Genetically engineered haemoglobin wrapped covalently with human serum albumins as an artificial O2 carrier†
Abstract
We describe the synthesis and O2 affinity of genetically engineered human adult haemoglobin (rHbA) wrapped covalently with recombinant human serum albumins (rHSAs) as an artificial O2 carrier used for a completely synthetic red blood cell (RBC) substitute. Wild-type rHbA [rHbA(wt)] expressed in yeast species Pichia pastoris shows an identical amino acid sequence and three-dimensional structure to those of native HbA. It is particularly interesting that two orientations of the prosthetic haem group in rHbA(wt) were aligned by gentle heating in the natural form. Covalent wrapping of rHbA(wt) with three rHSAs conferred a core–shell structured haemoglobin–albumin cluster: rHbA(wt)–rHSA3. Three variant clusters containing an rHbA mutant core were also created: Leu-β28 → Phe, Leu-β28 → Trp, and Leu-β28 → Tyr/His-β63 → Gln. Replacement of Leu-β28 with Trp decreased the distal space in the haem pocket, thereby yielding a cluster with moderately low O2 affinity which is nearly the same as that of human RBC.
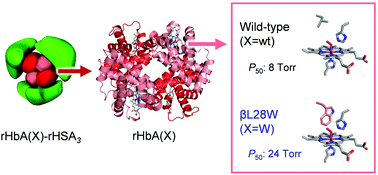


 Please wait while we load your content...
Please wait while we load your content...