Anion substitution in hydrogen-bonded organic conductors: the chemical pressure effect on hydrogen-bond-mediated phase transition†
Abstract
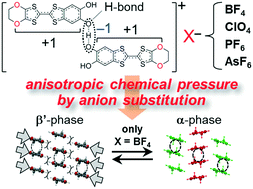
We have synthesized three kinds of novel hydrogen-bonded (H-bonded) organic conductor, β′-[H3(Cat-EDO-TTF)2]X (X = ClO4, PF6, AsF6; Cat-EDO-TTF = catechol-fused ethylenedioxytetrathiafulvalene), as anion-substituted analogues of the parent BF4 salt (X = BF4). These salts are all isostructural, however, only the BF4 salt shows a phase transition, upon which the molecular arrangement and physical properties are drastically changed in cooperation with bending of the H-bond. A systematic comparison of their crystal and electronic structures disclosed that a significant chemical pressure is generated by the present anion substitution, causing differences in their physical properties and phase transition nature. Interestingly, this chemical pressure is highly anisotropic, which exclusively changes the side-by-side interactions between the Cat-EDO-TTF skeletons in the π-stacking layer. Therefore, we conclude that this unique phase transition is caused by a cooperation of the intercolumnar side-by-side interactions with the π–π intra-dimer interactions and H-bond bending.

 Please wait while we load your content...
Please wait while we load your content...