterial-specific health and environmental risks, the EU's NANoREG decision-support frameworks, or the OECD's guidance tools. Such resources are currently more available in high-income countries; in less wealthy nations, equivalent software is often lacking, which may exacerbate disparities in safe nanowaste management.
Guiding principles for the management of nanowaste
We recently proposed six guiding principles for the management of nanowaste,14 based on the precautionary principle. Guiding principles are essential because regulations remain incomplete and often lack the specificity required for managing nanowaste; these principles provide a clear framework that laboratories and SMEs can apply immediately to reduce risks and ensure precautionary handling:1. Use an inclusive definition for nanomaterials including particles ≤500 nm in diameter and 2–50 μm in length and ≤3 μm in diameter for hazardous respirable fibers.19,20
2. Apply safe-by-design principles when designing and engineering novel nanomaterials.
3. Minimize upstream nanowaste generation, i.e., avoid producing nanowaste, including safe-by-design and in situ approaches where feasible.
4. Reduce aerosolization by suspending/embedding nanomaterials in liquid/solid matrices.
5. Treat unavoidable nanowaste as a hazardous material.
6. Where proven effective, apply low-energy inactivation routes (e.g., hydrolysis, embedding into solid matrices). Otherwise, render nanowaste inert by precipitation or pyrolysis at 1200–1300 °C in specialized waste treatment facilities.33
Determining categories
Nanowaste categories differ between laboratories, but it is generally useful to start by distinguishing liquid from solid waste. In line with global regulations on hazardous materials,34 waste can then be further classified according to material properties and hazards (solid, liquid, gas, biohazardous, toxic by inhalation, flammable, oxidizing, corrosive, acidic, basic, organic, inorganic).14,34,35 Importantly, classification must also account for whether nanomaterials are uncapped or capped/functionalized, as surface modifications (e.g., stabilizing agents, coatings, capping ligands) strongly influence their aggregation behavior, persistence, and hazard potential. A detailed discussion of these aspects is provided in our earlier work.4 The key rule is that only chemically compatible nanowastes may be collected in the same container, and the container itself must be compatible with the waste.34 Ideally, hazard classification should be carried out on an individual material basis. However, given the rapidly expanding diversity of nanomaterials and frequent data gaps, this is not always feasible. In such cases, grouping by material class (e.g., metal oxides, carbon-based nanomaterials, polymers) and applying the precautionary principle provides a pragmatic alternative until more specific data become available. Further subcategories depend on specific research activities and hazards. Special categories apply to radioactive materials, biohazardous contaminated materials (biosafety class ≥2), or other highly hazardous substances.36 Details on chemical incompatibilities and container requirements are provided in the section “collection and storage”.Although it is legally possible to define very large numbers of categories,14,34,35 this can be counterproductive: too many categories reduce compliance, especially if linked to lengthy documentation or non-functional websites (dead links). Instead, simple and standardized decision tools, such as decision matrices (Fig. 2), can help laboratory staff categorize nanowaste quickly and ensure integration with existing chemical waste management procedures. Where hazard information is lacking, the precautionary scheme in Fig. 2 provides a pragmatic fallback, applying the precautionary principle to ensure that nanowaste with unknown properties is provisionally treated as hazardous until further data are available.
For liquid nanowaste, categories include purely aqueous nanowaste, aqueous nanowaste mixed with biohazardous materials, nanowaste in non-halogenated organic solvents, and nanowaste in halogenated organic solvents. Nanomaterials in suspension are categorized as liquid waste even if minor flocculation or precipitation occurs, since the dominant phase remains liquid. Solid waste refers to powders or other dry materials and can be categorized into regular solid chemical or nanowaste and solid nanowaste mixed with biohazardous materials (Fig. 2 and Table 2).
| Type of waste | Example of a collection container |
|---|---|
| Solids | |
| Nanomaterial-contaminated only | White nanowaste bin |
| Nanomaterial-contaminated metal sharps (e.g., needles, knife blades) | White nanowaste bin (wrapped in paper before disposal) |
| Biohazardous solids contaminated with nanomaterials | Blue bins with yellow lid |
| Liquids | |
|---|---|
| Aqueous suspensions of nanomaterials, including sterilized liquid biohazardous nanowaste | White 10 L canister |
Special cases include sharps (e.g., needles, razor blades), glass (microscopy slides, pipettes, solvent bottles), paper and cardboard, solid metals, and highly hazardous materials (Table 2). Highly hazardous waste, such as electron microscopy fixatives (sodium cacodylate, osmium tetroxide, uranyl acetate, lead citrate), or chromium and mercury compounds, must be collected separately by specially trained personnel, under the supervision of a local safety officer (safety advisor).34 In some cases, nanowaste can be converted into less problematic categories.14,37 For example, sterilization (autoclaving, heat treatment, addition of disinfectants) can transform liquid biohazardous nanowaste into regular liquid nanowaste. Where autoclaving is used, closed-container systems must be available to prevent aerosolization of nanoparticles;14 since most laboratories lack such autoclaves, chemical treatments are usually preferred. For example, >600 mg L−1 peracetic acid (e.g., 2% Sekusept™ CLASSIC) can sterilize liquid nanowaste containing biohazardous materials. Embedding nanowaste into concrete has been proposed as another innovative inactivation method,38,39 but studies show that nanomaterials may be released during aging processes.40 Thus, new methods require independent validation before adoption. Similarly, embedding reactive nanomaterials into inexpensive polymers, or admixing inexpensive monomers to form a polymeric matrix, has been suggested as a potentially more sustainable containment option. However, as with concrete, such approaches require independent validation to ensure long-term safety before adoption.
When nanoscale properties are lost
In cases where the nanoscale nature of a material is demonstrably lost, the waste may be reassigned into conventional hazardous or biohazard categories rather than being treated as nanowaste. This loss of nanoscale properties can occur naturally (e.g., dissolution, aggregation, or the degradation of liposomes and micelles) or through deliberate treatment (e.g., sterilization, embedding, pyrolysis). Such reclassification provides a pragmatic route for reducing the burden of nanowaste management when nanospecific risks no longer apply. However, the transformation must be well-documented and scientifically justified, as incomplete or unstable degradation processes may still release nanoscale fractions.Collection and storage
The size, shape, color, and number of nanowaste containers are not legally standardized and therefore vary between laboratories (see section categories, Fig. 3 and 4).14 What is mandatory is that all containers are clearly labeled and compliant with at least national standards for hazardous goods storage, ideally also United Nations (UN) standards, to ensure safety and readiness for transport.34 As a further precaution, nanowaste should always be stored in a well-ventilated space, such as a chemical cupboard or cabinet, to minimize chronic personnel exposure and avoid the accumulation of volatile fumes. A key principle is the strict separation of chemically incompatible nanowaste during storage.14,35 This prevents dangerous incidents such as heat generation, fire, explosions, toxic fume release, or spills. The relevant incompatibilities can usually be derived from the bulk material's properties or, for liquid nanowaste, from the solvent used. Substances that must always be stored separately include strong acids, bases, redox-active materials, and solvents prone to forming explosive mixtures. In cases of uncertainty, global34,35 and local36,41 legislation may provide detailed guidance on hazardous goods management. Further examples and incompatibility rules are summarized in the SI. To minimize workplace risk, storage of flammable liquids or nanomaterial powders should be kept as low as practicable, especially where explosive dusts may form (see SI, “Specific Rules for Flammable Nanowaste”). | ||
| Fig. 3 Representative containers for nanowaste storage and transport. Adapted from Schwab et al. with permission from Springer Nature, Nature Nanotechnology, 18(4):317–321, copyright 2023.14 a: White bin for solid nanowaste. b: Blue bin for solid biohazardous nanowaste. c: Example of dual storage/transport container with ADR34 biohazard pictogram. The United Nations code 3291 stands for not otherwise specified clinical waste. d: White canister for aqueous nanowaste. e: Green container for nanomaterials in halogenated solvents. Contents must always be explicitly listed on the label.35,36 Waste colors shown are examples and not legally binding. For more details on labeling, refer to the section ‘labels for storage and transport’ and the SI section ‘details on labeling nanowaste’. | ||
At this stage, coordination with an institutional safety officer (or safety advisor) is essential, as transport regulations for hazardous goods differ from storage regulations and require professional oversight (see section Labels for storage and transport). Finally, because each laboratory faces unique conditions, waste practices should be tailored to local infrastructure and hazards. For example, Table 2 illustrates the containers used for four typical nanowaste categories in our own research setting.
Solid nanowaste
All solid materials containing nanomaterials or hazardous chemicals must be disposed of in sealable, approved containers such as the plastic bins shown in Fig. 3a–c. Once the containers reach ∼90% capacity, they must be sealed and transferred to a central special waste collection room (Fig. 4). Under no circumstances may solid nanowaste be discarded with ordinary household waste. Special solid waste includes any items that have come into contact with (bio)hazardous chemicals or nanomaterial powders or suspensions, such as empty chemical containers, disposable pipettes and tips, wipes, Petri dishes, chamber slides, microscope slides with cells, or well plates. If cleaning or sterilization is not possible, these items must be placed in designated nanowaste containers (Fig. 3a–c). Improper disposal (e.g., placing chemical-contaminated containers into unventilated municipal glass bins) poses serious risks to laboratory and cleaning personnel. For ergonomic reasons, solid waste may first be collected in smaller zipper bags inside the working area (e.g., a fume hood). However, once removed from the laboratory, these bags must be double-bagged and sealed before transfer to larger nanowaste containers, to prevent contamination during handling and transport. Direct disposal into containers located in the same laboratory does not require double-bagging. Solid biohazardous nanowaste (materials containing both nanomaterials and biohazardous material of class ≥2) poses additional challenges. Even trace amounts of biohazardous material require separate collection in containers labeled explicitly as “solid biohazardous nanowaste” (Fig. 3b, c and 2; Table 2). Unless a specialized autoclave capable of sterilizing closed containers is available (rare outside biological or medical laboratories), this waste can only be eliminated by certified special waste treatment companies. Once free of nanomaterials, biohazardous waste may be transferred to conventional autoclavable biohazardous waste streams (Table 2).Liquid nanowaste
Liquid suspensions containing nanomaterials must always be collected separately in a research laboratory setting. Ideally, they are stored in ∼10 L canisters that are easy to transport and clearly labeled as liquid nanowaste (Fig. 3d and e). These containers must be kept in a well-ventilated area, as they are frequently opened for disposal and may release aerosols or volatile chemicals. Even aqueous suspensions require ventilation because of the risk of nanoparticle aerosolization. Regulations for organic (flammable) nanowaste vary by country. For example, in Switzerland, containers for organic nanowaste (Fig. 3e) must be stored like other flammable liquids: inside a fire-rated, ventilated cabinet, with total stored volume not exceeding the institutional safety limit36 (e.g., 100 kg). As with solid nanowaste, liquid containers should never be filled beyond 90% capacity. Once full, they should be transferred to a central waste collection area (Fig. 4) for temporary storage until pick-up by a licensed waste treatment company. Pouring liquid nanowaste down the sink is strictly prohibited under all circumstances.Labels for storage and transport
All nanowaste containers must be clearly labeled (Fig. 5). At present, no globally harmonized system (GHS) exists specifically for nanowaste.14,34,35 Instead, labeling follows the rules for conventional chemical waste under international frameworks (see section “History and regulatory context”). It is essential to distinguish between storage and transport requirements. For storage, labels must follow the GHS35 and, in the EU, its CLP (Classification, Labelling and Packaging) adaptation. For transport, the Agreement concerning the International Carriage of Dangerous Goods by Road (ADR)34 applies. Containers certified for transport (Fig. 3b and c) require additional ADR pictograms and UN numbers. To simplify practice, laboratories may use transport-certified containers from the start and apply combined GHS/ADR labels (Fig. 5).14 At a minimum, storage labels must include: a clear title (e.g., Solid Nanowaste), a complete list of known ingredients (Composition), a signal word (Danger or Warning), applicable hazard statements (H-phrases, standardized ‘Hazard statements’ under the GHS) and precautionary statements (P-phrases, standardized ‘Precautionary statements’ under the GHS), GHS pictograms, quantity, and contact information (responsible person/lab).14,35,36 If SDS are available, they should be used to determine the correct signal words and pictograms. If SDS data are missing (common for novel nanomaterials), regulatory databases such as ECHA's Substance Infocards should be consulted. For transport labels, ADR pictograms and UN numbers must be clearly distinguished from storage labels and placed on the outermost visible container surface. ADR pictograms must be larger to ensure hazard recognition from a distance (Fig. 3c and 5).35 Packaging and labeling for transport are generally shared between the local safety officer and the waste treatment company.34 | ||
| Fig. 5 Example of a single-packaging nanowaste label. Adapted from Schwab et al.14 with permission from Springer Nature, Nature Nanotechnology, 18(4):317–321, copyright 2023. The label is compliant with the Globally Harmonized System (GHS)35 and the agreement concerning the International Carriage of Dangerous Goods by Road (ADR).34 Such combined labels can be used when nanowaste is both stored and transported in the same container.34,35 Blue text: elements required by different regulations. Blue dotted lines: transport elements required only by ADR (see section labels for storage and transport and SI, case study pilot-scale nanoparticle disposal). For storage-only containers, the transport section is not required. Abbreviations: UN = United Nations. n.o.s. = not otherwise specified. In this example, the country-specific information complies with Swiss regulations.37,41–43 If the waste contained a larger fraction of organic nanomaterials relative to inorganic nanomaterials, the UN code would change to 2811 (Toxic solid, inorganic, n.o.s.), and the VeVA code would be 16 03 05. | ||
Responsibility, however, remains with the waste producer until collection by the treatment facility. Clear, concise labeling is essential not only for compliance but also for safety. Well-designed labels ensure that laboratory staff, building managers, firefighters, and spill response teams can immediately recognize hazards during routine work or emergencies.
Protective measures and personal protective equipment (PPE)
In accordance with the hierarchy of control measures applied to hazardous substances, exposure to nanomaterials and nanowaste should be controlled first through engineering controls (e.g., enclosed processes, local exhaust ventilation), followed by administrative and organizational measures (e.g., standard operating procedures, training, restricted access). Personal protective equipment (PPE) constitutes the last line of measure and is applied where residual risks cannot be eliminated by engineering or administrative controls.Protective measures are required whenever nanomaterials or nanowaste are handled or present in the laboratory. Detailed guidelines exist for selecting appropriate protective strategies depending on nanomaterial type.16,18 At a minimum, laboratory coats, protective gloves, and safety goggles must be worn at all times, particularly when hazard data are incomplete or uncertain.18,44 Where available, the SDS provides guidance for selecting PPE. In the absence of an SDS, regulatory databases (e.g., ECHA Substance Infocards, https://echa.europa.eu/) should be consulted for information on the base materials.
Particular caution is required when using gloves. A widespread misconception is that nitrile gloves provide strong, long-term protection. In reality, most glove materials only protect for a limited time against splashes of nanomaterial suspensions or solvents. Details on glove permeability and breakthrough times are provided in the SI (“on the permeability of protective gloves”).
If there is a risk of aerosolization, additional measures are mandatory, including the use of an FFP3 respirator, hair cover, and shoe covers. Respirators must be treated as personal equipment and should not be shared. They must be fit-tested, properly maintained, and stored away from contaminated areas. Common misuse, such as wearing respirators for more than 2 hours without replacement, failing to replace expired cartridges, or skipping fit tests, can severely reduce protection. At the end of their life cycle, respirators and cartridges must be disposed of as solid nanowaste (see “Solid nanowaste”).
Pregnant or breastfeeding women, as well as women planning pregnancy, must undergo an occupational health risk assessment to evaluate workplace safety before being permitted to handle nanowaste. This precaution recognizes the particular uncertainties in the reproductive toxicity of many nanomaterials, including the limited availability of data on foetal exposure pathways. In situations of significant uncertainty, restricting exposure of pregnant workers may be considered an appropriate application of the Precautionary Principle.
Overall, PPE is the last line of defense within a comprehensive safety concept. Engineering controls (e.g., ventilated storage, fume hoods, sealed containers) and organizational measures (e.g., training, waste segregation, clear labeling) should always take priority. PPE complements, but never replaces, these upstream protections.
Conclusions, gap analysis, and outlook
This guideline provides a comprehensive overview of laboratory practices for the safe handling of nanowaste in research environments, particularly where small-to-medium volumes of diverse nanomaterials are used. In practice, this typically means milligrams to a few grams of solids per experiment, or milliliters to a few tens of milliliters of suspensions. These guidelines are therefore tailored to laboratory and SME settings rather than industrial waste streams. The main message is clear: the necessary laboratory tools and procedures already exist. What is often missing is their consistent application to nanomaterials, and this guideline demonstrates how to do so. Research institutions, SMEs, and nanomaterial manufacturers worldwide can therefore implement safe nanowaste management today. Nevertheless, several critical gaps must be addressed to harmonize practices and strengthen global governance:(a) A global consensus on nanowaste categorization, labeling, and assignment of clear and, ideally, nano-specific UN codes, GHS, and ADR pictograms, which remain ambiguous.
(b) Official guidelines on nanowaste management tailored to research and SME environments, potentially elaborated as a Technical Guideline under the Basel Convention.
(c) Strategies for environmentally sound nanowaste management in low- and middle-income countries; and
(d) Validated protocols for recycling or deactivating nanomaterials to eliminate nanoscale properties.
The Basel Convention offers a unique framework to address these issues and prevent fragmented or unequal approaches. Our analysis shows that nanowaste is already implicitly covered in its annexes, but explicit recognition and technical guidance would close current regulatory gray zones and accelerate harmonized practice. In parallel, pragmatic preventive procedures must be applied immediately. Delaying implementation risks unnecessary exposure of laboratory staff, waste handlers, and the environment to unknown hazards.
Importantly, regulators should also recognize that some nanomaterials, particularly biodegradable ones, may represent promising alternatives to persistent conventional chemicals (“forever chemicals”). Complex regulatory requirements, such as those under REACH, may unintentionally discourage innovation and transparency if they become overly burdensome and may lead some companies to underreport or avoid developing nano-enabled products. Future regulation should therefore be proportionate, clear, and enabling: strict enough to ensure protection but streamlined enough to foster safe-by-design innovation and proper declaration.
In summary, this tutorial review is intended as both an immediately usable manual for laboratory practice and as a draft technical basis for future Basel Convention Technical Guidelines. By bridging hands-on laboratory practice with international governance, we aim to support researchers, safety officers, and policymakers in advancing safe, harmonized, and sustainable nanowaste management worldwide.
Author contributions
B. R.-R., A. P.-F., and F. S. conceived the idea of the article. A. P.-F re-wrote the manuscript written by F. S. with contributions from B. B. K. who both gathered most of the research articles included in the references with contributions from B. R.-R. A. S. and T. M. contributed with their insights and experience as workplace hygienists and safety officers at the University of Fribourg and the Ecole Polytechnique Fédérale de Lausanne (EPFL), respectively.Disclaimer
The information in this guideline was compiled with utmost attention to correctness. Nevertheless, it is not a legal document and cannot replace local regulations that may differ from global regulations. No responsibility is taken for the correctness of the information and any resulting adverse implications. Any opinions, findings, conclusions, or recommendations expressed in this material are those of the authors. They do not necessarily reflect the views of the Swiss government. This work has not been subjected to review by the Swiss government, and no official endorsement should be inferred.Conflicts of interest
F. S. is an inventor on patent WO2020212526A1 (SiO2 nanoparticle plant biostimulant) and received funding via Innosuisse (38515.1 IP-EE). This work was also supported by the United Nations Institute for Training and Research (UNITAR), the Swiss Federal Office for the Environment, the Adolphe Merkle Foundation, and the University of Fribourg. The authors have no further competing interests to disclose and are responsible for the content and writing of the article.Data availability
No new data were generated or analysed during this study. Ref. 45–73 are cited in the supplementary information (SI).Supplementary information: additional information is provided in Fig. S1; Tables S1 and S2. Further, four case studies are presented, and more details on nanowaste management are provided in the sections Definition of terms, Characterization methods, What is nanowaste? In-house treatment and recycling to prevent upstream nanowaste, Specific rules for flammable nanowaste, Details on labeling nanowaste, and on the permeability of protective gloves. See DOI: https://doi.org/10.1039/d6en00013d.
Acknowledgements
F. S. was supported by Innosuisse (project No. 38515.1 IP-EE). This research was also supported by the Adolphe Merkle Foundation at the University of Fribourg. This guideline is loosely based on initial work by Yuki Umehara and Nico Bruns (Adolphe Merkle Institute). We thank Georg Karlaganis and Oliver Wootton from UNITAR for the insights they provided into the Basel Convention and global regulation, Michael Mayer for helpful discussions on institutional workplace safety, Anita Roulin for information on the coordination with the waste treatment company, Nicola Schäppi for excellent photography and graphic design support, Oliver Erni and Roger Marti for reviewing the information on the pilot-scale disposal of nanomaterial suspensions, and Dimitri Vanhecke for double-checking the risk assessment calculations for the disposal of transmission electron microscopy grids. We also thank the members of the BioNanomaterials group for constructive comments regarding the practicality of this guideline.References
- M. Scheringer, Nanoecotoxicology: Environmental risks of nanomaterials, Nat. Nanotechnol., 2008, 3, 322–323, DOI:10.1038/nnano.2008.145.
- A. A. Keller, S. McFerran, A. Lazareva and S. Suh, Global life cycle releases of engineered nanomaterials, J. Nanopart. Res., 2013, 15, 1692, DOI:10.1007/s11051-013-1692-4.
- T. Faunce and B. Kolodziejczyk, in G20 Insights - Policy Briefs, Nanowaste: Need for Disposal and Recycling Standards, 2017, [Accessed 23. December 2021], Available from: https://www.g20-insights.org/policy_briefs Search PubMed.
- E. Buitrago, et al., NanoSafe III: a user friendly safety management system for nanomaterials in laboratories and small facilities, Nanomaterials, 2021, 11, 2768, DOI:10.3390/nano11102768.
- F. Schwab, B. Rothen-Rutishauser and A. Petri-Fink, When plants and plastic interact, Nat. Nanotechnol., 2020, 15, 729–730, DOI:10.1038/s41565-020-0762-x.
- V. K. Sharma, J. Filip, R. Zboril and R. S. Varma, Natural inorganic nanoparticles – formation, fate, and toxicity in the environment, Chem. Soc. Rev., 2015, 44, 8410–8423, 10.1039/C5CS00236B.
- S. Griffin, et al., Natural Nanoparticles: A Particular Matter Inspired by Nature, Antioxidants, 2018, 7, 3 CrossRef PubMed.
- European Committee for Standardization (CEN), Brussels, Belgium, 2018.
- N. Musee, Nanowastes and the environment: potential new waste management paradigm, Environ. Int., 2011, 37, 112–128, DOI:10.1016/j.envint.2010.08.005.
- F. Part, G. Zecha, T. Causon, E. K. Sinner and M. Huber-Humer, Current limitations and challenges in nanowaste detection, characterisation and monitoring, Waste Manage., 2015, 43, 407–420, DOI:10.1016/j.wasman.2015.05.035.
- A. Campos and I. López, in Handbook of Environmental Materials Management, ed. C. M. Hussain, Springer International Publishing, 2019, ch. 161, pp. 2287–2314 Search PubMed.
- S. A. Mazari, et al., Nanomaterials: Applications, waste-handling, environmental toxicities, and future challenges – A review, J. Environ. Chem. Eng., 2021, 9, 105028, DOI:10.1016/j.jece.2021.105028.
- T. Y. Suman and D.-S. Pei, in Nanomaterials Recycling, ed. M. Rai and T. A. Nguyen, Elsevier, 2021, pp. 21–36 Search PubMed.
- F. Schwab, et al., The need for awareness and action in managing nanowaste, Nat. Nanotechnol., 2023, 18, 317–321, DOI:10.1038/s41565-023-01331-4.
- UNEP, in UNEP/CHW/OEWG.11/INF/24 Report on issues related to waste containing nanomaterials and options for further work under the Basel Convention, Secretariat of the Basel Convention, Geneva, Switzerland, 2018, [Accessed 14. October 2021], Available from: https://www.basel.int Search PubMed.
- V. Stuttgen, et al., The UCD nanosafety workshop (03 December 2018): towards developing a consensus on safe handling of nanomaterials within the Irish university labs and beyond – a report, Nanotoxicology, 2019, 13, 717–732, DOI:10.1080/17435390.2019.1621402.
- S. Keshavan, A. Petri-Fink and B. Rothen-Rutishauser, Understanding the Benefits and Risks of Sustainable Nanomaterials in a Research Environment, Chimia, 2024, 78, 397–402, DOI:10.2533/chimia.2024.397.
- A. Groso, A. Petri-Fink, B. Rothen-Rutishauser, H. Hofmann and T. Meyer, Engineered nanomaterials: toward effective safety management in research laboratories, J. Nanobiotechnol., 2016, 14, 21, DOI:10.1186/s12951-016-0169-x.
- P. A. Baron, NIOSH Manual of Analytical Methods (NMAM), National Institute for Occupational Safety and Health (NIOSH), 2016, pp. FI–2–FI–31 Search PubMed.
- G. Boulanger, et al., Quantification of short and long asbestos fibers to assess asbestos exposure: a review of fiber size toxicity, Environ. Health, 2014, 13, 1–18, DOI:10.1186/1476-069X-13-59.
- K. M. A. Perry, Diseases of the lung resulting from occupational dusts other than silica, Thorax, 1947, 2, 91–120, DOI:10.1136/thx.2.2.91.
- D. Hunter and K. M. A. Perry, Bronchiolitis resulting from the handling of bagasse, Br. J. Ind. Med., 1946, 3, 64–74, DOI:10.1136/oem.3.2.64.
- J. Templeton and P. Kohler, Summary of the meetings of the conferences of the parties to the Basel, Rotterdam, and Stockholm Conventions: 26-30 July 2021, Earth Negotiations Bulletin, 2021, vol. 15, pp. 1–11 CAS.
- Z. Wang, et al., We need a global science-policy body on chemicals and waste, Science, 2021, 371, 774–776, DOI:10.1126/science.abe9090.
- SBC, in Basel Convention on the control of transboundary movements of hazardous wastes and their disposal. International convention adopted 22 March 1989, entered into force on 5 May 1992, revised version in 2019, Secretariat of the Basel Convention, Basel, Switzerland, 2019, [Accessed 20. August 2021], Available from: https://www.basel.int/ Search PubMed.
- UNEP, in UNEP/CHW.14/28 Report of the Conference of the Parties to the Basel Convention on the Control of Transboundary Movements of Hazardous Wastes and Their Disposal on the work of its fourteenth meeting, Secretariat of the Basel Convention, Geneva, Switzerland, 2019, [Accessed 13. October 2021], Available from: https://www.basel.int Search PubMed.
- UNEP, in UNEP/CHW.13/28 Report of the Conference of the Parties to the Basel Convention on the Control of Transboundary Movements of Hazardous Wastes and Their Disposal on the work of its thirteenth meeting, Secretariat of the Basel Convention, Geneva, Switzerland, 2017, [Accessed 13. October 2021], Available from: https://www.basel.int Search PubMed.
- UNEP, in UNEP/CHW/OEWG.11/15 Report of the Open-ended Working Group of the Basel Convention on the Control of Transboundary Movements of Hazardous Wastes and Their Disposal on the work of its eleventh meeting, Secretariat of the Basel Convention, Geneva, Switzerland, 2018, [Accessed 14. October 2021], Available from: https://www.basel.int Search PubMed.
- UNEP, in UNEP/CHW.14/7/Add.1/Rev.1 General technical guidelines on the environmentally sound management of wastes consisting of, containing or contaminated with persistent organic pollutants, Secretariat of the Basel Convention, Geneva, Switzerland, 2019, [Accessed 14. October 2021], Available from: https://www.basel.int Search PubMed.
- R. A. Aitken, et al., RNC/RIP-oN3/FPR/1/FINAL Specific advice on exposure assessment and hazard/risk characterisation for nanomaterials under REACH (RIP-oN 3) - final project report, 2011.
- D. S. Jarvis and N. Richmond, Regulation and governance of nanotechnology in China: Regulatory challenges and effectiveness, Eur. J. Law Technol., 2011, 2, 1–11 Search PubMed.
- BAG, in Precautionary matrix for synthetic nanomaterials, Swiss Federal Office of Public Health Berne, Switzerland, 2018, [Accessed 25. March 2021], Available from: https://www.bag.admin.ch Search PubMed.
- Thommen-Furler, Switzerland, 2019, Available from: https://www.thommen-furler.ch/de/.
- UNECE, in ECE/TRANS/300 (Vol. I+II), ADR, applicable as from 1 January 2021, Agreement Concerning the International Carriage of Dangerous Goods by Road, United Nations Economic Commission for Europe, New York, USA, Geneva, Switzerland, 2020, [Accessed 25. November 2021], Available from: https://unece.org/transportdangerous-goods/adr-2021-files Search PubMed.
- UNECE, in ST/SG/AC.10/30/Rev.9, Globally harmonized system of classification and labelling of chemicals (GHS), United Nations Economic Commission for Europe, New York, USA; Geneva, Switzerland, 9th edn, 2021, [Accessed 25. November 2021], Available from: https://unece.org/transport/standards/transport/dangerous-goods/ghs-rev9-2021 Search PubMed.
- CH, in Lagerung gefährlicher Stoffe - Leitfaden für die Praxis, Januar 2018, Umweltfachstellen der Kantone, Frauenfeld, Switzerland, 2018, [Accessed 28. October 2021], Available from: https://www.safetycenter.ch Search PubMed.
- BAG, in Federal Office of Public Health (FOPH) Concept Paper September 2010, Umweltverträgliche und sichere Entsorgung von Abfällen aus Herstellung sowie industrieller und gewerblicher Verarbeitung von synthetischen Nanomaterialien, Swiss Confederation, Berne, Switzerland, 2010, [Accessed 19. March 2021], Available from: https://www.bag.admin.ch Search PubMed.
- S. A. Mostafa, M. M. El-Deeb, A. A. Farghali and A. S. Faried, Evaluation of the nano silica and nano waste materials on the corrosion protection of high strength steel embedded in ultra-high performance concrete, Sci. Rep., 2021, 11, 2617, DOI:10.1038/s41598-021-82322-0.
- A. S. Faried, S. A. Mostafa, B. A. Tayeh and T. A. Tawfik, Mechanical and durability properties of ultra-high performance concrete incorporated with various nano waste materials under different curing conditions, J. Build. Eng., 2021, 43, 102569, DOI:10.1016/j.jobe.2021.102569.
- N. Bossa, et al., Mechanisms limiting the release of TiO2 nanomaterials during photocatalytic cement alteration: the role of surface charge and porous network morphology, Environ. Sci.: Nano, 2019, 6, 624–634, 10.1039/c8en00700d.
- VeVA, in Swiss Federal Council Regulation No. 814.610 of 22 June 2005, version of January 1st 2020, Verordnung über den Verkehr mit Abfällen (VeVA), Swiss Confederation, Berne, Switzerland, 2005, [Accessed 19. March 2021], Available from: https://www.fedlex.admin.ch Search PubMed.
- ChemRRV, in Swiss Federal Council Regulation No. 814.81 of 18 May 2005, version of 25 February 2020, Verordnung zur Reduktion von Risiken beim Umgang mit bestimmten besonders gefährlichen Stoffen, Zubereitungen und Gegenständen (Chemikalien-Risikoreduktions-Verordnung, ChemRRV), Swiss Confederation, Berne, Switzerland, 2005, [Accessed 5. May 2020], Available from: https://www.fedlex.admin.ch Search PubMed.
- ChemV, in Swiss Federal Council Regulation No. 813.11 of 5 June 2015, version of 1st April 2020, Verordnung über den Schutz vor gefährlichen Stoffen und Zubereitungen (Chemikalienverordnung, ChemV), Swiss Confederation, Berne, Switzerland, 2015, [Accessed 5. May 2020], Available from: https://www.fedlex.admin.ch Search PubMed.
- OSHA, OSHA 3151-12R, Personal Protective Equipment, 2004, https://www.osha.gov/sites/default/files/publications/osha3151.pdf.
- D. M. Zalk and S. Y. Paik, in Assessing Nanoparticle Risks to Human Health, ed. G. Ramachandran, William Andrew Publishing, 2011, pp. 139–166 Search PubMed.
- A. Massari, M. Beggio, S. Hreglich, R. Marin and S. Zuin, Behavior of TiO2 nanoparticles during incineration of solid paint waste: a lab-scale test, Waste Manage., 2014, 34, 1897–1907, DOI:10.1016/j.wasman.2014.05.015.
- W. Yin, et al., Removal and recovery of silver nanoparticles by hierarchical mesoporous calcite: Performance, mechanism, and sustainable application, Environ. Res., 2020, 187, 109699, DOI:10.1016/j.envres.2020.109699.
- A. L. Holder, E. P. Vejerano, X. Zhou and L. C. Marr, Nanomaterial disposal by incineration, Environ. Sci.: Processes Impacts, 2013, 15, 1652–1664, 10.1039/c3em00224a.
- V. Oestreicher, C. S. Garcia, G. Soler-Illia and P. C. Angelome, Gold recycling at laboratory scale: from nanowaste to nanospheres, ChemSusChem, 2019, 12, 4882–4888, DOI:10.1002/cssc.201901488.
- A. P. Gondikas, et al., Release of TiO2 nanoparticles from sunscreens into surface waters: a one-year survey at the old Danube recreational lake, Environ. Sci. Technol., 2014, 48, 5415–5422, DOI:10.1021/es405596y.
- F. Schwabe, et al., Dissolved cerium contributes to uptake of Ce in the presence of differently sized CeO2-nanoparticles by three crop plants, Metallomics, 2015, 7, 466–477, 10.1039/c4mt00343h.
- D. Bossert, et al., A hydrofluoric acid-free method to dissolve and quantify silica nanoparticles in aqueous and solid matrices, Sci. Rep., 2019, 9, 7938, DOI:10.1038/s41598-019-44128-z.
- M. Miernicki, T. Hofmann, I. Eisenberger, F. von der Kammer and A. Praetorius, Legal and practical challenges in classifying nanomaterials according to regulatory definitions, Nat. Nanotechnol., 2019, 14, 208–216, DOI:10.1038/s41565-019-0396-z.
- Union, E. Commission Recommendation of 10 June 2022 on the definition of nanomaterial (Text with EEA relevance) 2022/C229/01, 2022, https://eur-lex.europa.eu/legal-content/EN/TXT/?uri=CELEX:32022H0614(01).
- K. Donaldson and P. J. Borm, The quartz hazard: a variable entity, Ann. Occup. Hyg., 1998, 42, 287–294, DOI:10.1093/annhyg/42.5.287.
- D. R. Boverhof, et al., Comparative assessment of nanomaterial definitions and safety evaluation considerations, Regul. Toxicol. Pharmacol., 2015, 73, 137–150, DOI:10.1016/j.yrtph.2015.06.001.
- M. El-Shetehy, et al., Silica nanoparticles enhance disease resistance in Arabidopsis plants, Nat. Nanotechnol., 2021, 16, 344–353, DOI:10.1038/s41565-020-00812-0.
- F. Schwab and M. Maceroni, Method of production of controlled release silica nanoparticles for plant growth and/or defense enhancement, 2020.
- B. Nowack and T. D. Bucheli, Occurrence, behavior and effects of nanoparticles in the environment, Environ. Pollut., 2007, 150, 5–22, DOI:10.1016/j.envpol.2007.06.006.
- L. E. Murr, E. V. Esquivel, J. J. Bang, G. de la Rosa and J. L. Gardea-Torresdey, Chemistry and nanoparticulate compositions of a 10,000 year-old ice core melt water, Water Res., 2004, 38, 4282–4296, DOI:10.1016/j.watres.2004.08.010.
- D. X. Flores-Cervantes, H. M. Maes, A. Schaffer, J. Hollender and H. P. Kohler, Slow biotransformation of carbon nanotubes by horseradish peroxidase, Environ. Sci. Technol., 2014, 48, 4826–4834, DOI:10.1021/es4053279.
- M. Xu, D. N. Futaba, T. Yamada, M. Yumura and K. Hata, Carbon nanotubes with temperature-invariant viscoelasticity from −196° to 1000°C, Science, 2010, 330, 1364–1368, DOI:10.1126/science.1194865.
- D. M. Mitrano, K. Mehrabi, Y. A. R. Dasilva and B. Nowack, Mobility of metallic (nano)particles in leachates from landfills containing waste incineration residues, Environ. Sci.: Nano, 2017, 4, 480–492, 10.1039/c6en00565a.
- L. Vinches, et al., Experimental evaluation of the penetration of TiO2 nanoparticles through protective clothing and gloves under conditions simulating occupational use, Nanosci. Methods, 2013, 2, 1–15, DOI:10.1080/21642311.2013.771840.
- L. Golanski, A. Guiot, F. Rouillon, J. Pocachard and F. Tardif, Experimental evaluation of personal protection devices against graphite nanoaerosols: fibrous filter media, masks, protective clothing, and gloves, Hum. Exp. Toxicol., 2009, 28, 353–359, DOI:10.1177/0960327109105157.
- NORTH, NORTH Chemical Resistance Guide, 2005, https://webfiles.ehs.ufl.edu/North.pdf.
- R. N. Phalen, T. Le and W. K. Wong, Changes in chemical permeation of disposable latex, nitrile, and vinyl gloves exposed to simulated movement, J. Occup. Environ. Hyg., 2014, 11, 716–721, DOI:10.1080/15459624.2014.908259.
- R. Arvidsson, Risk assessments show engineered nanomaterials to be of low environmental concern, Environ. Sci. Technol., 2018, 52, 2436–2437, DOI:10.1021/acs.est.8b00754.
- M. Younes, et al., Re-evaluation of sodium ferrocyanide (E 535), potassium ferrocyanide (E 536) and calcium ferrocyanide (E 538) as food additives, EFSA J., 2018, 16, e05374, DOI:10.2903/j.efsa.2018.5374.
- Y. Sekine, et al., Osmium evidence for synchronicity between a rise in atmospheric oxygen and Palaeoproterozoic deglaciation, Nat. Commun., 2011, 2, 502, DOI:10.1038/ncomms1507.
- G. Alves Miranda, et al., Proposal of novel Predicted No Effect Concentrations (PNEC) for metals in freshwater using Species Sensitivity Distribution for different taxonomic groups, Sci. Rep., 2025, 15, 8180, DOI:10.1038/s41598-025-92692-4.
- R. K. Iler, The Chemistry of Silica: Solubility, Polymerization, Colloid and Surface Properties and Biochemistry of Silica, Wiley, 1979 Search PubMed.
- S. V. Patwardhan, et al., Chemistry of aqueous silica nanoparticle surfaces and the mechanism of selective peptide adsorption, J. Am. Chem. Soc., 2012, 134, 6244–6256, DOI:10.1021/ja211307u.
| This journal is © The Royal Society of Chemistry 2026 |