pH-dependent transport of neonicotinoid pesticides in saturated soil: single and combined functions of rhamnolipid and biochar colloids
Lixia
Li
a,
Tao
Jia
a,
Zhichong
Qi
 b,
Usman
Farooq
b,
Usman
Farooq
 b,
Yanbin
Ma
bc,
Fanyan
Yang
bc,
Minghui
Lv
*a and
Taotao
Lu
b,
Yanbin
Ma
bc,
Fanyan
Yang
bc,
Minghui
Lv
*a and
Taotao
Lu
 *d
*d
aShandong Provincial Geo-mineral Engineering Exploration Institute, Shandong Provincial Bureau of Geology & Mineral Resources, Shandong Engineering Research Center for Environmental Protection and Remediation on Groundwater, Key Laboratory of Geological Disaster Risk Prevention and Control, Emergency Management Department of Shandong Province, Jinan 250353, China. E-mail: lmhyoxiang@163.com
bHenan Joint International Research Laboratory of Environmental Pollution Control Materials, College of Chemistry and Molecular Sciences, Henan University, Kaifeng 475004, China
cNingxia Fundamental Geological Survey Institute (Geological and Mineral Resources Center Laboratory of Ningxia), Geological Bureau of Ningxia Hui Autonomous Region, Yinchuan 750021, China
dCollege of Hydraulic Science and Engineering, Yangzhou University, Yangzhou, 225009, China. E-mail: Taotao.Lu@yzu.edu.cn
First published on 15th January 2026
Abstract
Applying biochar as an amendment for the remediation of neonicotinoid pesticide-contaminated soils is a promising way to reduce the environmental risks posed by these pollutants. Rhamnolipid, a widespread glycolipid biosurfactant in soils, may regulate the transport of biochar colloids and/or pesticides in soil–water environments. Currently, critical knowledge gaps remain regarding how biosurfactant/biochar affects neonicotinoid pesticide mobility. Herein, rhamnolipid was employed to explore its influences on neonicotinoid pesticide (acetamiprid or nitenpyram) mobility and biochar colloid-affected mobility of pesticides at variable solution pH levels (5.0–9.0). In the binary system, rhamnolipid restrained pesticide transport owing to the biosurfactants' bridging effects, forming soil–biosurfactant–pesticide ternary complexes; similarly, biochar colloids also inhibited pesticide mobility because of colloid–pesticide complex deposited on soil surfaces. Notably, the degree of the inhibiting impacts of biochar/biosurfactant varied with pesticide types (acetamiprid > nitenpyram), which was attributed to differences in the chemical features of pesticides (e.g., hydrophobicity). Interestingly, the pH-dependent inhibition effects followed the order pH 5.0 > pH 7.0 > pH 9.0, which were ascribed to the different deposition amounts of biosurfactant molecules or biochar colloids. Surprisingly, in the ternary system, adding biosurfactant weakened the repressive influences of biochar colloids on pesticide migration over a broad pH range of 5.0 to 9.0 because of the reduced retention of colloid-associated pesticides and the great mobility of free neonicotinoid pesticides. Additionally, the degree of rhamnolipid's suppressive effects declined as the pH value increased. These findings provide critical insights into the environmental behaviors and fate of neonicotinoid pesticides influenced by ubiquitous biosurfactants in biochar-amended soils.
Environmental significanceRhamnolipid is a widespread biosurfactant in natural environments. Investigation of its effects on biochar colloid-mediated transport of neonicotinoid pesticides is significant in assessing the fate of these contaminants when biochar is used as a soil amendment. This study clearly demonstrated that rhamnolipid and biochar colloids, alone and together, affected pesticide mobility with strong dependence on pH and pesticide type. Meanwhile, the presence of rhamnolipid weakened the inhibitory effects of biochar colloids on pesticide transport within the pH range of 5.0–9.0. These findings provide critical insights into the fate of neonicotinoid pesticides mediated by biochars and biosurfactants in soil–water environments. |
1. Introduction
Neonicotinoid pesticides, a class of systemic insecticides characterized by their nitroguanidine structure, are widely used on crops, forests, and home gardens because of their broad-spectrum efficacy, low mammalian toxicity, and high potency against insects.1,2 Notably, field studies demonstrate that crop systems typically absorb only 2–20% of the applied neonicotinoid, resulting in substantial release of these compounds into natural environments.3 Currently, these pesticides have been found to be widespread in various environmental and biological media (e.g., water, dust, soil, fruits, sediments, etc.).4–7 Meanwhile, neonicotinoid pesticides exhibit exceptional environmental persistence (e.g., the half-life of clothianidin ranges from 148 to 6931 days).8 Thus, as non-target insecticide species, long-term repeated use of these compounds is more likely to cause potential adverse effects on human health and ecosystems.9 It is worth noting that neonicotinoid insecticides with high aqueous solubility can infiltrate groundwater systems through multiple pathways, such as agricultural runoff, irrigation return flow, and vertical leaching,9 posing significant risks to groundwater quality.10 In view of the environmental effects of neonicotinoid insecticides, it is important to investigate the mobility and fate of these contaminants in water/soil systems.9The hydrologic transport of neonicotinoid insecticides in groundwater systems is largely affected by a variety of environmental factors, including soil features (e.g., organic carbon and/or clay contents),11–13 media types (e.g., soil matrix and quartz sand),14,15 solution chemistry,12 colloids/nanoparticles,12,16 exogenous organic substances,17,18 and field crops.19 For example, previous research observed that vegetation (e.g., prairie grasses) reduced neonicotinoid insecticide mobility in agricultural fields.13,20 Moreover, Wang et al. demonstrated that biochar amendment reduced acetamiprid mobility in sand grains stemming from the binding of biochar to this pesticide.12 Another study demonstrated significant vertical migration of imidacloprid (60 cm depth) in soils characterized by low organic matter content, low cation exchange capacity (CEC), sandy texture, and alkaline pH values.21 Considering the inherent variability and complexity of groundwater systems, more critical studies are required to reveal the transport features of neonicotinoid insecticides in aquifer systems, thereby enhancing the predictive understanding of their environmental fate.
Biochar, a carbon-rich material produced through pyrolysis of biomass under little or no oxygen conditions, has emerged as a potential, multifunctional, cost-effective carbon-based material for soil amendment, carbon sequestration, enhancement of agricultural productivity, and biowaste treatment.22 Biochar has a high affinity for organic compounds due to its multitude of advantages, such as high surface area, hierarchical porosity, and abundant functional groups.23 Thus, biochar can effectively remove or immobilize organic contaminants in water/soil environments.24 Notably, during environmental applications, biochar undergoes transformation through physical, chemical, and biological actions, leading to the release of nanoparticles and colloids from bulk biochar particles.25 There is ample field evidence that biochar colloids exhibit high mobility in soil–water systems.23,26–29 More importantly, biochar exhibits strong sorption capacity to neonicotinoid pesticides through synergistic mechanisms (e.g., hydrogen bonding, π–π interactions, hydrophobic partitioning, and electrostatic interaction).30–33 Consequently, the mobility of neonicotinoid pesticides through aquifers is possibly affected by the presence of biochar colloids when this carbon-based material is used to remediate pesticides in contaminated sites. Understanding the transport and fate of neonicotinoid pesticides in the presence of colloidal biochar in aquifers is of important interest in the environmental behaviors of these organic pollutants. Thus, to gain a more comprehensive understanding of the transport characteristics of neonicotinoid pesticides, further detailed studies are needed to evaluate key environmental factors—such as the presence of biosurfactants—that govern the co-transport of colloidal biochar and pesticides through aquifer systems.
Biosurfactants are bioavailable surface-active metabolites mainly produced during the vital movement of microorganisms (e.g., bacteria, fungi, and yeast).34 Currently, biosurfactants are commonly used for industrial applications, soil remediation, and therapeutic products.34,35 As a well-known biosurfactant, rhamnolipid is predominantly secreted by Pseudomonas aeruginosa (a ubiquitous pathogen).36 Rhamnolipid molecules are usually made up of hydrophilic parts (polysaccharides) and hydrophobic moieties (aliphatic hydrocarbons).37 This biosurfactant primarily exists as an anionic species across wide environmentally relevant pH conditions stemming from its low pKa values (4.3–5.6 (ref. 38)).39 Previous studies confirmed that rhamnolipid exhibited great binding capabilities toward organic pollutants, including pesticides, polycyclic aromatic hydrocarbons (PAHs), and antibiotics.40–43 Also, rhamnolipid could interact dynamically with aquifer media, subsequently altering its surface properties.44–47 Consequently, the presence of rhamnolipid is likely to alter the fate and transport of organic contaminants in subsurface systems.48–52 Meanwhile, this biosurfactant can also bind to biochars and change their surface physicochemical properties.53,54 In this situation, it is reasonable to believe that rhamnolipid may affect biochar–neonicotinoid interactions, ultimately impacting co-transport behaviors of colloidal biochar and neonicotinoid pesticides. Up to now, however, the effect of biosurfactants on the co-mobility of biochar colloids and neonicotinoid pesticides in subsurface has not been thoroughly studied, which is a critical knowledge gap for clarifying the fate and risks of neonicotinoid pesticides when biochars are applied for remediation of contaminated soils. Meanwhile, the mechanisms governing their co-transport behavior in systems containing biosurfactants also remain poorly understood. Systematic investigations are therefore required to expound biochar colloid-assisted transport of pesticides as influenced by ubiquitous biosurfactants in soil matrices.
In this work, acetamiprid and nitenpyram, two widely used neonicotinoid pesticides, were chosen as the targeted contaminants. Meanwhile, rhamnolipid was employed as a typical biosurfactant. Significantly, solution pH was a key factor controlling the mobility of biochar colloid-mediated pesticides, particularly when biosurfactants were present in the surrounding environment. The affinity of biochars to neonicotinoid pesticides and/or biosurfactants is heavily impacted by pH levels.12,16,55,56 The major objective of this study was to clarify the single or combined influences of a typical biosurfactant (i.e., rhamnolipid)/biochar colloids on the transport behaviors of neonicotinoid pesticides at various pH conditions. Meanwhile, the mechanisms underlying the transport features of the two pesticides as impacted by exogenous biosurfactant and/or biochar colloids were elucidated. This study provides available knowledge related to neonicotinoid pesticide mobility and fate mediated by carbon-based materials and biosurfactants in soil–water environments.
2. Materials and methods
2.1. Reagents
Generally, the topsoil is the growth and distribution area of the active area of microbial metabolism.57,58 Surface soil (0–20 cm) was collected from an agricultural field in Shangqiu, Henan province, China. Prior to the experiment, soil samples were air-dried at 25 °C, homogenized, and sieved (<2 mm) to remove impurities (e.g., plant residues and root fragments). The soil with a pH value of 7.61 had a texture composed of 6.6% clay, 78.4% silt, and 15.0% sand (the detailed procedure is provided in section S1, SI). The average particle size of this sample was 0.123 mm. The soil exhibited an organic carbon fraction (fOC) of 0.42% and a cation exchange capacity (CEC) of 11.3 cmol kg−1 (the detailed methodology is presented in section S2, SI). The average zeta potential (ζ) of soil particles was measured using a Nano-ZS90 Zetasizer (Malvern Instruments, UK).59Nitenpyram (≥99.7%) and acetamiprid (≥99.5%) were purchased from Shanghai Aladdin Bio-Chem Technology Co., Ltd. (China). Stock pesticide solutions (100 mg L−1) were prepared and stored in a refrigerator at 4 °C. All other chemicals, including HCl, NaOH, and NaCl, were of analytical grade.
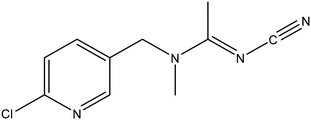
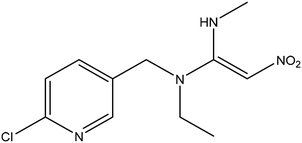
Rhamnolipid (>95%, 250 g L−1 stock solution) was also obtained from Aladdin Biochemical Technology Co., Ltd. The chemical structure of rhamnolipid molecule is shown in Fig. S1. A working solution with a concentration of 500 mg L−1 was prepared by diluting the stock solution with deionized (DI) water.
2.2. Preparation and characterization of biochars and biochar colloid suspensions
Chinese fir sawdust was chosen as the typical biomass. The biomass feedstock was rinsed with DI water to remove the surface impurities and then oven-dried at 80 °C for over 12 h. Afterward, a conventional quartz tube furnace was used to pyrolyze the samples at 450 °C for 3 h under a N2 atmosphere to ensure oxygen-limited conditions. The temperature was increased at a rate of 20 °C min−1. Following pyrolysis, the resulting biochar was allowed to cool naturally to room temperature, after which it was collected, mechanically milled, and fractionated through a 100-mesh sieve. The ash content quantification in the biochar was performed through complete combustion of biochar samples in a muffle furnace at 800 °C for at least 4 h. The elemental composition (C, N, H, O, S, wt%) of biochar was determined using a Flash EA1112 elemental analyzer (Thermo Fisher Scientific). Functional group characterization was performed using a Nicolet 6700 FTIR spectrometer.The biochar colloid stock suspensions were prepared following previously published methods.28 In summary, 2 g of biochar was dispersed in 500 mL of DI water and subjected to thorough mixing and 30 min of sonication. After 3 days of settling, the suspension was filtered through 0.45 μm membranes to isolate the colloidal fraction. The total organic carbon (TOC) concentration was quantified using a Shimadzu TOC-5000 analyzer (Japan). The measured TOC values were 123 mg C per L. Colloidal surface charge characteristics were evaluated through ζ-potential measurements performed on a ZetaPlus analyzer as mentioned above. The average hydrodynamic sizes (Dh) of colloid suspensions were quantified using dynamic light scattering analysis.
A portion of the biochar suspensions was freeze-dried for subsequent chemical characterization. The elemental composition and surface functional groups were characterized as mentioned above. Additionally, surface chemistry was analyzed by X-ray photoelectron spectroscopy (XPS, ESCALAB 250XI, Thermo Scientific). The colloid concentration (mg L−1) was obtained based on the measured TOC values and the carbon content percentages obtained from elemental analysis.28
2.3. Transport tests
Approximately 8.5 g of dry soil was packed into the column (10.3 mm inner diameter and 6.3 cm length), yielding a porosity of 0.43 ± 0.02. Column in- and outlet were packed with a layer of about 50 μm of nylon screen to prevent fine soil grains from exiting the column. Firstly, the column was saturated with 25 PV (porous volume) of DI water in an upward flow model using an LSP01-1A syringe pump (Longer Precision Pump Co., Ltd, Baoding, China). Soil columns were then leached with electrolyte solution (1 mM NaCl at the designated pH) for about 25 PV to stabilize the soil column. Afterward, the corresponding influent (i.e., 1 mg L−1 neonicotinoid pesticide (acetamiprid or nitenpyram) with or without biochar colloids/rhamnolipid) was introduced into the soil column. Generally, the concentrations of neonicotinoid pesticides in real environments are typically at the ng L−1 level.3 However, high neonicotinoid pesticide concentrations may occur in special scenarios, particularly following accidental leakage from production facilities. Therefore, the high concentrations used in this study (∼1 mg L−1) would likely represent a point-source contamination scenario (e.g., industrial spill or discharge). A steady flow was maintained during all the experiments (flow velocity: 0.135 cm min−1). The pH of the influent solutions was adjusted to the target value using 0.1 M HCl or NaOH solutions. Effluent samples were collected at 30 minute intervals using glass vials. The concentration of biochar colloids in the effluent was determined by measuring absorbance at 360 nm using a UV-vis spectrophotometer, with reference to a pre-established calibration curve (Fig. S2a). Moreover, the full-scan UV spectrums in Fig. S2b verified that neonicotinoid pesticides and rhamnolipid had negligible effects on colloid detection. Neonicotinoid pesticide concentrations were quantified using an HPLC (Alliance e2695, Waters) following solvent extraction. Detailed extraction procedures can be found in the SI (section S3).60,61 In order to ensure the quality of experimental results, all column mobility studies were performed in at least duplicate. The normalized concentration (C/C0) of biochar colloids or neonicotinoid pesticides was calculated from influent (C0) and effluent (C) measurements, with breakthrough curves plotted against PV passing through the column. For every column experiment, a new column filled with soil was used.2.4. Sorption tests
Batch adsorption experiments were conducted to evaluate the effects of rhamnolipid on neonicotinoid pesticide adsorption by biochar colloids across a range of pH conditions (5.0–9.0). The adsorbed quantity of neonicotinoid pesticide q (mg g−1) was obtained via mass balance calculations. Finally, the distribution coefficients (Kd, L g−1) for the neonicotinoid pesticide between colloids and water were calculated using the equation Kd = q/Ce, where Ce (mg L−1) is the pesticide concentration in the aqueous phase. Comprehensive methodological details are available in section S4 in the SI.Furthermore, adsorption experiments were also conducted to quantify the amount of adsorbed rhamnolipid on biochar colloids or soil particles (i.e., q (mg g−1)) based on the mass-balance approach as follows:
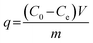 | (1) |
3. Results and discussion
3.1. Characteristics of bulk biochar and biochar colloids
The comparative elemental compositions between colloidal and bulk biochar forms are provided in Tables 1 and S1, respectively. Biochar colloids demonstrated higher oxygen content (26.69%) than bulk biochar (23.60%), indicating that the former contained a greater abundance of oxygen-containing functional groups.62 Furthermore, the O/C and H/C values of biochar colloids were 0.641 and 0.603, respectively (Tables 1 and S1). As demonstrated by the FTIR analysis in Fig. 1a and S3 and Table S2, both bulk biochar and biochar colloids exhibited abundant surface functional groups (e.g., –COOH, –OH, –NH2, and –C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O moieties).63 Also, XPS analysis quantitatively analyzed the surface functional groups of biochar colloids, with the relative abundance and distribution presented in Fig. 1b.
O moieties).63 Also, XPS analysis quantitatively analyzed the surface functional groups of biochar colloids, with the relative abundance and distribution presented in Fig. 1b.
| Colloids | Bulk element composition | O/Ca | H/Ca | Surface element compositionb | |||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| C (wt%) | O (wt%) | N (wt%) | H (wt%) | S (wt%) | C (at%) | O (at%) | N (at%) | S (at%) | |||
| a Bulk atomic ratio, O/C: atomic ratio of oxygen to carbon, H/C: atomic ratio of hydrogen to carbon. b Surface atomic ratio of oxygen to carbon. | |||||||||||
| BC colloids | 31.23 | 26.69 | 0.51 | 1.57 | 0.23 | 0.641 | 0.603 | 68.12 | 29.09 | 2.22 | 0.57 |
3.2. Impact of rhamnolipid on neonicotinoid pesticide mobility
Fig. 2 illustrates the inhibitory effects of rhamnolipid (20 mg L−1) on the mobility of the two pesticides. For example, the highest C/C0 values of acetamiprid decreased from 83.4% (without rhamnolipid) to 68.0% (with rhamnolipid) at pH 5.0 (Table 2). The restrained influences of rhamnolipid on neonicotinoid pesticide transport primarily stemmed from the following reasons. As illustrated in Fig. S1, the biosurfactant molecules had an amphiphilic structure comprising a hydrophobic fatty acid tail bonded to a polar hydrophilic head group.39 Generally, surfactant molecules were adsorbed onto soil primarily through ligand exchange, surface complexation, and/or hydrogen bonding between the hydrophilic head groups and soil surfaces.64 This interfacial arrangement positioned hydrophobic tails toward the aqueous phase.34,65 The adsorption results also quantitatively demonstrated rhamnolipid adsorption to soil particles (Table S3). In this case, the hydrophobic moieties of biosurfactant molecules contacted with neonicotinoid pesticide molecules via hydrophobic interactions (between the alkyl chains of rhamnolipid and the N-containing heterocyclic rings of pesticides)66,67 resulting in the enhanced deposition of pesticides. Furthermore, the neonicotinoid pesticide molecules could bind with rhamnolipid to form pesticide–rhamnolipid complexes via hydrophobic interactions in the influent systems.68,69 In this case, the hydrophilic groups of biosurfactant molecules that attached to neonicotinoid pesticides would deposit on the surfaces of soil grains, facilitating the retention of contaminants,34,70 that is, the bridging effects involved in the enhanced pesticide deposition by rhamnolipid (the formation of soil–rhamnolipid–pesticide ternary complexes).50,71 | ||
| Fig. 2 Effects of rhamnolipid on the transport of neonicotinoid pesticides in saturated soil columns under different pH conditions: (a, c, and e) acetamiprid transport at pH 5.0, 7.0, and 9.0, respectively (columns 1, 2, 5, 6, 9, and 10, Table 2); (b, d, and f) nitenpyram transport at pH 5.0, 7.0, and 9.0, respectively (columns 3, 4, 7, 8, 11, and 12, Table 2). | ||
| Influent properties | Effluent propertiesc | Mass retained in column of pesticided (%) | |||||||
|---|---|---|---|---|---|---|---|---|---|
| Column no. | Biochar colloids conc.a (mg L−1) | Neonicotinoid pesticides | Electrolyte solution | Rhamnolipid conc.a (mg L−1) | pH | Adsorbed mass of pesticideb (%) | C/C0_colloids (%) | C/C0_pesticides (%) | |
| a Conc. represents concentration. b Percentage of contaminant adsorbed to biochar colloids in the influent. The data were obtained based on the sorption experiments (section S4, SI). c Average value of last three data points of respective breakthrough curve. d Mass retained in column = 100 − effluent mass. | |||||||||
| 1 | — | Acetamiprid | 1 mM NaCl | — | 5.0 | — | — | 83.4 ± 0.4 | 23.9 ± 0.1 |
| 2 | — | Acetamiprid | 1 mM NaCl | 20 | 5.0 | — | — | 68.0 ± 0.3 | 39.1 ± 0.3 |
| 3 | — | Nitenpyram | 1 mM NaCl | — | 5.0 | — | — | 95.8 ± 1.5 | 12.6 ± 0.6 |
| 4 | — | Nitenpyram | 1 mM NaCl | 20 | 5.0 | — | — | 84.3 ± 0.1 | 23.9 ± 1.0 |
| 5 | — | Acetamiprid | 1 mM NaCl | — | 7.0 | — | — | 80.5 ± 0.8 | 25.6 ± 0.6 |
| 6 | — | Acetamiprid | 1 mM NaCl | 20 | 7.0 | — | — | 70.5 ± 0.7 | 38.3 ± 1.1 |
| 7 | — | Nitenpyram | 1 mM NaCl | — | 7.0 | — | — | 94.7 ± 0.5 | 14.6 ± 0.5 |
| 8 | — | Nitenpyram | 1 mM NaCl | 20 | 7.0 | — | — | 85.9 ± 0.1 | 22.4 ± 1.0 |
| 9 | — | Acetamiprid | 1 mM NaCl | — | 9.0 | — | — | 79.2 ± 0.4 | 28.7 ± 0.4 |
| 10 | — | Acetamiprid | 1 mM NaCl | 20 | 9.0 | — | — | 69.8 ± 0.5 | 36.0 ± 0.2 |
| 11 | — | Nitenpyram | 1 mM NaCl | — | 9.0 | — | — | 87.4 ± 0.7 | 17.9 ± 0.3 |
| 12 | — | Nitenpyram | 1 mM NaCl | 20 | 9.0 | — | — | 86.2 ± 0.7 | 22.9 ± 0.6 |
| 13 | 30 | Acetamiprid | 1 mM NaCl | — | 5.0 | 65.6 ± 0.3 | 24.3 ± 0.7 | 42.3 ± 0.2 | 61.1 ± 0.5 |
| 14 | 30 | Nitenpyram | 1 mM NaCl | — | 5.0 | 43.7 ± 0.1 | 27.7 ± 1.6 | 59.0 ± 0.9 | 45.2 ± 0.1 |
| 15 | 30 | Acetamiprid | 1 mM NaCl | — | 7.0 | 68.9 ± 1.2 | 30.0 ± 1.4 | 47.7 ± 0.1 | 57.2 ± 0.5 |
| 16 | 30 | Nitenpyram | 1 mM NaCl | — | 7.0 | 46.7 ± 0.5 | 37.0 ± 0.7 | 63.4 ± 1.1 | 41.1 ± 0.2 |
| 17 | 30 | Acetamiprid | 1 mM NaCl | — | 9.0 | 73.5 ± 1.0 | 45.9 ± 2.2 | 55.7 ± 0.4 | 52.0 ± 0.3 |
| 18 | 30 | Nitenpyram | 1 mM NaCl | — | 9.0 | 51.2 ± 0.8 | 43.5 ± 0.5 | 67.1 ± 0.5 | 35.8 ± 0.1 |
| 19 | 30 | Acetamiprid | 1 mM NaCl | 20 | 5.0 | 53.3 ± 1.2 | 27.6 ± 0.8 | 50.3 ± 0.2 | 51.9 ± 0.8 |
| 20 | 30 | Nitenpyram | 1 mM NaCl | 20 | 5.0 | 36.2 ± 0.6 | 32.9 ± 0.7 | 68.8 ± 0.2 | 35.6 ± 0.5 |
| 21 | 30 | Acetamiprid | 1 mM NaCl | 20 | 7.0 | 60.5 ± 0.7 | 41.1 ± 0.8 | 54.5 ± 0.3 | 50.0 ± 0.8 |
| 22 | 30 | Nitenpyram | 1 mM NaCl | 20 | 7.0 | 39.8 ± 1.3 | 42.1 ± 0.1 | 69.4 ± 0.4 | 35.1 ± 0.1 |
| 23 | 30 | Acetamiprid | 1 mM NaCl | 20 | 9.0 | 63.2 ± 2.2 | 50.0 ± 2.0 | 58.2 ± 0.5 | 47.9 ± 0.6 |
| 24 | 30 | Nitenpyram | 1 mM NaCl | 20 | 9.0 | 43.7 ± 1.7 | 45.9 ± 0.5 | 71.8 ± 0.7 | 34.3 ± 1.6 |
In order to quantitatively assess pH-dependent transport-inhibition effects of rhamnolipid on the two neonicotinoid pesticides, the apparent transport-inhibition index (Iinhibition_app) of this biosurfactant was operationally defined as:
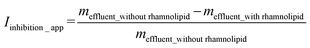 | (2) |
As demonstrated by the estimated Iinhibition_app value trend in Fig. 3a, the inhibitory impacts of biosurfactant on the mobility of a given neonicotinoid pesticide (acetamiprid or nitenpyram) increased with decreasing pH values. In the case of nitenpyram, the Iinhibition_app values declined from 0.199 (pH 5.0) to 0.171 (pH 7.0) and to 0.102 (pH 9.0), suggesting that the degree of the transport-inhibition effects of rhamnolipid was considerably pH-dependent. Meanwhile, the influences of rhamnolipid on acetamiprid mobility exhibited a similar tendency (Fig. 3a). The ζ-potential values in Table S4 confirmed the increasingly negative surface charges on soil grains with rising pH values. Consequently, biosurfactant adsorption onto soil grains decreased when the background solution pH values were elevated owing to the increased electrostatic repulsion between anionic head groups of rhamnolipid and porous media grains (see the sorption results in Table S3),52,60 leading to a weakened bridge effect induced by the presence of rhamnolipid,48,72 As a result, the biosurfactant's inhibiting ability on neonicotinoid pesticide mobility was more pronounced under acidic conditions compared to that under alkaline conditions.
 | ||
| Fig. 3 Comparison of the apparent transport-inhibition ability index (Iinhibition_app) of (a) rhamnolipid and (b) biochar colloids under different pH conditions. | ||
Moreover, the other interesting observation was that for a given pH value, the inhibitory mobility of neonicotinoid pesticides mediated by rhamnolipid was also related to pesticide types. Specifically, the trend of Iinhibition_app values demonstrated that rhamnolipid exhibited stronger transport inhibition for acetamiprid compared to nitenpyram (Fig. 3b). The observation might be ascribed to the contrasting hydrophobicity properties between the two neonicotinoid pesticides.73–75 As shown in Table 3, acetamiprid exhibited higher hydrophobicity than nitenpyram (as demonstrated by the log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Kow values, 0.8 vs. −0.66). In this situation, rhamnolipid exhibited more restrained effects on acetamiprid mobility than nitenpyram because of the stronger hydrophobic interactions between the former and the deposited rhamnolipid on the surfaces of soil grains.72
Kow values, 0.8 vs. −0.66). In this situation, rhamnolipid exhibited more restrained effects on acetamiprid mobility than nitenpyram because of the stronger hydrophobic interactions between the former and the deposited rhamnolipid on the surfaces of soil grains.72
Additionally, regardless of the presence or absence of rhamnolipid, the mobility of nitenpyram exceeded that of acetamiprid (Fig. S4). As a key soil composition, soil organic matter (SOM) contained abundant surface functional groups, such as –COOH, –NH2, and –OH as well as aromatic structures.76,77 Several prior studies reported that SOM played a significant role in the retention of neonicotinoid pesticides through mechanisms including hydrogen bonding and π–π stacking interactions as well as the partitioning of pesticides between the solution phase and SOM.11,78,79 Thus, the two neonicotinoid pesticides could strongly bind to SOM (in the absence of rhamnolipid) and/or rhamnolipid (in the presence of biosurfactant) during pesticide transport. In this case, acetamiprid with higher hydrophobicity exhibited stronger deposition because of the stronger hydrophobic interactions with SOM and/or rhamnolipid.
3.3. Impact of biochar colloids on the mobility of neonicotinoid pesticides
This work also examined the influences of biochar colloids on neonicotinoid pesticide mobility in soil columns (Fig. 4). The addition of biochar colloids inhibited neonicotinoid pesticide transport under all three pH conditions (5.0, 7.0, and 9.0). Such as, for acetamiprid, the breakthrough curves revealed that the highest C/C0 values declined from 80.5% (without colloids) to 47.7% (with 30 mg L−1 colloids) at pH 7.0 (Table 2). | ||
| Fig. 4 Effects of biochar colloids on the transport of neonicotinoid pesticides in saturated soil columns under different pH conditions: (a, c, and e) acetamiprid transport at pH 5.0, 7.0, and 9.0, respectively (columns 1, 13, 5, 15, 9, and 17, Table 2); (b, d, and f) nitenpyram transport at pH 5.0, 7.0, and 9.0, respectively (columns 3, 14, 7, 16, 11, and 18, Table 2). The left plots show the breakthrough curves of biochar colloids in each of the experiments, and the right plots show the breakthrough curves of neonicotinoid pesticides in the respective experiments. | ||
The observed colloid-enhanced retention of neonicotinoid pesticides can be attributed to the following two mechanistic explanations. On the one hand, biochar deposited on soil surfaces through multiple mechanisms, including electrostatic attraction (between positively charged iron–aluminum oxide compositions of soil particles and the negatively charged colloids (Table S5)), π–π and hydrophobic interaction (between the aromatic rings of biochar and SOM), and hydrogen bonding interaction (between the soil mineral hydroxyls (![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) Fe/Al–OH and
Fe/Al–OH and ![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) Si–OH) and deprotonated functional groups on biochar surfaces).23,25,80,81 Notably, biochar colloids exhibited high affinity toward neonicotinoid pesticides through hydrogen bonding interaction (between colloid surface functional groups and pesticide's –NO2 groups), π–π interaction, and hydrophobic partitioning (between the aromatic structures of biochar colloids and the pesticide's N-aromatic heterocyclic rings).30,31,73,74 The adsorption data of neonicotinoid pesticides to biochar colloids also provided additional evidence for their interaction (Fig. S5). It is worth noting that the mobility of pesticides (acetamiprid or nitenpyram) was greater than that of biochar colloids (Fig. 4). In this situation, the co-deposition of neonicotinoid pesticide–biochar colloid complexes onto the surfaces of soil particles facilitated contaminant retention.12,73 On the other hand, biochar colloids pre-deposited on soil surfaces offered additional sites for the retention of pesticide molecules during their co-transport, causing the hindered transport of pesticides with biochar colloids.12,82
Si–OH) and deprotonated functional groups on biochar surfaces).23,25,80,81 Notably, biochar colloids exhibited high affinity toward neonicotinoid pesticides through hydrogen bonding interaction (between colloid surface functional groups and pesticide's –NO2 groups), π–π interaction, and hydrophobic partitioning (between the aromatic structures of biochar colloids and the pesticide's N-aromatic heterocyclic rings).30,31,73,74 The adsorption data of neonicotinoid pesticides to biochar colloids also provided additional evidence for their interaction (Fig. S5). It is worth noting that the mobility of pesticides (acetamiprid or nitenpyram) was greater than that of biochar colloids (Fig. 4). In this situation, the co-deposition of neonicotinoid pesticide–biochar colloid complexes onto the surfaces of soil particles facilitated contaminant retention.12,73 On the other hand, biochar colloids pre-deposited on soil surfaces offered additional sites for the retention of pesticide molecules during their co-transport, causing the hindered transport of pesticides with biochar colloids.12,82
Similarly, to quantitatively compare the suppressed influences of biochar colloids on pesticide mobility, the Iinhibition_app values of biochar colloids were also obtained according to eqn (2) (Fig. 3b). Interestingly, for a given pH value, the Iinhibition_app value trend demonstrated that the inhibitory mobility of acetamiprid mediated by biochar colloids was greater than that of nitenpyram. The result might stem from the greater binding abilities of colloids to acetamiprid (Fig. S5), leading to more colloid–acetamiprid complex deposition on soil grain surfaces.82 Moreover, the restrained influences of colloids on neonicotinoid pesticide transport declined as the background solution pH values raised from 5.0 to 9.0. As shown in Fig. 3b, the Iinhibition_app values of acetamiprid declined from 0.489 at pH 5.0 to 0.425 at pH 7.0 and further to 0.327 at pH 9.0, indicating that the inhibitory abilities of biochar colloids to pesticide transport are greatly related to solution pH values. Note that pesticide-binding abilities of biochar colloids declined as pH was raised (Table 2). More importantly, biochar colloids demonstrated increased mobility at higher pH levels (Fig. S6). As a result, more biochar colloid-associated neonicotinoid pesticide molecules would break through columns at pH 9.0. Thus, the restrained influences of biochar colloids on neonicotinoid pesticide mobility were weakened with the increase of pH values.16
3.4. Rhamnolipid effects on co-transport of biochar colloids and neonicotinoid pesticides
As interpreted above, biochar colloids and biosurfactants individually inhibited neonicotinoid pesticide transport. Logically, the co-presence of biochar colloids and rhamnolipid would enhance neonicotinoid pesticide retention. Surprisingly, the addition of rhamnolipid weakened the restraining impacts of colloids on neonicotinoid pesticide transport within a broad pH range of 5.0–9.0. As demonstrated in Fig. 5b and Table 2, the total retained mass of nitenpyram (co-presence of 30 mg L−1 colloids) in columns declined from 61.1% (without rhamnolipid) to 51.9% (with 20 mg L−1 rhamnolipid) at pH 5.0. | ||
| Fig. 5 Effects of rhamnolipid on biochar colloid-mediated transport of neonicotinoid pesticides in saturated soil columns under different pH conditions: (a, c, and e) acetamiprid transport at pH 5.0, 7.0, and 9.0, respectively (columns 1, 13, 5, 15, 9, 17, 19, 21, and 23, Table 2); (b, d, and f) nitenpyram transport at pH 5.0, 7.0, and 9.0, respectively (columns 3, 14, 7, 16, 11, 18, 20, 22, 24, Table 2). The left plots show the breakthrough curves of biochar colloids in each of the experiments, and the right plots show the breakthrough curves of neonicotinoid pesticides in the respective experiments. | ||
The observed results arose from three interrelated mechanisms. Firstly, biosurfactant molecules could come in contact with biochar colloid/soil particle surfaces and induce negative charge enhancement on their surfaces (Tables S4 and S5). Additionally, the aggregate sizes of biochar colloids became smaller by adding rhamnolipid (see the Dh values in Table S5). Hence, the combined effects of the increased electrostatic repulsion and the reduced steric hindrance facilitated biochar colloid transport (Fig. 5).23,27,29 Consequently, a greater fraction of biochar colloid-associated neonicotinoid pesticides were likely to break through soil porous media, thereby weakening colloids' inhibitory impacts on pesticide mobility.16,83 Secondly, biochar colloids also exhibited binding capacities toward rhamnolipid (Table S6). Generally, rhamnolipid adsorbed onto biochar colloids through hydrophobic interactions between the hydrophobic structures of rhamnolipid and the aromatic rings of biochar.47,53,54 causing rhamnolipid's hydrophilic rhamnose moieties to be oriented toward the water system. As a result, rhamnolipid reduced neonicotinoid pesticide adsorption onto biochar colloids through competitive site occupation (as indicated by the trend of Kd values of neonicotinoid pesticides in Fig. S5).84 That is, more free acetamiprid or nitenpyram molecules remained in the solution systems and might break through soil columns, diminishing the suppressed influences of biochar colloids on pollutant mobility.16,84 Thirdly, the deposition site competition between rhamnolipid–biochar complexes and neonicotinoid pesticide molecules might also contribute to reducing the transport-inhibition effects of biochar colloids.85
Additionally and interestingly, the degree of the weakened effects of biosurfactant on the restrained influences of colloids on neonicotinoid pesticide mobility declined when the pH values were increased from 5.0 to 9.0. For example, as illustrated in Fig. 5 and Table 2, at pH values of 5.0, 7.0, and 9.0, the retained masses of nitenpyram declined by 21.2% (from 45.2% (with 30 mg L−1 colloids) to 35.6% (with 30 mg L−1 colloids and 20 mg L−1 rhamnolipid)), 14.6% (from 41.1% to 35.1%), and 3.5% (from 35.8% to 34.3%), respectively. Note that more rhamnolipid was deposited on biochar colloid surfaces at lower pH levels according to the earlier discussion (Tables S6). Meanwhile, the inhibitory impacts of rhamnolipid on the pesticide-binding abilities of biochar colloids were more remarkable under lower pH conditions due to the enhanced competitive adsorption (Table 2). Consequently, more free neonicotinoid pesticide molecules in water systems could possibly break through soil columns under acidic conditions (pH 5.0).16,17 Thus, the influences of rhamnolipid on neonicotinoid pesticide mobility mediated by biochar colloids were more pronounced under lower pH conditions.
4. Conclusions
This study systematically evaluated the impacts of rhamnolipid, a model biosurfactant, on neonicotinoid pesticide transport mediated by biochar colloids in soil across pH 5.0–9.0. In the absence of biochar colloids, the findings obviously demonstrated that this biosurfactant impeded the mobility of the two neonicotinoid pesticides via biosurfactant-mediated bridging effects. When biochar colloids and neonicotinoid pesticides co-presented in the influents, biochar colloids also reduced neonicotinoid pesticide mobility through the retention of colloid-associated pesticide and new retention sites for organic contaminants created by colloid deposition. Note that the degree of the inhibiting effects of biochar/biosurfactant on acetamiprid mobility was greater than that on nitenpyram transport due to the different pesticides' chemical features (e.g., hydrophobic property). Interestingly, the transport-inhibition effects of colloids or biosurfactants decreased as pH increased, which was primarily attributed to the reduced deposition of colloids/surfactants on soil surfaces with increasing pH values. In the ternary system containing biochar colloids, rhamnolipid, and neonicotinoid pesticides, the biosurfactant unexpectedly mitigated the restraining effect of colloids on pesticide mobility across a broad pH range (5.0–9.0). This phenomenon resulted from the decreased retention of colloid-associated pesticides and the enhanced mobility of free neonicotinoid pesticides. In addition, the mitigating effect of rhamnolipid on the influence of biochar colloids on pesticide mobility was more pronounced at lower pH levels because the biosurfactant more strongly suppressed the colloids' affinity for neonicotinoid pesticides at pH 5.0, resulting in the increased transport of free neonicotinoid pesticide molecules through the soil matrix. In conclusion, these results underscore the necessity of carefully evaluating the influence of ubiquitous biosurfactants on the transport of neonicotinoid pesticides mediated by biochar colloids when implementing biochar-based technologies for soil remediation aimed at mitigating the environmental risks of these emerging organic contaminants. Meanwhile, to develop a comprehensive understanding of neonicotinoid pesticide fate in groundwater systems, additional environmental factors—such as electrical conductivity and redox potential—that influence biochar colloid-mediated transport of pesticides in the presence of biosurfactants should be investigated. Notably, given the greater complexity of field conditions compared to controlled laboratory settings, findings from lab-scale experiments are insufficient to directly guide field remediation. Therefore, extensive field-scale testing on the efficacy of rhamnolipid for remediation should be a priority in future research.Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article have been included as part of the supplementary information (SI).Supplementary information: SI data to this article can be found in the online version of this article. See DOI: https://doi.org/10.1039/d5en00982k.
Acknowledgements
The project ZR2024QD164 was supported by the Shandong Provincial Natural Science Foundation and the National Natural Science Foundation of China (Grant No. 32401403).References
- H. A. Craddock, D. Huang, P. C. Turner, L. Quirós-Alcalá and D. C. Payne-Sturges, Trends in neonicotinoid pesticide residues in food and water in the United States, 1999–2015, Environ. Health, 2019, 18, 7 CrossRef PubMed.
- M. Alsafran, M. Rizwan, K. Usman, M. H. Saleem and H. A. Jabri, Neonicotinoid insecticides in the environment: A critical review of their distribution, transport, fate, and toxic effects, J. Environ. Chem. Eng., 2022, 10, 108485 CrossRef CAS.
- T. J. Wood and D. Goulson, The environmental risks of neonicotinoid pesticides: a review of the evidence post 2013, Environ. Sci. Pollut. Res., 2017, 24, 17285–17325 CrossRef CAS PubMed.
- K. J. Kuechle, E. B. Webb, D. Mengel and A. R. Main, Factors Influencing Neonicotinoid Insecticide Concentrations in Floodplain Wetland Sediments across Missouri, Environ. Sci. Technol., 2019, 53, 10591–10600 CrossRef CAS PubMed.
- S. Mohapatra, L. Siddamallaiah, N. Y. Matadha, V. R. Udupi, D. P. Raj and S. Gadigeppa, Dissipation of neonicotinoid insecticides imidacloprid, indoxacarb and thiamethoxam on pomegranate (Punica granatum L.), Ecotoxicol. Environ. Saf., 2019, 171, 130–137 CrossRef CAS PubMed.
- J. Radolinski, J. Wu, K. Xia, W. C. Hession and R. D. Stewart, Plants mediate precipitation-driven transport of a neonicotinoid pesticide, Chemosphere, 2019, 222, 445–452 CrossRef CAS PubMed.
- A. Wang, G. Mahai, Y. Wan, Y. Jiang, Q. Meng, W. Xia, Z. He and S. Xu, Neonicotinoids and carbendazim in indoor dust from three cities in China: Spatial and temporal variations, Sci. Total Environ., 2019, 695, 133790 CrossRef CAS PubMed.
- N. Simon-Delso, V. Amaral-Rogers, L. P. Belzunces, J. M. Bonmatin, M. Chagnon, C. Downs, L. Furlan, D. W. Gibbons, C. Giorio, V. Girolami, D. Goulson, D. P. Kreutzweiser, C. H. Krupke, M. Liess, E. Long, M. McField, P. Mineau, E. A. D. Mitchell, C. A. Morrissey, D. A. Noome, L. Pisa, J. Settele, J. D. Stark, A. Tapparo, H. Van Dyck, J. Van Praagh, J. P. Van der Sluijs, P. R. Whitehorn and M. Wiemers, Systemic insecticides (neonicotinoids and fipronil): trends, uses, mode of action and metabolites, Environ. Sci. Pollut. Res., 2015, 22, 5–34 CrossRef CAS PubMed.
- M. J. Berens, P. D. Capel and W. A. Arnold, Neonicotinoid insecticides in surface water, groundwater, and wastewater across land use gradients and potential effects, Environ. Toxicol. Chem., 2021, 40, 1017–1033 CrossRef CAS PubMed.
- M. Menon, R. Mohanraj and W. Sujata, Monitoring of Neonicotinoid Pesticides in Water-Soil Systems Along the Agro-Landscapes of the Cauvery Delta Region, South India, Bull. Environ. Contam. Toxicol., 2021, 106, 1065–1070 Search PubMed.
- S. K. Papiernik, W. C. Koskinen, L. Cox, P. L. Rice, S. A. Clay, N. R. Werdin-Pfisterer and K. A. Norberg, Sorption-desorption of imidacloprid and its metabolites in soil and vadose zone materials, J. Agric. Food Chem., 2006, 54, 8163–8170 CrossRef CAS PubMed.
- H. Wang, Y. Huang, C. Shen, J. Wu, A. Yan and H. Zhang, Co-transport of pesticide acetamiprid and silica nanoparticles in biochar-amended sand porous media, J. Environ. Qual., 2016, 45, 1749–1759 CrossRef CAS PubMed.
- L. E. Satkowski, K. W. Goyne, S. H. Anderson, R. N. Lerch, E. B. Webb and D. D. Snow, Imidacloprid sorption and transport in cropland, grass buffer, and riparian buffer soils, Vadose Zone J., 2018, 17, 1–12 CrossRef CAS.
- M. Arias-Estévez, E. López-Periago, E. Martínez-Carballo, J. Simal-Gándara, J.-C. Mejuto and L. García-Río, The mobility and degradation of pesticides in soils and the pollution of groundwater resources, Agric., Ecosyst. Environ., 2008, 123, 247–260 CrossRef.
- J. A. Leiva, N. K. Peter, K. T. Morgan, D. M. Kadyampakeni and F. Leonardo, Imidacloprid transport and sorption nonequilibrium in single and multilayered columns of immokalee fine sand, PLoS One, 2017, 12, e0183767 CrossRef PubMed.
- Y. Zhu, B. Wang, F. Xu, B. Xu, U. Farooq, W. Chen and Z. Qi, Mobility of nitenpyram through saturated soil porous media: Combined effects of biochar colloids and low-molecular-weight organic acids, J. Mol. Liq., 2025, 417, 126665 CrossRef CAS.
- B. Xu, L. Lu, M. Liu, Q. Zhang, U. Farooq, T. Lu, Z. Qi and C. Ge, Low-molecular-weight organic acids-mediated transport of neonicotinoid pesticides through saturated soil porous media: Combined effects of the molecular structures of organic acids and the chemical properties of contaminants, Chemosphere, 2024, 349, 140870 CrossRef CAS PubMed.
- B. Xu, B. Wang, M. Liu, L. Lu, Z. Chen, U. Farooq, N. Xing, R. Miao and Z. Qi, Amino acids-mediated transport of nitenpyram through saturated soil porous media: Combined roles of amino acid types and soil properties, J. Mol. Liq., 2024, 410, 125661 CrossRef CAS.
- J. Radolinski, J. Wu, K. Xia and R. Stewart, Transport of a neonicotinoid pesticide, thiamethoxam, from artificial seed coatings, Sci. Total Environ., 2018, 618, 561–568 CrossRef CAS PubMed.
- M. L. Hladik, S. Bradbury, L. A. Schulte, M. Helmers, C. Witte, D. W. Kolpin, J. D. Garrett and M. Harris, Neonicotinoid insecticide removal by prairie strips in row-cropped watersheds with historical seed coating use, Agric., Ecosyst. Environ., 2017, 241, 160–167 CrossRef CAS.
- M. A. Bajeer, S. M. Nizamani, S. T. H. Sherazi and M. I. Bhanger, Adsorption and leaching potential of imidacloprid pesticide through alluvial soil, Am. J. Anal. Chem., 2012, 03, 604–611 CrossRef.
- K. Qian, A. Kumar, H. Zhang, D. Bellmer and R. Huhnke, Recent advances in utilization of biochar, Renewable Sustainable Energy Rev., 2015, 42, 1055–1064 CrossRef CAS.
- W. Yang, Y. Wang, P. Sharma, B. Li, K. Liu, J. Liu, M. Flury and J. Shang, Effect of naphthalene on transport and retention of biochar colloids through saturated porous media, Colloids Surf., A, 2017, 530, 146–154 CrossRef CAS.
- D. Mohan, A. Sarswat, Y. S. Ok and C. U. Pittman, Organic and inorganic contaminants removal from water with biochar, a renewable, low cost and sustainable adsorbent – A critical review, Bioresour. Technol., 2014, 160, 191–202 CrossRef CAS PubMed.
- W. Yang, J. Shang, B. Li and M. Flury, Surface and colloid properties of biochar and implications for transport in porous media, Crit. Rev. Environ. Sci. Technol., 2019, 50, 2484–2522 CrossRef.
- K. Zhao, L. Gao, Q. Zhang and J. Shang, Accumulation of sulfamethazine and ciprofloxacin on grain surface decreases the transport of biochar colloids in saturated porous media, J. Hazard. Mater., 2021, 417, 125908 CrossRef CAS PubMed.
- Y. Li, Y. Zhao, K. Cheng and F. Yang, Effects of biochar on transport and retention of phosphorus in porous media: Laboratory test and modeling, Environ. Pollut., 2022, 297, 118788 CrossRef CAS PubMed.
- P. Ma, Z. Qi, X. Wu, R. Ji and W. Chen, Biochar nanoparticles-mediated transport of organic contaminants in porous media: dependency on contaminant properties and effects of biochar aging, Carbon Res., 2023, 2, 4 CrossRef CAS.
- S. Yang, Y. Cao, Z. Li, C. Ma, Y. Huang, D. Hu, H. Liu and X. Huangfu, Cotransport of aged biochar colloids and thallium(I) in water-saturated porous media: Impact of the ionic strength, pH and aging degree, Sci. Total Environ., 2024, 927, 172294 CrossRef CAS PubMed.
- P. Zhang, H. Sun, L. Min and C. Ren, Biochars change the sorption and degradation of thiacloprid in soil: Insights into chemical and biological mechanisms, Environ. Pollut., 2018, 236, 158–167 CrossRef CAS PubMed.
- P. Zhang, L. Min, J. Tang, M. K. Rafiq and H. Sun, Sorption and degradation of imidacloprid and clothianidin in Chinese paddy soil and red soil amended with biochars, Biochar, 2020, 2, 329–341 CrossRef.
- H. Cheng, G. Tang, S. Wang, J. Rinklebe, T. Zhu, L. Cheng and S. Feng, Combined remediation effects of biochar and organic fertilizer on immobilization and dissipation of neonicotinoids in soils, Environ. Int., 2022, 169, 107500 CrossRef CAS PubMed.
- X. Dong, Y. Chu, Z. Tong, M. Sun, D. Meng, X. Yi, T. Gao, M. Wang and J. Duan, Mechanisms of adsorption and functionalization of biochar for pesticides: A review, Ecotoxicol. Environ. Saf., 2024, 272, 116019 CrossRef CAS PubMed.
- X. Mao, R. Jiang, W. Xiao and J. Yu, Use of surfactants for the remediation of contaminated soils: A review, J. Hazard. Mater., 2015, 285, 419–435 CrossRef CAS PubMed.
- P. Narendra Kumar, T. H. Swapna, M. Y. Khan, G. Reddy and B. Hameeda, Statistical optimization of antifungal iturin A production from Bacillus amyloliquefaciens RHNK22 using agro-industrial wastes, Saudi J. Biol. Sci., 2017, 24, 1722–1740 CrossRef CAS PubMed.
- C.-Y. Hsu, Z. H. Mahmoud, U. A.-R. Hussein, D. Abduvalieva, F. H. Alsultany and E. Kianfar, Biosurfactants: Properties, applications and emerging trends, S. Afr. J. Chem. Eng., 2025, 53, 21–39 Search PubMed.
- R. Jaiswal, P. V. Chellam and R. Selvasembian, Critical review on revamping circular economy strategies for the co-production of biosurfactants and lipase from agro-industrial wastes through resource recovery and life cycle assessment, Biomass Bioenergy, 2025, 196, 107733 CrossRef CAS.
- A. Lebron-Paler, J. E. Pemberton, B. A. Becker, W. H. Otto, C. K. Larive and R. M. Maier, Determination of the acid dissociation constant of the biosurfactant monorhamnolipid in aqueous solution by potentiometric and spectroscopic methods, Anal. Chem., 2006, 78, 7649–7658 Search PubMed.
- Y. Tian, C. Boulangé-Lecomte, A. Benamar, N. Giusti-Petrucciani, A. Duflot, S. Olivier, C. Frederick, J. Forget-Leray and F. Portet-Koltalo, Application of a crustacean bioassay to evaluate a multi-contaminated (metal, PAH, PCB) harbor sediment before and after electrokinetic remediation using eco-friendly enhancing agents, Sci. Total Environ., 2017, 607–608, 944–953 CrossRef CAS PubMed.
- S. Jorfi, A. Rezaee, G.-A. Mobeh-Ali and N. A. Jaafarzadeh, Application of biosurfactants produced by Pseudomonas aeruginosa SP4 for bioremediation of soils contaminated by pyrene, Soil Sediment Contam., 2013, 22, 890–911 CrossRef.
- Y. Chen, Y. Hu, Q. Guo, J. Yan and W. Wu, Effect of cations on the solubilization/deposition of triclosan in sediment-water-rhamnolipid system, Chemosphere, 2016, 159, 465–472 CrossRef CAS PubMed.
- S. Curiel-Alegre, D. de la Fuente-Vivas, A. H. A. Khan, J. García-Tojal, B. Velasco-Arroyo, C. Rumbo, G. Soja, C. Rad and R. Barros, Unveiling the capacity of bioaugmentation application, in comparison with biochar and rhamnolipid for TPHs degradation in aged hydrocarbons polluted soil, Environ. Res., 2024, 252, 118880 CrossRef CAS PubMed.
- M. Negarestani, H. Shayesteh, S. Reisi, S. Tavassoli, H. Farimaniraad, A. Mollahosseini and A. Kheradmand, Natural and environmentally friendly rhamnolipid functionalized luffa fibers for adsorptive removal of pharmaceutical contaminant: Batch and fixed-bed column studies, Chem. Eng. Sci., 2024, 299, 120552 Search PubMed.
- F. Bordas, P. Lafrance and R. Villemur, Conditions for effective removal of pyrene from an artificially contaminated soil using Pseudomonas aeruginosa 57SJ rhamnolipids, Environ. Pollut., 2005, 138, 69–76 CrossRef CAS PubMed.
- S. Wang and C. N. Mulligan, Rhamnolipid biosurfactant-enhanced soil flushing for the removal of arsenic and heavy metals from mine tailings, Process Biochem., 2009, 44, 296–301 CrossRef CAS.
- T. D. Renfro, W. Xie, G. Yang and G. Chen, Rhamnolipid surface thermodynamic properties and transport in agricultural soil, Colloids Surf., B, 2014, 115, 317–322 CrossRef CAS PubMed.
- K. A. Vu, K. Tawfiq and G. Chen, Rhamnolipid transport in biochar-amended agricultural soil, Water, Air, Soil Pollut., 2015, 226, 256 CrossRef.
- J. Liu, P. K. Subramaniam, L. Martin, P. Grasel, C. Taylor and G. Chen, E. coli O157:H7 Desorption by rhamnolipid biosurfactant in water-unsaturated porous media, J. Adhes. Sci. Technol., 2012, 25, 1803–1818 CrossRef.
- S. Liao, C. Liu, D. Pinchbeck, N. L. Cápiro, J. D. Fortner, L. M. Abriola and K. D. Pennell, Effects of rhamnolipid biosurfactant on the dissolution and transport of silver nanoparticles in porous media, Environ. Sci.: Nano, 2021, 8, 2492–2506 RSC.
- Y. Jin, J. Chen, Q. Zhang, U. Farooq, T. Lu, B. Wang, Z. Qi and W. Chen, Biosurfactant-affected mobility of oxytetracycline and its variations with surface chemical heterogeneity in saturated porous media, Water Res., 2023, 244, 120509 CrossRef CAS PubMed.
- A. Abbasi, L. Qi and G. Chen, Transport of nanoscale zero-valent iron in the presence of rhamnolipid, Sci. Total Environ., 2024, 927, 172279 CrossRef CAS PubMed.
- Z. Chen, B. Wang, U. Farooq, T. Lu, Z. Qi, L. Zhang and R. Miao, pH-dependent transport of tetracycline in saturated porous media: Single and combined effects of surfactants and iron oxide colloids, J. Hazard. Mater., 2025, 490, 137829 CrossRef CAS PubMed.
- M. Zhen, H. Chen, Q. Liu, B. Song, Y. Wang and J. Tang, Combination of rhamnolipid and biochar in assisting phytoremediation of petroleum hydrocarbon contaminated soil using Spartina anglica, J. Environ. Sci., 2019, 85, 107–118 CrossRef CAS PubMed.
- M. Zhen, J. Tang, C. Li and H. Sun, Rhamnolipid-modified biochar-enhanced bioremediation of crude oil-contaminated soil and mediated regulation of greenhouse gas emission in soil, J. Soils Sediments, 2020, 21, 123–133 CrossRef.
- R. Zhao, X. Ma, J. Xu and Q. Zhang, Removal of the pesticide imidacloprid from aqueous solution by biochar derived from peanut shell, BioResources, 2018, 13, 5656–5669 CAS.
- C. Wu, M. Jiang, Z. Ye, Y. Cai, Y. Shen, H. Wang, Q. Lin, C. Shen, B. Hu and L. Lou, Exploring the recycling of bioleaching functional bacteria and sulfur substrate using the sulfur-covered biochar particles, Environ. Sci. Eur., 2020, 32, 70 CrossRef CAS.
- M. E. Kabir, C. Johansen and R. W. Bell, Subsoil rhizosphere modification by chickpea under a dry topsoil: implications for phosphorus acquisition, J. Plant Nutr. Soil Sci., 2015, 178, 904–913 CrossRef CAS.
- H. Sheng, P. Zhou, Y. Zhang, Y. Kuzyakov, Q. Zhou, T. Ge and C. Wang, Loss of labile organic carbon from subsoil due to land-use changes in subtropical China, Soil Biol. Biochem., 2015, 88, 148–157 CrossRef CAS.
- P. N. Mitropoulou, V. I. Syngouna and C. V. Chrysikopoulos, Transport of colloids in unsaturated packed columns: Role of ionic strength and sand grain size, Chem. Eng. J., 2013, 232, 237–248 CrossRef CAS.
- S. Irace-Guigand and J. J. Aaron, The role of organic colloids in herbicide transfer to rivers: a quantitative study of triazine and phenylurea interactions with colloids, Anal. Bioanal. Chem., 2003, 376, 431–435 CrossRef CAS PubMed.
- K. Sun, S. Dong, Y. Sun, B. Gao, W. Du, H. Xu and J. Wu, Graphene oxide-facilitated transport of levofloxacin and ciprofloxacin in saturated and unsaturated porous media, J. Hazard. Mater., 2018, 348, 92–99 CrossRef CAS PubMed.
- X. Qu, H. Fu, J. Mao, Y. Ran, D. Zhang and D. Zhu, Chemical and structural properties of dissolved black carbon released from biochars, Carbon, 2016, 96, 759–767 CrossRef CAS.
- Q. Fang, B. Chen, Y. Lin and Y. Guan, Aromatic and hydrophobic surfaces of wood-derived biochar enhance perchlorate adsorption via hydrogen bonding to oxygen-containing organic groups, Environ. Sci. Technol., 2013, 48, 279–288 CrossRef PubMed.
- W. H. Noordman, M. L. Brusseau and D. B. Janssen, Adsorption of a multicomponent rhamnolipid surfactant to soil, Environ. Sci. Technol., 2000, 34, 832–838 CrossRef CAS.
- P. Sun, K. Zhang, J. Fang, D. Lin, M. Wang and J. Han, Transport of TiO2 nanoparticles in soil in the presence of surfactants, Sci. Total Environ., 2015, 527–528, 420–428 CrossRef CAS PubMed.
- W. H. Noordman, J. W. Bruining, P. Wietzes and D. B. Janssen, Facilitated transport of a PAH mixture by a rhamnolipid biosurfactant in porous silica matrices, J. Contam. Hydrol., 2000, 44, 119–140 CrossRef CAS.
- T. G. Ambaye, F. Formicola, S. Sbaffoni, A. Franzetti and M. Vaccari, Insights into rhamnolipid amendment towards enhancing microbial electrochemical treatment of petroleum hydrocarbon contaminated soil, Chemosphere, 2022, 307, 136126 CrossRef CAS PubMed.
- J. C. Mata-Sandoval, J. Karns and A. Torrents, Effect of rhamnolipids produced bypseudomonas aeruginosa UG2 on the solubilization of pesticides, Environ. Sci. Technol., 2000, 34, 4923–4930 CrossRef CAS.
- H. T. Wattanaphon, A. Kerdsin, C. Thammacharoen, P. Sangvanich and A. S. Vangnai, A biosurfactant from Burkholderia cenocepacia BSP3 and its enhancement of pesticide solubilization, J. Appl. Microbiol., 2008, 105, 416–423 CrossRef CAS PubMed.
- G. Liu, H. Zhong, X. Yang, Y. Liu, B. Shao and Z. Liu, Advances in applications of rhamnolipids biosurfactant in environmental remediation: A review, Biotechnol. Bioeng., 2018, 115, 796–814 CrossRef CAS PubMed.
- H. Yu, G.-h. Huang, C.-j. An and J. Wei, Combined effects of DOM extracted from site soil/compost and biosurfactant on the sorption and desorption of PAHs in a soil–water system, J. Hazard. Mater., 2011, 190, 883–890 CrossRef CAS PubMed.
- M. Liu, L. Lu, Q. Zhang, U. Farooq, T. Lu, Y. Li, D. Li and Z. Qi, Insight into the inhibitory mechanism of rhamnolipid biosurfactant on the mobility of sulfonamide antibiotics in saturated soil porous media, Colloids Surf., A, 2024, 680, 132683 Search PubMed.
- P. Zhang, H. Sun, C. Ren, L. Min and H. Zhang, Sorption mechanisms of neonicotinoids on biochars and the impact of deashing treatments on biochar structure and neonicotinoids sorption, Environ. Pollut., 2018, 234, 812–820 Search PubMed.
- Y. Shi, S. Wang, M. Xu, X. Yan, J. Huang and H. Wang, Removal of neonicotinoid pesticides by adsorption on modified Tenebrio molitor frass biochar: Kinetics and mechanism, Sep. Purif. Technol., 2022, 297, 121506 CrossRef CAS.
- W. Yan, J. Zhang, C. Wang and Y. Xia, An urchin-shaped covalent organic framework with rich nitrogen for efficient removal of neonicotinoid insecticides in honey and fruits, Food Chem., 2023, 429, 136872 CrossRef CAS PubMed.
- O. K. Borggaard, P. E. Holm and B. W. Strobel, Potential of dissolved organic matter (DOM) to extract As, Cd, Co, Cr, Cu, Ni, Pb and Zn from polluted soils: A review, Geoderma, 2019, 343, 235–246 CrossRef CAS.
- Y. Audette, K. A. Congreves, K. Schneider, G. C. Zaro, A. L. P. Nunes, H. Zhang and R. P. Voroney, The effect of agroecosystem management on the distribution of C functional groups in soil organic matter: A review, Biol. Fertil. Soils, 2021, 57, 881–894 CrossRef CAS PubMed.
- Y. Li, P. Su, Y. Li, K. Wen, G. Bi and M. Cox, Adsorption-desorption and degradation of insecticides clothianidin and thiamethoxam in agricultural soils, Chemosphere, 2018, 207, 708–714 CrossRef CAS PubMed.
- A. K. Aseperi, R. Busquets, P. S. Hooda, P. C. W. Cheung and J. Barker, Behaviour of neonicotinoids in contrasting soils, J. Environ. Manage., 2020, 276, 111329 CrossRef CAS PubMed.
- K. G. I. D. Kumari, P. Moldrup, M. Paradelo, L. Elsgaard and L. W. de Jonge, Effects of biochar on dispersibility of colloids in agricultural soils, J. Environ. Qual., 2017, 46, 143–152 CrossRef CAS PubMed.
- Q. Meng, L. Jin, L. Cheng, J. Fang and D. Lin, Release and sedimentation behaviors of biochar colloids in soil solutions, J. Environ. Sci., 2021, 100, 269–278 CrossRef CAS PubMed.
- M. Liu, L. Lu, Z. Chen, B. Wang, Y. Li, Q. Zhang, U. Farooq and Z. Qi, Ferrihydrite colloid-mediated transport of sulfamerazine in saturated porous media: Combined roles of rhamnolipid and solution pH, Colloids Surf., A, 2024, 700, 134842 CrossRef CAS.
- G. Twagirayezu, H. Cheng, Y. Wu, H. Lu, S. Huang, X. Fang and O. Irumva, Insights into the influences of biochar on the fate and transport of pesticides in the soil environment: a critical review, Biochar, 2024, 6, 9 CrossRef CAS.
- X. Y. Yu, C. L. Mu, C. Gu, C. Liu and X. J. Liu, Impact of woodchip biochar amendment on the sorption and dissipation of pesticide acetamiprid in agricultural soils, Chemosphere, 2011, 85, 1284–1289 CrossRef CAS PubMed.
- D. Pan, X. Wu, P. Chen, Z. Zhao, F. Fan, Y. Wang, M. Zhu, J. Xue and Y. Wang, New insights into the interactions between humic acid and three neonicotinoid pesticides, with multiple spectroscopy technologies, two-dimensional correlation spectroscopy analysis and density functional theory, Sci. Total Environ., 2021, 798, 149237 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2026 |