Mixed matrix membranes decorated with in situ self-assembled polymeric nanoparticles driven by electrostatic interaction†
Abstract
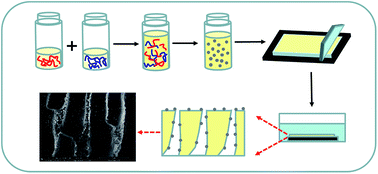
A novel ultrafiltration membrane is developed by incorporating in situ self-assembled polymeric nanoparticles into the membrane matrix. The chemical stability of the nanoparticles in regard to pH and temperature is explored via a rational method. The result indicates that the electrostatic interaction strength between the two polyelectrolytes can withstand a broad pH range (pH 1–11) and a relatively high temperature (80 °C). Meanwhile, the nanoparticle size and distribution in the membranes are investigated by scanning electron microscopy (SEM), which are proved to be affected by the polyelectrolyte concentration and molecular weight. The contact angle values of the prepared membranes show that the membrane hydrophilicity is improved by adding polymeric nanoparticles. Furthermore, the molecular weight cut-off and pore size of the membranes are increased with added nanoparticle loading. More importantly, the mixed matrix membranes exhibit simultaneous increases in permeability and rejection of contaminants compared with the pristine PES membrane, which is ascribed to the appropriate pore size and enhanced hydrophilicity of the membranes. Especially, the composite membrane mixed with 3 wt% polymeric nanoparticles displays the highest pure water flux (460 L m−2 h−1), which is 3 times that of the pristine PES membrane, and 97.6% rejection of BSA. This work provides a new strategy for preparing flexible polymeric nanoparticles and developing high-performance mixed matrix membranes for water treatment.


 Please wait while we load your content...
Please wait while we load your content...