Themed collection Physical chemistry at the cross-road of advanced oxide materials

Physical-chemistry at the cross-road of advanced oxide materials
The relevance of physical chemistry studies for understanding the properties of oxide materials and for their exploitation in several technological applications is highlighted by this high quality collection of papers.

Phys. Chem. Chem. Phys., 2013,15, 13236-13237
https://doi.org/10.1039/C3CP90102E
Oxidic materials: an endless frontier
Both microporous (left) and mesoporous (right) oxidic solids can be fashioned into high-performance heterogeneous single-site catalysts.

Phys. Chem. Chem. Phys., 2013,15, 13238-13242
https://doi.org/10.1039/C3CP52182F
Coupling physical chemical techniques with hydrotalcite-like compounds to exploit their structural features and new multifunctional hybrids with luminescent properties
Recent advances in physical–chemical characterization to study HTlc structure and to design and synthesise, via intercalation chemistry, new hybrid materials.

Phys. Chem. Chem. Phys., 2013,15, 13254-13269
https://doi.org/10.1039/C3CP51581H
Recent advances in visible-light-responsive photocatalysts for hydrogen production and solar energy conversion – from semiconducting TiO2 to MOF/PCP photocatalysts
This article reviews the recent advances in the development of visible-light-responsive photocatalysts designed for water splitting and solar energy conversion.

Phys. Chem. Chem. Phys., 2013,15, 13243-13253
https://doi.org/10.1039/C3CP51427G
Molecular-level characterization of the structure and the surface chemistry of periodic mesoporous organosilicates using DNP-surface enhanced NMR spectroscopy
DNP-SENS enables the discrimination of functionalized, surface and subsurface molecular subunits in a crystal-like periodic mesoporous organosilicate.

Phys. Chem. Chem. Phys., 2013,15, 13270-13274
https://doi.org/10.1039/C3CP00026E
Impact of Pt additives on the surface reactions between SnO2, water vapour, CO and H2; an operando investigation
The impact of Pt doping was investigated by a combination of simultaneously performed operando DRIFT (Diffuse Reflectance Infrared Fourier Transform) spectroscopy, DC resistance measurements and exhaust analysis by using a MS (Mass Spectrometer).

Phys. Chem. Chem. Phys., 2013,15, 19151-19158
https://doi.org/10.1039/C3CP52782D
Molecular theory of graphene oxide
Applied to graphene oxide, the molecular theory of graphene considers its oxide as a final product in the succession of polyderivatives related to a series of oxidation reactions involving different oxidants.
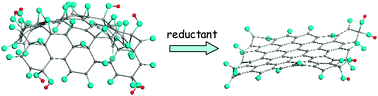
Phys. Chem. Chem. Phys., 2013,15, 13304-13322
https://doi.org/10.1039/C3CP00032J
On the adsorption/reaction of acetone on pure and sulfate -modified zirconias
In situ FTIR spectroscopy is employed to investigate the ambient temperature adsorption of acetone on various pure and sulfate-doped zirconia specimens.
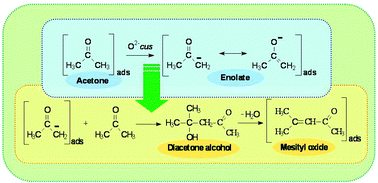
Phys. Chem. Chem. Phys., 2013,15, 13446-13461
https://doi.org/10.1039/C3CP50990G
Novel insights on magadiite disaggregation: a multitechnique study on thermal stability
H,Na-Magadiite prepared by disaggregation of CTA-magadiite displays a particular morphology and is thermally stable up to 1273 K; FTIR and NMR signals were found to indicate the interlayer interactions arising at high temperatures.

Phys. Chem. Chem. Phys., 2013,15, 13434-13445
https://doi.org/10.1039/C3CP51491A
Probing the surface of nanosheet H-ZSM-5 with FTIR spectroscopy
FTIR in situ adsorption of molecular hydrogen, carbon monoxide, water, methanol, pyridine and 2,4,6-trimethylpyridine (collidine) on nanosheet H-ZSM-5 is reported.

Phys. Chem. Chem. Phys., 2013,15, 13363-13370
https://doi.org/10.1039/C3CP51280K
Effect of ceria structural properties on the catalytic activity of Au–CeO2 catalysts for WGS reaction
Two gold based catalysts supported on ceria prepared by different methods (urea gelation coprecipitation, UGC, and coprecipitation, CP) have been synthesized and tested in the WGS reaction, showing quite different catalytic behaviors.
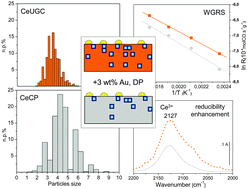
Phys. Chem. Chem. Phys., 2013,15, 13400-13408
https://doi.org/10.1039/C3CP51198G
The interactions of methyl tert-butyl ether on high silica zeolites : a combined experimental and computational study
The effect of physico-chemical properties of different dealuminated high silica zeolites on MTBE adsorption were studied by means of both experimental and computational approaches.

Phys. Chem. Chem. Phys., 2013,15, 13275-13287
https://doi.org/10.1039/C3CP51684A
Niobium–silica catalysts for the selective epoxidation of cyclic alkenes : the generation of the active site by grafting niobocene dichloride
Niobium-containing silica materials obtained by deposition via liquid-phase grafting or dry impregnation of niobocene(IV) dichloride are active and selective catalysts in the epoxidation of alkenes in the presence of aqueous hydrogen peroxide.
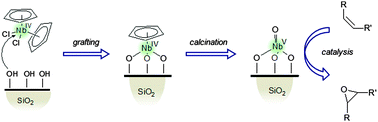
Phys. Chem. Chem. Phys., 2013,15, 13354-13362
https://doi.org/10.1039/C3CP51570B
Structural characterization and thermal and chemical stability of bioactive molecule–hydrotalcite (LDH) nanocomposites
Hydrotalcites intercalated with bioactive molecules were solved by X-ray powder diffraction, and their stability was studied by spectroscopic and thermogravimetric techniques.
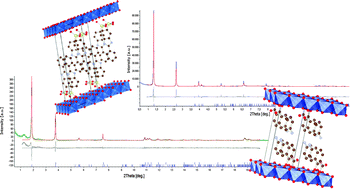
Phys. Chem. Chem. Phys., 2013,15, 13418-13433
https://doi.org/10.1039/C3CP51235E
Surface features of TiO2 nanoparticles : combination modes of adsorbed CO probe the stepping of (101) facets
Studies of CO adsorption on TiO2 materials with different morphologies identified frustrated translational CO modes as a probe of surface topography.
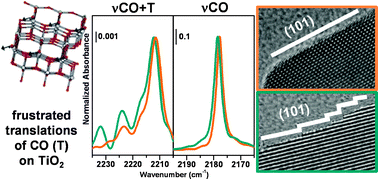
Phys. Chem. Chem. Phys., 2013,15, 13391-13399
https://doi.org/10.1039/C3CP51524A
Effect of water and ammonia on surface species formed during NOx storage–reduction cycles over Pt–K/Al2O3 and Pt–Ba/Al2O3 catalysts
Dissociative chemisorption of H2O and NH3 on Pt–Ba/Al2O3 and Pt–K/Al2O3 catalysts transforms bidentate nitrates and linear isocyanates into ionic species.

Phys. Chem. Chem. Phys., 2013,15, 13409-13417
https://doi.org/10.1039/C3CP51195B
A flexible, bolaamphiphilic template for mesoporous silicas
A series of mesoporous silicas with various morphologies and with either cylindrical or elliptical pores, arranged in one of several symmetries (c2mm, p2gg and p6mm), has been prepared using a novel symmetrical bolaamphiphile, able to self-organize into ribbon phases.
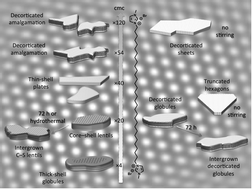
Phys. Chem. Chem. Phys., 2013,15, 13343-13353
https://doi.org/10.1039/C3CP51546J
Surface properties of alumino-silicate single-walled nanotubes of the imogolite type
A, B and C surfaces of imogolite-like NTs have different accessibilities to different gas molecules.
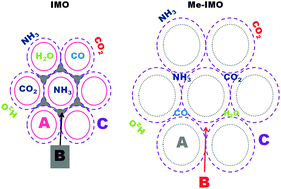
Phys. Chem. Chem. Phys., 2013,15, 13381-13390
https://doi.org/10.1039/C3CP51508G
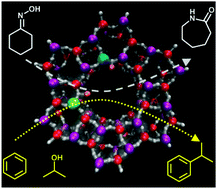
Investigating site-specific interactions and probing their role in modifying the acid-strength in framework architectures
Through the controlled promotion of site-specific interactions, extra framework zinc ions modify the Brønsted acidity in novel bimetallic nanoporous materials.
Phys. Chem. Chem. Phys., 2013,15, 13288-13295
https://doi.org/10.1039/C3CP51182K
Preparation of aluminum-containing mesoporous silica with hierarchical macroporous architecture and its enhanced catalytic activities
Al-containing porous silica with ordered mesoporous and hierarchical macroporous structure can serve as a solid acid catalyst for tetrahydropyranylation of linear alcohols with enhanced performance.

Phys. Chem. Chem. Phys., 2013,15, 13323-13328
https://doi.org/10.1039/C3CP51022K
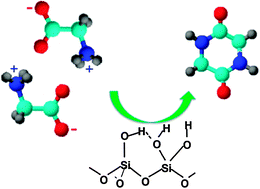
A comparative study of the catalysis of peptide bond formation by oxide surfaces
Amino acids often polymerise in the presence of inorganic surfaces but the process is more complicated than generally believed.
Phys. Chem. Chem. Phys., 2013,15, 13371-13380
https://doi.org/10.1039/C3CP51282G
The electronic structure of MgO nanotubes . An ab initio quantum mechanical investigation
The structural, vibrational and response properties of the (n,0) and (m,m) MgO nanotubes are computed by using a Gaussian type basis set, a hybrid functional (B3LYP) and the CRYSTAL09 code.

Phys. Chem. Chem. Phys., 2013,15, 13296-13303
https://doi.org/10.1039/C3CP50979F
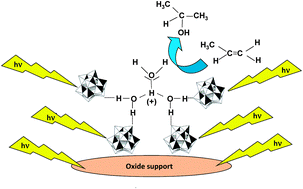
Keggin heteropolyacid H3PW12O40 supported on different oxides for catalytic and catalytic photo-assisted propene hydration
Catalytic and catalytic photo-assisted hydration of propene to form 2-propanol in gas–solid regime at atmospheric pressure and 85 °C was carried out by using a heteropolyacid (POM) supported on different oxides.
Phys. Chem. Chem. Phys., 2013,15, 13329-13342
https://doi.org/10.1039/C3CP51142A
About this collection
The relevance of physical chemistry studies for understanding the properties of oxide materials and for their exploitation in several technological applications is highlighted by this high quality collection of papers.