The effect of amide bond orientation and symmetry on the self-assembly and gelation of discotic tripeptides†
Abstract
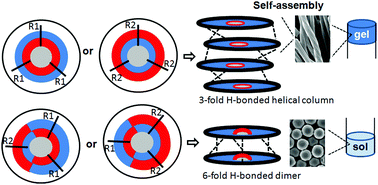
A series of discotic tripeptides containing a rigid aromatic core and L-phenylalanine have been developed. The orientation of the amide bonds yielded variations of the structure and self-assembly properties of the compounds. The aggregation behavior of the discotic tripeptides was studied by various spectroscopic techniques. The morphology of the resulting aggregates was studied by field emission electron microscopy and atomic force microscopy. These studies showed that the orientation of the amide bonds has a strong influence on the intermolecular interactions, resulting in huge differences in the aggregation properties, and morphology of the discotic tripeptides. Only the C3-symmetric discotic tripeptides formed organogels. The supramolecular aggregation mechanism of N-centered and C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O-centered discotic tripeptides for forming 3-fold intermolecular H-bonded helical column were the same, there was only a smaller enthalpy change due to the occurrence of longer distances for the N–H⋯O
O-centered discotic tripeptides for forming 3-fold intermolecular H-bonded helical column were the same, there was only a smaller enthalpy change due to the occurrence of longer distances for the N–H⋯O![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C bonds of the N-centered discotic tripeptide. Whereas, the C2-symmetric discotic tripeptides 2 and 3 adopted a 6-fold intermolecular H-bonded dimer structure. Thus, this report presents a valuable approach for the fine-tuning of the discotic tripeptide based functional material.
C bonds of the N-centered discotic tripeptide. Whereas, the C2-symmetric discotic tripeptides 2 and 3 adopted a 6-fold intermolecular H-bonded dimer structure. Thus, this report presents a valuable approach for the fine-tuning of the discotic tripeptide based functional material.


 Please wait while we load your content...
Please wait while we load your content...