Synthesis and evaluation of an agrocin 84 toxic moiety (TM84) analogue as a malarial threonyl tRNA synthetase inhibitor†
Abstract
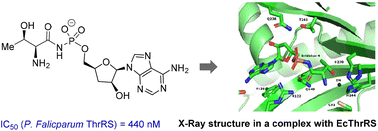
An analogue of a toxic moiety (TM84) of natural product agrocin 84 containing threonine amide instead of 2,3-dihydroxy-4-methylpentanamide was prepared and evaluated as a putative Plasmodium falciparum threonyl t-RNA synthetase (PfThrRS) inhibitor. This TM84 analogue features submicromolar inhibitory potency (IC50 = 440 nM) comparable to that of borrelidin (IC50 = 43 nM) and therefore complements chemotypes known to inhibit malarial PfThrRS, which are currently limited to borrelidin and its analogues. The crystal structure of the inhibitor in complex with the E. coli homologue enzyme (EcThrRS) was obtained, revealing crucial ligand–protein interactions that will pave the way for the design of novel ThrRS inhibitors.


 Please wait while we load your content...
Please wait while we load your content...