Single step aerosol assisted chemical vapor deposition of p–n Sn(ii) oxide–Ti(iv) oxide nanocomposite thin film electrodes for investigation of photoelectrochemical properties†
Abstract
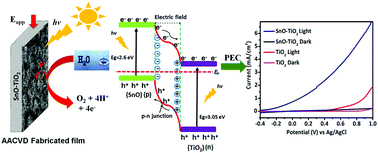
A homogeneous 1 : 1 solution of tetraisopropoxytitanium(IV) and [Sn(OAc)(dmae)]2 precursors in toluene was used at 450 °C, under argon, to deposit p–n-type tin(II) oxide–titanium(IV) oxide nanocomposite thin film electrodes by a single step aerosol assisted chemical vapor deposition (AACVD) technique. Field emission scanning electron microscopy (FESEM), X-ray diffractometry (XRD), Raman scattering, energy-dispersive X-ray spectrometry (EDX), X-ray photoelectron spectroscopy (XPS) and UV-vis absorption spectrophotometry were conducted to characterize the thin film electrodes. The deposited thin film electrodes were tested for their applications in water photolysis. A comparison of the photoelectrochemical properties of the deposited SnO–TiO2 composite electrodes with those of pristine TiO2 and SnO electrodes fabricated from tetraisopropoxytitanium(IV) and [Sn(OAc)(dmae)]2, respectively, indicates a higher photocatalytic activity as compared to pristine TiO2. This is possibly due to the incorporation of p-type SnO, which effectively promoted the separation of photogenerated charge carriers to rectify electron transfer from SnO to TiO2, leading to the separation of electron–hole pairs. The combined synergistic effect of p-type SnO and n-type TiO2, flower-like morphology and augmented ability to absorb solar light produced a significantly enhanced photocurrent of ∼4.3 mA cm−2 at 0.7 V vs. Ag/AgCl electrode. No obvious photocurrent decay was noted for prolonged stability measurements of up to 60 min under one sun illumination of 100 mW cm−2. With such a facile synthetic strategy and enhanced performance profile, the resulting material provides inspiration for producing new p–n-type composite photoanodes for better PEC performance using similar strategies.


 Please wait while we load your content...
Please wait while we load your content...