Sulfonic acid functionalized silica nanoparticles as catalysts for the esterification of linoleic acid†
Abstract
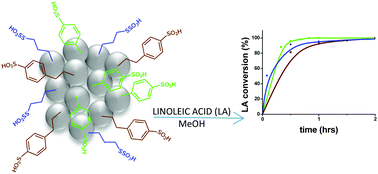
In this work we describe a comprehensive study on sulfonic acid functionalized silica nanoparticles (SiO2NPs) as novel catalysts for esterification of linoleic acid in the presence of methanol. SiO2NPs were covalently functionalized with propyl- or aryl-sulfonic acid groups (SO3H–) by several post-grafting methodologies through a sylilation reaction involving methoxy/ethoxy derivative organosilanes. Alkyl-sulfonic SiO2NPs were prepared by a two-step protocol using 3-mercaptopropyltrimethoxysilane followed by oxidation using H2O2 (1) or HNO3 (2) and chlorosulfonation (3). Aryl sulfonic acid SiO2NPs were prepared by a two-step protocol using phenyltriethoxysilane (a) and phenyltrimethoxysilane (b), followed by chlorosulfonation (4a and 4b) and sulfonation (5a and 5b); direct silylation with (4-chlorosulfonylphenyl)ethyltrimethoxysilane allowed the preparation of 6. The physicochemical characterization of all SO3H–SiO2NPs showed that the nanocatalysts 3, 4b and 6 exhibited the highest acidity capacity (estimated by EA sulphur content and potentiometric titration), with pHpzc lower than 2.0, suggesting that the use of either methoxy derivative organosilanes and chlorosulfonation steps played key roles in the surface acidity fine-tuning of SiO2NPs. These catalysts exhibited excellent performance in the heterogeneous esterification of linoleic acid with 100% conversion in a 2 h reaction time, with rate constants in the range of 1.80–5.55 h−1 and TOF values in the interval 53–498 h−1. The nanocatalyst 6 showed the highest stability and reusability for 5 catalytic cycles.


 Please wait while we load your content...
Please wait while we load your content...