Is ReO3 a mixed ionic–electronic conductor? A DFT study of defect formation and migration in a BVIO3 perovskite-type oxide†
Abstract
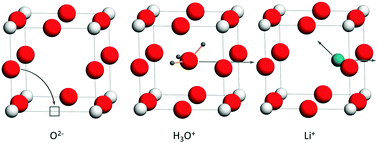
Density-functional-theory (DFT) calculations within the generalised gradient approximation (GGA) were used to examine the behaviour of point defects in the cubic BVIO3 perovskite-type oxide, ReO3. Energies of reduction and of hydration were calculated, and the results are compared with literature data for ABO3 perovskite oxides. The activation energies of migration were determined for O2−, H+, Li+, Na+, K+ and H3O+. An occupied A site in ReO3 is found to be beneficial to oxide-ion migration by a vacancy mechanism as well as to proton migration by a Grotthuss mechanism. Na+, K+ and H3O+ exhibit activation energies of migration higher than 2 eV, whereas Li+ is characterised by a very low migration barrier of 0.1 eV. Reasons for this behaviour are discussed. Our results suggest that H+, O2−, and especially Li+, are highly mobile ions in ReO3.


 Please wait while we load your content...
Please wait while we load your content...