Local structure of the metal–organic perovskite dimethylammonium manganese(ii) formate†
Abstract
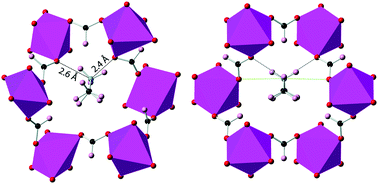
We report total neutron scattering measurements on the metal–organic perovskite analogue dimethylammonium manganese(II) formate, (CD3)2ND2[Mn(DCO2)3]. Reverse Monte Carlo modelling shows that, in both the disordered high-temperature and ordered low-temperature phases, the ammonium moiety forms substantially shorter hydrogen bonds (N⋯O = 2.4 Å and 2.6 Å) than are visible in the average crystal structures. These bonds result from a pincer-like motion of two adjacent formate ions about the dimethylammonium ion in such a way that the framework can adjust independently to the positions of nearest-neighbour dimethylammonium ions. At low temperatures the shortest hydrogen bond is less favourable, apparently because it involves a greater distortion of the framework. Furthermore, in the high-temperature phase, in addition to the three disordered nitrogen positions expected from the average crystal structure, there appear to be also smaller probability maxima between these positions, corresponding to orientations in which the dimethylammonium is hydrogen-bonded to the two oxygen atoms of a single formate ion. The spontaneous strain across the phase transition reveals a contraction of the framework about the dimethylammonium cation, continuing as the material is cooled below the transition temperature. These results provide direct evidence of the local atomic structure of the guest-framework hydrogen bonding, and in particular the distortions of the framework responsible for the phase transition in this system.
- This article is part of the themed collection: Flexibility and Disorder in Metal-Organic Frameworks

 Please wait while we load your content...
Please wait while we load your content...