Glyco-functionalized dinuclear rhenium(i) complexes for cell imaging†
Abstract
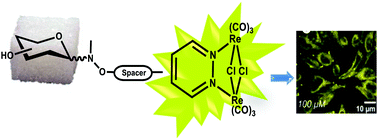
The design, synthesis and photophysical characterization of four new luminescent glycosylated luminophores based on dinuclear rhenium complexes, namely Glyco-Re, are described. The derivatives have the general formula [Re2(μ-Cl)2(CO)6(μ-pydz-R)] (R-pydz = functionalized 1,2-pyridazine), where a sugar residue (R) is covalently bound to the pyridazine ligand in the β position. Different synthetic pathways have been investigated including the so-called neo-glycorandomization procedure, affording stereoselectively glyco-conjugates containing glucose and maltose in a β anomeric configuration. A multivalent dinuclear rhenium glycodendron bearing three glucose units is also synthesized. All the Glyco-Re conjugates are comprehensively characterized and their photophysical properties and cellular internalization experiments on human cervical adenocarcinoma (HeLa) cells are reported. The results show that such Glyco-Re complexes display interesting bio-imaging properties, i.e. high cell permeability, organelle selectivity, low cytotoxicity and fast internalization. These findings make the presented Glyco-Re derivatives efficient phosphorescent probes suitable for cell imaging application.


 Please wait while we load your content...
Please wait while we load your content...