Rapid synthesis of bicyclic lactones via palladium-catalyzed aminocarbonylative lactonizations†
Abstract
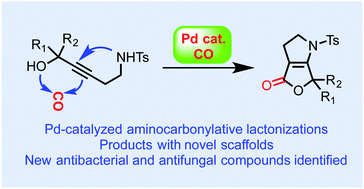
A novel and efficient palladium-catalyzed aminocarbonylative lactonization of amino propargylic alcohols has been developed to provide rapid access to various bicyclic lactones especially dihydropyrrole-fused furanones, which are novel structures and have not been explored in biological and medicinal settings. This method can also be used to access β-lactone products such as 16. Preliminary biological evaluations revealed that compounds 13h and 13s demonstrated promising activity against Clostridium difficile and compounds 13h, 13k, 13s, and 16b showed activity against several important fungal pathogens.
- This article is part of the themed collection: 2017 Emerging Investigators


 Please wait while we load your content...
Please wait while we load your content...