Synthesis, characterization and application of a MnFe2O4@poly(o-toluidine) nanocomposite for magnetic solid-phase extraction of polycyclic aromatic hydrocarbons†
Abstract
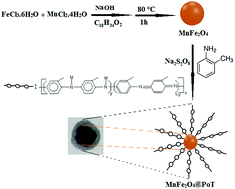
A nanocomposite of poly(o-toluidine)-coated MnFe2O4 magnetic nanoparticles with a core–shell structure was synthesized by the chemical co-precipitation method and employed as a magnetic adsorbent for solid-phase extraction of ten polycyclic aromatic hydrocarbons (PAHs). The as-prepared nanocomposite was characterized by Fourier transform infrared (FT-IR) spectroscopy, energy dispersive X-ray (EDX) spectroscopy, field emission scanning electron microscopy (FE-SEM), transmission electron microscopy (TEM), X-ray diffraction (XRD) spectrometry, vibrating sample magnetometry (VSM) and Brunauer–Emmett–Teller (BET) analysis for confirmation of their composition, morphology, size, phase, magnetic properties and specific surface area, respectively. Several important parameters influencing the extraction efficiency were investigated in order to establish the optimum conditions for the determination of PAHs in aqueous samples in combination with a gas chromatography-flame ionization detector (GC-FID). Under the optimized conditions, the calibration curves were linear in the concentration range of 0.05–100 ng mL−1 and 0.01–100 ng mL−1. Limits of detection of 0.3–5.5 pg mL−1 were achieved. The intra- and inter-day precisions (N = 5) and reproducibilities (N = 3) were from 1.4–7.1%, 2.1–9.2% and 2.4–11.7% at 0.5 ng mL−1 concentration, respectively. The method has been successfully applied to the analysis of trace PAHs in real water samples and the recoveries for the spiked samples were in the range of 88–105%. The experimental results showed that the proposed procedure was simple, convenient and fast to analyze PAHs.


 Please wait while we load your content...
Please wait while we load your content...