Highly efficient synthesis of benzodioxins with a 2-site quaternary carbon structure by secondary amine-catalyzed dual Michael cascade reactions†
Abstract
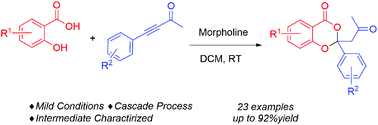
Salicylic acids and substituted ynones were employed as substrates to afford a class of valuable 4H-benzo[d][1,3]dioxin-4-ones with a 2-site quaternary carbon structure in up to 92% yield by secondary amine-catalyzed dual Michael cascade reactions under mild reaction conditions. The α,β-unsaturated ketone as the key intermediate in the cascade process was successfully separated and characterized. As a result, a new reaction route for ynone species is demonstrated, which is totally different from the existing allenamine activation model.
- This article is part of the themed collection: Synthetic methodology in OBC

 Please wait while we load your content...
Please wait while we load your content...