Encapsulation of a hexaaza macrocyclic nickel(ii) complex in zeolite Y: an experimental and theoretical investigation†
Abstract
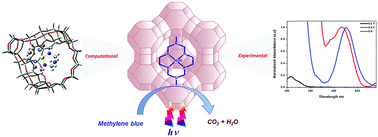
In this study, a 1,8-dimethyl-1,3,6,8,10,13-hexaazacyclotetradecane nickel(II) complex was encapsulated in the supercages of zeolite Y for the photocatalytic removal of methylene blue under visible light. The nickel(II) macrocyclic complex catalysts are known for their photooxidation property. Heterogenization of the homogenous catalyst is essential for their outstanding activity and reusability. The flexible ligand method was adopted for the encapsulation of the complex into the zeolite Y. The neat complex and encapsulated complex were characterized by DRS UV-vis, FTIR, powder XRD, cyclic voltammetry, BET and FESEM. The energy levels and transition probability of the encapsulated metal complex were varied due to the constrained environment of the zeolite. Density Functional Theory (DFT) calculations were performed to correlate with the experimental results for both the neat and encapsulated complexes. The DFT results reveal that the changes in the structural parameters and HOMO–LUMO energies offer useful insights into the nature of the host–guest complex and its reactivity. Besides this, the zeolite encapsulated complex shows photocatalytic activity towards the degradation of methylene blue under visible light irradiation with reusability. The mechanistic details on the photocatalytic activity were also proposed.


 Please wait while we load your content...
Please wait while we load your content...