The virulence factor LecB varies in clinical isolates: consequences for ligand binding and drug discovery†
Abstract
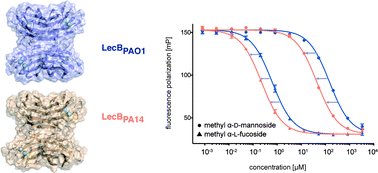
P. aeruginosa causes a substantial number of nosocomial infections and is the leading cause of death of cystic fibrosis patients. This Gram-negative bacterium is highly resistant against antibiotics and further protects itself by forming a biofilm. Moreover, a high genomic variability among clinical isolates complicates therapy. Its lectin LecB is a virulence factor and necessary for adhesion and biofilm formation. We analyzed the sequence of LecB variants in a library of clinical isolates and demonstrate that it can serve as a marker for strain family classification. LecB from the highly virulent model strain PA14 presents 13% sequence divergence with LecB from the well characterized PAO1 strain. These differences might result in differing ligand binding specificities and ultimately in reduced efficacy of drugs directed towards LecB. Despite several amino acid variations at the carbohydrate binding site, glycan array analysis showed a comparable binding pattern for both variants. A common high affinity ligand could be identified and after its chemoenzymatic synthesis verified in a competitive binding assay: an N-glycan presenting two blood group O epitopes (H-type 2 antigen). Molecular modeling of the complex suggests a bivalent interaction of the ligand with the LecB tetramer by bridging two separate binding sites. This binding rationalizes the strong avidity (35 nM) of LecBPA14 to this human fucosylated N-glycan. Biochemical evaluation of a panel of glycan ligands revealed that LecBPA14 demonstrated higher glycan affinity compared to LecBPAO1 including the extraordinarily potent affinity of 70 nM towards the monovalent human antigen Lewisa. The structural basis of this unusual high affinity ligand binding for lectins was rationalized by solving the protein crystal structures of LecBPA14 with several glycans.
- This article is part of the themed collection: Global challenges: Health & Food


 Please wait while we load your content...
Please wait while we load your content...