Electrocatalytic performance of a zinc sulphide nanoparticles-modified carbon paste electrode for the simultaneous determination of acetaminophen, guanine and adenine†
Abstract
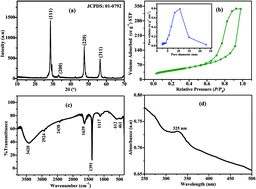
The simultaneous electroanalysis of acetaminophen (AC), guanine (G) and adenine (A) was successfully achieved on the zinc sulphide nanoparticles-modified carbon paste electrode (ZnS NPs/CPE) in phosphate buffer solution (PBS). Zinc sulphide nanoparticles (ZnS NPs) were prepared through a simple co-precipitation method and characterized via various spectroscopic techniques. The detailed studies confirmed that the as-synthesized ZnS NPs grew as high-density crystalline structures and they exhibit good electrocatalytic properties. The proposed method is selective and sensitive toward the development of an electrochemical sensor. The modified electrode showed acceptable reproducibility and stability under optimal conditions with high-accessible surface area and antifouling properties. The as-synthesized ZnS NPs enhanced the oxidation peak current and reduced the peak potential of AC, G and A. Furthermore, the sensor showed two linear ranges over the concentration from 1 to 15 μM and 15 to 120 μM. Based on the calibration plot, the limit of detection (LOD) was calculated and found to be 0.041, 0.036 and 0.031 μM for AC, G and A, respectively.


 Please wait while we load your content...
Please wait while we load your content...