Effect of donor modification on the photo-physical and photo-voltaic properties of N-alkyl/aryl amine based chromophores†
Abstract
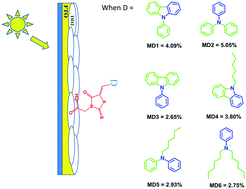
Six push–pull sensitizers (MD1 to MD6) having rhodanine-3-acetic acid acting as an electron withdrawing group and N-alkyl/aryl amine as a donor moiety were planned and prepared to rationalize the influence of donor alteration on absorption/emission properties and photon to current conversion efficiency (η). The structures of all the sensitizers were ascertained by 1H-NMR, 13C-NMR, mass spectroscopy and CHN analysis. The photophysical parameters of these sensitizers were recorded in nine solvents having different polarities to study the solvatochromic behavior of these dyes. Nanocrystalline mesoporous TiO2 based DSSCs were constructed by employing MD1 to MD6. The overall photon to current conversion efficiency was obtained in the range from 2.75% to 5.06% where the MD2 chromophore shows the highest efficiency of 5.06%. All efficiencies were correlated with the structural modification of the donor moiety to establish a structure–property relationship. The improved light to power conversion efficiency of MD2 is because of its high short circuit current Jsc (12.70 mA), high resistance in electron recombination and longer lifetime of injected electrons. The rigidity of the donor moiety (MD1) results in stabilization of the HOMO energy level, which increases the band gap and shows a decrease in efficiency along with a blue shift in the photophysical properties. N-Alkylation of the donor moiety (MD4–MD6) causes an increase in the molar absorptivity but affects the conjugation and shows a negative impact on efficiency. Lack of planarity in the pi-conjugation (MD3) has a negative impact on the photophysical and photovoltaic properties. An extensive DFT study reveals that the experimental absorption of all the dyes has very good correlation with theoretical vertical excitation obtained by time-dependent density functional theory (TD-DFT).


 Please wait while we load your content...
Please wait while we load your content...