Targeting antioxidant pathways with ferrocenylated N-heterocyclic carbene supported gold(i) complexes in A549 lung cancer cells†
Abstract
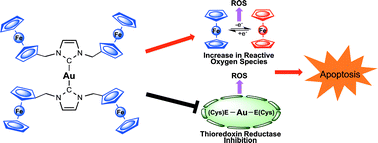
Ferrocene containing N-heterocyclic carbene (NHC) ligated gold(I) complexes of the type [Au(NHC)2]+ were prepared and found to be capable of regulating the formation of reactive oxygen species (ROS) via multiple mechanisms. Single crystal X-ray analysis of bis(1-(ferrocenylmethyl)-3-mesitylimidazol-2-ylidene)-gold(I) chloride (5) and bis(1,3-di(ferrocenylmethyl)imidazol-2-ylidene)-gold(I) chloride (6) revealed a quasi-linear geometry around the gold(I) centers (i.e., the C–Au–C bond angle were measured to be ∼177° and all the Au–Ccarbene bonds distances were in the range of 2.00 (7)–2.03 (1) Å). A series of cell studies indicated that cell proliferation inhibition and ROS generation were directly proportional to the amount of ferrocene contained within the [Au(NHC)2]+ complexes (IC50 of 6 < 5 < bis(1-benzyl-3-mesitylimidazol-2-ylidene)-gold(I) chloride (4)). Complexes 4–6 were also confirmed to inhibit thioredoxin reductase as inferred from lipoate reduction assays and increased chelatable intracellular zinc concentrations. RNA microarray gene expression assays revealed that 6 induces endoplasmic reticulum stress response pathways as a result of ROS increase.
- This article is part of the themed collection: Global challenges: Health & Food

 Please wait while we load your content...
Please wait while we load your content...