Tricyclanos: conformationally constrained nucleoside analogues with a new heterotricycle obtained from a d-ribofuranose unit†
Abstract
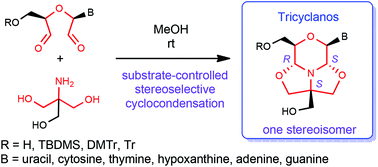
A novel type of nucleoside analogue in which the sugar part is replaced by a new tricycle, 3,7,10-trioxa-11-azatricyclo[5.3.1.05,11]undecane has been prepared by substrate-controlled asymmetric synthesis. 1,5-Dialdehydes obtained from properly protected or unprotected uridine, ribothymidine, cytidine, inosine, adenosine and guanosine by metaperiodate oxidation reacted readily with tris(hydroxymethyl)aminomethane to provide the corresponding tricyclic derivatives with three new stereogenic centers. Through a double cyclisation cascade process the tricyclic compounds were obtained in good to high yields, with very high diastereoselectivity. Formation of one stereoisomer, out of the eight possible, was observed in all cases. The absolute configuration of the new stereotriad-containing tricyclic systems was aided by conventional NMR experiments followed by chemical shift calculations using an X-ray crystal structure as reference that was in good agreement with H–H distances obtained from a new ROESY NMR method. The synthesis was compatible with silyl, trityl and dimethoxytrityl protecting groups. A new reagent mixture containing ZnCl2, Et3SiH and hexafluoroisopropanol was developed for detritylation of the acid-sensitive tricyclano nucleosides.


 Please wait while we load your content...
Please wait while we load your content...