Different effects of water molecules on CO oxidation with different reaction mechanisms†
Abstract
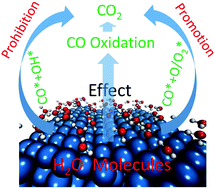
The effects of water molecules (promotion/prohibition) on CO oxidation remain debated. Herein, using density functional theory calculations, we demonstrate that water molecules can facilitate the CO + O/O2 oxidation process, but prohibit the CO + OH oxidation process, which is consistent with the experimental finding that water molecules have two distinct effects on CO oxidation. For the CO + O/O2 oxidation mechanisms, we find that the reactants were pushed towards each other due to the steric effect of the water molecules, which decreases the reaction barriers and promotes the CO + O/O2 oxidation process. For the CO + OH oxidation mechanisms, water molecules increase the stability of the COOH* intermeditae by H-bonds and van der Waals forces, which increase the barriers of the COOH* transformation process and the COOH*-tra dissociation process, and prohibit the CO + OH oxidation process. These results clarify the different effects of water molecules on CO oxidation and shed light on catalyst usage in the CO oxidation industry.


 Please wait while we load your content...
Please wait while we load your content...