Application of syringe to syringe dispersive micro-solid phase extraction using a magnetic layered double hydroxide for the determination of cadmium(ii) and lead(ii) ions in food and water samples†
Abstract
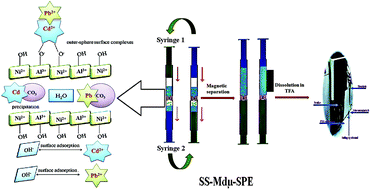
In this research work, for the first time, a new mode of dispersive solid-phase extraction named syringe to syringe magnetic dispersive micro-solid phase extraction is introduced as an efficient, simple, rapid, and eco-friendly sample extraction method. A nickel–aluminum layered double hydroxide coated on magnetic nanoparticles (Fe3O4@Ni–Al–CO3-LDH), as an efficient nanosorbent, was synthesized and successfully applied for the concurrent extraction of trace amounts of Cd2+ and Pb2+ ions in food and water samples prior to their determination using a microsampling-flame atomic absorption spectrometry technique. After extraction and magnetic separation, the elution step was easily performed through dissolving the magnetic layered double hydroxide (LDH) containing the target analytes in an acidic solution. Also the use of a magnetic LDH overcomes the difficulty of phase separation that decreases the extraction time. The optimum conditions efficiently evaluated via the response surface methodology (RSM) were pH 7.0, 13.7 mg of the nanosorbent, 16 extraction cycles, 125 μL of 3.8 mol L−1 trifluoroacetic acid, and an ultrasonic time of 75 s. According to the optimum conditions obtained in above, good linear ranges (0.75–35 ng mL−1 for Cd2+ ions and 4.0–370 ng mL−1 for Pb2+ ions) with coefficient of determination (R2) values higher than 0.995 were obtained. The limits of detection were found to be 0.25 and 1.0 ng mL−1 for Cd2+ and Pb2+, respectively. The preconcentration factor for the target analytes was 80 in a 10 mL sample solution. The obtained results indicated the high quality of the developed procedure for the much-needed quantification of the aforementioned metal ions in food and water samples.
- This article is part of the themed collection: Analytical Methods Recent HOT articles


 Please wait while we load your content...
Please wait while we load your content...